Abstract
Invasive plants can affect native plants through competition or allelopathy, and researchers often use pot experiments as a tool to measure the strength of these interactions. Recently, such pot experiments provided inconsistent estimates of the impact and allelopathic potential of invasive knotweed, one of the world’s most successful plant invaders. We suspected that the inconsistencies may be explained by the use of different substrates in different experiments. To test this, we conducted an experiment in which knotweed competed pairwise with five common native European species in several different substrates: two compost-based potting substrates and two natural soils, with or without extra fertilizer added. To test for allelopathy, we added activated carbon to half of the pots. We found that knotweed was generally much more successful, and there was much more evidence for its allelopathy, when tested in artificial potting substrates than in natural soils. Furthermore, addition of extra fertilizer decreased the dominance of knotweed and changed patterns of allelopathy. The physicochemical properties of potting soil, such as lower bulk density, higher pore space, permeability and nitrogen content may better allow rhizomes to penetrate and/or allelochemicals to be produced and diffused. If artificial substrates generally exaggerate dominance and allelopathy also in other invasive plants, then many previous studies may have overestimated the potential impact of invaders, and the results of these experiments should be interpreted with caution. To avoid misleading results, experiments that test the competitive or allelopathic impact of invasive plants should be done with natural soils, preferably from the targeted habitats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Competition experiments and estimates of competition intensity are a cornerstone of classic plant ecology, and fundamental for understanding plant communities (Keddy 2001; MacDougall and Turkington 2004; Grime 2006). Indices of competition intensity are often used for predicting community structure, and they are generally based on competitor identity (Weigelt and Jolliffe 2003) but not on the environment in which competition takes place, i.e. they are species- but not environment-specific (but see Carlyle et al. 2010 for a counterexample). However, the environment of course plays a role in plant–plant interactions. Neighbour effects among plants can range from facilitation in harsh environments (e.g., Lortie and Callaway 2006; Wipf et al. 2006; Maestre et al. 2009) to intense competition in benign, resource-rich environments (Bertness and Callaway 1994; Craine 2005; Coomes et al. 2009). Oftentimes, pot experiments are used to estimate the strength of plant–plant interactions and to establish the dominance hierarchy of different species. Pot experiments provide better control than experiments in the field, but they can be plagued by artefacts caused by the limited pot volumes (de Kroon 2007) or unrealistic physicochemical properties of the substrate (Passioura 2006; Dalling et al. 2013). Taken together, previous research suggests that substrate should be taken into account when testing for plant competition with pot experiments.
The study of plant competition is particularly active in invasion biology. Invasive plants are plant species have been introduced by humans to new areas outside of their native range, and that have become extremely abundant and cause ecological and economic problems in their new ranges (Vila et al. 2011; Pyšek et al. 2012). Huge research efforts are spent to understand the impact and mechanisms of dominance of invasive plants. For instance, we now know that exotic plants which become invasive are often good competitors (Davidson et al. 2011; Dawson et al. 2012) and profit particularly strongly from increased resource availability (Blumenthal et al. 2009). Another mechanism of invader dominance can be allelopathy, the root exudation of organic compounds that harm neighbors, either directly, or indirectly by influencing soil microbes (Callaway and Aschehoug 2000; Wurst et al. 2010; Wardle et al. 2011). Furthermore, if exotics employ chemicals which are coevolutionarily novel to the native plants, allelopathy can be regarded as a “novel weapon” (Callaway and Ridenour 2004). However, the production of secondary compounds is costly (Cipollini et al. 2003; Karban 2011) and plants might be able to produce them only where sufficient resources are available. In conclusion, nutrient availability in soils can influence the impact of plant invaders both directly, through resource competition, as well as indirectly, by altering allelopathic potential.
Invasive knotweeds (Fallopia japonica, F. sachalinensis and their hybrid Fallopia × bohemica; Polygonaceae) are some of the most successful and most problematic current plant invaders (Williams et al. 2010). The species were introduced to Europe from East Asia at the end of the nineteenth century and have become very dominant plant invaders of the European and North American temperate zone (Bailey and Connolly 2000). They grow extremely vigorously, spread clonally through rhizomes, and often form monospecific stands that leave little room for native plants, and alternative ecosystems in many fundamental ways (Gerber et al. 2008; Dassonville et al. 2011).
