Abstract
Purpose
This study examined clinical breast exam (CBE) and mammography surveillance in long-term young breast cancer survivors (YBCS) and identified barriers and facilitators to cancer surveillance practices.
Methods
Data collected with a self-administered survey from a statewide, randomly selected sample of YBCS diagnosed with invasive breast cancer or ductal carcinoma in situ younger than 45 years old, stratified by race (Black vs. White/Other). Multivariate logistic regression models identified predictors of annual CBEs and mammograms.
Results
Among 859 YBCS (n = 340 Black; n = 519 White/Other; mean age = 51.0 ± 5.9; diagnosed 11.0 ± 4.0 years ago), the majority (> 85%) reported an annual CBE and a mammogram. Black YBCS in the study were more likely to report lower rates of annual mammography and more barriers accessing care compared to White/Other YBCS. Having a routine source of care, confidence to use healthcare services, perceived expectations from family members and healthcare providers to engage in cancer surveillance, and motivation to comply with these expectations were significant predictors of having annual CBEs and annual mammograms. Cost-related lack of access to care was a significant barrier to annual mammograms.
Conclusions
Routine source of post-treatment care facilitated breast cancer surveillance above national average rates. Persistent disparities regarding access to mammography surveillance were identified for Black YBCS, primarily due to lack of access to routine source of care and high out-of-pocket costs.
Implications
Public health action targeting cancer surveillance in YBCS should ensure routine source of post-treatment care and address cost-related barriers. Clinical Trials Registration Number: NCT01612338.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The number of cancer survivors in the U.S. will reach 18 million by 2022 [1]. Breast cancer survivors constitute about 22% of this population due to the high incidence of the disease, with more than 2.7 million currently living in the US [1]. Approximately 25% are diagnosed when they are under 50 years old; these women constitute a growing clinical population of young breast cancer survivors (YBCS) [2]. Early onset breast cancer presents several challenges, including tumors that are more aggressive, higher recurrence rates, twofold higher risk for new primary tumors, and delays in diagnosis primarily due to lack of screening guidelines for younger women, resulting in increased mortality [2,3,4,5]. Black YBCS are a particularly vulnerable clinical population due to higher incidence of the disease; 33% are diagnosed younger than 45 years old compared to 21% of White patients [6, 7]. Black YBCS experience a higher incidence of triple-negative breast cancer, a subtype with poorer survival [8], and are twice as likely to die from the disease compared to White YBCS, contributing to racial disparities in breast cancer mortality [3]. After controlling for biology, there are significant racial differences in barriers accessing care, which affect survival for Black and White YBCS [9].
Breast Cancer Survivorship Care Guidelines developed by the American Cancer Society (ACS) and the American Society of Clinical Oncology (ASCO) provide recommendations for post-treatment care aiming to improve the overall health and quality of life of affected women [10]. Surveillance for breast cancer recurrence includes a detailed cancer-related history, an annual physical examination after the first 5 years of primary therapy, an annual clinical breast exam (CBE), and an annual mammogram to screen for local recurrence and/or a new primary cancer. Annual screening with Magnetic Resonance Imaging (MRI) as an adjunct to mammography increases significantly breast cancer survival in women who carry a pathogenic variant in Breast Cancer 1 (BRCA1) or Breast Cancer 2 (BRCA2) genes [11], women with a genetic syndrome associated with breast cancer, i.e., Li-Fraumeni or Cowden syndrome, and their untested first-degree relatives, and women with a personal history of breast cancer [12]. Adherence to mammography surveillance in breast cancer survivors older than 65 years of age was reportedly between 78 and 85% [13,14,15], but was as low as 44% among YBCS [16,17,18,19]. Black and Hispanic survivors reported lower rates of clinic visits and mammography surveillance [18,19,20].
Monitoring breast cancer surveillance of YBCS helps planning adequate quantity, quality, and coordination of cancer care, improves YBCS’ survival and overall quality of life, and meets recommendations of the National Academy of Science, Engineering, and Medicine regarding delivering high-quality cancer care [21, 22]. The purpose of the study is to describe breast cancer surveillance of long-term YBCS and examine demographic, clinical, and psychosocial predictors, and perceived barriers and facilitators to annual CBE and annual mammograms.
Theoretical framework
The Theory of Planned Behavior (TPB) has been used extensively to predict and explain health behaviors such as mammography screening [23] and physical activity in breast cancer survivors [24, 25]. According to the TPB, knowledge and attitudes toward a behavior, subjective norms, perceived control, and intention predict actual health behavior [26]. The TPB was expanded to include family support as a form of social support, which facilitates decision-making for genetic testing and mammography screening [27, 28]. Figure 1 presents the expanded TPB.
