Abstract
As recently as 2009 the number of introductions recorded for South Africa comprised 22 marine and estuarine species. This review aims to reassess the diversity and scale of introduced marine and estuarine species in the region. Accurate taxonomic and systematic work, broad review of historical records and new sampling surveys across selected marine habitats conducted by a team of local and international experts has effectively revealed the presence of previously misidentified, overlooked, or new introductions. A total of 86 introduced and 39 cryptogenic species are recognized, increasing known numbers four and twofold respectively within 1 year, although the current assessment is far from fully comprehensive. Additional species were revealed within the historic literature (76%), from surveys conducted post-2005 (11%) and following taxonomic resolution (13%). Temporal analyses confirmed discovery rates were increasing over time. Ship fouling and ballast water were the dominant vector pathways, accounting for 48 and 38% respectively. Spatial analyses revealed patterns of bioinvasion to be significantly higher on the west coast compared to the other coastal regions. Overall, 53% of introductions were concentrated within harbour areas with only 4 open-coast invaders detected at present. Introduced species found in the cool and warm-temperate provinces of the west and south coast mainly originated from the northern hemisphere (65%). In contrast, introductions located in the sub-tropical and tropical provinces of the east coast mainly originated from the southern hemisphere (18%), with the remaining 17% of introduced species being of unknown origin. The research approach described has proven pivotal, contributing massively toward revealing the true scale and patterns of bioinvasion for a developing region within a relatively short period of time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The frequency of human-mediated marine introductions is increasing globally and their role and importance as agents of global change is becoming ever more apparent (Ruiz et al. 1997; Sala et al. 2000; Wonham and Carlton 2005). As a result, considerable resources have been directed into research on this topic. However, a few regions, such as Europe, North America, New Zealand, and Australia dominate the literature in terms of reporting the presence and impacts of marine introduced species at a variety of spatial scales (Byers 2009; Carlton 1996; Cranfield et al. 1998; Leppakoski and Olenin 2000; Levings et al. 2002; Hewitt et al. 2004; Ruiz et al. 1997, 1999, 2000; Sala et al. 2000). In part this domination is due to the greater availability of financial, scientific and in particular taxonomic resources in these regions.
Obtaining a realistic ‘fix’ on the number of marine introductions within a region can be a challenging task (Carlton 2009). However, it has been attempted across a number of regions (Carlton 1987; Castilla and Neill 2009; Cohen and Carlton 1998; DeFelice et al. 2001; Ferreira et al. 2009; Galil and Zenetos 2002; Gollasch and Nehring 2006; Hewitt et al. 2004; Leppakoski et al. 2009; Orensanz et al. 2002; Ruiz et al. 2000; Seo and Lee 2009). Ruiz et al. (2000) recorded 298 marine and estuarine introduced species in North America, a comparatively well-known region. However, the authors suggest that the real number of introductions could easily be 600–900 species and thus the reported figure should not be interpreted as accurate and final. Coles et al. (1999) reported 101 introduced species from Pearl Harbour on Oahu Island, Hawaii. Ten years later, Carlton and Eldredge (2009) report nearly twice that number for the same region, based almost entirely on retrospective historical analysis and greater taxonomic resolution, rather than on new introductions that had occurred post 1999. Thus even in well-studied areas, the diversity of introduced species is rarely adequately resolved.
The situation is of even greater concern in regions of the world where the state of knowledge of the marine biota remains relatively poor, or where there has been less historical interest in invasions (Nuñez and Pauchard 2010), as a result of several confounding reasons. These include (i) systematic and taxonomic challenges, (ii) access issues in order to undertake new sampling surveys across a variety of marine habitats and (iii) availability of historic records (Carlton 1996, 2003, 2009; Wonham and Carlton 2005). Denmark, Japan, Uruguay/Argentina, the Azores and Chile have recently reported totals of 18, 25, 31, 33 and 51 marine introductions respectively (Castilla and Neill 2009; Hewitt et al. 2004; Jensen and Knudsen 2005; Orensanz et al. 2002; Otani 2004). Based on the aforementioned reasons, Carlton (2009) suggests that the actual number of introductions in these regions is probably 5–10 times the number reported. In addition, many regions fail to report cryptogenic species (Carlton 1996, 2009). Cryptogenics represent ‘red lights’ as they emphasize the potential depth and breadth of a region’s cryptic introduction history. It is critical to call attention to the many species that have been assumed to be native without compelling evidence otherwise substantial underestimates will be made when determining the potential scale of regional and global invasions (Carlton 1996, 2009).