Previous research first suggested that allelopathy might play a significant role in the competitive superiority of invasive knotweeds (Siemens and Blossey 2007; Murrell et al. 2011), but follow-up experiments (Parepa et al. 2012, 2013a, 2014) could not confirm these findings, and often found knotweed to be less dominant, in spite of similar experimental set-ups. A key difference between previous studies, however, was that some had used commercial potting substrates whereas others had used natural soils. Since soil characteristics are likely to affect allelopathic potential, we suspected that the inconsistent findings may have been caused by the use of different substrates. To test this, we conducted an experiment in which invasive knotweed competed with five different common native competitors, in a range of different substrates. Specifically, we asked to what degree estimates of knotweed dominance and allelopathy depended on the substrate used, and on the levels of nutrient availability.
Methods
Experiment
We conducted a competition experiment in which invasive hybrid knotweed (Fallopia × bohemica) competed pairwise with five native European forbs, Geranium robertianum, Geum urbanum, Silene dioica, Symphytum officinale and Urtica dioica, in four different main substrates: (1) a regular, compost-based potting soil (Florabella, Klasmann-Deilmann GmbH, Geeste, Germany), (2) a research substrate mix (15:6:2 compost:sand:vermiculite) which had also been used in the Murrell et al. (2011) study, (3) an agricultural field soil mixed 1:1 with sand, and (4) a natural soil collected from a typical, but so far uninvaded, riverside habitat in Switzerland. For two of the substrates, the research mix and the field soil, we created two different nutrient levels by adding a slow release fertilizer (Osmocote Pro 5–6 M, Everris International BV, Geldermalsen, Netherlands) at 5 g/L. Finally, we tested for allelopathy in each of these substrates by mixing it with analytical-grade activated carbon (Charcoal Activated powder extra pure, Merck KGA, Darmstadt, Germany) at a concentration of 20 mL/L. Altogether there were 12 different treatments (four substrates, two of them with two nutrient levels, and each with or without AC) that were tested with five different neighbor species, and each species × treatment combination replicated 10 times, for a total of 12 × 5 × 10 = 600 pots. The natural soil as well as the knotweed rhizomes were collected along the river Birs near Delémont, Switzerland. The seeds of the native plants came from a commercial supplier of wild-collected seeds (Rieger-Hofmann GmbH, Blaufelden-Raboldshausen, Germany). We filled 1-L pots with the substrate, and in each we planted one 8–10 cm piece of knotweed rhizome with two intact nodes. The native species were germinated and pre-cultivated in a greenhouse, and were planted into the pots as seedlings, one per pot, when the knotweed began to sprout from the rhizomes. Since there were some differences in plant size at the time of planing, we measured the length of the longest leaf of each native, as well as the total shoot length of each knotweed, just after planting. The pots were placed in a garden bed in the Botanical Garden of Bern, Switzerland, in a fully randomized order. After 3 months, we harvested the aboveground biomass of each plant individually, dried it at 70 °C for 72 h and weighed it.
Statistical analyses
To test for substrate effects on knotweed dominance and allelopathy, we analyzed the % native biomass of the total pot biomass, using a linear model in R (R Core Team 2014) that included the fixed effects of species identity (5 levels), substrate type (4 levels), fertilizer (2 levels), addition of activated carbon (2 levels), as well as their interactions. To account for differences in the initial sizes of competitors, we included the initial sizes of natives and knotweed as covariates. If the native-invader biomass ratio significantly increased or decreased with activated carbon addition, this indicated allelopathic effects of knotweed or native species, respectively. We used post hoc Tukey tests to determine, for each species and substrate, whether AC had a significant effect.
Results
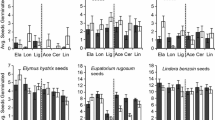
The competitive success of knotweed depended not only on its initial size, but also on the identity of the neighbour species and on the substrate it was competing in (significant main effects of species and substrate in Table 1). Out of the five native species, Geum and Symphytum were the strongest competitors and achieved around 50 % biomass in natural soils, whereas the other three species did not achieve more than 25 % in any of the substrates (Fig. 1). Across all neighbour species, knotweed was generally more successful in the two artificial potting substrates than in field soil or natural soil (Fig. 1). The competitive hierarchy of the natives changed with substrate (significant species × substrate interaction in Table 1). In natural soils, Geum and Symphytum were most successful, however their performance was strongly reduced in the artificial potting substrates. Urtica, in contrast, was little affected by substrate and was the most successful competitor in the compost-based substrate (Fig. 1).
