Abstract
Objective
To determine: (1) the correlation of prostate cancer incidence and mortality with groundwater boron and selenium concentrations; and (2) the impact of boron on prostate cancer cell proliferation during co-treatment with alternative chemo-preventative agents, along with boron pre-treatment effects on cell sensitivity to ionizing radiation.
Methods
For regression analysis, data on prostate cancer incidence and mortality were obtained from the Texas Cancer Registry, while groundwater boron and selenium concentrations were derived from the Texas Water Development Board. Cultured DU-145 prostate cancer cells were used to assess the impact of boric acid on cell proliferation when applied in combination with selenomethionine and genistein, or preceding radiation exposure.
Results
Groundwater boron levels correlated with a decrease in prostate cancer incidence (R = 0.6) and mortality (R = 0.6) in state planning regions, whereas selenium did not (R = 0.1; R = 0.2). Growth inhibition was greater during combined treatments of boric acid and selenomethionine, or boric acid and genistein, versus singular treatments. 8-day boric acid pre-exposure enhanced the toxicity of ionizing radiation treatment, while dose-dependently decreasing the expression of anti-apoptotic protein Bcl-2.
Conclusions
Increased groundwater boron concentrations, across the state of Texas, correlate with reduced risk of prostate cancer incidence and mortality. Also, boric acid improves the anti-proliferative effectiveness of chemo-preventative agents, selenomethionine and genistein, while enhancing ionizing radiation cell kill.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The development of public health strategies, for the prevention and control of prostate cancer, has been hindered by a gap in our understanding of factors responsible for the large geographical disparity in disease risk. It is estimated that there were 232,090 new U.S. cases in 2005, along with significant regional variability in incidence and mortality [1].
Environmental exposure, by way of dietary intake, is receiving much attention as a contributor to prostate cancer prevention [2]. Several natural, plant-derived products are currently in clinical prevention trials, including: (n−3) polyunsaturated fatty acids, flaxseed, vitamin E, selenomethionine (SeM), soy protein isolate, isoflavones, genistein, lycopene and low-fat diets [3–5]. The largest prostate cancer clinical trial, SELECT, will determine the effectiveness of SeM and vitamin E supplementation in 35,000 men, over a 12-year period [2, 4].
Recently, boron (B), a metalloid element like selenium (Se), has been identified as a prostate cancer chemo-preventative agent. In a study based on the NHANES III database, the risk of prostate cancer was reported to be inversely proportional to dietary intake of B [6]. Studies have also shown that boric acid (BA), the chemical form of B in physiological fluids, reduced the growth of LNCaP-seeded tumors in nude mice, along with IGF-1 tissue concentrations and serum levels of prostate specific antigen (PSA) [7, 8]. Finally, using human prostate cancer cell lines, BA inhibited cell proliferation dose-dependently in androgen-dependent (LNCaP) and androgen-independent (DU-145 and PC-3) cells, without inducing cell death [9].
The B content of plants reflects both the soil concentrations in which they are grown, and the B levels of water applied during irrigation. B serves as an essential nutrient for plant growth, pollen tube formation and flowering, with deficiencies contributing to crop failures worldwide. B essentiality in plants is based on its role in maintaining the structure of scaffolding polysaccharides in the pectin of the cell wall that enable cells to resist the high turbid pressure associated with cell elongation. Monomers of the complex carbohydrate, rhamnogalacturonan, are cross-linked using borate-diol diester linkages between the two hydroxyl groups, located on apiose sugars of opposing polysaccharide chains [10, 11]. The rhamnogalacturonan II (RG-II) dimer is present in all human food plants, while being particularly abundant in vegetables and seed crops (legumes, nuts and carrots), as well as plant-based beverages (wine, fruit drinks and coffee). Furthermore, a borate transporter has been identified which traffics BA from plant roots to shoots [12].
In U.S. adult males, 31–70 years of age, the average daily intake of B from foods is 1.4 mg/day. Drinking water can contribute significantly to this value; yet unlike diet, groundwater chemistry at a particular location may be stable for long time periods, and its usage is often restricted to narrow geographical areas, thus making it a reputable data source [13, 14]. A recent study documented high B and Se concentrations in South Texas groundwater supplies, prompting us to correlate statewide groundwater B and Se levels with geographically variant prostate cancer risk [15]. In addition, we explored the biological relevance of this observation in studies comparing the anti-proliferative properties of BA exposure, in combination with SeM, genistein and radiation treatments, in human prostate cancer cells.