Methods
Design, sample, and procedures
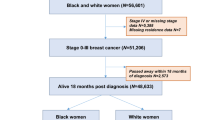
The study presents baseline data from a prospective randomized trial designed to increase breast cancer surveillance and use of cancer genetic services among YBCS and their unaffected relatives. In this paper, we present YBCS’s breast cancer surveillance practices; use of cancer genetic services among Black YBCS has been reported elsewhere [29]. Methodological details of the study and the recruitment process have been described in detail [28, 30]. A random sample of YBCS was selected from the Michigan Cancer Surveillance Program and was recruited with three mailed attempts. YBCS were eligible to participate if they were diagnosed with unilateral or bilateral invasive breast cancer or ductal carcinoma in situ (DCIS); Michigan residents at the time of diagnosis; between 20 and 45 years old at the time of diagnosis; between 25 and 64 years old at the time of study; not pregnant, incarcerated, or institutionalized; and able to read English and provide informed consent [28]. The study oversampled for Black versus White/Other YBCS (stratification for race) to increase participation of minority YBCS. Approximately 7% of YBCS of other ethnic/racial background identified in the cancer registry (e.g., Hispanic, American Indian, Arab American, Asian) were grouped with White/Other YBCS, because due to their small numbers they could not form a separate stratum. Eligible YBCS who returned a signed consent form and a completed baseline survey were included in the study. The Institutional Review Boards of the University of Michigan, the Michigan Department of Health and Human Services, and the Scientific Advisory Board of the Michigan Cancer Surveillance Program approved the study.
Measures
Table 1 provides a detailed description of the instruments used in the self-administered baseline survey. The instruments measured constructs of the expanded TPB. All measures were valid and reliable in this sample (Cronbach’s alpha > 0.71). The survey also collected self-reported demographic (age, education, marital status, employment, income, insurance, access to healthcare services) and clinical characteristics. YBCS’ race was recorded from the cancer registry, since this was the main criterion for sample stratification. The outcome variables were self-reported frequency of CBE and mammography, which were categorized as adherent or non-adherent with ASCO (2013) surveillance recommendations [31]. Although ASCO Breast Cancer Survivorship Care Guidelines have been recently revised [10], we used the 2013 guidelines that included recommendation for an annual CBE and an annual mammogram, because these were relevant at the time YBCS completed the baseline survey. YBCS responded to the following questions: “A mammogram is an x-ray of each breast to look for breast cancer. Have you ever had a mammogram? If yes, how often did you have a mammogram over the past 12 months?” and “A CBE is when a doctor or nurse checks the breast(s) for a lump. Have you ever had a CBE? If yes, how often did you have a CBE over the past 12 months?” YBCS’ responses were further dichotomized into having “none” or “at least one” CBE and mammogram within the past 12 months (Yes or No).
Statistical analyses
Descriptive statistics (frequencies, percentages, means, and standard deviations) describe the sample. Pearson’s r correlations in bivariate analyses examined relationships between demographic and clinical characteristics, psychosocial variables, and having at least one annual CBE and mammogram. Two different logistic regression models identified factors associated with having at least one annual CBE and mammogram. The model assessing mammography surveillance did not include YBCS with double mastectomy due to absence of mammary tissue. Two tailed p values were calculated and the level of significance was set at p < 0.05. A power analysis conducted with PASSv.14 software determined that a sample of n = 205 would achieve 80% power to detect significant predictors (Effect size = 0.5) of adherence to annual CBE and mammography in the logistic regression analyses. SPSS, Version 20.0, Chicago, SPSS Inc. was used for data analyses.
Results
Sample
Among 3000 YBCS initially identified from the Michigan Cancer Surveillance Program, the response rate was 27.5% for Black YBCS and 38.6% for White/Other YBCS. Details about the recruitment process have been reported [30]. Table 2 summarizes the demographic and clinical characteristics of the YBCS in the sample. YBCS have been diagnosed on average 11.0 ± 4.0 years prior to the study. Their mean age at the time of the study was 51.0 ± 5.9 years; range 30–63 years; mean age at first breast cancer diagnosis was 40.0 ± 4.7 years; and the mean age of their first mammogram was 36.0 ± 6.0 years. White/Other YBCS were significantly more likely to report family history of breast and ovarian cancer and a double mastectomy compared to Black YBCS. Finally, 58 YBCS reported a BRCA1 (n = 19) or BRCA2 (n = 14) or other mutation (n = 7); having a family member with a known mutation (n = 6); or having a known hereditary cancer syndrome (e.g., Li Fraumeni) (n = 12).
Despite overall high rates of health insurance, Black YBCS had significantly more barriers accessing healthcare services compared to White/Other YBCS. Survey items assessing access to healthcare showed that, compared to White/Other YBCS, Black YBCS were significantly more likely to report not having health insurance; not having a routine source of care; and not getting medical care in the past 12 months due to high out-of-pocket costs.
Frequency of CBE and mammography
Most YBCS in the study reported having CBEs (85.9%; n = 723) and mammograms annually (89.1%; n = 541, excluding YBCS with double mastectomy). There was no difference between Black and White/Other YBCS for self-reported CBE (85.7%; n = 281 vs. 86.0%; n = 442). However, Black YBCS were significantly less likely to report having an annual mammogram compared to White/Other YBCS (85.7%; n = 251 vs. 91.8%; n = 356; X2 = 6.386, p = 0.012).
Frequency of annual MRI
Among the 58 YBCS who reported having a mutation, a known hereditary cancer syndrome, or being a member of a mutation-harboring family, n = 27 did not have a double mastectomy. Among these YBCS n = 8 reported having an annual MRI, while n = 19 reported that it has been over a year since their last MRI. Among YBCS who were not mutation carriers and did not have a double mastectomy, n = 72 reported having an MRI the past 12 months and n = 186 reported that it has been more than 12 months since their last MRI. Due to the small sample, further analyses were not pursued.