Competitive balances between invasive knotweed (Fallopia × bohemica; dark bars) and five different native species (light bars) when grown in four different substrates, two with or without NPK fertilizer added. Arrows indicate cases where addition of activated carbon significantly altered the balance between native and invader, with dark arrows indicating knotweed allelopathy, and light arrows indicating allelopathic effects of natives
Overall, adding extra nutrients tended to be benefit the natives, but the effects were species- and substrate dependent (significant fertilizer [substrate] × species interaction in Table 1). In the artificial potting substrate, adding nutrients had a strong positive effect on Silene, whereas it had only minor or even negative effects on the other species. In the field soil, nutrient addition increased the success of Geranium and, to a lesser extent, Geum and Urtica, but it had little effect on the other two species.
Addition of activated carbon to the substrate often significantly affected knotweed success, but the direction and magnitude of the effect depended on substrate and fertilization, and the native species tested (significant AC interactions in Table 1). In eight out of the 30 species by substrate combinations (black arrows in Fig. 1), the addition of activated carbon significantly decreased the dominance of knotweed, which indicates knotweed allelopathy. Seven of these eight cases of allelopathy were observed in artificial potting substrates, whereas there was only one case in field soil and none in natural soil.
All native species suffered from knotweed allelopathy, but in different substrates: Geranium was affected in all artificial substrates, Silene in all unfertilized artificial substrates, and Symphytum and Urtica only in the fertilized Research Mix. In contrast, Geum was affected only in field soil. Across all native species, the strongest and most frequent allelopathic effects occurred in the fertilized Research Mix, with up to three-fold increases of native plant biomass after addition of AC (Fig. 1).
In five out of 30 species by substrate combinations, addition of activated carbon significantly decreased the dominance of knotweed (grey arrows in Fig. 1), which indicated that under certain environmental conditions, native species could also exert allelopathic effects on knotweed.
Discussion
Experimental tests of plant competition and allelopathy are important for understanding plant communities, and for estimating the ecological impact of invasive plants. However, estimates of plant–plant interactions may depend on the environment in which they are obtained. Here, we find that the dominance of exotic invasive knotweed over native plant species is generally much higher, and there is more evidence for knotweed allelopathy, when tested in artificial potting substrates rather than natural soils, which suggests that experiments with artificial substrates may strongly overestimate the potential impact of invasive plants.
Knotweed dominance was up to three times higher in artificial substrates than in natural substrates. One possible explanation for the different outcomes of plant–plant interactions in different substrates is that they may differ in their soil biota. Soil biota appear to play an important role not only in knotweed invasion (Parepa et al. 2013a), but also in the invasion success of several other introduced plants (e.g., Mangla and Callaway 2008; Scharfy et al. 2010). Exotic plants may experience more positive plant-soil interactions either because they encounter less antagonists than the native plants (Klironomos 2002) or because they benefit more from mutualistic soil biota (Reinhart and Callaway 2004; Sun and He 2010). However, soil biota could explain our results only if the frequency of antagonists of knotweed, but not those of natives plants, is higher in natural soils, or if the antagonists of natives are more frequent in artificial soils. Alternatively, knotweed could encounter more mutualists in artificial substrates. However, none of these scenarios sound very likely.
Another explanation for increased knotweed dominance in artificial substrates could be that differences in their physicochemical properties facilitate knotweed invasion. We can rule out nutrient availability as mechanism, since nutrient addition rather decreased knotweed success. Thus, other substrate properties must be responsible. For instance, compared to natural soils, artificial potting substrates usually have a much improved aeration and water retention, and knotweed may be better able to take advantage of these favorable conditions (see also Parepa et al. 2013b). Artificial substrates usually also have more favorable mechanical properties. They have lower bulk density and higher pore space and permeability, which may greatly facilitate the shoot emergence of knotweed, as well as the penetration of the soil by its rhizomes.
