Abstract
Shifting and permanent cultivation, selective logging, cattle production and coffee plantations are among the most important factors in montane cloud forest conversion and disturbance. Although shaded-coffee plantations can contribute to the preservation of local species richness, abundance of organisms could be determined by habitat resource availability in agricultural landscapes. We compared abundance of Sturnira and Artibeus bats (Phyllostomidae, Stenodermatinae), in shade coffee plantations and disturbed cloud forest fragments, which represent habitats with different chiropterochorous plant density. We also investigated the relationship between bat species abundance and food plant richness, abundance and diversity. We captured 956 bats, 76% in cloud forest fragments and 24% in shaded coffee plantations. Abundance of Sturnira spp. (small bats) was greater in cloud forest than in coffee plantations, but Artibeus spp. (large bats) abundance was similar in both habitats. Chiropterochorous plant abundance was positively related with bat abundance for Sturnira spp., while chiropterochorous plant richness and diversity were negatively related for Artibeus spp. This suggests that frugivorous bats with different morphological and ecological characteristics respond differentially to anthropogenic activities. For landscape management purposes, the maintenance and augmentation of diverse food resources, for frugivorous bats with different foraging requirements in coffee plantations, will benefit the resilience of bats to modification of their natural habitat.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The contribution of transformed landscapes to diversity conservation has been widely recognized; particularly in tropical forest mosaics comprised of forest fragments and crops such as shaded coffee plantations (Bhagwat et al. 2008; Gardner et al. 2009). Studies have found that shaded coffee plantations with low management—rustic or traditional polyculture (sensu Moguel and Toledo 1999)—maintain species richness near to that of the forest, particularly for non-forest specialist species (Philpott et al. 2008; Manson et al. 2008a, b). Forest fragments in a matrix of cattle pasture can contain a species richness approaching that of a landscape dominated by continuous forest, especially for highly mobile taxa such as bats and birds (Estrada et al. 2004; Medina et al. 2007). In the case of bats, even if species richness is similar between natural forests and shaded-coffee plantations, the abundance of many species is normally lower in coffee crops (Sosa et al. 2008). However, few studies have specifically investigated the changes in resource abundance and other factors which presumably underlie these patterns (Willig et al. 2007; Sosa et al. 2008).
Abundance is an important attribute of ecological communities, and low individual numbers of species can be interpreted as habitat loss or degradation (Fahrig 2003). Bat abundance is influenced by the availability of resources (Hodgkison et al. 2004; Dumont 2005). Temporal and spatial variation of food distribution within landscapes, and different energy requirements among bat species, are likely to influence how they perceive and use habitat patches within landscapes, and affect abundance patterns at the species level (Allen et al. 1999; Duchamp et al. 2007; Gagné et al. 2008; Pinto and Keitt 2008; Klingbeil and Willig 2009).
New World leaf-nosed bats (Phyllostomidae) are relatively abundant and are frequently captured when using mist-nets. They have been recognized for their taxonomic and ecological diversity, and particularly for their diet diversification (Gardner 1977). Frugivores constitute more than half of the 49 genera in this family (Nowak et al. 1994), and provide the critical ecosystem service of seed dispersal for regenerating open and abandoned areas and maintaining natural forest succession in the Neotropics (Galindo-González et al. 2000; Muscarella and Fleming 2007). Many species of frugivorous phyllostomids are sympatric (Giannini and Kalko 2004), such that they have developed mechanisms of fruit resource partitioning including vertical stratification of foraging areas and dietary specialization (Dumont 1999; Giannini and Kalko 2004).
In the mountainous region of central Veracruz, Mexico, Stenodermatinae fruit-eating bats are the most common phyllostomids, especially Artibeus intermedius, A. jamaicensis, Sturnira ludovici, and S. lilium (Pineda et al. 2005; Sosa et al. 2008). These sympatric species differ in morphology, size, displacement capacity in addition to home range and abundance of fruit species in their diets (Kalko 1999; Dumont 2005; Hernández-Montero 2009). Artibeus species have the largest body size (50–73 g) and maximum linear movement (8 km; Galindo-González 1998; Bernard and Fenton 2003). Artibeus spp. consume fruit from a great variety of late successional tropical trees, the majority in the families Anacardiaceae (more than five spp.), Lauraceae (two spp.), and Moraceae (more than 10 spp.); and exotic trees like Syzigium jambos and Musa paradisiaca. They also occasionally eat fruits on pioneer species of shrubs and trees like Piper spp., Solanum spp. and Cecropia obtusifolia (Ortega and Castro-Arellano 2001; Flores 2005; Hernández-Montero 2009). Conversely, species of Sturnira are smaller (20–25 g), have shorter linear movements (<3 km), and consume mainly early succession fruits, such as those of Solanum spp. and Piper spp. (Hernández-Conrique et al. 1997; Loayza and Loiselle 2008; Mello et al. 2008; Hernández-Montero 2009).