There are regions where the number of reported bioinvasions is sufficiently high to facilitate a spatio-temporal bioinvasive patterns analysis. To date, these have been conducted at both local and pan-regional scales for marine species (Carlton 2003; Castilla and Neill 2009; Fofonoff et al. 2009; Hayden et al. 2009; Rilov and Galil 2009; Sliwa et al. 2009; Wonham and Carlton 2005). Vector types, dispersal pathways, source regions, taxonomic composition, spatial distribution, receiving habitat types, measured impacts and rates of successful introduction have all been investigated (Carlton 2003, 2009; Hewitt et al. 2009; Miller and Ruiz 2009; Minchin et al. 2009; Ruiz and Hewitt 2002; Wilson et al. 2009; Wonham and Carlton 2005). These analyses are an important step toward understanding the patterns and processes behind successful introductions (Byers 2009; Carlton 1996; Lonhart 2009; Occhipinti-Ambrogi 2007; Olyarnik et al. 2009; Ruiz and Hewitt 2002). Moreover, cross-regional comparisons will facilitate bioinvasion predictions on a global scale and the formulation of effective management policies (Bax et al. 2004; Carlton 1996, 2009; Kolar and Lodge 2001). However, it is recognized that sound interpretation is dependent on the quality of baseline knowledge available and that, in the interim, assessing spatio-temporal patterns using the uneven data available needs to be undertaken with caution (Ruiz and Hewitt 2002).
In South Africa, there has been limited long-term historical focus on bioinvasions within the marine environment. Although there have been several ecological studies on conspicuous invasions, such as the western European crab Carcinus maenas (Hampton and Griffiths 2007; Le Roux et al. 1990) and the Mediterranean mussel Mytilus galloprovincialis (Bownes and McQuaid 2009; Branch and Steffani 2004; Branch et al. 2010; Rius and McQuaid 2009), far less work has been invested in resolving the potential scale of invasions that may have occurred over the past several centuries. Several progressive publications over the past two decades have offered estimates of numbers for both introduced and cryptogenic species (Griffiths 2000; Griffiths et al. 1992; Robinson et al. 2005) the most recent giving estimated numbers of 22 and 18 species respectively (Griffiths et al. 2009). However, it is suspected, a priori, that these numbers are substantial underestimates and that a more thorough investigation would reveal a far greater scale of bioinvasions within the region, which would in turn facilitate an assessment of bioinvasion patterns.
South Africa provides a clear example of a region where confounding factors have hindered progress when assessing the diversity of marine bioinvaders. One major hurdle is a lack of pre-invasion information. Given that the region lies along major shipping routes, a well documented vector of marine bioinvasions (Carlton and Hodder 1995; Carlton 2003; Carlton and Cohen 2003; Occhipinti-Ambrogi and Savini 2003; Wonham et al. 2000; Wonham and Carlton 2005) it is virtually certain that marine introductions consistently took place over the first 400 years of European colonial history. Whereas by 1699 there were 46 exotic plants already established in South Africa (Wells et al. 1986), no formal research endeavours on the introduced marine fauna and flora extend to these early periods.
Besides historical data gaps, Robinson et al. (2005) note that large areas of the South African coastline remain unexplored in terms of marine introductions. Similarly, not all coastal habitats have been investigated equally, if at all. In addition, there are few marine taxonomists available in South Africa to make authoritative identifications, even of native species. Additional taxonomic complications further confound interpretation of faunal and floral history. For example, species carried around the world by ships were frequently described over and over again as regional endemics in their areas of introduction, leading to individual introduced species “hiding” around the world under many different names (Carlton 2009).
This body of research aims to rectify the situation by, firstly applying a wider range of investigatory approaches in order to reveal a far greater diversity of introduced species than previously recognized within the region. Secondly, a preliminary spatio-temporal assessment of the extent of South African patterns of bioinvasion will be presented.
Materials and methods
Re-assessment of the South African bioinvasion inventory
A combination of approaches were employed to examine in detail the potential biogeographic histories and affinities of marine and estuarine taxa. The previous work of Griffiths et al. (2009) was expanded to capture additional nineteenth century literature (peer reviewed publications and monographs) and grey literature (government reports and maritime records) previously overlooked. Grey literature sources were used judiciously and only when the levels of expertise and scholarship could be adequately assessed. Voucher specimens and unpublished species records stored within the Iziko South African Museum Marine Collection, Cape Town, were examined.
