Abstract
Purpose
Breast cancer is the most common cancer and leading cause of cancer death in women. Body composition parameters, especially those related to muscle, have become a growing focus of cancer research. In this review, we summarize the literature on breast cancer and muscle parameters as well as combine their outcomes for overall survival (OS), time to tumor progression (TTP), and chemotherapy toxicity in a meta-analysis.
Methods
A systematic search of the literature for randomized controlled trials and observational studies was conducted on MEDLINE, Cochrane CENTRAL, and EMBASE through May 1, 2019. Two reviewers independently searched and selected. Meta-analysis was conducted using a random-effects model. The risk of bias was evaluated using the Newcastle–Ottawa quality assessment for cohorts and GRADE summary of findings tool from Cochrane.
Results
A total of 754 articles were screened from which 6 articles and one abstract were selected. Using skeletal muscle index (SMI), patients classified as sarcopenic had a 68% greater mortality risk compared to non-sarcopenic patients (HR 1.68 95% CI 1.09–2.59, 5 studies) (p = .02) (i2 = 70%). Low muscle density was not predictive of OS (HR 1.44 95% CI 0.77–2.68, 2 studies) (p = .25) (i2 = 87%). Patients with sarcopenia (56%) had more grade 3–5 toxicity compared to non-sarcopenic (25%) (RR 2.17 95% CI 1.4–3.34, 3 studies) (p = .0005) (i2 = 0%). TTP was nearly 71 days longer in advanced/metastatic patients classified as non-sarcopenic compared to patients with sarcopenia (MD − 70.75 95% CI − 122.32 to − 19.18) (p = .007) (i2 = 0%).
Conclusion
Our synthesis of the literature shows that patients with sarcopenia have more severe chemotherapy toxicity as well as shorter OS and TTP, and that low muscle density is prognostic of OS for women with metastatic breast cancer. Our findings suggest that in clinical practice, body composition assessment is valuable as a prognostic parameter in breast cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most common cancer and leading cause of cancer deaths in women. World Health Organization (WHO) data from 2018 report over 2 million new cases per year worldwide about 627,000 deaths from breast cancer [1]. In the United States, there were about 250,000 new cases and over 40,000 deaths per year [2, 3] in 2016. The majority of breast cancers are diagnosed at an early stage (I–III) and 20–30% will eventually develop metastases [4]. Breast cancer research leading to more effective treatment options has reduced mortality in all stages of disease [5], and the pursuit of new prognostic parameters coupled with personalized therapies holds the promise of further reductions in recurrence and mortality.
Body composition parameters, especially muscle quantity and density, have become a growing focus of research in cancer prognosis. Sarcopenia is the progressive degeneration of muscle mass [6] and is a well-known condition in older persons [7]. It is typically assessed using diagnostic imaging techniques such as computed tomography (CT), dual-energy X-ray absorptiometry (DEXA), and magnetic resonance imaging (MRI) or bioelectrical impedance analysis (BIA). Systematic reviews and meta-analysis of sarcopenia in patients with cancer have shown that sarcopenia is associated with poorer survival in pancreatic [8], esophageal [9], gastric [10], colorectal [11], and lung cancer [12]. Muscle density is another measure of body composition and pertains to fatty infiltration of the muscle [13]. It can be assessed indirectly through CT imaging evaluation of mean skeletal muscle density (SMD) expressed in Hounsfield Units (HU) [14]. Similar to sarcopenia, low SMD is associated with a poorer prognosis in multiple cancers, [15] specifically colorectal [14], pancreatic [16], and ovarian [17] cancer.
Starting in 2009 but especially over the past 3 years, a growing number of studies have evaluated body composition and prognosis in women with breast cancer. However, there have been no meta-analyses correlating body composition parameters with key outcome events in patients with breast cancer. The aim of this systematic review and meta-analysis is to summarize the literature and evaluate the strength of the evidence for the prognostic value of sarcopenia and low muscle density in breast cancer prognosis, severe chemotherapy toxicity, time to tumor progression, and OS. Should these measures prove to be important prognostic parameters, they could be incorporated into clinical practice to assist in personalized treatment decisions and better outcomes.