In the mountainous region of central Veracruz, coffee and cattle production have been important economic activities for over a century (Nestel 1995; Montagut-González 1999). Both landform and climate determined the regionalization of sugar cane and coffee crops, as well as pasture for cattle grazing, along an altitudinal gradient originally occupied by primary montane cloud forest. In general, but with some overlap, sugar cane is cultivated in the lower zones (below 800 m asl), shaded-coffee in the intermediate zone (700–1,400 m asl), and pastureland in the upper zone (above 1,400 m asl) (Hoffmann 1993). Most cloud forest fragments remain in the upper zone so they are normally located within a matrix of pastureland. All of these remnant fragments experience a certain degree of disturbance, most often selective logging and gathering of firewood. In contrast, coffee plantations are surrounded by a more heterogeneous matrix composed of additional coffee plantations, secondary forest, villages, roads, and other crops including forage grasses (see Table 1).
Coffee plantations and disturbed montane cloud forest fragments differ in plant structure and composition (Muñiz-Castro et al. 2006; Manson et al. 2008a). The shade in coffee plantations may be provided by native species of trees such as Inga jinicuil, Quercus sp., Liquidambar styraciflua, Trema micrantha, Clethra macrophylla, C. mexicana, or several non-native species like Inga vera, Musa paradisiaca, Syzygium jambos, Eriobotrya japonica. Such plantations are thus classified as commercial polycultures (sensu Moguel and Toledo 1999). Shrubs and herbs in coffee plantations are regularly pruned; the removal of the herbaceous layer in coffee plantations stands in stark contrast to disturbance regimes in cloud forest fragments associated with selective logging, in which early successional shrubs and trees dominate the understory. The remnant forest fragments consequently contain shrubs such as Piper auritum, Piper hispidum, Piper lapathifolium, Solanum spp., and trees such as Carpinus caroliniana, Trema micranta, and Acacia pennatula (Muñiz-Castro et al. 2006). These, among other vegetation differences in structure and composition, could have an effect on the abundance of bats with different morphology, size, home range and preferred food.
In this paper, we compare the abundance response of four common frugivorous bats (Artibeus jamaicensis, A. intermedius, Sturnira ludovici, S. lilium) in two transformed forest habitats: disturbed montane cloud forest fragments and shade coffee plantations—hereafter called forest fragments and coffee plantations. We explore how these habitats differ in density of chiropterochorous plants and, consequently, in abundance of frugivorous bats. We also investigate the relationship between bat abundance and certain ecological attributes (richness, abundance and Shannon diversity) of plant species, hereafter referred to as chiropterochorous plants, whose fruits are consumed—and seeds dispersed—by bats. We expect that: (1) The abundance of Sturnira spp. (with small body size, limited displacement capacity, small home range and shrub specialized feeding) will be higher in cloud forests than in coffee plantations, whereas the abundance of Artibeus spp. (large body size, greater displacement capacity, wide home range and generalized feeding) will be comparable between the two habitats; (2) the richness, abundance and diversity (H′) of chiropterochorous plants, for both small and large bat species, will be greater in the cloud forest compared with coffee plantations; and (3) the abundance of bat species will be positively associated with the richness, abundance and diversity of chiropterochorous plants.
Study sites and methods
Study sites
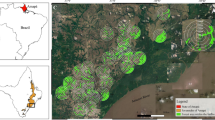
The study was carried out in the municipalities of Xalapa and San Andrés Tlalnehuayocan, in the mountainous region of central Veracruz (19°31′19″ to 19°29′46″ N, 96°59′30″ to 96°54′36″ W). The landscape arrangement in the region did not allow us to select study sites in two landscapes ideal for comparison: coffee plantations surrounded by a matrix of cloud forest, and forest fragments surrounded by a matrix of coffee plantations, however, we compared sites immersed in two relatively common transformed landscapes in the region. Four cloud forest fragments (surrounded mainly by pastures) and four coffee plantations of commercial polyculture (immersed in a matrix mainly of secondary forest) were selected using the following criteria: all sites of similar altitude for comparison of bat diversity (Sosa et al. 2008), and replicate sites located >1 km apart, with a minimum of 7 km in distance between habitat types, to avoid capturing the same individuals in both habitats. In the selection of coffee plantations, we also considered that the area of any forest fragment surrounding a coffee plantation could be not greater than the area of the selected site (Fig. 1; Table 1). Furthermore, during visits to the selected coffee plantations, we verified that the understory of the contiguous forest patches was absent, thus discounting the possibility that small frugivorous bats were being attracted by these patches. Table 1 shows the sampled area (range covered by nets) within forest fragments and coffee plantations.