Comparative invasion biogeography
Firstly, we identified marine and estuarine species that were recognized as bioinvasions elsewhere, but had not yet been recognized as introduced to South Africa using the aforementioned historic literature and through cross-checking lists of South African marine species with lists of species considered to be introduced in other climatically comparable regions. This included regions at similar latitudes in the southern hemisphere, for example Australia, New Zealand and South America, but also in northern latitudes, such as North America and Europe. The question was posed as to whether selected taxa recognized in South Africa, whose systematic status appeared to be reasonably resolved, had highly disjunct global distributions. It should be noted that large suites of microscopic species (protozoans, nematodes, rotifers, diatoms, dinoflagellates) do not lend themselves to this approach, as their global status has largely been defined based upon the morphospecies concept, rather than genetic verification. Secondly, South African species with questionable systematic status were investigated and evidence as to their modern systematic status derived from re-examination of voucher specimens as well as paleontological, archeological, historic, biogeographic, genetic and systematic studies (taxonomic resolution). The criteria of Carlton (1996), Chapman (1988) and Chapman and Carlton (1991) were applied to determine if species identified should be allocated introduced or cryptogenic status.
Under-considered habitats and rapid assessment surveys
Numerous introductions are found in habitats that are rarely or insufficiently explored (Carlton 2009). Through rapid assessment surveys (field surveys), the aim was to reveal species that were recognizable as bioinvasions in under-considered habitats. Sampling was conducted in September 2008 at locations on the southern and western coasts of South Africa. These were (i) Table Bay Harbour, Cape Town, (ii) Milnerton Lagoon, Table Bay, (iii) Langebaan Lagoon, Saldanha Bay and (iv) Zandvlei Lagoon, False Bay, Cape Town. For new species records, the date of first collection (DOC) was recorded as the sampling date.
Fouling and wood-boring communities were sampled in Table Bay Harbour (Cape Town). The emphasis was placed on fauna associated with gribble (Limnoria)-infested wood. Peices of wood were removed from wooden structures within the harbour and taken to the laboratory. At Milnerton Lagoon, Table Bay, quadrats were placed randomly along the strandline habitat located in the supralittoral zone. All debris (inclusive of decomposing kelp) and samples of sand from within the quadrats were removed and returned to the laboratory. At Langebaan Lagoon, Saldanha Bay, quadrats were placed randomly along sandy beach and marsh habitats. Organic material and samples of sand/mud from within the quadrats were removed and returned to the laboratory. At Zandvlei Lagoon, False Bay, Cape Town, the emphasis was on the tubeworm Ficopomatus enigmaticus and the associated biota found within the reefs formed by the tubeworm. Several sections of reef were removed from within the water and returned to the laboratory. Organisms across all size ranges were extracted from all samples and identified through examination under microscopes. Voucher specimens were preserved in 70% ethanol for long-term archival purposes and where necessary, for distribution to systematists.
Species categorization
The date of first collection (DOC) or date of publication (DOP) when DOC was not available was derived from several sources, inclusive of peer reviewed literature, museum collections, and field sampling post-2008. Native regions (regions of origin) were determined from highly specialized taxonomic literature and the biogeographic patterns of sister taxa. For cosmopolitan species, the origin was classified as ‘unknown’. Transport vector (vector pathway) assignment was based on both life history knowledge and the date/site of introduction as extracted from peer reviewed literature records. Analyses were based on subsets of species with the exclusion of cryptogenic species, depending on available data for each parameter. Introduced species counts were analyzed for temporal trends and distributional patterns for regions of origin, taxonomic groups, invasion pathways, vector and habitat type.
Temporal patterns analyses
Temporal analyses are currently confounded by a lack of ability to accurately assess rate of introduction. At this stage, the majority of South African bioinvasions are linked to the DOC rather than date of actual introduction, which may in fact be decades or centuries before. Therefore, using DOC’s, species counts were regressed against collection year since 1840 (this being the earliest collection decade known) in order to establish the rate of discovery. Discovery rate was tested using a least squares linear fit regression (y = α + βx) where β is the rate of change of the rate of discovery. The null hypothesis, which assumes a constant rate of discovery over the 170 year period, will be rejected if P < 0.05. If β < 0, this indicates the rate is increasing over the 170 year period. In addition, the mean number of introduced species collected per decade was calculated across 30 year periods, in order to compare discovery rates during periods where introductions were collected as a bi-product of maritime surveys or indigenous biodiversity surveys (1840–1990) relative to where introductions were collected as a product of focused bioinvasive research (1990–2010).