Materials and methods
Search strategy
This systematic review is registered at PROSPERO (international database of prospectively registered systematic reviews) [18] (CRD42019131280). With the help and guidance of the University of North Carolina Health Sciences Library in the use of Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia), two reviewers (GFA and KN) independently performed a search of the literature in several databases (PubMed/Medline, Cochrane Central Register for Clinical Trials, and EMBASE) with a publication cut-off date of May 1, 2019. References from published systematic reviews of sarcopenia that included breast cancer patients were included in the review.
The search strategy (see Supplementary Material/Appendix 1 for search terms) followed the PICO framework [19], using combined terms and MeSH (Medical Subject Headings of the National Library of Medicine) descriptors and the following criteria:
-
Population: women diagnosed with breast neoplasia +18 years old
-
Interest: patients with sarcopenia or low skeletal muscle density
-
Comparison or control: non-sarcopenic or normal skeletal muscle density patients
-
Outcomes: primary outcome—overall survival. Secondary outcomes: treatment toxicity and time to tumor progression
-
Study design: observational or randomized controlled trial (including abstracts)
-
Timing: any time after diagnosis
Two independent reviewers (GFA and KN) selected the articles, extracted the data, and analyzed the data. Any discrepancies were resolved by consensus between the reviewers or after discussion with a third author (GRW). The reviewers evaluated the title and abstract for all studies that were identified through the COVIDENCE search strategy. Full texts were evaluated when there was insufficient information in the title and abstract to make a decision about inclusion or exclusion. References in reviewed and excluded articles were examined to identify studies that may not have been identified through the primary search strategy. The search was not limited to the English language. A list of potential studies for inclusion in the systematic review was generated through this process.
Data extraction
Extracted data included details regarding authors, year of publication, country of the study population, inclusion/exclusion criteria (patient characteristics), and stage of cancer. Data were also extracted regarding the definition of sarcopenia (cut-points) and study outcomes (e.g., overall survival, toxicity, time to tumor progression).
Data synthesis and statistical analysis
A random-effects meta-analysis model was applied to take into account that patient populations in the included studies were in different disease stages and received different treatments. Estimation of a common effect size was not possible in light of the heterogeneity of the studies. Therefore, inverse-variance weighting was used to pool estimates from the included studies. Rev-Man 5.3 was used (Cochrane Collaboration) to combine the results across studies. Heterogeneity was evaluated using Q test (c2 Chi-square test) to assess whether observed differences in results are compatible with chance alone. A low p value (or a large Chi squared statistic relative to its degree of freedom) provides evidence of heterogeneity (variation in effect estimates beyond chance) and is expressed in the i2 statistic [20, 21]. According to the Cochrane handbook, [22] a guide to interpretation of the i2 statistic is as follows: 0% to 39%—might not be important, 40% to 59%—may represent moderate heterogeneity, 60% to 89%—may represent substantial heterogeneity, 90% to 100%—considerable heterogeneity. The importance of the observed value of i2 depends on the magnitude and direction of effects and the strength of evidence for heterogeneity. Statistical significance was defined at the 0.05 level.
Dichotomous data were used to assess inverse variance and risk ratio (RR). Generic inverse variance was expressed in log hazard ratios (HR) with 95% confidence intervals (CI) for overall survival. For continuous data, we used standard deviation (SD) that was either available in the text or calculated using data from the text, expressed as mean difference (MD) between groups with 95% CI. Subgroup analysis was done according to cancer stage (e.g., stage I–III vs. metastatic) when possible.
Outcome definitions
Outcomes included (a) overall survival (OS)—time from sarcopenia diagnosis until death from any cause, (b) time to tumor progression (TTP)—length of time from date of diagnosis or start of treatment for a disease until the disease starts to worsen or spread to other parts of the body (not applicable to early stages), and (c) toxicity grades 3–5 according to National Cancer Institute Common Toxicity Criteria for Adverse Events (NCI-CTCAE; Version 4.03) including hematologic toxicity (neutropenia, thrombocytopenia, anemia), febrile neutropenia, and common non-hematologic toxicities such as neurotoxicity and gastrointestinal (GI) toxicity (stomatitis, diarrhea, vomiting).