Factors associated with annual CBE
Table 3 shows the bivariate analyses and the multivariate logistic regression model of factors associated with YBCS having at least one annual CBE. Positive predictors of having an annual CBE in the bivariate analyses were education, income, insurance, having a routine source of care, knowledge of breast cancer risk factors and breast cancer genetics, perceived family and healthcare provider expectations to engage in breast cancer surveillance, motivation to comply with these expectations, self-efficacy managing breast cancer, confidence using healthcare services for CBE, and intention to have a CBE. Cost-related lack of access to care was negatively associated with frequency of CBE. In the multivariate logistic regression model, the most significant predictor of having an annual CBE was having a routine source of care followed by confidence in using healthcare services for CBE, perceived family and perceived healthcare provider expectations for breast cancer surveillance, and intention to have a CBE. We examined whether there was a difference in reported annual CBE between women with invasive breast cancer and those with a DCIS diagnosis. For these analyses, we excluded n = 101 women who reported having both diagnoses (i.e., had breast cancer more than once). There was a significant difference in annual CBE between YBCS with invasive breast cancer and those with DCIS, with the latter group less likely to report an annual CBE (X2 = 4.19, p = 0.041).
Factors associated with annual mammography
Table 4 shows the bivariate analyses and the multivariate logistic regression model of factors associated with YBCS having at least one annual mammogram. Positive predictors of having an annual mammogram in the bivariate analyses were age, education, income, insurance, having a routine source of care, knowledge of breast cancer risk factors and breast cancer genetics, perceived provider expectations to engage in breast cancer surveillance, motivation to comply with family member and provider expectations, confidence using healthcare services for mammograms, family support in illness, and intention to have a mammogram. Black race recorded from the registry, cost-related lack of access to care, and reporting more mammography barriers were negatively associated with having an annual mammogram. In the multivariate logistic regression model, the most significant predictor of having an annual mammogram was having a routine source of care followed by motivation to comply with recommendations from healthcare providers, confidence using healthcare services for mammograms, and intention to have a mammogram. Lack of access to care due to high out-of-pocket costs was negatively associated with having an annual mammogram. We examined whether there was a difference in reported annual mammogram between women with invasive breast cancer and those with a DCIS diagnosis. For these analyses, we excluded n = 193 women who reported having both diagnoses and double mastectomy. There was no significant difference in annual mammogram between YBCS with invasive breast cancer and those with DCIS (X2 = 0.03, p = 0.875).
The Decisional Balance Scale for Mammography did not identify significant differences between Black and White/Other YBCS in overall access to mammograms. However, a factor analysis involving the 23 items of the scale revealed that Black YBCS were significantly less likely than White/Other YBCS to perceive benefits from having an annual mammogram (1.75 ± 0.94 vs. 2.07 ± 1.21, t test = 3821, p < 0.001), and significantly less likely to follow recommendations from healthcare providers (1.70 ± 0.87 vs. 2.05 ± 1.01, t test = 4855, p < 0.001).
Discussion
The study reported engagement in CBE and mammography in a randomly selected sample of long-term YBCS with adequate representation of Black YBCS. Black and White/other YBCS in our study reported having had a CBE and a mammogram within the past 12 months above the national average for women 40 years and older (66.8%) [32]. Other studies have also reported high prevalence rates of surveillance mammograms in breast cancer survivors of different racial and ethnic backgrounds [13, 18, 33, 34]. However, this is among the first studies making a comprehensive comparison of CBE and mammography surveillances practices among long-term Black and White/Other YBCS.
The most significant barrier for post-treatment surveillance mammograms for YBCS was cost-related lack of access to care. A cancer diagnosis may cause significant financial burdens and high out-of-pocket costs may further deter YBCS from accessing surveillance care. Compared to adults without barriers accessing healthcare, those who lack health insurance, have coverage gaps, or delay care because of limited personal finances might face increased risks for poor physical and mental health, and overall premature mortality [35]. After the installment of the 2010 Patient Protection and Affordable Care Act, adults living below the poverty level in states that expanded Medicaid, such as the state of Michigan, were more likely to have a routine source of care and to not have unmet healthcare needs due to high out-of-pocket costs [36]. Extended healthcare coverage may have contributed to high rates of mammography surveillance among Black and White/Other YBCS in our study, which were above the national average for both racial/ethnic groups. The Health Michigan plan that was put in action on April 2014 has also granted access to cancer surveillance to more than 650,000 previously uninsured individuals, the majority of which come from minority groups [37]. The proposed dismissal of ACA after the fiscal year 2020 will likely make it more difficult for Black and White/Other YBCS living below or at poverty line from crucial access to cancer surveillance services.
The most significant facilitator of annual CBEs and annual mammograms was having a routine source of care. Mammography surveillance in breast cancer survivors normally requires a healthcare provider recommendation [13]. Breast cancer survivors are more likely to have mammograms when they receive care from a specialist, such as an oncologist, surgeon, or gynecologist [14, 15, 17, 19, 33]. Those receiving care from an oncologist were more likely to perceive that their care was well coordinated and were more confident in decisions made by their oncologist compared to those receiving care from primary care providers [34]. We also identified that women diagnosed with DCIS were less likely to report an annual CBE compared to those diagnosed with invasive breast cancer; data did not support the equivalent association for annual mammography. Given that Black women with DCIS are more likely to experience a higher risk of second breast cancer [38], and despite the importance of mammography in active surveillance of DCIS patients [39], not all racial/ethnic minority DCIS patients receive adequate follow-up surveillance [40]. Taken together, these findings highlight that source of post-treatment care could have significant implications for the implementation of recommendations and improvement of the overall health of affected women.
