Abstract
Cadmium (Cd) is known to cause various disorders in the testis, and metallothionein (MT) is known as a protein, which has a detoxification function for heavy metals. However, the changes of Fe, Cu, and Zn distribution in the testis induced by Cd exposure have not been well examined. Moreover, only a few studies have been reported on the localization of MT after Cd exposure. In this study, we have investigated the changes of Fe, Cu, and Zn distribution in Cd-exposed testis by a newly developed in air micro-Particle Induced X-ray Emission (PIXE) method. Also, we examined the distribution of MT expression in testis. In the testis of Cd-treated rats with significant increases of lipid peroxidation, the sertoli cell tight junction was damaged by Cd exposure, resulting from disintegration of the blood testis barrier (BTB). Evaluation by in air micro-PIXE method revealed that Cd and Fe distribution were increased in the interstitial tissues and seminiferous tubules. The histological findings indicated that the testicular tissue damage was advanced, which may have been caused by Fe flowing into seminiferous tubules followed by disintegration of the BTB. As a result, Fe was considered to enhance the tissue damage caused by Cd exposure. MT was detected in spermatogonia, spermatocytes, and Sertoli’s cells in the testis of Cd-treated rats, but was not detected in interstitial tissues. These results suggested that MT was induced by Cd in spermatogonia, spermatocytes, and Sertoli’s cells, and was involved in the resistance to tissue damage induced by Cd.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A heavy metal, cadmium (Cd), is used for industrial purposes throughout the world, and has been discharged into the environment in large quantities. It is also well known that Cd is toxic to various cells and organs in humans and other living things (Robards and Worsfold 1991). In particular, the testis is highly sensitive to Cd toxicity, and hemorrhagic necrosis of testicular tissues occurs in a short period after Cd exposure (Parizek and Zahor 1956; Nolan and Shaikh 1986; Saygi et al. 1991; Xu et al. 1999).
It has been reported that lipid peroxidation produced by Cd exposure were involved in testicular tissue damage (Omaye 1975; Sugawara and Sugawara 1984; Manca et al. 1991; Oteiza et al. 1999).
Furthermore, Cd administration consequently to increase the resistance to the metal by scavenging reactive oxygen species (ROS), and iron (Fe), copper (Cu), and zinc (Zn) levels in testicular tissues were significantly changed by Cd exposure (Ochi et al. 1983; Maitani and Suzuki 1986; Wahba and Waalkes 1990; Manca et al. 1991; Koizumi et al. 1992; Koizumi and Li 1992; Hatcher et al. 1995; Mikhailova et al. 1997; Shaikh et al. 1999; Yiin et al. 1999; Gaubin et al. 2000; Croute et al. 2005).
However, the mechanism of distribution changes of Cd, Fe, Cu, and Zn levels in cells of testicular tissues induced by Cd exposure has not been well evaluated. In this study, we have examined the distribution of these heavy metals in Cd-exposed testicular tissues by in air micro-Particle Induced X-ray Emission (PIXE) method.
Generally, in the PIXE method, the characteristic X-rays generated by the charged-particle irradiation of specimens were measured (Sven et al. 1995), but in the in air micro-PIXE method recently developed and used in this study, the characteristic X-rays emitted from specimens generated by a thin charged-particle beam (diameter, 1–2 μm) were measured, and the distribution of metal elements in cells could be more precisely detected (Sakai et al. 1999, 2002, 2005).
On the other hand, it has been reported that a heavy metal-binding protein, metallothionein (MT) is localized in the testis. It is a low molecular weight (6,600) protein, rich in cysteine (S) (30%), which binds and detoxifies heavy metals, such as Zn, Cu, and Cd, interacting with cysteine residues (Kagi and Kojima 1987).
Metallothionein was observed in spermatogenic and Sertoli’s cells of seminiferous tubules with mature sperms in the testis without heavy metal exposure, but not detected in Leydig’s cells (Nishimura et al. 1990; Tohyama et al. 1994).