Quality assessment
The Newcastle–Ottawa Quality (NOQ) [23] assessment form for cohort analysis was used by two independent researchers (GFA and GRW) to assess methodological quality and standard of outcome reporting in the included studies [24]. The quality of evidence was assessed using the GRADE (Cochrane Group) analysis of findings which summarizes the level of evidence and the relative or absolute impact of each analyzed outcome.
Results
Literature search
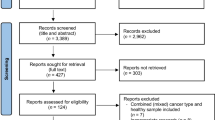
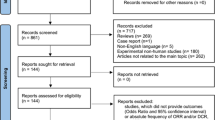
A total of 754 articles were identified through the search strategy. Figure 1 presents the PRISMA diagram (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) [25]. After duplicates were removed, the two primary reviewers screened titles and abstracts for 657 articles. For the articles that remained after the initial screen, 21 full texts were reviewed for eligibility. Most articles were excluded because they did not include information on outcomes selected for our review or did not include comparison groups [26,27,28,29,30,31,32,33,34]. Ultimately, seven studies were selected for inclusion in the systematic review—six articles [29, 35,36,37,38,39] and one abstract [40] with a total of 4065 patients.
Overview of included studies
Table 1 provides an overview of the included studies. Patients in four studies were early-stage breast cancer [35, 36, 38] and in four studies they were advanced/metastatic [29, 37, 39, 40]. Two studies focused on capecitabine/taxane treatment [29, 39] (both in patients with metastatic breast cancer), while all other studies entailed multiple chemotherapy treatments. Six studies used lumbar L3 CT scans to assess sarcopenia [29, 35,36,37, 39, 40] and one study used DEXA scan [38]. The mean proportion of patients with sarcopenia was 39.8% (range 25–66.9%).
Body composition definitions
Sarcopenia using Skeletal Muscle Index (SMI) and cut-points varied across the studies. Three studies used < 41 cm2/m2 [29, 36, 37], one study used < 40 cm2/m2 [35], one study used < 38.5 cm2/m2 [39] and in one study the sarcopenia cut-point was not defined [40]. The seventh study used DEXA with a cut-point of < 5.45 kg/m2 [38]. Low skeletal muscle density (SMD) using mean attenuation in Hounsfield Units (HU) was assessed in three studies, with one study using < 37.8 HU [35] and two using < 41 HU for BMI < 25 and < 33 HU for BMI > 25 [37, 39] as cut-points.
Primary outcome
Overall survival
Using skeletal muscle index (SMI) for all patients in five studies, patients classified as sarcopenic had a 68% greater mortality risk compared to patients with non-sarcopenic patients (HR 1.68 95% CI 1.09–2.59, 5 studies) (p = .02) (i2 = 70%) (Fig. 2). Subgroup analysis by stage showed that sarcopenia was of prognostic value in early breast cancer (p = .05) but not prognostic in metastatic (p = .44).
Low muscle density was not predictive of overall survival (HR 1.44 95% CI 0.77–2.68, 2 studies) (p = .25) (i2 = 87%) (Fig. 3). However, in subgroup analysis, low muscle density was significantly related to shortened survival in patients with metastatic breast cancer (p = .0009) but not for early breast cancer (p = .38).
Secondary outcomes
Metastatic patients with sarcopenia (56%) had more grade 3–5 toxicity compared to patients classified as non-sarcopenic (25%) (RR 2.17 95% CI 1.4–3.34, 3 studies) (p = .0005) (i2 = 0%) (Fig. 4).
Time to tumor progression in patients with advanced/metastatic breast cancer was nearly 71 days longer in non-sarcopenic compared to sarcopenic patients (Mean Deviation − 70.75 95% CI − 122.32 to − 19.18) (p = .007) (i2 = 0%) (Fig. 5).
Quality assessment
Table 2 summarizes the NOQ results for studies included in the review. Six studies were rated “good” [29, 35,36,37,38,39] and one “fair” [40]. The grade summary of findings (Table 3) shows that the certainty of the effect estimate was moderate for all outcomes.