Materials and methods
B and Se water sampling
Data for linear regression analysis of average B and Se, in groundwater samples of Texas counties and state planning regions, was derived from the Groundwater Database of the Texas Water Development Board, http://www.twdb.state.tx.us. Sampling dates varied at wells evaluated for dissolved B (1939–2004) and dissolved Se (1964–2004). Multi-sampled wells were included in the analysis. The template for the B groundwater distribution map was acquired from the Texas Association of Regional Councils (TARC) website, http://www.txregionalcouncil.org.
Texas cancer statistics
Cancer incidence and mortality data have been provided by the Texas Cancer Registry, Cancer Epidemiology and Surveillance Branch, Texas Department of State Health Services, 1100 W. 49th Street, Austin, Texas, 78756, http://www.dshs.state.tx.us/tcr, or (512) 458–7,523. Prostate cancer incidence and mortality rates (1997–2001), for each county and state planning region were derived from the Texas Cancer Registry.
Cell culture
DU-145 prostate cancer cells, donated by Dr. Allan Pantuck, were cultured in RPMI 1640 media (Invitrogen) supplemented with 10% FBS, penicillin/streptomysin (100 U/ml; 100 μg/ml), and l-glutamine (200 mM) (Gemini Bioproducts, Woodlands, CA).
Experimental culture
BA and SeM stock solutions were prepared in ultra-pure water. Genistein stock was prepared in DMSO, while solvent concentration in media (0.1%) was pre-determined to be nontoxic. Prior toxicity evaluation of reagents, for doses imposing a 25–50% reduction in cell growth, led to the concentrations applied during experimentation. Experimental cells were cultured for eight days in treated media, with media replacement occurring daily. On day 8, cells were counted with a hemacytometer, while utilizing Trypan blue (Invitrogen) for exclusion of dead cells.
Ionizing radiation exposure
DU-145 cells were cultured in BA-supplemented media (0, 100, and 500 μM) for 8 days, in 12-well culture plates (Fisher), with daily media replacement. On day 8, media was aspirated partially and adherent cells were exposed to 1, 3 and 5 Gy’s of ionizing radiation from a 137Cs gamma ray source. Following exposure, cells were trypsinized, re-seeded onto 601 × 5 mm plates (1–3 × 104 cells/plate), and cultured for 9 days in low-B media. Adherent colonies were washed with PBS, fixed in methanol (10 min), allowed to air-dry following methanol removal, submerged in Giemsa stain (30 s), washed with PBS (8 min), and rinsed with de-ionized water. Stained colonies were counted without microscopic aid.
Western blot
DU-145 cells, in the presence of BA (0–1,000 μM), were cultured for seven days on 100 × 20 mm tissue culture plates, with daily media replacement. On day seven, near-confluent monolayers were washed with PBS, scraped off with a rubber policeman, and centrifuged for 5 min at 1,200 rpm. Pellets were lysed (250 mM NaCl, .1% NP40, 50 mM HEPES (pH 7.0), 5 mM EDTA, 1 mM DTT, 10% protease inhibitor mixture (Sigma # P8340)), sonicated, and incubated for 40 min at 4°C for protein extraction. 30 μg of protein was loaded per lane and separated on a 10% stacking, 12% separating (SDS-PAGE) gels for 30 min at 100 V, followed by 1 h at 200 V. Proteins were transferred onto nitrocellulose membranes for 4 h at 40 V, 4°C. Following overnight blocking of membranes (4 g nonfat dry milk, 38 mM Tris base, 125 mM NaCl 2.5, 100 μl Tween 20, 100 ml ddH2O), a 2-h primary antibody exposure (1/800 dilution) was applied, with nitrocellulose membranes subsequently being washed for 10 min in PBS/Tween 20 (0.1%). Membranes were then probed with a secondary antibody for 1 h (1/1,000 dilution), followed by 3 × 10 min washes in PBS/Tween 20 (0.1%). Membranes were subsequently submerged in ECL detection reagent (Amersham Int), wrapped in cellophane and exposed to X-ray film (Fuji). Bcl-2 and B-actin antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA).