In the Cd-administered testis, MT was observed in Sertoli’s and interstitial cells (Danielson et al. 1982). However, only a few studies have reported the localization of MT after Cd treatment and its roles in the testis.
In this study, we examined the levels of Cd, Zn, Cu, Fe, and lipid peroxidation, which are considered to be involved in testicular tissue damage caused by Cd exposure. Also, the changes in the distribution of Fe, Cu, Zn, and Cd in testicular tissues were examined by the in air micro-PIXE method. We also examined the MT expression in testicular tissues induced by Cd exposure.
Methods
Experimental animals
Male Wistar Sprague–Dawley rats were obtained from Saitama Experimental Animals Supply (Saitama, Japan), weighing 300–350 g. These rats were maintained at 20–25°C under a 12-h light-dark cycle, and divided into experimental groups (Cd administration) (n = 4 in each group) and control group (n = 4).
A single intraperitoneal administration of 1 mg/kg of Cd was administered using CdCl2 5/2H2O dissolved in saline, and the animals were sacrificed under deep ether anesthesia at 6, 12, and 24 h after Cd administration. In the control group, a single administration of the same volume of saline as in the Cd group was administered intraperitoneally, and the animals were sacrificed under deep ether anesthesia at 24 h after administration. In each group, the testis were removed immediately after the animals were sacrificed, and stored at −80°C. Part of the testicular tissues was fixed in 10% formalin, and histologically examined. All animals were treated under the guidelines for the care and use of laboratory animals of the university of Gunma, Japan.
Analysis by in air micro-PIXE method
The removed testis were frozen at −80°C, and 15-μm frozen sections were prepared using a cryostat. The sections were placed on a metal-free mylar sheet (10 μm), and dried at room temperature. Measurement was performed by in air micro-PIXE apparatus at Japan Atomic Energy Agency, Takasaki Advanced Radiation Research Institute.
Proton beams with a diameter of 1–2 μm generated at 3 MeV by a single end accelerator were used to irradiate sections for 45 min, and the characteristic X-rays emitted from each element were measured.
Measurement of heavy metal concentrations in testicular tissues
Two ml of concentrated nitric acid was added to 0.1 g of testicular tissues, and heated at 120°C for 48 h. The residues were dissolved in 1% nitric acid, and the concentration of metals was measured by Inductively Coupled Plasma Mass Spectroscopy (ICP-MS) (ELAN6100, Perkin Elmer, Yokohama, Japan). The mass/charge ratio (m/z) was assumed to be 114 for Cd, 66 for Zn, 57 for Fe, and 65 for Cu. As standard reference material, NBS-bovine liver No. 1577 (Washington, DC, USA) was used. The recovery was 95–99% for Cd, 83–91% for Zn, 110–115% for Fe, and 105–111% for Cu.
Measurement of thiobarbituric acid reactive substances (TBARS)
The malondialdialdehyde (MDA) formation of lipid peroxides in the testicular tissues was measured by thiobarbituric acid assay (Ohkawa et al. 1979).
Homogenization of 0.1 g of testicular tissues in 1.0 ml of 0.25 M cold sucrose was performed using a Teflon homogenizer. Subsequently, 0.2 ml of the solution was stirred with 0.2 ml of 8.1% SDS, 1.5 ml of 20% acetic acid, 1.5 ml of 0.8% thiobarbituric acid, and 0.6 ml of Milli-Q water, and reacted at 95°C for 1 h. The solution was cooled, stirred with 5.0 ml of a butanol/pyridine mixture (15:1 v/v), and centrifuged at 1,600g for 10 min. The absorbance of the supernatant was measured at 532 nm. Standard MDA (1,1,3,3-tetramethoxypropane) was used, and the results are expressed as MDA nmol/wet weight (g).
Measurement of MT concentration
The testicular tissues were homogenized in 4 volume of 10 mM Tris–HCl, pH 7.4 using a Teflon homogenizer. The homogenate were centrifuged at 20,000g for 30 min, and supernatant fraction was heated for 5 min in a boiling water bath. The heated samples were centrifuged at 20,000g for 30 min.