Discussion
This systematic review and meta-analysis evaluated the evidence of body composition—specifically sarcopenia and low muscle density. An important finding is that adverse outcomes varied between women with early breast cancer and those with metastatic breast cancer. Specifically, SMI was prognostic for mortality risk in early breast cancer but not in metastatic, while low muscle density was prognostic for OS in women with metastatic but not with early breast cancer. Risk for grade 3–5 chemotherapy toxicity was significant for both early and metastatic breast cancer.
Interest continues to grow in exploring multiple body composition parameters of prognosis in patients with cancer [8, 10, 16]. This interest is evident in the publishing dates for most studies included in our meta-analysis. It is also evident in a recently published systematic review of sarcopenia and breast cancer using CT scans that included 15 articles [41] and other recent publications exploring the influence of body composition in breast cancer patients [30]. Research will continue to expand as advances in the use artificial intelligence for body imaging analysis [42, 43] make the assessment of multiple parameters of body composition increasingly reliable and accessible in clinical practice.
Sarcopenia (low SMI) was the first body composition parameter to provide independent prognostic information in cancer patients in general (e.g., overall survival, chemotherapy toxicity, surgical toxicity) and is the reason for its widespread use in body composition research. Our finding that sarcopenia is prognostic for OS, TTP, and high-grade chemotherapy toxicity in breast cancer has been observed in prior meta-analyses of sarcopenia in other cancer types [8, 10]. However, our subgroup analyses showed that sarcopenia was not always significant in early breast cancer compared to metastatic breast cancer. We hypothesize that the small number of patients in each group may be affecting these results. Of note, two studies not included in our analysis because they did not have data necessary for this analysis showed linear correlation between muscle density and mortality as well as chemotherapy toxicity in patients with breast cancer [28, 44].
It has been hypothesized that sarcopenia is prognostic because muscle acts as an energy storage compartment which may be used in catabolic periods such as cancer and chemotherapy [45]. The impact of sarcopenia may be due to a combination of vulnerability to cancer and its treatment, due to low physical reserves or in more advanced cases due to sub-optimal treatment options in patients with limited physical reserve [7]. Low SMD, which reflects high muscle fat content rather than low muscle mass, has been associated with poor survival in some cancers [14,15,16, 36] as it promotes higher systemic inflammation and insulin resistance which may reduce body defenses and stimulate neoplasia growth [46, 47]. Our evaluation did not find a statistically significant difference for OS in low SMD compared to normal SMD patients with breast cancer; however, this may be due to the heterogeneity of outcomes and the small number of studies eligible for our analysis. A large observational study showed that low muscle attenuation was associated with dose reductions and that women whose doses were reduced had a higher chance of dying from breast cancer [48].
Body composition markers other than SMI and SMD have been analyzed for outcomes in a variety of cancer populations. For example, Weinberg et al. [33] combined SMI and SMD to create skeletal muscle gauge (SMG) which was shown to be a strong predictor of outcomes in patients with cancer [29, 44]. Visceral adipose tissue (VAT) area, which increases insulin resistance and inflammation [49], is associated with poor outcomes in patients with colorectal cancer who are receiving bevacizumab or undergoing surgery [50, 51]. A recent abstract showed that low muscle density is associated with dose reductions in patients with breast cancer receiving taxanes [48]. One study also showed that VAT correlated with OS [27]; however, other studies have not associated VAT with prognosis [31, 36]. A large observational trial found an association between low subcutaneous adipose tissue area and prognosis for patients with various solid neoplasia [52], and in breast cancer an increase in both VAT and SAT was associated with poor survival [53]. Despite studies identifying sarcopenic obesity as a risk factor for poor outcomes in multiple cancers [54], our search identified only one study to date in breast cancer that analyzed sarcopenic obesity [37] and which showed poor overall survival in women with metastatic breast cancer.