Study area in the central mountainous region of Veracruz, Mexico. “A” corresponds to pasture matrix (forest patches; see text and Table 1). “B” corresponds to heterogeneous matrix (coffee patches). Gray polygons represent cloud forest, black polygons represent shade coffee plantations and white areas represent pastures. Each study site has an identifying mark. “F” indicates forest fragments and “C” indicates coffee plantations (F1: Abundio, F2: Agüita Fría, F3: Luis Hernández, F4: Valderrama-Navarro, C1: Antonio Fernández, C2: Juan, C3: Timoteo Zavaleta, C4: Epifanio Sánchez)
A GIS was created and, using a supervised classification (from a LANDSAT-7 2000 satellite image), the matrix of the study sites was characterized. Coffee plantations were separated from secondary forest using the census of coffee plantations conducted by the Secretaría de Agricultura, Ganadería, Desarrollo Rural y Pesca (2000; Fig. 1). A 44 ha wide buffer around the centre of the sampling area in each habitat and replicate was delimited to quantify the proportion of area occupied by each land use. Data is presented in Table 1.
Data collection
Fruiting of cloud forest chiropterochorous plants in this region is not concentrated in any particular season; consequently, bat sampling was conducted over an 11-month period in order to include the three seasons recognized in the region (Soto-Esparza and Gómez-Columna 1990; Williams-Linera 2007). Each cloud forest fragment and coffee plantation was sampled on one night per month (11 nights per site, giving a total of 44 nights in each of the two land use types), avoiding nights around the full-moon.
Bats were captured with eight mist nets (9 m long × 2.6 m high) placed in the understory of each plantation or forest fragment. Nets were opened at dusk for 5 h and checked every 30 min. Bats have a stratified foraging behavior (Kalko 1999); however, to move within forests they use “corridors”, areas without vegetation that may facilitate their movements (Caras and Korine 2009). Therefore, four 15–30 m interspaced pairs of mist nets were placed across various manmade corridors in an area of approximately 1.6 ha in each forest or plantation patch (Table 1). Sampling nights were alternated between habitat types, to disperse possible meteorological effects among samples. Bats were identified using field keys (Medellín et al. 1997). Because no conclusive evidence exists to the contrary (Guerrero et al. 2003; Redondo et al. 2008), we recognized A. intermedius as a valid species, following the most recent nomenclature for the terrestrial mammals of Mexico (Ramírez-Pulido et al. 2005). Abundance was quantified as the total number of individuals captured per forest fragment or coffee plantation. Before release, each bat was tagged with a numbered plastic collar (Security A-ties® Fasteners) to avoid inclusion of recaptures in the counts. Sampling effort was expressed as net m2 h sensu Straube and Bianconi (2002).
To estimate the abundance of chiropterochorous plants in each study site, we made direct counts of plants in ten 2 × 50 m strip transects per site (totalling 0.1 ha per site), randomly established in each cloud forest and coffee plantation. Trees or shrubs of height ≥1 m, and rooted within the transects, were recorded and identified. Plant material from those species that could not be determined in the field was collected for subsequent determination in the XAL herbarium at the Instituto de Ecología, A.C. Plants recorded in this study were classed as chiropterochorous for each bat species based on the list of Geiselman et al. (2002), and the bat fecal analysis recorded by Hernández-Montero (2009), from the same study site (Table 2). Because the respective consumption by A. jamaicensis and S. lilium of Coffea arabica and Musa paradisiaca fruit is merely the subject of anecdotal reports, these species were not considered as chiropterochorous plants for the purposes of this study.
Data analyses
We compared bat abundance between habitats (cloud forest and coffee plantation; n = 4 patches for each habitat) at both species and genus level (i.e. Sturnira spp.), using a Mann–Whitney U-test. Diversity of chiropterochorous plants was estimated by the Shannon–Wiener index (H′). Plant richness, abundance and diversity were also compared between habitats using a Mann–Whitney U-test. Non-parametric tests were used as data did not conform to the assumptions for an equivalent parametric test.
Pooling data over all sampling nights for each of the eight patches (irrespective of habitat), the possible association between bat abundance and chiropterochorous-plant richness, abundance and diversity were assessed using Pearson or Spearman correlation depending on data distribution normality. Because not all fruit species are shared by the four studied bat species, the number of chiropterochorous plants varied for each bat species in each patch, in the correlation analyses.
Results
A total of 82,368 net m2 h, resulted in 956 captures of the studied species (see list of all bat species captured and numbers in Supplementary Material). Seventy-six percent of all individuals were captured in cloud forest, and 24% in the coffee plantations. The four most frequently captured bat species were Sturnira ludovici (n = 727 individuals), S. lilium (n = 103), Artibeus intermedius (n = 80) and A. jamaicensis (n = 46). The recaptured percentage per species was 15% (cloud forest) and 6% (coffee plantations) for Sturnira ludovici, 8% (cloud forest) and 13% (coffee plantations) for S. lilium, and 0% for A. intermedius and A. jamaicensis in both habitats. In addition, there were no recaptures in a different habitat than the initial-capture habitat.