Spatial patterns analysis
Distribution ranges were not inferred from single point location records, in order not to assume that a species occurs between two known points, such as two ports (Mead et al. 2011). The coastline of South Africa can be divided into separate biogeographical regions with transition zones in-between (Fig. 1). The bioregions are defined based on climatic and oceanographic regimes combined with known species assemblages present, summaries of which are found in several sources but in this case are adapted from Lombard (2004). To test if regional distribution of introduced species was dependent on region of origin, vector pathway, habitat or species number (and an ∝ value of 0.05) species count data were arranged into contingency tables and the Chi-squared statistic applied (Zar 2004).
Species uniqueness per biogeographic region or transition zone was determined. The number of species shared between bioregions and those that were unique to a specific bioregion were identified and tallied. The Jaccard Similarity Index was used to identify similarity between regions (%) based on the numbers of shared and unique species present within a bioregion. The following formula was applied: S AB = A n B/A u B, where S = Similarity, A and B represent bioregions or transition zones being compared, A n B represents the total number of unique species found across both region A and B and A u B represents the total number of shared species found in both region A and B.
Results
Bioinvasion inventory
In total, 86 introduced and 39 cryptogenic marine and estuarine species were resolved for South Africa (Table 1). This is a major re-assessment of the previous inventory and expands substantially on the 22 and 18 species reported previously (Griffiths et al. 2009). Of the 64 species added as introductions, 76% were sourced from within the historic literature, 11% were identified following field surveys conducted post-2008 and 13% were revealed following taxonomic resolution. Of the 21 cryptogenic species newly resolved, 64% were sourced from within the historic literature, 14% were identified following field surveys conducted post-2008 and 22% were revealed following taxonomic resolution (Fig. 2). All taxa were identified to species level and Mead et al. (2011) provides the species-by-species treatment of the introduced and cryptogenic biota in a monographic format.
No fewer than 14 of the species in Table 1 were originally mistakenly re-described as new endemic species after they had arrived in South Africa (Table 2). Five species in the South African marine fauna were retained as introduced, although they were last collected over 50 years ago (Table 3) because they are either in locations or in habitats that have not been thoroughly re-explored for that specific species since the original record. Two additional species are recognized as introductions, but to date are only present within closed aquaculture facilities (Table 1). One bryozoan species, Membranipora membranacea, was removed from previous lists of introduced species (Griffiths et al. 2009; Robinson et al. 2005), as it is now recognized as a previously undescribed inidigenous species, M. rustica (Florence et al. 2008).
Rate of discovery
This analysis was based on 71 of the possible 86 species records as date of first collection was not determined for 15 species. As the number of species records accumulated over the past 170 years (Fig. 3a), the rate of discovery has increased over time (P = 0.25; confidence range of β = 0.007–0.1; R 2 = 0.27; Fig. 3b). Species collection rates were lower in periods where introductions were collected as bi-products of maritime or indigenous biodiversity surveys (1840–1990), compared to focused bioinvasive research (1990–2010). During the maritime and biodiversity surveys, the average species addition rate between 1840 and 1940 was 1.9 species per decade, rising to 3.2 species per decade between 1940 and 1990 (Fig. 3). During the period of focused invasive research, between 1990 and 2010, the average species addition rate was 17 species per decade (Fig. 3). Of the total known number of marine introductions to date (86 species), Robinson et al. (2005) revealed 11.7%, Griffiths et al. (2009) revealed 14.1% and the current re-assessment revealed 74.2%.
Biogeography
This analysis was based on 80 of the possible 86 introductions, as distributional records were not determined for 4 species and 2 species were located within aquaculture facilities. The highest numbers of introductions (55 species) were reported from the cool-temperate region (Fig. 1), hereafter refered to as the west coast. The lowest numbers of introductions (15 species) were reported from the tropical region (Fig. 1), hereafter refered to as the north-east coast. According to Chi-square analysis, indigenous regions, invasion pathways, habitat type, taxonomic groupings and shared or unique species differed significantly among regions (P < 0.001; Table 4). The west coast has the highest proportion of introductions that are unique to the region (42%; 55 species), followed by the sub-tropical region (Fig. 1), hereafter refered to as the east coast (26%; 31 species). The transition zones (Fig. 1) located in between bioregions (False Bay and East London) have the highest proportion of species shared across regions, with 94% (35 species) and 100% (21 species) respectively (Fig. 4). The Jaccard Similarity Index revealed that there were dissimilarities between regions based on respective shared and unique species compositions. The west coast shared the highest similarities (40–60%) with all other biogeographic regions and transition zones. The transition zones were most dissimilar to each other (96%).