Most studies included in our analysis were rated as good on the Newcastle–Ottawa Quality assessment of cohorts. However, the GRADE summary of findings was moderate in all outcomes either because of high heterogeneity or the limited the number of trials available for analysis, thereby creating a high risk for bias. It is of concern that several excellent studies could not be included in our analysis because of the heterogeneous manner in which their data were assessed. For example, two trials could not be included in our analysis because they did not provide cut-points for defining patients with sarcopenia or low muscle density [28, 44]. In another study, the outcome “time to progression” could not be evaluated because low SMD was not defined [29]. We also note that both sarcopenia and low muscle density had some variation in cut-points among the studies that could influence final results. A consensus on definitions and cut-points would improve the quality of future studies and the trustworthiness of results. Despite the existence of European [55] and Asian [56] guidelines for sarcopenia, the lack of consensus in other parameters remains problematic for body composition research in oncology [7].
Conclusion
To our knowledge, this is the first systematic review with a meta-analysis pertaining to the importance of muscle mass parameters in women with breast cancer. Our findings suggest that in clinical practice body composition assessment could prove valuable as a prognostic parameter in breast cancer. Future research is needed to explore additional body composition measures, such as visceral adipose tissue area, subcutaneous adipose tissue area, and sarcopenic obesity to deepen our understanding of the extent to which body composition affects outcomes in women with breast cancer. Further research is also needed to understand the mechanisms by which body composition affects cancer outcomes.
Abbreviations
- SMI:
-
Skeletal muscle index
- SMD:
-
Skeletal muscle density
- SMG:
-
Skeletal muscle gauge
- VAT:
-
Visceral adipose tissue
- SAT:
-
Subcutaneous adipose tissue
- CT:
-
Computer tomography
- MRI:
-
Magnetic resonance imaging
- DEXA:
-
Dual X-ray absorptiometry
- BIA:
-
Bioelectrical impedance analysis
References
WHO cancer data (2018). https://www.who.int/news-room/fact-sheets/detail/cancer
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Torre LA, Siegel RL, Ward EM et al (2016) Global cancer incidence and mortality rates and trends–an update. Cancer Epidemiol Biomark Prev 25:16–27
Veronesi U, Marubini E, Del Vecchio M et al (1995) Local recurrences and distant metastases after conservative breast cancer treatments: partly independent events. J Natl Cancer Inst 87:19–27
Gobbini E, Ezzalfani M, Dieras V et al (2018) Time trends of overall survival among metastatic breast cancer patients in the real-life ESME cohort. Eur J Cancer 96:17–24
Rosenberg IH (1997) Sarcopenia: origins and clinical relevance. J Nutr 127:990S–991S
Williams GR, Rier HN, McDonald A et al (2019) Sarcopenia & aging in cancer. J Geriatr Oncol 10:374–377
Mintziras I, Miligkos M, Wachter S et al (2018) Sarcopenia and sarcopenic obesity are significantly associated with poorer overall survival in patients with pancreatic cancer: systematic review and meta-analysis. Int J Surg 59:19–26
Boshier PR, Heneghan R, Markar SR et al (2018) Assessment of body composition and sarcopenia in patients with esophageal cancer: a systematic review and meta-analysis. Dis Esophagus 1:1. https://doi.org/10.1093/dote/doy047
Kamarajah SK, Bundred J, Tan BHL (2019) Body composition assessment and sarcopenia in patients with gastric cancer: a systematic review and meta-analysis. Gastric Cancer 22:10–22
Sun G, Li Y, Peng Y et al (2018) Can sarcopenia be a predictor of prognosis for patients with non-metastatic colorectal cancer? A systematic review and meta-analysis. Int J Colorectal Dis 33:1419–1427
Deng HY, Hou L, Zha P et al (2019) Sarcopenia is an independent unfavorable prognostic factor of non-small cell lung cancer after surgical resection: a comprehensive systematic review and meta-analysis. Eur J Surg Oncol 45:728–735