Sturnira spp. abundance was higher in cloud forest fragments than in coffee plantations (Fig. 2) at both genus (W = 16; d.f. = 1; P < 0.05) and species level (Sturnira ludovici, W = 16; d.f. = 1; P < 0.05; Sturnira lilium, W = 16; d.f. = 1; P < 0.05). There was no difference in Artibeus spp. abundance between habitats, at either genus or species level (P > 0.05; Fig. 2).
Fruits consumed by bats came from understory and canopy plant species in both habitats. However, in cloud forest the abundance of chiropterochorous plants was higher in the understory than in the canopy, while the opposite occurred in coffee plantations (Table 2). Abundance of chiropterochorous plants was greater in cloud forest than in coffee plantations, for both Sturnira ludovici (W = 16; d.f. = 1; P < 0.05) and S. lilium (W = 16; d.f. = 1; P < 0.05) and, consequently, at the genus level (Sturnira spp.; W = 16; d.f. = 1; P < 0.05). No significant differences in chiropterochorous plant richness and diversity were recorded between habitats at the species or genus level for Sturnira spp. (P’s > 0.05). The richness, abundance and diversity of chiropterochorous plants assigned to Artibeus spp. did not differ (P > 0.05) between habitats.
Bat abundance was positively correlated to chiropterochorous plant abundance at genus level for Sturnira (r s = 0.76; d.f. = 7; P < 0.05); and at species level for S. lilium (r = 0.76; d.f. = 7; P < 0.05). In the case of S. ludovici, this positive correlation was close to statistical significance (r = 0.67; d.f. = 7; P = 0.07; Fig. 3). On the other hand, no correlation (P > 0.05) was found between abundance of Sturnira spp. and richness and Shannon diversity of chiropterochorous plants. Richness and diversity of fruits eaten by Artibeus spp. were negatively correlated with Artibeus spp. abundance (plant richness: r = −0.78; d.f. = 7; P < 0.05; diversity: r = −0.85; d.f. = 7; P < 0.05). However, at the species level, these relationships only remain significant for Artibeus intermedius (plant richness: r = −0.74; d.f. = 7; P < 0.05; diversity: r = −0.73; d.f. = 7; P < 0.05).
Discussion
Factors explaining bat abundance patterns
Our main results show that the abundance of small frugivores (Sturnira spp.), and their food resources, declined in coffee plantations, whereas the abundance of large frugivores (Artibeus spp.) and their resources did not differ between the two habitat types. This implies that the abundance of small bats was affected by the low density of chiropterochorous plants in coffee plantations. Thus, while our first hypothesis was proved, our second and third hypotheses proved to be true only for the Sturnira species. Some traits of the autoecology of these species may partially explain these results. Sturnira spp. depend on roosts in tall trees, and on the understory for food provision (Evelyn and Stiles 2003; Mello et al. 2008), therefore individuals of these species may prefer forest fragments. In contrast, large frugivorous bats like A. jamaicensis and A. intermedius do not depend so heavily on big trees with holes to roost, as they can roost in palm fronds and the foliage of canopy and subcanopy trees (Ortega and Castro-Arellano 2001; Evelyn and Stiles 2003; Muñoz-Romo et al. 2008). The ability of Artibeus species to use a variety of habitats, including shade coffee plantations, can be attributed to their long-distance movements and their broader diet (Ortega and Castro-Arellano 2001; Galindo-González 1998; Bernard and Fenton 2003; Gorresen and Willig 2004).
Likewise, the greater abundance of chiropterochorous plants for Sturnira in forest fragments explains the greater abundance of these bats in this habitat. Human activities in the forest fragments, such as selective logging, increase the presence of pioneer shrubs and chiropterochorous plants (i.e. Piper spp. and Solanum spp.) which, in coffee plantations, are frequently weeded out in plantation maintenance. This suggests a strong relationship between Sturnira bats and the understory food items in disturbed cloud forests, as has been reported for tropical semi-deciduous forest fragments (Schulze et al. 2000). In the studied region, this relationship is reinforced by the pasture matrix surrounding most forest fragments, which renders other fruit resources unavailable for small bats such as Sturnira. Although this relationship has been proposed as an indicator of disturbance, its utility as an indicator would be restricted to forest fragments because the dramatic disturbance caused by pruning in coffee plantations can completely eliminate the understory species of bat-fruit, reducing the abundance of small bats. Thus, our results suggest that patterns of forest fragmentation and agroecosystem management affect bat communities differentially. This effect is, however, also influenced by disturbance at habitat and patch level (Franklin and Lindenmayer 2009).