Statistical analysis
SigmaStat 3.1 statistical software (Systat Software, Point Richmond, CA) was utilized for the following analyses: one-way analysis of variance, paired t-test, and linear regression analysis with correlation.
Results
Texas B and Se groundwater levels & prostate cancer rates
Linear regression analysis of prostate cancer incidence and mortality rates, as a function of average B and Se groundwater levels per state planning region, is shown in Fig. 3. Prostate cancer incidence and mortality was inversely correlated with B groundwater levels, with R values of 0.63 and 0.598, respectively (Fig. 1A, B). The predicted linear regression passed both ANOVA and t-tests (p-value < 0.002). In contrast, linear regression analysis of prostate cancer incidence and mortality, as a function of groundwater Se levels (Fig. 1C, D), showed no statistically relevant correlation, with R values of 0.144 and 0.154, respectively; while failing both ANOVA and t-tests (p-value > 0.4).
Scatter plots of mean groundwater B (A, B) and Se (C, D) concentrations in Texas state planning regions, as a function of prostate cancer incidence and mortality. y-axis represents the average annual age-adjusted (to the 2,000 U.S. standard population) prostate cancer incidence and mortality rates per 100,000 people. Dotted line represents 95% confidence interval, while solid line represents the linear regression
Cell proliferation
BA (250 μM) was evaluated singularly and in combination with SeM (15 μM) and genistein (15 μM), with respect to proliferative inhibition of DU-145 cells (Fig. 2). Cell proliferation, when cultured in the presence of SeM and BA, was significantly reduced relative to either compounds treatment alone; while pre- and post-treatment with BA had negligible inhibitory effects, relative to one another. As with SeM, BA and genistein co-administration induced a semi-additive proliferative inhibition; whereas pretreatment with BA sensitized cells to genistein toxicity, while the reverse treatment was less toxic.
Effects of 8-day exposure to 250 μM BA, in combination with 15 μM selenomethionine (SeM), (A) and 60 μM genistein (Gen), (B), on the proliferation of cultured DU-145 prostate cancer cells. Cells were treated with BA alone (BA); with the alternative compound alone (SeM or Gen); with both compounds simultaneously (BA + SeM or Gen); and in sequence, with BA treatment for the 1st 4 days followed by alternative compound for the last 4 days (BA ▸SeM or Gen), and the reverse sequence (SeM or Gen ▸BA). Mean ( sem, n = 6. *Statistically significant from single exposures (p-value < 0.003). **Statistically significant from reverse sequence exposure (p-value < 0.04)
Ionizing radiation sensitivity
DU-145 cells exposed to ionizing radiation (1, 3, and 5 Gy), following 8-days in culture with 0–500 μM BA, displayed enhanced sensitivity to induced gross toxicity (Fig. 3A). Relative to control cells, BA pre-treatment dose-dependently reduced the number of viable cells capable of seeding and proliferating into detectable colonies. Also, following a 7-day exposure to BA (0–1,000 μM), DU-145 cells displayed a dose-dependent reduction in Bcl-2 protein expression (Fig. 3B).
Effects of 8-day BA pre-treatment on the killing efficiency of ionizing radiation, and Bcl-2 protein expression, in DU-145 prostate cancer cells. (A), BA pre-treatments of 0 μM (X), 100 μM (■), and 500 μM (▲) were applied prior to ionizing radiation exposure (0–5 Gy); mean ± sem, n = 4. *Statistically significant from 0 μM pre-treatment at like radiation exposure (p-value > .005). (B) Cells given 7-day treatments of BA (0–1,000 μM) were evaluated for Bcl-2 protein expression by western blot
Discussion
The consistent quality of groundwater, in equilibrium with its environment, makes it an attractive choice for assessing health effects displaying strong geographic associations. In Texas, groundwater provides 60% of the water supply for drinking and irrigation, and varies in B concentration across the state (30–16,200 μg B/L) [5]. This wide range provided an opportunity to assess prostate cancer risk, as a function of groundwater B concentrations. The geographical distribution of groundwater B across the state of Texas is indicated with regions of higher B in darker hues (Fig. 4). Areas with relatively high groundwater B concentrations occupy portions of central and south Texas. These central and southern areas overlap portions of the Trinity Group and Gulf Coast Aquifers, respectively. Both aquifers comprise sediment deposited in fluvial, deltaic, and shallow marine environments. Portions of the Trinity Group also contain carbonates and evaporites. Brackish seawater trapped in marine sediment and evaporites are possible sources of B [16]. A previous investigation attributed high groundwater B concentrations in south Texas to naturally occurring, brackish water in the Gulf Coast Aquifer [15].