Williams GR, Deal AM, Muss HB et al (2017) Skeletal muscle measures and physical function in older adults with cancer: sarcopenia or myopenia? Oncotarget 8:33658–33665
Kroenke CH, Prado CM, Meyerhardt JA et al (2018) Muscle radiodensity and mortality in patients with colorectal cancer. Cancer 124:3008–3015
Martin L, Birdsell L, Macdonald N et al (2013) Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol 31:1539–1547
Akahori T, Sho M, Kinoshita S et al (2015) prognostic significance of muscle attenuation in pancreatic cancer patients treated with neoadjuvant chemoradiotherapy. World J Surg 39:2975–2982
Aust S, Knogler T, Pils D et al (2015) Skeletal muscle depletion and markers for cancer Cachexia are strong prognostic factors in epithelial ovarian cancer. PLoS ONE 10:e0140403
Centre for Reviews and Dissemination (CRD): PROSPERO: internation prospective register for systematic reviews, University of York, UK, 2019
Schardt C, Adams MB, Owens T et al (2007) Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inform Decis Mak 7:16
Higgins JP, Thompson SG, Deeks JJ et al (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Polus S, Pieper D, Burns J et al (2017) Heterogeneity in application, design, and analysis characteristics was found for controlled before-after and interrupted time series studies included in Cochrane reviews. J Clin Epidemiol 91:56–69
Higgins JP, Altman DG, Gotzsche PC et al (2011) The Cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Margulis AV, Pladevall M, Riera-Guardia N et al (2014) Quality assessment of observational studies in a drug-safety systematic review, comparison of two tools: the Newcastle-Ottawa Scale and the RTI item bank. Clin Epidemiol 6:359–368
von Elm E, Altman DG, Egger M et al (2014) The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg 12:1495–1499
Moher D, Liberati A, Tetzlaff J et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62:1006–1012
Blauwhoff-Buskermolen S, Langius JAE, Becker A et al (2017) The influence of different muscle mass measurements on the diagnosis of cancer cachexia. J Cachexia Sarcopenia Muscle 8:615–622
Del Fabbro E, Parsons H, Warneke CL et al (2012) The relationship between body composition and response to neoadjuvant chemotherapy in women with operable breast cancer. Oncologist 17:1240–1245
Mazzuca F, Onesti CE, Roberto M et al (2018) Lean body mass wasting and toxicity in early breast cancer patients receiving anthracyclines. Oncotarget 9:25714–25722
Shachar SS, Deal AM, Weinberg M et al (2017) Skeletal muscle measures as predictors of toxicity, hospitalization, and survival in patients with metastatic breast cancer receiving taxane-based chemotherapy. Clin Cancer Res 23:658–665
Sheean P, Gomez-Perez S, Joyce C et al (2019) Body composition, serum biomarkers of inflammation and quality of life in clinically stable women with estrogen receptor positive metastatic breast cancer. Nutr Cancer 71:981–991
Song EJ, Lee CW, Jung SY et al (2018) Prognostic impact of skeletal muscle volume derived from cross-sectional computed tomography images in breast cancer. Breast Cancer Res Treat 172:425–436
Versteeg KS, Blauwhoff-Buskermolen S, Buffart LM et al (2018) Higher muscle strength is associated with prolonged survival in older patients with advanced cancer. Oncologist 23:580–585
Weinberg MS, Shachar SS, Muss HB et al (2018) Beyond sarcopenia: characterization and integration of skeletal muscle quantity and radiodensity in a curable breast cancer population. Breast J 24:278–284
Rossi F, Valdora F, Barabino E et al (2019) Muscle mass estimation on breast magnetic resonance imaging in breast cancer patients: comparison between psoas muscle area on computer tomography and pectoralis muscle area on MRI. Eur Radiol 29:494–500
Caan BJ, Cespedes Feliciano EM, Prado CM et al (2018) Association of muscle and adiposity measured by computed tomography with survival in patients with nonmetastatic breast cancer. JAMA Oncol 4:798–804
Deluche E, Leobon S, Desport JC et al (2018) Impact of body composition on outcome in patients with early breast cancer. Support Care Cancer 26:861–868