The concentration of MT in the testicular tissues were measured by the enzyme-linked immunosorbent assay (ELISA) method (Cousins 1991).
Microtiter plates were coated with antigen solution (MT standard, Sigma-Aldrich, Steinheim, Germany) solution containing 1 μg/ml in phosphate buffered saline (PBS) with 2-mercaptoethanol (10 μl/ml) was diluted 1:10(V/V) with 0.05 M carbonate buffer, pH 9.6; 100 μl/well). After incubated at 4°C for overnight, plates were washed three times with PBS containing 0.5% Tween 20 (PBS-T). A blocking solution (PBS-T containing 1% BSA) was added to each well and left for 1 h at room temperature, and the plates were washed with PBS-T.
Metallothionein standard or heated samples were diluted PBS-T containing 1% BSA and were added each well (50 μl/well). After this, 50 μl of a 1/3,000 of rabbit polyclonal anti-MT antibody (primary antibody) was added to each well. This antibody was a polyclonal antibody recognizing MT-1 and MT-2 of rabbits, humans, and rats, of which epitope was the N terminal (1–7 amino acid) of MT (Nakajima et al. 1991).
After incubated at room temperature for 3 h, plates were washed three times with PBS-T.
After this, 100 μl of a 1/1,000 dilution of goat anti-rabbit IgG conjugated to alkaline phosphatase (secondary antibody) (Sigma-Aldrich) was added to each well. After incubated at room temperature for 30 min, and the plates were washed three times with PBS-T. An appropriate quantity of p-nitrophenyl phosphate. After sufficient color development, measure absorbance at 405 nm.
MT immunostaining
The removed testis were fixed in 10% formalin and embedded in paraffin. Sections were produced at a thickness of 3 μm, and HE staining or MT immunostaining of the sections was performed. In MT immunostaining, after treatment with 0.3% hydrogen peroxide/methanol solution for 30 min to inhibit the intrinsic peroxidase reaction, the sections were vibrated in 0.01 M citrate buffer (pH 6.0) for 5 min in a microwave oven for better reaction of the antibody.
Subsequently, the specimens were reacted with rabbit polyclonal anti-MT antibody as the primary antibody for 2 h.
Histofine Simple Stain MAX-PO (MALTI) (Nichirei Corporation, Toksyo, Japan) was used as the secondary antibody, and reacted for 1 h. To develop color, 0.02% 3,3′-diamino-benzidine (DAB) was used.
Statistical analysis
All data are expressed as the means ± SD, and analyzed by Dr. SPSS II (SPSS Inc., Tokyo, Japan). Statistical analysis was performed by Dunnett’s test, and a value of P < 0.05 was regarded as significant.
Results
Analysis of heavy metals by in air micro-PIXE method
A two-dimensional P map provides a good representation of the physical shape of the cell, so a counter plot of the P map can be drawn (Nagamine et al. 2006). In the control group, Zn and Cu were widely distributed in the seminiferous tubules, and a small amount of Fe was detected (Fig. 1a).
Heavy metal maps of testis by in air micro-PIXE analysis. Since the P map provides a good representation of the physical shape of the cell, a counter plot of the P map was drawn (background, gray). Saline-injected animals were sacrificed at 24 h (control) (a). Rats given a single intraperitoneal injection of Cd (1 mg/kg) were sacrificed at 6 h (b), 12 (c) and 24 h (d). Purple line is 100 μm
In the experimental groups, larger amounts of Fe were detected in the interstitial tissues and seminiferous tubules in 6 h after Cd administration than in the control group. Cd was detected in the interstitial tissues and seminiferous tubules, and was widely distributed in the seminiferous tubules in particular (Fig. 1b).
Cd was widely distributed in the interstitial tissues and seminiferous tubules. Cu accumulation was detected on the testicular basement membrane 12 h after Cd administration, and Fe, Zn, and Cd were colocated (Fig. 1c).
At 24 h after Cd administration, Fe and Cd were widely distributed in the seminiferous tubules, and large amounts of Zn and Cd were colocated in the seminiferous tubules. Cu accumulation was detected in the seminiferous lumens, and Fe, Zn, and Cd were collocated (Fig. 1d).