One of the limitations of this study was that large cities, including Houston, Dallas-Fort Worth and Austin, receive the majority of their drinking water from surface sources of inconsistent quality. A second limitation was the number of Afro-American and Hispanic men residing in rural counties and planning regions was too small to allow analysis of prostate cancer rates in ethnic sub-populations, as a function of the state’s groundwater geography. Higher rates of prostate cancer were not observed in areas of low median family income in south Texas where groundwater levels of boron were high. In addition to potential ethnic and demographic cofounders, environmental factors could also contribute to the observed cancer rates. For example, poor air quality in the Houston area of southeast Texas could contribute to higher observed cancer rates in that region.
In Texas, counties are grouped into state planning regions for the purpose of water supply and quality planning, health resource distribution, and assessment of the geographic adequacy of facilities, providers, and cancer services [17]. We compiled prostate cancer incidence and mortality rates using data from the Texas Cancer Registry [18], and B groundwater concentrations from the Texas Water Development Board [19], for the 24 planning regions of the state. When groundwater concentrations were plotted relative to the state planning regions, statistical analysis showed the data were normally distributed, and the inverse correlations of prostate cancer incidence (R = 0.6) and mortality (R = 0.6), with groundwater B, were highly significant (p < 0.002). Planning region B groundwater concentrations, along with the empirically-derived water consumption values of 3.6 l/d for males between 51 and 70 y, predict a B intake ranging from 619 μg B/d to 11,979 μg B/d in regions utilizing groundwater as their major drinking water source [20]. This 19-fold variance in B intake coincided with a 37% difference in prostate cancer incidence, and falls within the safe range of B ingestion even after adding in the dietary component of 1.4 mg B/d [14].
The correlation between groundwater Se and prostate cancer incidence and mortality rates was non-significant. Consumption of 3.6 l/d would have provided a mean inorganic Se intake of 36 μg/day with a range of 10–98.6 μg/day. The recommended dietary intake of Se is 55 μg/day so this range should have provided a sufficient gradient to observe an effect of inorganic Se on prostate cancer incidence should one exist [21]. Others have reported a decrease in the incidence of prostate cancer in men living in regions of low soil Se content subsequent to dietary supplementation with an organic form of Se, SeM [22]. Until the mechanism is discovered through which Se achieves its protective effect, a more explicit explanation for the absence of an effect cannot be provided. The favored explanation, at present, is that Se protects through its antioxidant enzymes, primarily the glutathione peroxidases. However, Se is also anti-angiogenic and protects against basement membrane thickening [23, 24].
Knowledge of the relative effectiveness of anti-cancer agents is important for understanding both their mode of action, and identifying possible inhibitory interactions with alternative treatments. The potential of BA to interfere with SeM and genistein’s capacity to inhibit cell proliferation was studied using DU-145 human prostate cancer cells. Rather than blocking their induced growth inhibition, BA exhibited a semi-additive effect when administered simultaneously with SeM and genistein. Although sequential treatments with SeM and BA did not display statistical differences in effectiveness, BA treatment followed by genistein enhanced proliferative inhibition. Radiation therapy is frequently used, alone and in combination with chemical reagents, for treatment of both localized and metastatic prostate tumors [25]. Cell kill resulting from gamma irradiation of DU-145 prostate cancer cells has been shown to be enhanced when cells are pre-treated with genistein and selenite [26, 27]. The same positive effect occurred when cells were pretreated with BA, prior to ionizing radiation exposure. Because a 137Cs source of gamma irradiation was utilized for exposures, rather than a pulse of neutrons, 10B nuclei fractionization was not a contributor to the observed toxicity, as occurs during the administration boron neutron capture therapy [28].