Number of species with a unique or shared distribution for known South African marine introductions. Note: East London and False Bay are transitional zones between bioregions (refer to Fig. 1)
Regions of origin and vector pathways
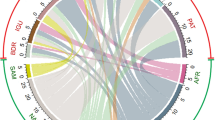
The region of origin analysis was based on 70 of the possible 86 introductions, as origins were not determined for 16 species and therefore classed as ‘unknown’. The majority of introduced species were native to the northern hemisphere (65%) although 18% originated from the southern hemisphere (Fig. 5). Northern hemisphere species have established themselves on the cool-temperate west coast and warm-temperate south-east coast (Fig. 1), plus the transition zones either side. Conversely, the majority of southern hemisphere species have established all along the warmer east coast.
The vector pathway analysis was based on all of the possible 86 introductions. An overwhelming proportion of species (94%) were introduced unintentionally with a very small percentage (6%) being imported intentionally or through mariculture practice (Fig. 6). Ship fouling (48%) and ballast water (38%) are the commonest vectors of marine introduced species to the South African coast (86%) with 51% of species potentially arriving at the South African coastline through multiple vectors. The west coast and adjacent transitional zone (False Bay) had introduced species vectored by all the pathways listed, inclusive of introduction through oil rigs. Ship boring species had arrived predominantly on the north-east coast, as well as the west coast and False Bay, although in relatively low numbers compared to the other listed vectors. This is probably attributable to the fact the materials used to build ships have changed historically, limiting the temporal window of opportunity for boring species. The south-east coast has the highest number of species arriving through the mariculture industry, reflective of the number of mariculture facilities present within this province.
Habitat and taxonomic distribution
The habitat and taxonomic analysis was based on all of the possible 86 introductions. The marine introduced species of South Africa are currently known from 11 habitats (Fig. 7). A large proportion of introductions (53%) were found within harbours (most South African harbours are not located within estuaries), with a further 30% found equally between rocky shores and estuaries and a small percentage found offshore (Fig. 7). Whereas introduced species found on rocky shores were evident consistently across all regions, estuarine species were mainly concentrated on the east and north-east coasts.
Marine introduced species were distributed over 17 taxonomic groups (Table 1). The cnidarians, annelids, crustaceans, molluscs and chordates made up over three quarters of the species (Fig. 8). The cnidarians (13 species) consisted of anthozoans (2 species) and hydrozoans (11 species) and the molluscs (12 species) comprised of gastropods (7 species) and bivalves (5 species). The crustaceans (22 species) primarily consisted of isopods (6 species) and 11 species of amphipod (Table 1). There was one fish, an estuarine species, Cyprinus carpio and the echinoderms included one echinoid and one ophiuroid. Algal records consisted of two green algae plus three red algae and there was one flowering plant reported, a dune dwelling species, Ammophila arenaria (Table 1). The earliest collection records of introduced species in South Africa are of the alga, Chlorophyta prolifera, the bryozoans, Bugula flabellata and B. dentata, the estuarine fish, Cyprinus carpio, the dune plant, Ammophila arenaria and the crustacean, Chelura terebrans. All these species were reported between 1846 and 1888 (Table 1). Impacts are only known for 5% of all the bioinvaders listed within the current inventory, with the majority of studies concentrating on one or two of the more conspicuous species. The total number of marine introduced species within the current inventory represents 0.7% of the total known marine biodiversity for South Africa, based on an updated version of the marine species list compiled by Gibbons et al. (1999).
Discussion
The present study primarily showed that expanding the approaches used within the methods resulted in a large increase in the number of introduced and cryptogenic species recognized within the South African region. More specifically, the advances of the present re-assessment can be divided into: (i) A temporal approach that further contributes to our understanding of the colonization history of introduction in the studied region and (ii) spatial assessments that revealed differences within the patterns of regional bioinvasions.
The bioinvasion inventory: a temporal perspective
The current re-assessment resulted in the ‘earliest collection date’ for an introduced species being pushed back over a century to 1846 (the alga, Cladophora prolifera) rather than 1955 as previously recorded (Robinson et al. 2005). This clearly indicates that marine introductions started establishing in the South African marine environment a long time ago and is comparable to the earliest records of Denmark (1895), the Azores (1887) and Chile (1864) (Carlton 2009).