Motivation to comply with recommendations from healthcare providers and confidence using healthcare services were significantly associated with annual CBE and mammograms. Open communication with providers following a breast cancer diagnosis was also positively associated with self-efficacy to engage in cancer surveillance [41]. Self-efficacy is a key resource for YBCS associated with better mental health [42] and higher quality of life [43]. YBCS with less self-efficacy to manage breast cancer and to navigate the healthcare system are a group at risk for adverse outcomes and warrants further assessment and early intervention.
Perceived expectations of family members were positively associated with having a CBE. This finding is consistent with studies identifying support from family and friends as a facilitator of post-treatment care, in addition to faith and church support for Black breast cancer survivors [44, 45]. The equivalent association between perceived expectations of family members and annual mammography was not identified in the data. One possible explanation is that there are more barriers associated with having a mammogram compared to a CBE, which can be completed during a routine office visit. While family support reinforces seeking a provider visit for a CBE, it may not be able to address mammography-related barriers, such as cost and lack of provider recommendation. This finding also suggests that the expanded TPB identified important family-related factors associated with engaging in breast cancer surveillance. It has significant theoretical and clinical implications because it emphasizes the importance of engaging family members in post-treatment care of YBCS.
More than 50% of Black YBCS in our study reported having an income less than $40,000. Black YBCS were also significantly more likely to report barriers accessing healthcare services due to lack of health insurance, lack of a routine source of care, and high out-of-pocket costs, which likely explains why rates of annual mammography among Black YBCS were significantly lower compared to White/Other YBCS in the study. These findings are consistent with studies reporting that among 4535 breast cancer survivors, Blacks and Hispanics were more likely to be non-adherent to mammography surveillance guidelines and to report cost-related barriers accessing care compared to White breast cancer survivors [20, 46]. In a primarily White sample of 3965 breast cancer survivors identified from a healthcare plan registry in the western U.S. region, 79% received surveillance mammograms the first year after their diagnoses, which decreased to 63% 10 years after diagnosis, especially for women diagnosed younger than 40 years old [19]. Given that our sample was on average 11 years post-diagnosis, long-term Black YBCS may be significantly less likely to benefit from breast cancer surveillance. Black YBCS in our study were also less likely to perceive benefits from having an annual mammogram, suggesting a significant need for cancer survivorship education in this population. One possible explanation for this finding may relate to lack of a routine source of care among Black YBCS.
Although the American Cancer Society [47], the American College of Radiology [48], and the Society of Breast Imaging [49] recommend breast MRI for screening women with a lifetime breast cancer risk greater than 20–25%, these recommendations state that MRI “may be considered” for women who have a personal history of breast cancer; that is, for women with intermediate breast cancer risk (lifetime breast cancer risk 15–20%). Therefore, little is known about the role of breast MRI in post-treatment surveillance programs, as this imaging method is expensive, lacks availability, requires contrast media injection for adequate imaging, and neither the technique nor interpretive criteria for breast MRI are standardized [50].
While this study examined adherence to CBE and mammography as post-treatment surveillance, the recent ACS/ASCO Breast Cancer Survivorship Care Guidelines provide holistic recommendations, including education and counseling about signs and symptoms of local or regional recurrence, education and counseling about the importance of adhering to adjuvant (antiestrogen) endocrine therapy, risk evaluation and genetic counseling for women with risk factors for hereditary cancer, screening for local recurrence or new primary cancers with mammography and MRI for those who meet criteria, assessment and management of physical and psychosocial long-term and late effects of treatment, general health promotion to address obesity, physical activity, nutrition, and smoking cessation, and care coordination including the implementation of a survivorship care plan [10]. The survivorship period presents an opportunity for healthcare providers to promote the health of YBCS and to reduce the burden and breast cancer mortality for this population. Each clinical encounter can identify “teachable moments” to further engage this population in the ACS/ASCO Breast Cancer Survivorship Care Guidelines. A teachable moment is a naturally occurring health event that can motivate individuals to adopt risk-reducing health behaviors [51]. It is vital that healthcare providers assess and reduce barriers to timely survivorship care by ensuring that YBCS have access to care plans with information about their cancer, cancer treatment, and follow-up care. This is especially important for Black YBCS who are more likely to have poor survival. Studies supported use of low-cost mobile mammography units, clinics with evening hours, use of churches to support preventive care, and innovative reminder systems for women and their supportive family members [41, 52,53,54,55]. The Commission on Cancer requires survivorship care plans as a minimum standard [56]. Healthcare providers, especially those who care for Black YBCS, should ensure that their survivorship care plan addresses all current surveillance recommendations, including genetic testing when appropriate and health promotion behaviors, focusing on education and psychosocial support, necessary to improve outcomes during cancer survivorship [10]. Future studies should make comprehensive assessments to ensure that surveillance plans are consistent with these guidelines, and develop culturally appropriate interventions that recognize the unique challenges and assets across diverse YBCS groups to improve survivorship care.