Bcl-2 has been established as an anti-apoptotic protein and one that conveys resistance to radiotherapy in prostate cancer patients [29, 30]. Supporting Bcl-2’s role in ionizing radiation-induced cell death, curcumin acts both as a down-regulator of Bcl-2 expression and a sensitizer to ionizing radiation kill in PC-3 prostate cancer cells [31]. We examined Bcl-2 expression in DU-145 cells subjected to prolonged BA treatments, and found protein concentrations to be reduced in a dose-dependent manner. This suggests that BA functions as a sensitizer to radiation treatment by inhibiting Bcl-2. The cell death most likely occurred through a pathway that did not involve Bcl-2 because DU-145 cells treated with either a Bcl-2 antisense oligonucleotide or BA undergo proliferation inhibition without cell death [9, 32, 33].
In conclusion, groundwater B concentrations are inversely correlated with prostate cancer incidence and mortality rates in the state of Texas. When applied in combination with SeM and genistein, BA enhances the ability of either reagent to inhibit DU-145 cell proliferation, while prior treatment of cells with BA sensitizes to ionizing radiation kill.
References
Jemal A, Murray T, Ward E, et al (2005). Cancer statistics. CA Cancer J Clin 55(1):10–30
Willis MS, Wians FH (2004) The role of nutrition in preventing prostate cancer: a review of the proposed mechanism of action of various dietary substances. Clinica Chimica Acta 330: 57–83
Djavan B, Zlotta A, Schulman C, et al (2004) Chemotherapeutic prevention studies of prostate cancer. J Urol 171(2 Pt 2): S10–S13
Klein EA, Thompson IM, Lippman SM, et al (2001) SELECT: the next prostate cancer prevention trial. Selenium and Vitamin E Cancer Prevention Trial. J Urol 166(4): 1311–1315
Cui Y, Winton MI, Zhang ZF, et al (2004) Dietary boron intake and prostate cancer risk. Oncol Rep 11(4): 887–892
Gallardo-Williams MT, Chapin RE, King PE, et al (2004) Boron supplementation inhibits the growth and local expression of IGF-1 in human prostate adenocarcinoma (LNCaP) tumors in nude mice. Toxicol Pathol 32: 73–78
Gallardo-Williams MT, Maronpot RR, Wine RN, Brunssen SH, Chapin RE (2003) Inhibition of the enzymatic activity of prostate specific antigen by BA and 3-nitrophenyl boronic acid. Prostate 54: 44–49
Barranco WT, Eckhert CD (2004) BA inhibits human prostate cancer cell proliferation. Cancer Lett 216(1): 21–9
Kobayashi M, Matoh T, Azuma J (1996) Two chains of rhamnogalacturonan II are cross-linked by borate-diol ester bonds in higher plant cell walls. Plant Physiol 110(3): 1017–1020
O’Neill MA, Warrenfeltz D, Kates K, et al (1997) Rhamnogalacturonan-II, a pectic polysaccharide in the walls of growing plant cell, forms a dimer that is covalently cross-linked by a borate ester. In vitro conditions for the formation and hydrolysis of the dimer. J Biol Chem 271(37): 22923–22930
Takano J, Noguchi K, Yasumori M, et al (2002) Arabidopsis boron transporter for xylem loading. Nature 420(6913): 337–340
Anderson DL, Cunningham WC, Lindstrom TR (1994) Concentrations and intakes of H, B, S, K Na, Cl, and NaCl in foods. J Food Comp Anal 7: 59–82
Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. A report of the panel on micronutrients, subcommittees on upper reference levels of nutrients and of interpretation and use of dietary reference intakes, and the standing committee on the scientific Evaluation of dietary reference Intakes. (2001) Food and Nutrition Board, Institute of Medicine, National Academy Press, Washington, D.C., p C-13
Hudak PF (2004) Boron and selenium contamination in south Texas groundwater. J Environ Sci Health A Tox Hazard Subst Environ Eng 39(11–12):2827–2834
Hem JD (1985) Study and interpretation of the chemical characteristics of natural water. U.S. Geological Survey Water Supply Paper 2254
http://www.tcc.state.tx.us/tcplan/goal2/goal2_obje_frames.html
http://www.dshs.state.tx.us/tcr, accessed May 31, 2005
http://www.twdb.state.tx.us, accessed May 31, 2005
Panel on Dietary Reference Intakes for Electrolytes and Water (2004) Dietary reference intakes for water, potassium, sodium, chloride, and sulfate. National Academies Press, Washington, D.C., p 494.