Based on a model developed by Wonham and Pachepsky (2006), an exponential increase in detection rates would be the expected (null) trend for the region. However, the authors recognize that data sets spanning short temporal periods may not reveal a ‘true’ best fit model, due to record limitation. In addition, their model assumes regular (non-random), standardized surveys have been sustained within the region in question over a period of decades, which is not the case for South Africa. Therefore, at this stage, it is only possible to identify that rate of discovery is increasing. Under similar assumptions, Solow and Costello (2004) clearly demonstrate that it is possible to have an accelerating rate of detection without an increasing rate of invasion and that the former does not necessarily prove or indicate the latter. Therefore, although the South African discovery rate is increasing over time, it cannot be implied from these results that introduction rate is also increasing. The records that will facilitate accurate assessment of bioinvasion rates start with and will continue from the 2000s, where prior absence can be demonstrated. What is clear is that an increase in effort per unit time has been highly effective in revealing marine introductions. The peak in the number of species revealed per decade coincides with the period of focused bioinvasive research within marine habitats spanning 1990 to the present day (Griffiths 2000; Griffiths et al. 1992, 2009; Robinson et al. 2005), the largest contributor being this re-assessment.
Although a collection date has been identified for the majority of introductions in the inventory, many of these species could have arrived decades and even centuries earlier. The date of collection is primarily tied to the expertise of the early taxonomists working within the region and thus an unquantifiable effort bias exists within the data. Cohen and Carlton (1998) removed over one-third of known introductions from their temporal analyses because they were artifacts of effort bias. Despite the fact that South Africa is a region that has sustained approximately 400 years of shipping history, little baseline data pertaining to the status of marine introductions is available.
The design and implementation of a well-planned investigative programme has paid dividends in terms of making dramatic progress within a very short space of time. In 1 year, the current re-assessment revealed three quarters of the introduced and cryptogenic species within South Africa. This substantially increased the known resolution by over four and twofold respectively from the last publication (Griffiths et al. 2009). According to Carlton (2009), the number of introductions within the South African region could easily be as high as 220 species. Through continued use of the methods applied within this re-assessment, it may be possible to reveal these introductions within a decade, based on the fact 64 introduced species were added over a 12-month period spanning 2009 and 2010.
In comparison, New Zealand added 40 introduced and 27 cryptogenic species over a period of 9 years between 1998 and 2007 (Hayden et al. 2009). This coincided with the commencement of targeted surveillance aimed at identifying marine introduced species (Hewitt et al. 2004) and represents an average rate of 4.4 introduced species and 1.8 cryptogens added per year. Australia increased the number of introductions two-fold over a period of 14 years following the establishment of the CSIRO Centre for Research on Introduced Marine Pests in 1994 (Sliwa et al. 2009) which represents an average rate of 4.3 introduced species added per year. Although South Africa has added 1.7 introductions and 0.9 cryptogens on average, per year from 1990 to 2010, the reality is that the additions were not temporally equal, with the majority of species added as a result of the current re-assessment. Thus, this provides strong motivation for other regions to initiate similar programs aimed at effectively and efficiently detecting marine introductions and cryptogenic species.
While we report a far wider diversity of marine and estuarine introductions than previously recorded, we emphasize that even this work remains a preliminary assessment in which we have identified only part of the real number of introductions. For example, a great many more species of sponges, hydroids, flatworms, polychaetes, bryozoans and other taxonomically-challenging groups (Gibbons et al. 1999) are neither clearly recognized as indigenous nor introduced at this stage. Through continued application of the methodologies utilized within this re-assessment, combined with finer-scale morphological and genetic work aimed at resolving taxonomic issues, it is predicted that many more species will be discovered within the region, in line with the predictions of Carlton (2009). Thus, we caution those making global assessments to avoid assuming that the number of non-native marine species in South Africa is sufficiently well-known to invite conclusive comparisons with other regions at this stage (such as attempted by Molnar et al. 2008).
A recent study by Nuñez and Pauchard (2010) revealed that the challenge in forming global strategies to deal with biological invasions is the fact that there are differences in data quality and availability between developed and developing regions, hindering comparative analyses. In order for scientists to fully understand the potential depth and breadth of this striking global phenomenon, it is imperative that the key issues of commitment to monitoring and vigilance and implementation of standardized assessments across areas (e.g., Hewitt et al. 2009; Sliwa et al. 2009) are addressed.
Spatial patterns of bioinvasion
Based on the current inventory, the northern hemisphere appears to be the main source of introduced species into the region with temperate species located from the west coast to the south-east coast. In contrast, those species originating from southern hemisphere regions appear to have more success establishing on the east and north-east coast. This is despite the fact there are major ports located all along the coastline that would have been exposed to centuries of shipping history from all global regions. Thus, it would initially appear that the combined climatic and oceanographic regimes that form the environmental backdrop within the different biogeographic regions (Bustamante and Branch 1996; Emanuel et al. 1992; Lombard 2004; Sink and Branch 2005) are influential in bioinvasion success and spread along the South African coastline. Interestingly, a high number of introduced species are shared across bioregions, the majority of these being found on the west coast and within the transitional zone between the west and south-east coast, where there is a well-documented overlap of indigenous species (Griffiths and Branch 1991). This suggests that there are introduced species which can exhibit high levels of plasticity, adapting to a range of temperate conditions, thus increasing establishment success (Occhipinti-Ambrogi 2007).
