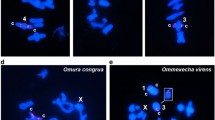
Abstract
We investigate regularities and restrictions in chromosome location of ribosomal RNA genes, analysed by fluorescent in situ hybridization (FISH), and their phenotypic expression assessed by nucleolus formation at first meiotic prophase cells, analysed by silver impregnation, in 49 grasshopper species. High variation was found for rDNA location between species within most genera analysed. The mean haploid number of rDNA loci detected by FISH was 2.47, but some species had up to 10 loci. Chromosome distribution of rDNA loci differed between the Gomphocerinae and Oedipodinae subfamilies, most loci being proximal to the centromere in the former and distal to it in the latter. Chromosomes 2, 3 and X frequently carried rDNA in Gomphocerinae species with 2n♂=17 chromosomes, whereas chromosomes 6 and 9 were the most frequent rDNA locations in the Oedipodinae. About 13% of the 126 rDNA loci detected by FISH were silent, although this figure might be even higher. The comparison of FISH and silver-impregnation results also suggested the existence of cryptic NORs, i.e. those forming small nucleoli with no apparent presence of rDNA revealed by FISH. This was especially clear after the same cells in two species were sequentially treated with both silver impregnation and FISH. The abundance of silent and cryptic loci might thus suggest that rDNA spreads through grasshopper genomes by the Dubcovsky and Dvorak mechanism—that is, the transposition of a few rRNA genes to new chromosome locations, their amplification giving rise to new NORs, and the elimination of the old NORs. The cryptic NORs might correspond to nascent NORs, i.e. a few rRNA gene copies moved to new locations, whereas the inactive rDNA loci might correspond to those being in the process of elimination.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bugrov A, Novikova O, Mayorov V, Adkison L, Blinov A (2006) Molecular phylogeny of Palaearctic genera of Gomphocerinae grasshoppers (Orthoptera, Acrididae). System Entomol 31: 362–368.
Cabrero J, Camacho JPM (1986) Cytogenetic studies in Gomphocerine grasshoppers. 2. Chromosomal location of active Nucleolar Organizing Regions. Can J Genet Cytol 28: 540–544.
Cabrero J, Bugrov A, Warchalowska-Sliwa E, López-León MD, Perfectti F, Camacho JPM (2003) Comparative FISH analysis in five species of Eyprepocnemidine grasshoppers. Heredity 90: 377–381.
Cai Q, Zhang DM, Liu ZL, Wang XR (2006) Chromosomal localization of 5S and 18S rDNA in five species of subgenus Strobus and their implications for genome evolution of Pinus. Ann Bot 97: 715–722.
Castilho A, Heslop-Harrison JS (1995) Physical mapping of 5S and 18S-25S rDNA and repetitive DNA sequences in Aegilops umbellulata. Genome 38: 91–96.
Castro J, Rodriguez S, Arias J, Sánchez L, Martínez P (1994) A population analysis of Robertsonian and Ag-NOR polymorphisms in brown trout (Salmo trutta). Theor Appl Genet 89: 105–111.
Castro J, Rodriguez S, Pardo BG, Sánchez L, Martínez P (2001) Population analysis of an unusual NOR-site polymorphism in brown trout (Salmo trutta L.). Heredity 86: 291–302.
Contreras D, Chapco W (2006) Molecular phylogenetic evidence for multiple dispersal events in gomphocerine grasshoppers. J Orth Res 15: 91–98.
Cuñado N, De la Herrán R, Santos JL, Rejón CR, Garrido-Ramos MA, Rejón MR (2000) The evolution of the ribosomal loci in the subgenus Leopoldia of the genus Muscari (Hyacinthaceae). Plant Syst Evol 221: 245–252.
Childs G, Maxson R, Cohn RH, Kedes L (1981) Orphons: dispersed genetic elements derived from tandem repetitive genes of eucaryotes. Cell 23: 651–663.
Datson PM, Murray BG (2006) Ribosomal DNA locus evolution in Nemesia: transposition rather than structural rearrangement as the key mechanism? Chromosome Res 14: 845–857.
Dobigny G, Ozouf-Costaz C, Bonillo C, Volobouev V (2002) “Ag-NORs” are not always true NORs: new evidence in mammals. Cytogenet Genome Res 98: 75–77.
Dubcovsky J, Dvorak J (1995) Ribosomal RNA multigene loci - Nomads of the Triticeae genomes. Genetics 140: 1367–1377.
Goodpasture C, Bloom SE (1975) Visualization of nucleolar organizer regions in mammalian chromosomes using silver stain. Chromosoma 53: 37–50.
Gosálvez J, López-Fernández C, Bella LJ, Butlin RK, Hewitt GM (1988) A hybrid zone between Chorthippus parallelus parallelus and Chorthippus parallelus erythropus (Orthoptera, Acrididae)—chromosomal differentiation. Genome 30: 656–663.
Gu ZJ, Hua X (2003) Physical mapping of the 18S-26S rDNA by fluorescent in situ hybridization (FISH) in Camellia reticulata polyploid complex (Theaceae). Plant Sci 164: 279–285.
Guimond A, Moss T (1999) A ribosomal orphon sequence from Xenopus laevis flanked by novel low copy number repetitive elements. Biol Chem 380: 167–174.
Hanson RE, IslamFaridi MN, Percival EA et al. (1996) Distribution of 5S and 18S-28S rDNA loci in a tetraploid cotton (Gossypium hirsutum L) and its putative diploid ancestors. Chromosoma 105: 55–61.