Concentration of heavy metals in testicular tissues
The concentration of Cd in testicular tissues increased after Cd administration, and was significantly higher in the experimental groups after Cd administration than in the control group (P < 0.01) (Fig. 2a). The concentration of Fe was significantly higher in the experimental group 24 h after Cd administration than in the control group (P < 0.05) (Fig. 2b). The concentration of Zn in the testis increased after Cd administration, and was significantly higher in the experimental groups after Cd administration than in the control group (P < 0.01) (Fig. 2c). The concentration of Cu was significantly higher in the experimental group 24 h after Cd administration than in the control group (P < 0.01) (Fig. 2d).
Metal concentration in testis measured by ICP-MS. Saline-injected animals were sacrificed at 24 h (control, 0 h). Rats given a single intraperitoneal injection of Cd (1 mg/kg) were sacrificed at 6, 12, and 24 h after injection. All values are the mean ± SD (n = 4). *P < 0.05 versus control, **P < 0.01 versus control. (a) Cd, (b) Fe, (c) Zn, and (d) Cu
Concentration of TBARS
The concentration of TBARS in the testicular tissues increased after Cd administration, and was significantly higher in the experimental groups after Cd administration than in the control group (P < 0.05) (Fig. 3).
Thiobarbituric acid reactive substances (TBARS) concentration in testis. Saline-injected animals were sacrificed at 24 h (control, 0 h). Rats given a single intraperitoneal injection of Cd (1 mg/kg) were sacrificed at 6, 12, 24 h after injection. All values are the mean ± SD (n = 4). *P < 0.05 versus control
Concentration of MT
The concentration of MT in testicular tissues increased after Cd administration, and was significantly higher in the experimental groups after Cd administration than in the control group (P < 0.01) (Fig. 4).
Histological findings
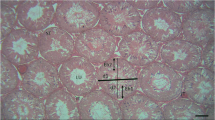
In the control group, a large number of mature sperm were observed in the seminiferous tubules, filled with spermatoblasts, spermatocytes, and Sertoli’s cells (Fig. 5a). In the experimental groups, the number of sperm in the seminiferous tubules was decreased in 6 h after Cd administration, and the lumens were dilated by the decrease in the number of cells (Fig. 5b). At 12 h after Cd administration, the seminiferous tubules and their basement membrane were detached, and they were atrophied by a marked decrease in the number of cells (Fig. 5c). Disintegration of the interstitial tissues and marked decreases in the numbers of cells and sperm in the seminiferous tubules were observed 24 h after Cd administration (Fig. 5d).
Immunohistostaining
In the specimens reacted with the MT antibody in the control group, weak MT expression in the peripheral regions of the seminiferous tubules was observed. MT expression in the spermatogonia, spermatocytes, and Sertoli’s cells was also observed, however, no MT expression was observed in the interstitial tissues (Fig. 6a).
MT localization in testis of control rats and Cd-treated rats by immunochemical staining. Saline-injected animals were sacrificed at 24 h (control) (a). Rats given a single intraperitoneal injection of Cd (1 mg/kg) were sacrificed at 6 h (b), 12 h (c), and 24 h (d) after injection. (1) Sertoli cell, (2) spermatogonia, (3) spermatocytes, (4) sperm, and (5) Leydig cells, interstitial tissues
Six hours after Cd administration, strong MT expression was observed in all regions of the seminiferous tubules. MT expression was strongest in the spermatogonia, spermatocytes, and Sertoli’s cells in particular, however, no MT expression was detected in the interstitial tissues (Fig. 6b). At 12 h after Cd administration, weak MT expression was observed in all regions of the seminiferous tubules, and was detected in the spermatogonia, spermatocytes, and Sertoli’s cells (Fig. 6c). At 24 h after Cd administration, MT expression was not observed in the peripheral regions of the seminiferous tubules because of severe damage to the testiculus organization (Fig. 6d).