Although the forest fragments were larger than the coffee plantations, our sampling method makes it unlikely that patch size influenced our results. We estimated bat density by applying the same netting effort of almost the same area in every patch (Table 1), following the assumption that bat distribution is random within patches. Furthermore, recent studies suggest that patch size could only be important, in determining number and abundance of species, when patches are on a matrix that constitutes a completely inhospitable habitat (Prugh et al. 2008). This is not the case in the mountainous region of central Veracruz. For instance, when we compare bat captures on fragments of contrasting size (for example F2 and F3 in Fig. 1), captures are similar; Conversely, when we compare fragments of comparable size (F1 and F3) there are ten times more captures in F3 than in F1 (see Supplementary Material).
More research is needed to explain the negative and surprising relationship between Artibeus spp. abundance and chiropterochorous plants richness and diversity. It has been proposed that A. jamaicensis consumes more food items from the canopy than from the understory (Geiselman et al. 2002). Therefore, the low concentration of canopy food items and homogeneous vegetation structure in shade coffee plantations (Saldaña-Vázquez 2008) could not explain the observed negative relationship. We hypothesize that Artibeus bats are common in shaded-coffee plantations because the vegetation structure allows them to fly easily. Artibeus wing morphology and body size restricts its manoeuvrability in cluttered scenarios like a disturbed forest, where space between plant individuals is reduced (Stockwell 2001; Caras and Korine 2009). In coffee plantations of the studied region they have been captured more often in mist nets hung at the sub-canopy, where an ample flight-passage is available, than at the understory level (V. J. Sosa, unpublished data). The longer distance movements of Artibeus might allow them to exploit a variety of food resources from several habitats such as living fences, backyard orchards, and even isolated trees (Galindo-González et al. 2000); or from those that are spatially concentrated, such as Ficus trees—absent in our plantations, but common in other plantations (Manson et al. 2008a). We cannot rule out that the presence of bat roosts and other chiropterochorous plants (not yet recorded as items of the bat diet) in the secondary forest fragments and other wooded habitats adjacent to coffee farms, could contribute—if sufficiently abundant—to the lack of association of Sturnira ludovici and Artibeus abundance with their food items. Further research must measure chiropterochorous plant abundance within the patch and its contiguous matrix habitat. The question of whether Artibeus and Sturnira bats use coffee plantations mainly as a stepping-stone habitat or for foraging and day-roosting, is currently under study. In general, our results support the notion that Artibeus species (large bats) forage at a scale larger than the size of cloud forest fragments and coffee plantations in our study region, whereas Sturnira species (small bats) forage at a smaller scale. This is supported by the results of recaptures: none for Artibeus and all for Sturnira in the same habitat of first capture.
Our results on bat abundance contrast to the results of a similar study conducted in the coffee region of Quindío in Colombia (Numa et al. 2005). In this region, the overall abundance of common frugivorous bats (Artibeus jamaicensis, A. lituratus, Sturnira lilium, S. ludovici) in lower montane forest patches was 28% of the abundance in coffee plantations. In forest patches, the abundance of large species (Artibeus) was one half and one-third of the abundance of associated non-shaded and shaded coffee, respectively. On the contrary, small species (Sturnira) were doubly abundant in forest than in associated coffee or comparably abundant between forest and shaded coffee. This lack of consistence in results could be due to the different landscape context in which both studies were conducted. In the Quindío study, the three compared habitats could be found in two types of matrix, one dominated by associated coffee and another one dominated by shaded-coffee. Forest fragments in the Quindío region are small and scarce and surrounded by these two types of landscape matrix. Unfortunately, in that study, the fruit resource available for bats in the compared habitats was not quantified; thus, its importance in explaining bat species richness and abundance cannot be evaluated. According to available studies, it seems that landscape context plays a determinant role for bat diversity distribution in habitats of varying degrees of tree cover in coffee regions of the Neotropics.
Implications for conservation
We now relate our results to the conservation of frugivorous bats in a fragmented landscape. As important seed dispersers, Stenodermatinae bats are considered, together with bird dispersers and seed sources, as part of the regeneration potential of Neotropical forests (Muscarella and Fleming 2007). We have seen that, in coffee plantations, pruning understory shrubs that bear small fruits apparently reduces the abundance of small bats. Thus, in a landscape dominated by coffee plantations, the maintenance of viable populations of these bats would depend on management practices that promote understory shrubs. This can hardly be implemented in most small plantations (<5 ha), which constitute the majority within the region. However, the heterogeneous matrix of coffee landscapes could counterbalance the low density of understory-food shrubs in plantations by offering these shrubs in other forested marginal habitats such as fallows, living fences, secondary vegetation patches, etc.