Rier HN, Jager A, Sleijfer S et al (2017) Low muscle attenuation is a prognostic factor for survival in metastatic breast cancer patients treated with first line palliative chemotherapy. Breast 31:9–15
Villasenor A, Ballard-Barbash R, Baumgartner K et al (2012) Prevalence and prognostic effect of sarcopenia in breast cancer survivors: the HEAL Study. J Cancer Surviv 6:398–406
Prado CM, Baracos VE, McCargar LJ et al (2009) Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res 15:2920–2926
Lee HJ, Kim HS, Kim WY et al (2019) Sarcopena as a predicting factors for chemotherapy in advanced breast cancer patients. Eur J Surg Oncol 45:e31
Rossi FVF, Bignotti B, Torri L, Succio G, Tagniafico AS (2019) Evaluation of body Computer Tomography-determined sarcopenia in breast cancer patients and clinical outcomes: a systematic review. Cancer Treatment Res Commun 21:100154
Dabiri S, Popuri K, Cespedes Feliciano EM et al (2019) Muscle segmentation in axial computed tomography (CT) images at the lumbar (L3) and thoracic (T4) levels for body composition analysis. Comput Med Imaging Graph 75:47–55
Popuri K, Cobzas D, Esfandiari N et al (2016) Body composition assessment in axial CT images using FEM-based automatic segmentation of skeletal muscle. IEEE Trans Med Imaging 35:512–520
Shachar SS, Deal AM, Weinberg M et al (2017) Body composition as a predictor of toxicity in patients receiving anthracycline and taxane-based chemotherapy for early-stage breast cancer. Clin Cancer Res 23:3537–3543
Gonzalez MC, Heymsfield SB (2017) Bioelectrical impedance analysis for diagnosing sarcopenia and cachexia: what are we really estimating? J Cachexia Sarcopenia Muscle 8:187–189
Goodpaster BH, Thaete FL, Simoneau JA et al (1997) Subcutaneous abdominal fat and thigh muscle composition predict insulin sensitivity independently of visceral fat. Diabetes 46:1579–1585
Miljkovic I, Cauley JA, Wang PY et al (2013) Abdominal myosteatosis is independently associated with hyperinsulinemia and insulin resistance among older men without diabetes. Obesity 21:2118–2125
Feliciano E.C. LV, Chen W, Prado C., Shachar SS, Alexeeff S, Caan BJ: adiposity, muscle mass and delays and dose reduction on adjuvant taxane based chemotherapy for breast cancer. AACR2019 abstract:3284, 2019
Banerji MA, Faridi N, Atluri R et al (1999) Body composition, visceral fat, leptin, and insulin resistance in Asian Indian men. J Clin Endocrinol Metab 84:137–144
Goulart A, Malheiro N, Rios H et al (2019) Influence of visceral fat in the outcomes of colorectal cancer. Dig Surg 36:33–40
Guiu B, Petit JM, Bonnetain F et al (2010) Visceral fat area is an independent predictive biomarker of outcome after first-line bevacizumab-based treatment in metastatic colorectal cancer. Gut 59:341–347
Ebadi M, Martin L, Ghosh S et al (2017) Subcutaneous adiposity is an independent predictor of mortality in cancer patients. Br J Cancer 117:148–155
Bradshaw PT, Cespedes Feliciano EM, Prado CM et al (2019) Adipose tissue distribution and survival among women with nonmetastatic breast cancer. Obesity 27:997–1004
Hopanci Bicakli D, Cehreli R, Ozveren A et al (2019) Evaluation of sarcopenia, sarcopenic obesity, and phase angle in geriatric gastrointestinal cancer patients: before and after chemotherapy. Turk J Med Sci 49:583–588
Cruz-Jentoft AJ, Baeyens JP, Bauer JM et al (2010) Sarcopenia: european consensus on definition and diagnosis: report of the european working group on sarcopenia in older people. Age Ageing 39:412–423
Chen LK, Liu LK, Woo J et al (2014) Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc 15:95–101
Acknowledgements
We thank for the help and guidance on our search from the University of North Carolina Health Sciences Library.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
This study did not entail direct contact with humans and therefore did not entail obtaining informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aleixo, G.F.P., Williams, G.R., Nyrop, K.A. et al. Muscle composition and outcomes in patients with breast cancer: meta-analysis and systematic review. Breast Cancer Res Treat 177, 569–579 (2019). https://doi.org/10.1007/s10549-019-05352-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05352-3