Strengths and limitations
Self-reported surveillance behaviors may be less reliable compared to data collected from medical records or insurance claims, but take into account treatment received from a variety of sources. While the sample size was large and Black YBCS were oversampled, response rates among Black and White/Other YBCS were low for both racial/ethnic groups although typical for large population-based studies. Sample stratification for race enhanced our ability to make comparisons between Black and White/Other YBCS in the study, but has limitations, namely the inability to derive nationally representative statistics from the combined samples of Black and White/Other YBCS. Despite these limitations, the study advanced the body of knowledge for breast cancer surveillance of long-term YBCS and further highlights the survivorship needs of Black YBCS, who are a vulnerable population. YBCS who accepted participation are self-selected, and are more likely to engage with the healthcare system. Most likely there is a larger disparity between Black and White/Others YBCS, since those who did not accept participation may also be less likely to engage with the healthcare system.
Conclusions
Most YBCS reported engaging in breast cancer surveillance consistent with national recommendations a decade post-diagnosis. However, the rate of annual mammography for Black YBCS was lower compared to the White/Other YBCS. The expanded TPB led to the identification of multiple important predictors of breast cancer surveillance for YBCS and emphasized the need to engage family members in their care. Future studies should address comprehensively the breast cancer survivorship care needs of YBCS, and especially of Black YBCS who are a vulnerable population experiencing higher disease incidence and mortality. The survivorship period is an ideal time to address these needs and promote YBCS’ health. Monitoring YBCS’ cancer surveillance provides information that can help plan adequate quantity, quality, and coordination of cancer care to meet recommendations of the National Academy of Science, Engineering, and Medicine for delivering high-quality post-treatment care.
References
de Moor JS, Mariotto AB, Parry C, Alfano CM, Padgett L, Kent EE, Forsythe L, Scoppa S, Hachey M, Rowland JH (2013) Cancer survivors in the United States: prevalence across the survivorship trajectory and implications for care. Cancer Epidemiol Biomark Prev 22(4):561–570. https://doi.org/10.1158/1055-9965.epi-12-1356
King M-C, Levy-Lahad E, Lahad A (2014) Population-based screening for BRCA1 and BRCA2: 2014 Lasker Award. JAMA 312(11):1091–1092
McCarthy AM, Yang J, Armstrong K (2015) Increasing disparities in breast cancer mortality from 1979 to 2010 for US black women aged 20 to 49 years. Am J Public Health 105(Suppl 3):S446–448. https://doi.org/10.2105/ajph.2014.302297
Pal T, Vadaparampil ST (2012) Genetic risk assessments in individuals at high risk for inherited breast cancer in the breast oncology care setting. Cancer Control 19(4):255–266
Rosenberg SM, Newman LA, Partridge AH (2015) Breast cancer in young women: rare disease or public health problem? JAMA Oncol 1(7):877–878
Daly B, Olopade OI (2015) A perfect storm: how tumor biology, genomics, and health care delivery patterns collide to create a racial survival disparity in breast cancer and proposed interventions for change. CA Cancer J Clin 65(3):221–238. https://doi.org/10.3322/caac.21271
Danforth DN Jr (2013) Disparities in breast cancer outcomes between Caucasian and African American women: a model for describing the relationship of biological and nonbiological factors. Breast Cancer Res 15(3):208. https://doi.org/10.1186/bcr3429
Kumar P, Aggarwal R (2016) An overview of triple-negative breast cancer. Arch Gynecol Obstet 293(2):247–269
Ademuyiwa FO, Gao F, Hao L, Morgensztern D, Aft RL, Ma CX, Ellis MJ (2015) US breast cancer mortality trends in young women according to race. Cancer 121(9):1469–1476. https://doi.org/10.1002/cncr.29178
Runowicz CD, Leach CR, Henry NL, Henry KS, Mackey HT, Cowens-Alvarado RL, Cannady RS, Pratt-Chapman ML, Edge SB, Jacobs LA, Hurria A, Marks LB, LaMonte SJ, Warner E, Lyman GH, Ganz PA (2016) American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. J Clin Oncol 34(6):611–635. https://doi.org/10.1200/jco.2015.64.3809
Evans DG, Kesavan N, Lim Y, Gadde S, Hurley E, Massat NJ, Maxwell AJ, Ingham S, Eeles R, Leach MO, Howell A, Duffy SW (2014) MRI breast screening in high-risk women: cancer detection and survival analysis. Breast Cancer Res Treat 145(3):663–672. https://doi.org/10.1007/s10549-014-2931-9
Tadros A, Arditi B, Weltz C, Port E, Margolies LR, Schmidt H (2017) Utility of surveillance MRI in women with a personal history of breast cancer. Clin Imaging 46:33–36. https://doi.org/10.1016/j.clinimag.2017.06.007