As with other regions, the chances of an introduction being initially introduced by certain vector pathways is likely to have peaked in the past, whereas others will have increased over time (Wonham and Carlton 2005). Wilson et al. (2009) attempted to represent the temporal windows and peaks of vectors within South Africa. However, these windows were broad estimations of vector pulses over time with an assessment of their potential role in successful establishment of introduced species. Thus, while Wilson et al. (2009) show dry ballast persisting as a vector well past the 1950s, this vector ceased in the mid-twentieth century; they show ballast water as commencing in 1900, when it was in regular use by the 1880s in ships arriving in South Africa, and they show mariculture as beginning in the mid-1700s, whereas this vector is a relatively new phenomena (last two or three decades) on South African shores.
One also needs to be aware of the adaptive nature of introduced species with regards to vector pathways. For example, the boring bivalve, Martesia striata, is able to bore into ABS (Acrylonitrile butadiene styrene) pipes (Jenner et al. 2003). In addition, pelagic plastics are becoming an increasing concern as the latest modern vector for bryozoans (Winston 1982; Barnes and Milner 2005). In South Africa, it appears both ballast water (since 1880s) and ship fouling (since 1600s) are the dominant invasive pathways with mariculture becoming more prominent. If linked to information related to the evolution of the shipping and mariculture through time and space, these vectors are highly likely to be on the increase in line with these industries.
The majority of the introduced species recorded are invertebrates which is consistent with other studies (Cohen and Carlton 1998; Ruiz et al. 2000; Wonham and Carlton 2005). The low number of macroalgal species identified is most probably due to sampling biases, especially given the high algal diversity and historically limited access in the sub-tropical and tropical provinces of the east coast (Stegenga et al. 1997). Many taxa that are not represented within the current inventory are likely to be the result of sampling and taxonomic bias. Many taxa, such as Nematoda, are undersurveyed or have not been surveyed at all due to the lack of taxonomic expertise (Gibbons et al. 1999) and introduced species certainly lie undetected within such groups (Carlton 2009).
Although we recognize that more work is needed to fully reveal all South African bioinvasions in order to facilitate conclusive comparisions with other regions, a preliminary comparison with similar assessments reveals some interesting concurrences and contrasts between regional bioinvasion patterns. Rilov and Galil (2009) report that up to 95% of Mediterranean bioinvaders have tropical (Indo-Pacific) origins which is in line with that region’s shipping history. South African bioinvasions also appear to originate from historical shipping regions such as Europe. Hayden et al. (2009) reported that the discovery rate of marine introductions was on the increase within New Zealand with ballast water and ship fouling as major invasive pathways, which is in agreement with our South African findings. Mariculture is recognized as an emerging vector pathway in both Korea and South Africa (Seo and Lee 2009) and as with South Africa, the majority of Australian and South East Pacific bioinvaders are dominated by the annelids, molluscs, crustacea and chordates (Castilla and Neill 2009; Sliwa et al. 2009).
At the present time, we recognize that the patterns of biological invasions described here represent a coarse measure, limited by the information available on known introductions in the region. However, the current research can be used to effectively re-direct and focus regional research, ensuring a comprehensive coverage that parallels studies undertaken in well-studied regions globally, as well as act as a role model approach for developing regions with similar constraints.