Hewitt GM (1979) Grasshoppers and Crickets. Berlin: Gebrüder Borntraeger.
Li DY, Zhang XY (2002) Physical localization of the 18S-5 center dot 8S-26S rDNA and sequence analysis of ITS regions in Thinopyrum ponticum (Poaceae: Triticeae): implications for concerted evolution. Ann Bot 90: 445–452.
López-León MD, Neves N et al. (1994) Possible origin of a B chromosome deduced from its DNA composition using double FISH technique. Chromosome Res 2: 87–92.
López-León MD, Cabrero J, Camacho JPM (1995) Changes in NOR activity pattern in the presence of supernumerary heterochromatin in the grasshopper Eyprepocnemis plorans. Genome 38: 68–74.
López-León MD, Cabrero J, Camacho JPM (1999) Unusually high amount of inactive ribosomal DNA in the grasshopper Stauroderus scalaris. Chromosome Res 7: 83–88.
Maluszynska J, Heslop-Harrison JS (1993) Physical mapping of rDNA loci in Brassica species. Genome 36: 774–781.
McClintock B (1934) The relationship of a particular chromosomal element to the development of the nucleoli in Zea mays. Z Zellforsch 21: 294–328.
Munro J, Burdon RH, Leader DP (1986) Characterization of a human orphon 28 S ribosomal DNA. Gene 48: 65–70.
Oakes M, Nogi Y, Clark MW, Nomura M (1993) Structural alterations of the nucleolus in mutants of Saccharomyces cerevisiae defective in RNA polymerase I. Mol Cell Biol 13: 2441–2455.
Pedrosa-Harand A, de Almeida CCS, Mosiolek M, Blair M, Schweizer D, Guerra M (2006) Extensive ribosomal DNA amplification during Andean common bean (Phaseolus vulgaris L.) evolution. Theor Appl Genet 112: 924–933.
Raskina O, Belyayev A, Nevo E (2004) Activity of the En/Spm-like transposons in meiosis as a base for chromosome repatterning in a small, isolated, peripheral population of Aegilops speltoides Tausch. Chromosome Res 12: 153–161.
Rooney AP, Ward TJ (2005) Evolution of a large ribosomal RNA multigene family in filamentous fungi: Birth and death of a concerted evolution paradigm. Proc Natl Acad Sci U S A 102: 5084–5089.
Rufas JS, Iturra P, De Souza W, Esponda P (1982) Simple silver staining procedures for the location of nucleolus and nucleolar organizer under light and electron microscopy. Arch Biol 93: 267–274.
Rufas JS, Esponda P, Gosálvez J (1985) NOR and nucleolus in the spermatogenesis of acridoid grasshoppers. Genetica 66: 139–144.
Salcedo FJ, Viseras E, Camacho JPM (1988) The B chromosomes of Locusta migratoria. III. Effects on the activity of nucleolar organizer regions. Genome 30: 387–394.
Santos JL, Fox DP (1988) A study of nucleolus organiser regions (NORs) in the subfamily gomphocerinae (Acrididae; Orthoptera) by means of an acridine orange staining procedure. Genet (Life Sci Adv) 7: 27–32.
Schubert I (1984) Mobile nucleolus organizing regions (NORs) in Allium (Liliaceae S-Lat)—inferences from the specifity of silver staining. Plant Syst Evol 144: 291–305.
Schubert I, Wobus U (1985) In situ hybridization confirms jumping nucleolus organizing regions in Allium. Chromosoma 92: 143–148.
Shan FC, Yan GJ, Plummer JA (2003) Cytoevolution of Boronia genomes revealed by fluorescent in situ hybridization with rDNA probes. Genome 46: 507–513.
Shishido R, Sano Y, Fukui K (2000) Ribosomal DNAs: an exception to the conservation of gene order in rice genomes. Mol Gen Genet 263: 586–591.
Taketa S, Ando H, Takeda K, Ichii M, Von Bothmer R (2005) Ancestry of American polyploid Hordeum species with the I genome inferred from 5S and 18S-25S rDNA. Ann Bot 96: 23–33.
Thomas HM, Harper JA, Meredith MR, Morgan WG, King IP (1997) Physical mapping of ribosomal DNA sites in Festuca arundinacea and related species by in situ hybridization. Genome 40: 406–410.
Viseras E, Camacho JPM (1984) Polysomy in Omocestus bolivari—endophenotypic effects and suppression of nucleolar organizing region activity in the extra autosomes. Canad J Genet Cytol 26: 547–556.
Viseras E, Cabrero J, Talavera M, Camacho JPM (1991) C-banding and NOR location variations in oedipodine grasshoppers. Cytobios 68: 165–177.
Zurita F, Jiménez R, Burgos M, Díaz de la Guardia RD (1998) Sequential silver staining and in situ hybridization reveal a direct association between rDNA levels and the expression of homologous nucleolar organizing regions: a hypothesis for NOR structure and function. J Cell Sci 111: 1433–1439.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Supplementary indormation on populations analysed for both rDNA and NOR activity locations, and the corresponding references. (DOC 196KB)
Rights and permissions
About this article
Cite this article
Cabrero, J., Camacho, J.P.M. Location and expression of ribosomal RNA genes in grasshoppers: Abundance of silent and cryptic loci. Chromosome Res 16, 595–607 (2008). https://doi.org/10.1007/s10577-008-1214-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-008-1214-x