On the other hand, while disturbed cloud forests in the pasture landscape offer food and roosting sites to small frugivorous bats, they are ecologically isolated when surrounded by a pasture matrix. In this landscape, bat conservation could be enhanced if both the connectivity between forest patches and patch size are increased. By confining cattle, the edge and extent of the forest patch can increase at the expense of the pasture. Of greater difficulty is the establishment of forested corridors, although riparian vegetation can function very well in the region. Simple rules for cattle producers to extend and maintain riparian zones and living fences would also contribute to bat conservation in the pasture dominated landscape. In addition, maintaining the connectivity of remnant forest fragments in this landscape would allow the movement of bats dispersing seeds of pioneer plants, and thereby increasing the possibility of passive forest restoration (Holl 1999; Muñiz-Castro et al. 2006).
The abundance of large frugivorous bats (Artibeus spp.) is not affected by habitat fragmentation or coffee management practices in the studied region. It seems that the current structure of the fragmented landscapes (either with a pasture or heterogeneous matrix), still allows them to forage and find day-roosts in diverse types of forested man-made habitats, thanks to their greater range. However, their densities could be increased by favouring connectivity in the pasture landscape and chiropterochorous trees in the canopy of coffee plantations. Coffee growers should be encouraged to implement these conservation actions through a program of publicity that shows the benefits of conserving biodiversity in coffee plantations in order to access more profitable alternative markets for environmentally friendly coffee.
In summary, our research showed that frugivorous bats with different body sizes and feeding ecologies have differential abundance responses to rural anthropogenic (coffee production and selective logging) activities. The management practices of coffee growing, such as pruning of the understory, are associated with a decrease in the abundance of small bats, but not of large bats. More research is still needed regarding the distribution of food and roost resources for bats in agroforestry systems and other man-made habitats. These bats provide the critical ecosystems service of seed dispersal for regenerating open and abandoned areas and maintaining natural forest succession. It is therefore of crucial importance that management at the landscape scale can provide the assemblages of frugivorous bats (large and small) with the food items and roost availability becoming ever more necessary in the face of modification to their natural habitats.
References
Allen CR, Forys EA, Holling CS (1999) Body mass patterns predict invasions and extinctions in transforming landscapes. Ecosystems 2:114–121
Bernard E, Fenton MB (2003) Bat mobility and roosts in a fragmented landscape in central Amazonia, Brazil. Biotropica 35:262–277
Bhagwat SA, Willis KJ, Birks HJB, Whittaker RJ (2008) Agroforestry: a refuge for tropical biodiversity? TREE 23:261–267
Caras T, Korine C (2009) Effect of vegetation density on the use of trails by bats in a secondary tropical rain forest. J Trop Ecol 25:97–101
Duchamp JE, Arnett EB, Larson MA, Swihart RK (2007) Ecological considerations for landscape-level management of bats. In: Lacki MJ, Hayes JP, Kurta A (eds) Conservation and management of bats in forests. John Hopkins University Press, Baltimore
Dumont ER (1999) The effect of food hardness on feeding behaviour in frugivorous bats (Phyllostomidae): an experimental study. J Zool Lond 248:219–229
Dumont ER (2005) Bats and fruit: an ecomorphological approach. In: Kunz TH, Fenton BM (eds) Bat ecology. University Chicago Press, Chicago
Estrada A, Coates-Estrada R, Meritt D Jr (2004) Anthropogenic landscape changes and avian diversity at Los Tuxtlas, Mexico. Biodivers Conserv 6:19–43
Evelyn MJ, Stiles DA (2003) Roosting requirements of two frugivorous bats (Sturnira lilium and Artibeus intermedius) in fragmented Neotropical forest. Biotropica 35:405–418
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515
Flores JJ (2005) Artibeus intermedius. In: Ceballos G, Oliva G (eds) Los mamíferos de México. Fondo de Cultura Económica y Comisión Nacional para la Biodiversidad, México, DF
Franklin JF, Lindenmayer DB (2009) Importance of matrix habitats in maintaining biological diversity. PNAS 106:349–350
Gagné SA, Proulx R, Fahrig L (2008) Testing Holling’s textural-discontinuity hypothesis. J Biogeogr 35:2149–2150