Buist DS, Bosco JL, Silliman RA, Gold HT, Field T, Yood MU, Quinn VP, Prout M, Lash TL (2013) Long-term surveillance mammography and mortality in older women with a history of early stage invasive breast cancer. Breast Cancer Res Treat 142(1):153–163. https://doi.org/10.1007/s10549-013-2720-x
Field TS, Doubeni C, Fox MP, Buist DS, Wei F, Geiger AM, Quinn VP, Lash TL, Prout MN, Yood MU, Frost FJ, Silliman RA (2008) Under utilization of surveillance mammography among older breast cancer survivors. J Gen Intern Med 23(2):158–163. https://doi.org/10.1007/s11606-007-0471-2
Keating NL, Landrum MB, Guadagnoli E, Winer EP, Ayanian JZ (2006) Factors related to underuse of surveillance mammography among breast cancer survivors. J Clin Oncol 24(1):85–94. https://doi.org/10.1200/jco.2005.02.4174
Katz ML, Donohue KA, Alfano CM, Day JM, Herndon JE 2nd, Paskett ED (2009) Cancer surveillance behaviors and psychosocial factors among long-term survivors of breast cancer. Cancer and Leukemia Group B 79804. Cancer 115(3):480–488. https://doi.org/10.1002/cncr.24063
Sabatino SA, Thompson TD, Richardson LC, Miller J (2012) Health insurance and other factors associated with mammography surveillance among breast cancer survivors: results from a national survey. Med Care 50(3):270–276. https://doi.org/10.1097/MLR.0b013e318244d294
Shelby RA, Scipio CD, Somers TJ, Soo MS, Weinfurt KP, Keefe FJ (2012) Prospective study of factors predicting adherence to surveillance mammography in women treated for breast cancer. J Clin Oncol 30(8):813–819. https://doi.org/10.1200/jco.2010.34.4333
Wirtz HS, Boudreau DM, Gralow JR, Barlow WE, Gray S, Bowles EJ, Buist DS (2014) Factors associated with long-term adherence to annual surveillance mammography among breast cancer survivors. Breast Cancer Res Treat 143(3):541–550. https://doi.org/10.1007/s10549-013-2816-3
Advani PS, Ying J, Theriault R, Melhem-Bertrand A, Moulder S, Bedrosian I, Tereffe W, Black S, Pini TM, Brewster AM (2014) Ethnic disparities in adherence to breast cancer survivorship surveillance care. Cancer 120(6):894–900. https://doi.org/10.1002/cncr.28490
Institute of Medicine (2013) Addressing the challenges of an aging, population. Delivering high-quality cancer care: charting a new course for a system in crisis. National Academies Press, Washington, DC. https://doi.org/10.17226/18359
Howell D, Hack TF, Oliver TK, Chulak T, Mayo S, Aubin M, Chasen M, Earle CC, Friedman AJ, Green E, Jones GW, Jones JM, Parkinson M, Payeur N, Sabiston CM, Sinclair S (2012) Models of care for post-treatment follow-up of adult cancer survivors: a systematic review and quality appraisal of the evidence. J Cancer Surviv 6(4):359–371. https://doi.org/10.1007/s11764-012-0232-z
Taymoori P, Moshki M, Roshani D (2014) Facilitator psychological constructs for mammography screening among Iranian women. Asian Pac J Cancer Prev 15(17):7309–7316
Forbes CC, Blanchard CM, Mummery WK, Courneya K (2015) Prevalence and correlates of strength exercise among breast, prostate, and colorectal cancer survivors. Oncol Nurs Forum 42(2):118–127. https://doi.org/10.1188/15.onf.42-02ap
Trinh L, Mutrie N, Campbell AM, Crawford JJ, Courneya KS (2014) Effects of supervised exercise on motivational outcomes in breast cancer survivors at 5-year follow-up. Eur J Oncol Nurs 18(6):557–563. https://doi.org/10.1016/j.ejon.2014.07.004
Ajzen I (1991) The theory of planned behavior. Organ Behav Hum Decis Process 50(2):179–211. https://doi.org/10.1016/0749-5978(91)90020-T
Katapodi MC, Facione NC, Miaskowski C, Dodd MJ, Waters C (2002) The influence of social support on breast cancer screening in a multicultural community sample. Oncol Nurs Forum 29(5):845–852. https://doi.org/10.1188/02.onf.845-852
Katapodi MC, Northouse LL, Schafenacker AM, Duquette D, Duffy SA, Ronis DL, Anderson B, Janz NK, McLosky J, Milliron KJ, Merajver SD, Duong LM, Copeland G (2013) Using a state cancer registry to recruit young breast cancer survivors and high-risk relatives: protocol of a randomized trial testing the efficacy of a targeted versus a tailored intervention to increase breast cancer screening. BMC Cancer. https://doi.org/10.1186/1471-2407-13-97
Jones T, Lockhart JS, Mendelsohn-Victor KE, Duquette D, Northouse LL, Duffy SA, Donley R, Merajver SD, Milliron KJ, Roberts JS, Katapodi MC (2016) Use of cancer genetics services in African-American young breast cancer survivors. Am J Prev Med 51(4):427–436. https://doi.org/10.1016/j.amepre.2016.03.016
Katapodi MC, Duquette D, Yang JJ, Mendelsohn-Victor KE, Anderson B, Nikolaidis C, Mancewicz E, Northouse LL, Duffy S, Ronis D, Milliron KJ, Probst-Hensch N, Merajver SD, Janz NK, Copeland G, Roberts SJ (2017) Recruiting families at risk for hereditary breast and ovarian cancer from a statewide cancer registry: a methodological study. Cancer Causes Control 28(3):191–201. https://doi.org/10.1007/s10552-017-0858-2