Dietary reference intakes for vitamin C, vitamin E, selenium, and carotenoids (2000) Selenium. National Academies Press, Washington, D.C., p. 284.
Clark LC, Dalkin B, Krongrad A, et al (1998) Decreased incidence of prostate cancer with selenium supplementation: results of a double-blind cancer prevention trial. Br J Urol 81: 730–734
Eckhert CD, Lockwood MK, Shen B (1993) Influence of selenium on the microvascular circulation of the retina. Microvas Res 45: 74–82
Thornber JM, Eckhert CD (1984) Protection against sucrose induced retinal capillary damage in the Wistar rat. J Nutr 114:1070−1075
Bischof M, Abdollahi A, Gong P, et al (2004) Triple combination of irradiation, chemotherapy (pemetrexed), and VEGFR inhibition (SU5416) in human endothelial and tumor cells. Int J Radiat Oncol Biol Phys 60(4):1220–1232
Yan SX, Ejima Y, Sasaki R, et al (2004) Combination of genistein with ionizing radiation on androgen-independent prostate cancer cells. Asian J Androl 6(4): 285–290
Husbeck B, Peehl DM, Knox SJ (2005) Redox modulation of human prostate carcinoma cells by selenite increases radiation-induced cell killing. Free Radic Biol Med 38(1):50–57
Bendel P (2005) Biomedical applications of 10B and 11B NMR. NMR Biomed 18(2): 74–82
Rudner J, Jendrossek V, Belka C (2002) New insights in the role of Bcl-2 Bcl-2 and the endoplasmic reticulum. Apoptosis 7(5): 441–447
Rosser CJ, Reyes AO, Vakar-Lopez F et al (2003) Bcl-2 is significantly overexpressed in localized radio-recurrent prostate carcinoma, compared with localized radio-naive prostate carcinoma. Int J Radiat Oncol Biol Phys 56(1): 1–6
Chendil D, Ranga RS, Meigooni D, Sathishkumar S, Ahmed MM (2004) Curcumin confers radiosensitizing effect in prostate cancer cell line PC-3. Oncogene 23(8): 1599–1607
Campbell MJ, Dawson M, Koeffler HP (1998) Growth inhibition of DU-145 prostate cancer cells by a Bcl-2 antisense oligonucleotide is enhanced by N-(2-hydroxyphenyl) all-trans retinamide. Br J Cancer 77(5): 739–744
Raffo A, Lai JC, Stein CA, et al (2004) Antisense RNA down-regulation of bcl-2 expression in DU145 prostate cancer cells does not diminish the cytostatic effects of G3139 (Oblimersen). Clin Cancer Res May 10(9): 3195–3206
Acknowledgments
We thank Dr. Allan Pantuck and Randy Kallilew for their expertise concerning the culturing of prostate cancer cells, along with Kurt Hafer and Cecelia Chan for aiding in the administration of ionizing radiation. Funding for this research was provided by: DOD prostate idea grant DAMD17–03-1-0067 (CD Eckhert) and UC TRS&TP (WT Barranco). In the spirit of full disclosure, we declare that since 1997 the National Institutes of Health has without exception declined funding of all grant applications submitted by CE on the role of boron in biology or cancer with recent applications triaged prior to full review.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s10552-007-9023-7.
Rights and permissions
About this article
Cite this article
Barranco, W.T., Hudak, P.F. & Eckhert, C.D. Evaluation of ecological and in vitro effects of boron on prostate cancer risk (United States). Cancer Causes Control 18, 71–77 (2007). https://doi.org/10.1007/s10552-006-0077-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10552-006-0077-8


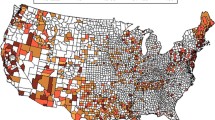




 ), 400–600 μg/l (
), 400–600 μg/l ( ), 600–800 μg/l (
), 600–800 μg/l ( ),800–1,000 μg/l (
),800–1,000 μg/l ( ), > 1,000 μg/l (
), > 1,000 μg/l ( )
)