Galindo-González J (1998) Dispersión de semillas por murciélagos: Su importancia en la conservación y regeneración del bosque tropical. Acta Zoológica Mexicana (n.s.) 73:57–74
Galindo-González J, Guevara S, Sosa VJ (2000) Bat-and bird-generated seed rains at isolated trees in pastures in a tropical rainforest. Conserv Biol 14:1693–1703
Gardner AL (1977) Feeding habits. In: Baker JR, Jones KJ, Carter CD (eds) Biology of bats of the New World Family Phyllostomidae part II. Special publications. The Museum Texas Tech University, Lubbock
Gardner TA, Barlow J, Chazdon R, Ewers RM, Harvey CA, Peres CA, Sodhi NS (2009) Prospects for tropical forest biodiversity in a human-modified world. Ecol Lett 12:561–582
Geiselman CK, Mori SA, Blanchard F (2002) Database of neotropical bat/plant interactions. http://www.nybg.org/botany/tlobova/mori/batsplants/database/dbase_frameset.htm
Giannini NP, Kalko EKV (2004) Trophic structure in a large assemblage of phyllostomid bats in Panama. Oikos 105:209–220
Gorresen PM, Willig RM (2004) Landscape responses of bats to habitat fragmentation in Atlantic forest of Paraguay. J Mammal 85:688–697
Guerrero JA, De Luna E, Sánchez-Hernández C (2003) Morphometrics in the quantification of character state identity for the assessment of primary homology: an analysis of character variation of the genus Artibeus (Chiroptera: Phyllostomidae). Biol J Linn Soc 80:45–55
Hernández-Conrique D, Iñiguez-Dávalos LI, Storz JF (1997) Selective feeding by phyllostomid fruit bats in a subtropical montane cloud forest. Biotropica 29:376–379
Hernández-Montero JR (2009) Riqueza, abundancia, equidad y composición de plantas dispersadas por murciélagos filostómidos en cafetales bajo sombra y fragmentos de bosque mesófilo de montaña del centro de Veracruz. Dissertation, Universidad Veracruzana
Hodgkison R, Balding ST, Zubaid A, Kunz TH (2004) Temporal variation in the relative abundance of fruit bats (Megachiroptera: Pteropodidae) in relation to the availability of food in a lowland Malaysian Rain Forest. Biotropica 36:522–533
Hoffmann O (1993) Rumbos y paisajes de Xico, geografía de un municipio de la sierra Veracruzana. ORSTOM e Instituto de Ecología, A. C. Xalapa, Ver
Holl K (1999) Factors limiting tropical rain forest regeneration in abandoned pasture: seed rain, seed germination, microclimate, and soil. Biotropica 31:229–242
Kalko EKV (1999) Organization and diversity of tropical bat communities through space and time. Zoology 101:281–297
Klingbeil BT, Willig MR (2009) Guild-specific responses of bats to landscape composition and configuration in fragmented Amazonian rainforest. J Appl Ecol 46:203–213
Loayza AP, Loiselle AB (2008) Preliminary information on the home range and movement patterns of Sturnira lilium (Phyllostomidae) in a naturally fragmented landscape in Bolivia. Biotropica 40:630–635
Manson RH, Hernández-Ortiz V, Gallina S, Mehltreter K (eds) (2008a) Agroecosistemas Cafetaleros de Veracruz: Biodiversidad, manejo y conservación. Instituto de Ecología, A. C., (INECOL) e Instituto Nacional de Ecología (INE-SEMARNAT), Xalapa, Ver
Manson RH, Sosa VJ, Contreras A (2008b) Efectos del manejo sobre la biodiversidad: síntesis y conclusiones. In: Manson RH, Hernández-Ortiz V, Gallina S, Mehltreter K (eds) Agroecosistemas Cafetaleros de Veracruz: Biodiversidad, Manejo y Conservación. Instituto de Ecología, A. C., (INECOL) e Instituto Nacional de Ecología (INE-SEMARNAT), Xalapa, Ver
Medellín RA, Arita H, Sánchez O (1997) Identificación de los murciélagos de México: Clave de campo. Publicaciones Especiales. Asociación Mexicana de Mastozoología A. C., Mexico, DF
Medina A, Harvey CA, Merlo DS, Vilchez S, Hernández B (2007) Bat diversity and movement in an agricultural landscape in Matiguás, Nicaragua. Biotropica 39:120–128
Mello MAR, Kalko EKV, Silva WR (2008) Diet and abundance of the bat Sturnira lilium (Chiroptera) in a Brazilian montane Atlantic forest. J Mammal 89:485–492
Moguel P, Toledo VM (1999) Biodiversity conservation in traditional coffee systems of Mexico. Conserv Biol 13:11–21
Montagut-González R (1999) Factors that contributed to the expansion of cattle ranching in Veracruz, Mexico. Mex Stud 15:101–130
Muñiz-Castro MA, Williams-Linera G, Rey-Benayas JM (2006) Distance effect from cloud forest fragments on plant community structure in abandoned pastures in Veracruz, Mexico. J Trop Ecol 22:431–440
Muñoz-Romo M, Herrera EA, Kunz TH (2008) Roosting behavior and group stability of the big fruit-eating bat Artibeus lituratus (Chiroptera: Phyllostomidae). Mamm Biol 73:214–221