We used activated carbon to test the degree to which allelopathy plays a role in the interactions between invasive knotweed and native plants. We found that the effects of adding activated carbon strongly depended on the substrate, and we interpret this as substrate-dependent allelopathy. However, there are some potential problems with the use of activated carbon: First, some types of activated carbon may add significant amounts of nutrients to the substrate (Lau et al. 2008). Second, it has been shown that activated carbon can affect plant–microbe interactions such as mycorrhization, presumably through interfering with the signaling between plants and their associated microbes, even in the absence of interspecific competitors (e.g., Wurst et al. 2010). Both mechanisms could thus alter plant growth without any allelopathy between species. While it is unlikely that nutrient addition played a role in our study, as the activated carbon we used was of high purity, we cannot rule this out, nor the direct effects of activated carbon, as observed by Wurst et al. (2010).
If we assume that the observed effects of activated carbon indicate allelopathy, then there was much more evidence for allelopathy in the artificial substrates, but hardly any in the natural substrates. These results confirm and at the same time reconcile the contradictory findings from previous experiments where knotweed had first been found to exert strong allelopathic effects in potting substrate (Murrell et al. 2011), but then very little evidence for allelopathy had been found in experiments with field soil (Parepa et al. 2012, 2013a, 2014). Our experiment shows that these contradictory findings are likely due to the use of the different substrates, and it confirms the results of another recent study that could not verify allelopathic effects of alleged allelopathic plants invaders under field conditions (Del Fabbro et al. 2014). Our experiment does not allow us to identify the pathways of allelopathy, whether it is acting directly (Kabouw et al. 2010) or indirectly through soil biota (Weisshuhn and Prati 2009; Wurst et al. 2010). It also cannot tell us the mechanism by which allelopathy is reduced in natural soils, but it is conceivable that the clay particles and minerals in natural soils have a greater capacity to adsorb allelochemicals (Dao 1987), or that soil organisms decompose a fraction of the allelopathic compounds before they reach their target (Kaur et al. 2009). In any case, our study suggests that allelopathy tests with artificial substrates overestimate allelopathic potential of invasive plants. The decreasing support for allelopathy and novel weapons of invasive plants (Jeschke et al. 2012) may be the result of a trend towards more realistic allelopathy studies during the past few years.
Interestingly, we found that the evidence for allelopathy was strongest when the artificial substrates were additionally fertilized. This finding is consistent with the idea that allelochemicals—just as herbivore defense (Herms and Mattson 1992; Karban 2011)—are metabolically costly, and that there exists a trade-off between resource allocation to growth versus allelopathy (see also Rasher and Hay 2014). Our results are in line with Kovarova et al. (2010, 2011) who also found that the production of secondary compounds in knotweed depends on nutrient supply. Most likely, invasive knotweed can “afford” to be more allelopathic when resources are abundant, and this may contribute to their superiority in nutrient-rich environments.
Conclusions
Our experiment demonstrates that experimental estimates of the dominance and allelopathic effects of an invasive plant can strongly depend on the substrate in which the plants are tested. We found that invasive knotweed was generally more dominant over native plants, and there was more evidence for its allelopathic potential, when tested in artificial substrates, particularly under high-nutrient conditions, than when tested in natural soils. Thus, while invasive knotweed appears to possess the ability to exert allelopathic interference, this ability may often not show in the real world because unfavorable soil characteristics limit the action of allelochemicals, or a lack of resource prevents their production.
Pot experiments such as the one in our study are frequently used to estimate the potential impact of invasive plants, and it is not uncommon to test competitive ability and impact of invasive plants. If artificial potting substrates generally exaggerate dominance and allelopathy also in other invasive plants, then many previous studies may have overestimated the potential impact of invaders, and the results of these experiments should be interpreted with caution. To avoid misleading results, experiments that test the competitive or allelopathic impact of invasive plants should be done with natural soils, preferably from the targeted habitats.