Incubation of tissue sections with the anti-MT antibody and MT totally nullified the MT immunostaining (Fig. 7a, b).
Discussion
Cd administration generates ROS in the cellular level (Yang et al. 1997; Wang et al. 2004), and associated to decrease of reduced glutathione and increase of lipid peroxidation (Abe et al. 1994; Shaikh et al. 1999; Bagchi et al. 1996; Gaubin et al. 2000; Croute et al. 2005). Furthermore, it has been reported that hemorrhagic necrosis and apoptosis in the testis caused by Cd treatment decreased antioxidizing enzymes (Omaye 1975; Sato et al. 1983; Klimczak et al. 1984; Sugawara and Sugawara 1984; Koizumi et al. 1992; Koizumi and Li 1992; Sarkar et al. 1998; Oteiza et al. 1999).
In the present study, Cd treatment induced an increase of lipid peroxidation in the testis, as evidenced by the increase of TBARS levels. As a result, Cd administration increased lipid peroxidation, and damage to testicular tissues, suggesting that ROS generated by Cd exposure caused testicular tissue damage.
Acute Cd administration decreased testicular Zn and elevated Fe (Maitani and Suzuki 1986; Yiin et al. 1999). However, only a few studies have been reported the distribution of Cd, Fe, Zn, and Cu in testicular tissues after Cd treatment. The importance of metal in Cd exposure in the testis was suggested, and the distribution of Cd, Fe, Zn, and Cu in testicular tissues was examined by the newly developed in air micro-PIXE method.
Present study showed that Cd was increased in all regions of the seminiferous tubules by the in air micro-PIXE method after Cd administration. The concentration of Cd was demonstrated in testicular tissues analyzed by ICP-MS. Histologically, disintegration of the testicular basement membrane and atrophy of the seminiferous tubules were observed 12 h after Cd administration.
At 24 h after Cd administration, Fe accumulation was detected in the seminiferous tubules by the in air micro-PIXE method. The concentration of Fe was demonstrated in testicular tissues analyzed by ICP-MS. The histological findings indicated that testicular tissue damage was advanced. The blood testis barrier (BTB) mainly consists of the TJ of Sertoli’s cells, the BTB was damaged by Cd exposure (Aoki and Hoffer 1978; Hew et al. 1993; Xu et al. 1996; Russell 1997; Chung and Cheng 2001; Wong et al. 2004). The results of the present study suggested that the TJ was damaged by Cd exposure, resulting in disintegration of the BTB. At 24 h after Cd administration, the histological findings indicated that testicular tissue damage was advanced, which may have been caused by Fe flowing into seminiferous tubules followed by disintegration of the BTB, and Fe toxicity in free radical-mediated tissue damage has been reported (Fraga and Oteiza 2002). As a result, Fe was considered to enhance the tissue damage caused by Cd treatment.
It was also indicated that the amount of Fe was small in the control group, but was increased in interstitial tissues after Cd administration. In Leydig’s cells, oxidant-induced breakage of single-strand DNA was enhanced by Fe (II), and testicular tissue damage was also enhanced by Fe (Koizumi et al. 1992; Fraga and Oteiza 2002). Since Fe is involved in oxidative damage, this metal was considered to enhance the interstitial tissue damage caused by Cd treatment.
Furthermore, 12 h after Cd administration, Cu accumulation was observed in the basement membrane region of the seminiferous tubules by the in air micro-PIXE method, and Fe, Cd, and Zn were colocated. It was thought that such metals participated in the disintegration of the BTB.
Zn was increased in all regions of the seminiferous tubules by the in air micro-PIXE method after Cd administration. The concentration of Zn was demonstrated in testicular tissues analyzed by ICP-MS. We observed that Cd and Zn were colocated in seminiferous tubules by the in air micro-PIXE method after Cd administration. Since MT can bind these metals (Kagi and Kojima 1987), the detected Cd and Zn may be those bound to MT. Furthermore, it has been reported that Zn could suppress lipid peroxidation caused by Cd (Girotti et al. 1985; Zago et al. 2000; Zago and Oteiza 2001; Villanueva et al. 2005). It was thought that Zn was involved in the resistance to Cd-induced tissue damage.