Khatcheressian JL, Hurley P, Bantug E, Esserman LJ, Grunfeld E, Halberg F, Hantel A, Henry NL, Muss HB, Smith TJ, Vogel VG, Wolff AC, Somerfield MR, Davidson NE (2013) Breast cancer follow-up and management after primary treatment: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol 31(7):961–965. https://doi.org/10.1200/jco.2012.45.9859
Center for Disease Control and Prevention (2015) Mammography and Breast Cancer. https://www.cdc.gov/nchs/fastats/mammography.htm. Accessed 11 Feb 2017
Doubeni CA, Field TS, Ulcickas Yood M, Rolnick SJ, Quessenberry CP, Fouayzi H, Gurwitz JH, Wei F (2006) Patterns and predictors of mammography utilization among breast cancer survivors. Cancer 106(11):2482–2488. https://doi.org/10.1002/cncr.21893
Risendal BC, Sedjo RL, Giuliano AR, Vadaparampil S, Jacobsen PB, Kilbourn K, Baron A, Byers T (2016) Surveillance and beliefs about follow-up care among long-term breast cancer survivors: a comparison of primary care and oncology providers. J Cancer Surviv 10(1):96–102. https://doi.org/10.1007/s11764-015-0454-y
Mujahid MS, Janz NK, Hawley ST, Griggs JJ, Hamilton AS, Graff J, Katz SJ (2011) Racial/ethnic differences in job loss for women with breast cancer. J Cancer Surviv 5(1):102–111. https://doi.org/10.1007/s11764-010-0152-8
Okoro CA, Zhao G, Fox JB, Eke PI, Greenlund KJ, Town M (2014) Surveillance for health care access and health services use, adults aged 18-64 years: Behavioral Risk Factor Surveillance System, United States. MMWR Surveill Summ 66(7):1–42. https://doi.org/10.15585/mmwr.ss6607a1
Ayanian JZ, Ehrlich GM, Grimes DR, Levy H (2017) Economic effects of medicaid expansion in Michigan. N Engl J Med 376(5):407–410
Stark A, Stapp R, Raghunathan A, Yan X, Kirchner HL, Griggs J, Newman L, Chitale D, Dick A (2012) Disease-free probability after the first primary ductal carcinoma in situ of the breast: a comparison between African-American and White-American women. Breast Cancer Res Treat 131(2):561–570. https://doi.org/10.1007/s10549-011-1742-5
Grimm LJ, Ghate SV, Hwang ES, Soo MS (2017) Imaging features of patients undergoing active surveillance for ductal carcinoma in situ. Acad Radiol 24(11):1364–1371. https://doi.org/10.1016/j.acra.2017.05.017
López ME, Kaplan CP, Nápoles AM, Livaudais JC, Hwang ES, Stewart SL, Bloom J, Karliner L (2013) Ductal carcinoma in situ (DCIS): posttreatment follow-up care among Latina and non-Latina White women. J Cancer Surviv 7(2):219–226. https://doi.org/10.1007/s11764-012-0262-6
Gathirua-Mwangi WG, Monahan PO, Stump T, Rawl SM, Skinner CS, Champion VL (2016) Mammography adherence in African-American women: results of a randomized controlled trial. Ann Behav Med 50(1):70–78. https://doi.org/10.1007/s12160-015-9733-0
Ellis KR, Janevic MR, Kershaw T, Caldwell CH, Janz NK, Northouse L (2017) The influence of dyadic symptom distress on threat appraisals and self-efficacy in advanced cancer and caregiving. Support Care Cancer 25(1):185–194. https://doi.org/10.1007/s00520-016-3385-x
Northouse LL, Mood D, Kershaw T, Schafenacker A, Mellon S, Walker J, Galvin E, Decker V (2002) Quality of life of women with recurrent breast cancer and their family members. J Clin Oncol 20(19):4050–4064. https://doi.org/10.1200/jco.2002.02.054
Thompson HS, Littles M, Jacob S, Coker C (2006) Posttreatment breast cancer surveillance and follow-up care experiences of breast cancer survivors of African descent: an exploratory qualitative study. Cancer Nurs 29(6):478–487
Haynes-Maslow L, Allicock M, Johnson LS (2016) Cancer support needs for African American breast cancer survivors and caregivers. J Cancer Educ 31(1):166–171. https://doi.org/10.1007/s13187-015-0832-1
Palmer NR, Weaver KE, Hauser SP, Lawrence JA, Talton J, Case LD, Geiger AM (2015) Disparities in barriers to follow-up care between African American and White breast cancer survivors. Support Care Cancer 23(11):3201–3209. https://doi.org/10.1007/s00520-015-2706-9
Saslow D, Boetes C, Burke W, Harms S, Leach MO, Lehman CD, Morris E, Pisano E, Schnall M, Sener S, Smith RA, Warner E, Yaffe M, Andrews KS, Russell CA (2007) American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin 57(2):75–89
Mainiero MB, Lourenco A, Mahoney MC, Newell MS, Bailey L, Barke LD, D’Orsi C, Harvey JA, Hayes MK, Huynh PT, Jokich PM, Lee SJ, Lehman CD, Mankoff DA, Nepute JA, Patel SB, Reynolds HE, Sutherland ML, Haffty BG (2016) ACR appropriateness criteria breast cancer screening. J Am Coll Radiol 13(11s):R45–r49. https://doi.org/10.1016/j.jacr.2016.09.021
Lee CH, Dershaw DD, Kopans D, Evans P, Monsees B, Monticciolo D, Brenner RJ, Bassett L, Berg W, Feig S, Hendrick E, Mendelson E, D’Orsi C, Sickles E, Burhenne LW (2010) Breast cancer screening with imaging: recommendations from the Society of Breast Imaging and the ACR on the use of mammography, breast MRI, breast ultrasound, and other technologies for the detection of clinically occult breast cancer. J Am Coll Radiol 7(1):18–27. https://doi.org/10.1016/j.jacr.2009.09.022