References
Bailey JP, Connolly AP (2000) Prize winners to pariahs: a history of Japanese knotweed s.l. Polygonaceae in the Bristish Isles. Watsonia 23:93–110
Bertness MD, Callaway R (1994) Positive interactions in communities. Trends Ecol Evol 9:191–193
Blumenthal D, Mitchell CE, Pyšek P, Jarosik V (2009) Synergy between pathogen release and resource availability in plant invasion. PNAS 106:7899–7904
Callaway RM, Aschehoug ET (2000) Invasive plants versus their new and old neighbors: a mechanism for exotic invasion. Science 290:521–523
Callaway RM, Ridenour WM (2004) Novel weapons: invasive success and the evolution of increased competitive ability. Front Ecol Environ 2:436–443
Carlyle CN, Fraser LH, Turkington R (2010) Using three pairs of competitive indices to test for changes in plant competition under different resource and disturbance levels. J Veg Sci 21:1025–1034
Cipollini D, Purrington CB, Bergelson J (2003) Costs of induced responses in plants. Basic Appl Ecol 4(1):79–89
Coomes DA, Kunstler G, Canham CD, Wright E (2009) A greater range of shade-tolerance niches in nutrient-rich forests: an explanation for positive richness-productivity relationships? J Ecol 97:705–717
Craine JM (2005) Reconciling plant strategy theories of Grime and Tilman. J Ecol 93:1041–1052
Dalling JW, Winter K, Andersen KM, Turner BL (2013) Artefacts of the pot environment on soil nutrient availability: implications for the interpretation of ecological studies. Plant Ecol 214:329–338
Dao TH (1987) Sorption and mineralization of plant phenolic-acids in soil. ACS Symp Ser 330:358–370
Dassonville N, Guillaumaud N, Piola F, Meerts P, Poly F (2011) Niche construction by the invasive Asian knotweeds (species complex Fallopia): impact on activity, abundance and community structure of denitrifiers and nitrifiers. Biol Invasions 13:1115–1133
Davidson AM, Jennions M, Nicotra AB (2011) Do invasive species show higher phenotypic plasticity than native species and if so, is it adaptive? A meta-analysis. Ecol Lett 14:419–431
Dawson W, Rohr RP, van Kleunen M, Fischer M (2012) Alien plant species with a wider global distribution are better able to capitalize on increased resource availability. New Phytol 194:859–867
de Kroon H (2007) Ecology-How do roots interact? Science 318:1562–1563
Del Fabbro C, Guesewell S, Prati D (2014) Allelopathic effects of three plant invaders on germination of native species: a field study. Biol Invasions 16:1035–1042
Gerber E, Krebs C, Murrell C, Moretti M, Rocklin R, Schaffner U (2008) Exotic invasive knotweeds (Fallopia spp.) negatively affect native plant and invertebrate assemblages in European riparian habitats. Biol Conserv 141:646–654
Grime JP (2006) Trait convergence and trait divergence in herbaceous plant communities: mechanisms and consequences. J Veg Sci 17:255–260
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:283–335
Jeschke JM, Aparicio LM, Haider S, Heger T, Lortie CJ, Pyšek P, Strayer DL (2012) Support for major hypotheses in invasion biology is uneven and declining. NeoBiota 14:1–20
Kabouw P, Nab M, van Dam NM (2010) Activated carbon addition affects substrate pH and germination of six plant species. Soil Biol Biochem 42:1165–1167
Karban R (2011) The ecology and evolution of induced resistance against herbivores. Funct Ecol 25:339–347
Kaur H, Kaur R, Kaur S, Baldwin IT (2009) Taking ecological function seriously: soil microbial communities can obviate allelopathic effects of released metabolites. PLoS ONE 4:e4700
Keddy PA (2001) Competition. Kluwer, Dordrecht
Klironomos JN (2002) Feedback with soil biota contributes to plant rarity and invasiveness in communities. Nature 417:67–70
Kovarova M, Bartunkova K, Frantik T, Koblihova H, Prchlova K, Vosatka M (2010) Factors influencing the production of stilbenes by the knotweed, Reynoutria × bohemica. BMC Plant Biol 10:19
Kovarova M, Frantik T, Koblihova H, Bartunkova K, Nyvltova Z, Vosatka M (2011) Effect of clone selection, nitrogen supply, leaf damage and mycorrhizal fungi on stilbene and emodin production in knotweed. BMC Plant Biol 11:14