With regard to MT expression, showing the high sensitivity of the testis to Cd toxicity. MT was detected in the spermatogonia, spermatocytes, and Sertoli’s cells of the control group. MT expression was observed in Sertoli’s cells and spermatogonia in seminiferous tubules (Nishimura et al. 1990; Tohyama et al. 1994). Our study confirmed these results.
Six hours after Cd administration, the concentration of Cd and MT in the testis peaked, but histological findings indicated that testicular tissue damage was weakly observed. MT was strongly expressed in the spermatogonia, spermatocytes, and Sertoli’s cells 6 h after Cd administration. Since weak MT expression in spermatogonia, spermatocytes, and Sertoli’s cells in the control group and strong MT expression in 6 h after Cd administration were observed, it was concluded that Cd induced MT expression in these cells. It was suggested that Cd-induced MT in spermatogonia, spermatocytes, and Sertoli’s cells was involved in the resistance to Cd-induced tissue damage.
Danielson et al. (1982) reported that MT was detected in interstitial cells in Cd administration experiments, but no MT expression was observed both in the control and Cd administration groups in the present study. This was probably because the MT induced in our study was lower due to the smaller dose of Cd compared to that of Danielson et al. or the difference of specificity of the MT antibody used in the studies.
In conclusion, after Cd administration, significant increases of lipid peroxidation are observed. Evaluation by the in air micro-PIXE method revealed that Fe distribution was increased in the interstitial tissues and seminiferous tubules. Furthermore, the histological findings indicated that testicular tissue damage was advanced, which may have been caused by Fe flowing into seminiferous tubules followed by disintegration of the BTB. As a result, Fe was considered to enhance the tissue damage caused by Cd exposure.
In the testis of Cd-treated rats, MT was detected in spermatogonia, spermatocytes, and Sertoli’s cells, but was not detected in interstitial tissues. These results suggested that MT was induced by Cd in spermatogonia, spermatocytes, and Sertoli’s cells, and was involved in the resistance to tissue damage induced by Cd.
References
Abe T, Konishi T, Katoh T, Hirano H, Matsukuma K, Kashimura M, Higashi K (1994) Induction of heat shock 70mRNA by cadmium is mediated by glutathione suppressive and non-suppressive triggers. Biochim Biophys Acta 28:29–36
Aoki A, Hoffer AP (1978) Reexamination of the lesions in rat testis caused by cadmium. Biol Reprod 18:579–591
Bagchi D, Bagchi M, Hassoun EA, Stohs SJ (1996) Cadmium induced excretion of urinary lipid metabolites, DNA damage, glutathione depletion, and hepatic lipid peroxidation in Sprague–Dawley rats. Biol Trace Elem Res 52:143–154
Cousins RJ (1991) Measurement of human metallothionein by enzyme-linked immunosorbent assay. Methods Enzymol 205:131–140
Chung NP, Cheng CY (2001) Is cadmium chloride-induced inter-Sertoli tight junction permeability barrier disruption a suitable in vitro model to study the events of junction disassembly during spermatogenesis in the rat testis? Endocrinology 142:1878–1888
Croute F, Beau B, Murat JC, Vincent C, Komatsu H, Obata F, Soleilhavoup JP (2005) Expression of stress-related genes in a cadmium-resistant A549 human cell-line. J Toxicol Environ Health A 68:703–718
Danielson KG, Ohi S, Huang PC (1982) Immunochemical detection of metallothionein in specific epithelial cells of rat organs. Proc Natl Acad Sci USA 79:2301–2304