Muscarella R, Fleming TH (2007) The role of frugivorous bats in tropical forest succession. Biol Rev 82:573–590
Nestel D (1995) Coffee in Mexico: international market, agricultural landscape and ecology. Ecol Econ 15:165–178
Nowak R, Kunz TH, Pierson ED (1994) Walker’s bats of the World. JHU Press, Baltimore
Numa C, Verdú JR, Sánchez-Palomino P (2005) Phyllostomid bat diversity in a variegated coffee landscape. Biol Conserv 122:151–158
Ortega J, Castro-Arellano I (2001) Artibeus jamaicensis. Mamm Species 662:1–9
Philpott SM, Wayne JA, Armbrecth I, Bichier P, Diestch TV, Gordon C, Greenberg R, Perfecto I, Reynoso-Santos R, Soto-Pinto L, Tejeda-Cruz C, Williams-Linera G, Valenzuela J, Zolotoff JM (2008) Biodiversity loss in Latin American coffee landscapes: review of the evidence on ants, birds, and trees. Conserv Biol 22:1093–1105
Pineda E, Moreno C, Escobar F, Halffter G (2005) Frog, bat and dung beetle diversity in the cloud forest and coffee agroecosystems of Veracruz, Mexico. Conserv Biol 19:400–410
Pinto N, Keitt T (2008) Scale-dependent responses to forest cover displayed by frugivore bats. Oikos 117:1725–1731
Prugh LR, Hodgesb KE, Sinclairc ARE, Brasharesa JS (2008) Effect of habitat area and isolation on fragmented animal populations. PNAS 105:20770–20775
Ramírez-Pulido J, Arroyo-Cabrales J, Castro-Campillo A (2005) Estado actual y relación nomenclatural de los mamíferos terrestres de México. Acta Zoológica Mexicana (n.s.) 21:21–82
Redondo RAF, Brina LPS, Silva RF, Ditchfield AD, Santos FR (2008) Molecular systematics of the genus Artibeus (Chiroptera: Phyllostomidae). Mol Phylogenet Evol 49:44–58
Saldaña-Vázquez RA (2008) Comparación de la diversidad de murciélagos filostómidos en fragmentos de bosque mesófilo de montaña y cafetales de sombra, del centro de Veracruz. Dissertation, Instituto de Ecología, A. C
Schulze M, Seavy N, Whitacre D (2000) A comparison of the phyllostomid bat assemblages in undisturbed Neotropical forest and in forest fragments of a slash-and-burn farming mosaic in Petén, Guatemala. Biotropica 32:174–184
Sosa VJ, Hernández-Salazar E, Hernández-Conrique D, Castro-Luna AA (2008) Murciélagos. In: Manson R, Hernández-Ortiz V, Gallina S, Mehltreter K (eds) Agroecosistemas cafetaleros de Veracruz: Biodiversidad, manejo y conservación. Instituto de Ecología, A. C., (INECOL) e Instituto Nacional de Ecología (INE-SEMARNAT), Xalapa, Ver
Soto-Esparza M, Gómez-Columna M (1990) Atlas climático del municipio de Xalapa. Instituto de Ecología, A. C, Xalapa, Ver
Stockwell EF (2001) Morphology and flight manoeuvrability in New World leaf-nosed bats (Chiroptera: Phyllostomidae). J Zool 254:505–514
Straube FC, Bianconi G (2002) Sobre a grandeza e a unidade utilizada para estimar esforço de captura com utilização de redes de neblina. Chiroptera Neotrop 8:150–152
Williams-Linera G (2007) El bosque de niebla del centro de Veracruz: Ecología, historia y destino en tiempos de fragmentación y cambio climático. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad e Instituto de Ecología A.C, Xalapa, Ver
Willig MR, Presley SJ, Bloch CP, Hice CL, Yanoviak SP, Diaz MM, Chauca LA, Pacheco V, Weaver SC (2007) Phyllostomid bats of lowland Amazonia: effects of habitat alteration on abundance. Biotropica 39:737–746
Acknowledgements
We acknowledge scholarship support to RAS-V from Secretaría de Medio Ambiente y Recursos Naturales (BIOCAFE 2002-01-C01-00194) and Consejo Nacional de Ciencia y Tecnología (CONACyT) of Mexico. We are grateful to A. Tauro, A. A. Castro-Luna, V. Pérez, A. García, A. Albalat, I. Corrales, M. Márquez, and J. Galindo-González for their valuable help with fieldwork; A. C. Hernández-Rojas and C. Gallardo-Hernández for help in the identification of plants in the field; G. Murcia-Flores and C. M. Durán-Espinosa for help in the identification of plants in XAL-herbarium. We thank the owners of the coffee farms and forest fragments for allowing us working on their property. We received valuable comments and suggestions from Federico Villalobos, Erich Fisher and Luis F. Aguirre. Comments of two anonymous reviewers substantially improved the focus and organization of the paper. James Pearce and Keith MacMillan revised the English.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saldaña-Vázquez, R.A., Sosa, V.J., Hernández-Montero, J.R. et al. Abundance responses of frugivorous bats (Stenodermatinae) to coffee cultivation and selective logging practices in mountainous central Veracruz, Mexico. Biodivers Conserv 19, 2111–2124 (2010). https://doi.org/10.1007/s10531-010-9829-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-010-9829-6