Morris EA, Liberman L, Ballon DJ, Robson M, Abramson AF, Heerdt A, Dershaw DD (2003) MRI of occult breast carcinoma in a high-risk population. AJR Am J Roentgenol 181(3):619–626. https://doi.org/10.2214/ajr.181.3.1810619
McBride CM, Emmons KM, Lipkus IM (2003) Understanding the potential of teachable moments: the case of smoking cessation. Health Educ Res 18(2):156–170
Browder C, Eberth JM, Schooley B, Porter NR (2015) Mobile mammography: an evaluation of organizational, process, and information systems challenges. Healthcare (Amst) 3(1):49–55. https://doi.org/10.1016/j.hjdsi.2014.12.001
Harrison RV, Janz NK, Wolfe RA, Tedeschi PJ, Chernew M, Stross JK, Huang X, McMahon LF Jr (2003) Personalized targeted mailing increases mammography among long-term noncompliant medicare beneficiaries: a randomized trial. Med Care 41(3):375–385. https://doi.org/10.1097/01.mlr.0000053020.30060.f2
Holt CL, Lee C, Wright K (2008) A spiritually based approach to breast cancer awareness: cognitive response analysis of communication effectiveness. Health Commun 23(1):13–22. https://doi.org/10.1080/10410230701626919
Russell KM, Champion VL, Monahan PO, Millon-Underwood S, Zhao Q, Spacey N, Rush NL, Paskett ED (2010) Randomized trial of a lay health advisor and computer intervention to increase mammography screening in African American women. Cancer Epidemiol Biomark Prev 19(1):201–210. https://doi.org/10.1158/1055-9965.epi-09-0569
Greenlee H, Molmenti CL, Crew KD, Awad D, Kalinsky K, Brafman L, Fuentes D, Shi Z, Tsai WY, Neugut AI, Hershman DL (2016) Survivorship care plans and adherence to lifestyle recommendations among breast cancer survivors. J Cancer Surviv 10(6):956–963. https://doi.org/10.1007/s11764-016-0541-8
Katapodi MC, Aouizerat BE (2005) Do women in the community recognize hereditary and sporadic breast cancer risk factors? Oncol Nurs Forum 32(3):617–623
Wang C, Gonzalez R, Milliron KJ, Strecher VJ, Merajver SD (2005) Genetic counseling for BRCA1/2: a randomized controlled trial of two strategies to facilitate the education and counseling process. Am J Med Genet A 134A(1):66–73. https://doi.org/10.1002/ajmg.a.30577
Katapodi MC, Dodd MJ, Lee KA, Facione NC (2009) Underestimation of breast cancer risk: influence on screening behavior. Oncol Nurs Forum. https://doi.org/10.1188/09.ONF.306-314
Katapodi MC, Northouse LL, Milliron KJ, Liu G, Merajver SD (2013) Individual and family characteristics associated with BRCA1/2 genetic testing in high-risk families. Psychooncology 22(6):1336–1343. https://doi.org/10.1002/pon.3139
Johnson Vickberg SM (2001) Fears about breast cancer recurrence. Cancer Pract 9(5):237–243. https://doi.org/10.1046/j.1523-5394.2001.009005237.x
Vickberg SMJ (2003) The Concerns About Recurrence Scale (CARS): a systematic measure of women’s fears about the possibility of breast cancer recurrence. Ann Behav Med 25(1):16–24
Rakowski W, Andersen MR, Stoddard AM, Urban N, Rimer BK, Lane DS, Fox SA, Costanza ME (1997) Confirmatory analysis of opinions regarding the pros and cons of mammography. Health Psychol 16(5):433–441
Bandura A (1989) Human agency in social cognitive theory. Am Psychol 44(9):1175–1184
Fletcher KA, Lewis FM, Haberman MR (2010) Cancer-related concerns of spouses of women with breast cancer. Psychooncology 19(10):1094–1101. https://doi.org/10.1002/pon.1665
Ajzen I, Fishbein M (1980) Understanding attitudes and predicting social behaviour. Prentice Hall, Englewood Cliffs
Acknowledgements
Joan Such Lockhart, PhD, RN, CORLN, AOCN, CNE, ANEF, FAAN, and Sr. Rosemary Donley, PhD, APRN from Duquesne University School of Nursing, 600 Forbes Avenue, Pittsburgh, PA 15282, USA for providing support and guidance to Dr. Tarsha Jones while she completed her doctoral studies at Duquesne University School of Nursing. Jenna McLosky, CGC, Cancer Genomics Program—Michigan Department of Health and Human Services for patient and relative identification, recruitment, and assessment of eligibility.
Funding
Centers for Disease Control and Prevention, 5U48DP001901-03, PI: M.C. Katapodi. Robert Wood Johnson Foundation, Nurse Faculty Scholars Award 68039, PI: M.C. Katapodi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Tarsha Jones, Debra Duquette, Meghan Underhill, Kari E. Mendelsohn-Victor, Beth Anderson, Chang Ming, Kara J. Milliron, Glenn Copeland, Nancy K. Janz, Laurel L. Northouse, Sonia M. Duffy, Sofia D. Merajver, and Maria C. Katapodi declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Jones, T., Duquette, D., Underhill, M. et al. Surveillance for cancer recurrence in long-term young breast cancer survivors randomly selected from a statewide cancer registry. Breast Cancer Res Treat 169, 141–152 (2018). https://doi.org/10.1007/s10549-018-4674-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-4674-5