Fraga CG, Oteiza PI (2002) Iron toxicity and antioxidant nutrients. Toxicology 180:23–32
Girotti AW, Thomas JP, Jordan JE (1985) Inhibitory effect of zinc(II) on free radical lipid peroxidation in erythrocyte membranes. J Free Radic Biol Med 1:395–401
Gaubin Y, Vaissade F, Croute F, Beau B, Soleilhavoup J, Murat J (2000) Implication of free radicals and glutathione in the mechanism of cadmiuminduced expression of stress proteins in the A549 human lung cell-line. Biochim Biophys Acta 1495:4–13
Hatcher EL, Chen Y, Kang YJ (1995) Cadmium resistance in A549 cells correlates with elevated glutathione content but not antioxidant enzymatic activities. Free Radic Biol Med 19:805–812
Hew KW, Heath GL, Jiwa A, Welsh MJ (1993) Cadmium in vivo causes disruption of tight junction-associated microfilaments in rat Sertoli cell. Biol Reprod 49:840–849
Johansson SAE, Campbel JL, Malmqvist KG (eds) (1995) Particle-induced X-ray emission spectrometry (PIXE). ISBN: 978-0-471-58944-0, Hardcover 451 pages, August 1995, John Wiley & Sons (Hardcover) pte. Ltd., USA, pp. 109–110
Kagi JH, Kojima Y (1987) Chemistry and biochemistry of metallothionein. Experientia Suppl 52:25–61
Klimczak J, WisniewskaKnypl JM, Kolakowski J (1984) Stimulation of lipid peroxidation and heme oxygenase activity with inhibition of cytochrome P-450 monooxygenase in the liver of rats repeatedly exposed to cadmium. Toxicology 32:267–276
Koizumi T, Li ZG (1992) Role of oxidative stress in single-dose, cadmium-induced testicular cancer. J Toxicol Environ Health 37:25–36
Koizumi T, Li ZG, Tatsumoto H (1992) DNA damaging activity of cadmium in Leydig cells, a target cell population for cadmium carcinogenesis in the rat testis. Toxicol Lett 63:211–220
Maitani T, Suzuki KT (1986) Essential metal contents and metallothionein-like protein in testes of mice after cadmium administration. Toxicology 40:1–12
Manca D, Ricard AC, Trottier B, Chevalier G (1991) Studies on lipid peroxidation in rat tissues following administration of low and moderate doses of cadmium chloride. Toxicology 67:303–323
Mikhailova MV, Littlefield NA, Hass BS, Poirier LA, Chou MW (1997) Cadmium-induced 8-hydroxydeoxyguanosine formation, DNA strad breaks and antioxidant enzyme activities in lymphoblastoid cells. Cancer Lett 115:141–148
Nagamine T, Kusakabe T, Takada H et al (2006) Interferon beta-induced changes in metallothionein expression and subcellular distribution of zinc in HepG2 cells. Cytokine 34:312–319
Nakajima K, Suzuki K, Otaki N, Kimura M (1991) Epitope mapping of metallothionein antibodies. Methods Enzymol 205:174–189
Nishimura H, Nishimura N, Tohyama C (1990) Localization of metallothionein in the genital organs of the male rat. J Histochem Cytochem 38:927–933
Nolan CV, Shaikh ZA (1986) The vascular endothelium as a target tissue in acute cadmium toxicity. Life Sci 39:1403–1409
Ochi T, Ishiguro T, Ohsawa M (1983) Participation of active oxygen species in the induction of DNA single-strand scissions by cadmium chloride in cultured Chinese hamster cells. Mutat Res 122:169–175
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Omaye ST (1975) Effect of cadmium chloride on the rat testicular soluble selenoenzyme, glutathione peroxidase. Res Commun Chem Pathol Pharmacol 12:695–711
Oteiza PI, Adonaylo VN, Keen CL (1999) Cadmium-induced testis oxidative damage in rats can be influenced by dietary zinc intake. Toxicology 137:13–22
Parizek J, Zahor Z (1956) Effect of cadmium salts on testicular tissue. Nature 177:1036
Russell LD (1977) Movement of spermatocytes from the basal to the adluminal compartment of the rat testis. Am J Anat 148:313–328