Lau JA, Puliafico KP, Kopshever JA, Steltzer H, Jarvis EP, Schwarzlander M, Strauss SY, Hufbauer RA (2008) Inference of allelopathy is complicated by effects of activated carbon on plant growth. New Phytol 178:412–423
Lortie CJ, Callaway RM (2006) Re-analysis of meta-analysis: support for the stress-gradient hypothesis. J Ecol 94:7–16
MacDougall AS, Turkington R (2004) Relative importance of suppression-based and tolerance-based competition in an invaded oak savanna. J Ecol 92:422–434
Maestre FT, Callaway RM, Valladares F, Lortie CJ (2009) Refining the stress-gradient hypothesis for competition and facilitation in plant communities. J Ecol 97:199–205
Mangla S, Callaway RM (2008) Exotic invasive plant accumulates native soil pathogens which inhibit native plants. J Ecol 96:58–67
Murrell C, Gerber E, Krebs C, Parepa M, Schaffner U, Bossdorf O (2011) Invasive knotweed afects native plants through allelopathy. Am J Bot 98:38–43
Parepa M, Schaffner U, Bossdorf O (2012) Sources and modes of action of invasive knotweed allelopathy: the effects of leaf litter and trained soil on the germination and growth of native plants. NeoBiota 13:15–30
Parepa M, Fischer M, Bossdorf O (2013a) Environmental variability promotes plant invasion. Nat Commun 4:1604
Parepa M, Schaffner U, Bossdorf O (2013b) Help from under ground: soil biota facilitate knotweed invasion. Ecosphere 4:31
Parepa M, Fischer M, Krebs C, Bossdorf O (2014) Hybridization increases invasive knotweed success. Evolut Appl 7:413–420
Passioura JB (2006) The perils of pot experiments. Funct Plant Biol 33:1075–1079
Pyšek P, Jarosik V, Hulme PE, Pergl J, Hejda M, Schaffner U, Vila M (2012) A global assessment of invasive plant impacts on resident species, communities and ecosystems: the interaction of impact measures, invading species’ traits and environment. Glob Change Biol 18:1725–1737
Rasher DB, Hay ME (2014) Competition induces allelopathy but suppresses growth and anti-herbivore defence in a chemically rich seaweed. Proc R Soc B 281:20132615
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Reinhart KO, Callaway RM (2004) Soil biota facilitate exotic Acer invasions in Europe and North America. Ecol Appl 14:1737–1745
Scharfy D, Guesewell S, Gessner MO, Venterink HO (2010) Invasion of Solidago gigantea in contrasting experimental plant communities: effects on soil microbes, nutrients and plant-soil feedbacks. J Ecol 98:1379–1388
Siemens TJ, Blossey B (2007) An evaluation of mechanisms preventing growth and survival of two native species in invasive bohemian knotweed (Fallopia × bohemica, Polygonaceae). Am J Bot 94:776–783
Sun ZK, He WM (2010) Evidence for enhanced mutualism hypothesis: Solidago canadensis plants from regular soils perform better. PLoS ONE 5:e15418
Vila M, Espinar JL, Hejda M, Hulme PE, Jarosik V, Maron JL, Pergl J, Schaffner U, Sun Y, Pyšek P (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708
Wardle DA, Karban R, Callaway RM (2011) The ecosystem and evolutionary contexts of allelopathy. Trends Ecol Evol 26:655–662
Weigelt A, Jolliffe P (2003) Indices of plant competition. J Ecol 91:707–720
Weisshuhn K, Prati D (2009) Activated carbon may have undesired side effects for testing allelopathy in invasive plants. Basic Appl Ecol 10:500–507
Williams F, Eschen R, Harris A, Djeddour D, Pratt C, Shaw RS, Varia S, Lamontagne-Godwin J, Thomas SE, Murphy ST (2010) The economic cost of invasive non-native species on Great Britain. CABI Publishing, Wallingford
Wipf S, Rixen C, Mulder CPH (2006) Advanced snowmelt causes shift towards positive neighbour interactions in a subarctic tundra community. Glob Change Biol 12:1496–1506
Wurst S, Vender V, Rillig MC (2010) Testing for allelopathic effects in plant competition: does activated carbon disrupt plant symbioses? Plant Ecol 211:19–26
Acknowledgments
We thank Steffen Boch for helpful comments on a previous version of the manuscript. This study was supported by the Swiss National Science Foundation (Project 31003A_122408).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Parepa, M., Bossdorf, O. Testing for allelopathy in invasive plants: it all depends on the substrate!. Biol Invasions 18, 2975–2982 (2016). https://doi.org/10.1007/s10530-016-1189-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-016-1189-z