Robards K, Worsfold P (1991) Cadmium: toxicology and analysis. Analyst 16:549–568
Sakai T, Naitoh Y, Kamiya T et al (1999) An external ion microbeam for studies of biological samples. Biol Trace Elem Res Winter 71–72:77–82
Sakai T, Kamiya K, Oikawa M, Sato T, Tanaka A, Ishii K (2002) JAERI Takasaki in-air micro-PIXE system for various applications. Nucl Instrum Methods Phys Res B 190:271–275
Sakai T, Oikawa M, Satoh T, Nagamine T, Moon HD, Nakazato K, Suzuki K (2005) New in-air micro-PIXE system for biological applications. Nucl Instrum Methods Phys Res B 231:112–116
Sarkar S, Yadav P, Bhatnagar D (1998) Lipid peroxidative damage on cadmium exposure and alterations in antioxidant system in rat erythrocytes: a study with relation to time. Biometals 11:153–157
Sato M, Yamanobe K, Nagai Y (1983) Sex-related differences in cadmium-induced lipid peroxidation in the rat. Life Sci 33:903–908
Saygi S, Deniz G, Kutsal O, Vural N (1991) Chronic effects of cadmium on kidney, liver, testis, and fertility of male rats. Biol Trace Elem Res 31:209–214
Shaikh ZA, Vu TT, Zaman K (1999) Oxidative stress as a mechanism of chronic cadmium hepatotoxicity and renal toxicity and protection by antioxidants. Toxicol Appl Pharmacol 154:253–256
Sugawara N, Sugawara C (1984) Selenium protection against testicular lipid peroxidation from cadmium. J Appl Biochem 6:199–204
Tohyama C, Nishimura N, Suzuki JS, Karasawa M, Nishimura H (1994) Metallothionein mRNA in the testis and prostate of the rat detected by digoxigenin-labeled riboprobe. Histochemistry 101:341–346
Villanueva O, Vigueras RM, Hernandez R, Chavira R, Cardenas M, Villa A, Murphy E (2005) Zinc-induced survival of Leydig cells in Fischer rats (Rattus norvegicus) treated with cadmium chloride. Comp Med 55:533–538
Wahba ZZ, Waalkes MP (1990) Cadmium-induced route-specific alterations in essential trace element homeostasis. Toxicol Lett 54:77–81
Wang Y, Fang J, Leonard SS, Rao KM (2004) Cadmium inhibits the electron transfer chain and induces reactive oxygen species. Free Radic Biol Med 11:1434–1443
Wong CH, Mruk DD, Lui WY, Cheng CY (2004) Regulation of blood-testis barrier dynamics: an in vivo study. J Cell Sci 117:783–798
Xu C, Johnson JE, Singh PK, Jones MM, Yan H, Carter CE (1996) In vivo studies of cadmium-induced apoptosis in testicular tissue of the rat and its modulation by a chelating agent. Toxicology 107:1–8
Xu G, Zhou G, Jin T, Zhou T, Hammarstrom S, Bergh A, Nordberg G (1999) Apoptosis and p53 gene expression in male reproductive tissues of cadmium exposed rats. Biometals 12:131–139
Yang CF, Shen HM, Shen Y, Zhuang ZX, Ong CN (1997) Cadmium-induced oxidative cellular damage in human fetal lung fibroblasts (MRC-5 cells). Environ Health Perspect 105:712–716
Yiin SJ, Cheru CL, Sheu JY, Lin TH (1999) Cadmium-induced lipid peroxidation in rat testes and protection by selenium. Biometals 12:353–359
Zago MP, Verstraeten SV, Oteiza PI (2000) Zinc in the prevention of Fe2+-initiated lipid and protein oxidation. Biol Res 33:143–150
Zago MP, Oteiza PI (2001) The antioxidant properties of zinc: interactions with iron and antioxidants. Free Radic Biol Med 31:266–274
Acknowledgments
This study was supported by a grant for young researchers of twenty-first century COE program of Gunma University, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kusakabe, T., Nakajima, K., Suzuki, K. et al. The changes of heavy metal and metallothionein distribution in testis induced by cadmium exposure. Biometals 21, 71–81 (2008). https://doi.org/10.1007/s10534-007-9094-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-007-9094-7