References
Barnes DKA, Milner P (2005) Drifting plastic and its consequences for sessile organism dispersal in the Atlantic Ocean. Mar Biol 146:815–825
Bax N, Williamson A, Aguero M, Gonzalez E, Geeves W (2004) Marine invasive alien species: a threat to global biodiversity. Mar Policy 27:313–323
Bownes SJ, McQuaid CD (2009) Mechanisms of habitat segregation between an invasive and an indigenous mussel: settlement, post-settlement mortality and recruitment. Mar Biol 156:991–1006
Branch GM, Steffani CN (2004) Can we predict the effects of alien species? A case history of the invasion of South Africa by Mytilus galloprovincialis Lamarck. J Exp Mar Biol Ecol 300:189–215
Branch GM, Odendaal F, Robinson TB (2010) Competition and facilitation between the alien mussel Mytilus galloprovincialis and indigenous species: moderation by wave action. J Exp Mar Biol Ecol 383:65–78
Bustamante RH, Branch GM (1996) Large scale patterns and trophic structure of Southern African rocky shores: the roles of geographic variation and wave exposure. J Biogeogr 23:339–351
Byers JE (2009) Competition in marine invasions. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 245–260
Carlton JT (1987) Patterns of transoceanic marine biological invasions in the Pacific. Ocean Bull Mar Sci 41:452–465
Carlton JT (1996) Pattern, process and prediction in marine invasion ecology. Biol Conserv 78:96–106
Carlton JT (2003) Community assembly and historical biogeography in the North Atlantic Ocean: the potential role of human-mediated dispersal vectors. Hydrobiologia 503:1–8
Carlton JT (2009) Deep invasion ecology and the assembly of communities in historical time. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 13–56
Carlton JT, Cohen AN (2003) Episodic global dispersal in shallow water marine organisms. The case history of the European shore crabs Carcinus maenus and C. aestuarii. J Biogeogr 30:1809–1820
Carlton JT, Eldredge LG (2009) Marine bioinvasions of Hawai’i. The introduced and cryptogenic marine and estuarine animals and plants of the Hawaiian Archipelago. Bishop Museum Bulletins in Cultural and Environmental Studies 4. Bishop Museum Press, Honolulu, 202 pp
Carlton JT, Hodder J (1995) Biogeography and dispersal of coastal marine organisms: experimental studies on a replica of a 16th century sailing vessel. Mar Biol 121:721–730
Castilla JC, Neill PE (2009) Marine bioinvasions in the southeastern Pacific: status, ecology, economic impacts, conservation, and management. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 439–457
Chapman JW (1988) Invasions of the northeast Pacific by Asian and Atlantic gammaridean amphipod crustaceans, including a new species of Corophium. J Crustac Biol 8:364–382
Chapman JW, Carlton JT (1991) A test of criteria for introduced species: the global invasion by the isopod Synidotea laevidorsalis (Miers, 1881). J Crustac Biol 11:386–400
Cohen AN, Carlton JT (1998) Accelerating invasion rate in a highly invaded estuary. Science 279:555–558
Coles SL, DeFelice RC, Eldredge LC, Carlton JT (1999) Historical and recent introductions of nonindigenous marine species into Pearl Harbor, Oahu, Hawaiian Islands. Mar Biol 135:147–158
Cranfield HJ, Gordon DP, Willan RC, Marshall BA, Battershill CN, Francis NP, Nelson WA, Glasby CJ, Read JB (1998) Adventive marine species in New Zealand. NIWA Technical Report 34, 48 pp
DeFelice RC, Eldredge LG, Carlton JT (2001) Non-indigenous marine invertebrates. In: Eldredge LG, Smith CM (eds) A guidebook of introduced marine species in Hawaii. Bishop Museum Technical Report 21, 70 pp
Emanuel BP, Bustamante RH, Branch GM, Eekhout S, Odendaal SJ (1992) A zoogeographic and functional approach to the selection of marine reserves on the west coast of South Africa. S Afr J Mar Sci 12:341–354
Ferreira CEL, de Oliveira Ribeiro Junqueira A, Villac MC, Lopes RM (2009) Marine bioinvasions in the Brazilian coast: brief report on history of events, vectors, ecology, impacts, and management of non-indigenous species. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 459–477
Florence WK, Hayward PJ, Gibbons MJ (2008) Taxonomy of shallow-water Bryozoa from the west coast of South Africa. Afr Nat Hist 3:1–58
Fofonoff PW, Ruiz GM, Hines AH, Steves BD, Carlton JT (2009) Four centuries of biological invasions in tidal waters of the Chesapeake Bay region. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 479–506
Galil BS, Zenetos A (2002) A sea change—exotics in the eastern Mediterranean. In: Leppakoski E, Gollasch S, Olenin S (eds) Invasive aquatic species of Europe: distributions, impacts and management. Kluwer, Dordrecht, pp 325–336
Gibbons MJ, Abiahy BB, Angel M, Assuncao CML, Bartsch I, Best P, Biseswar RJ, Bradford-Grieve JM, Branch W, Burreson E, Cannon L, Casanova JP, Channing A, Child CA, Compagno L, Cornelius RFS, Dadon JR, David JHM, Day J, Della-Croce N, Emschermann P, Erseus C, Esnal G, Gibson R, Griffiths CL et al (1999) The taxonomic richness of South Africa’s marine fauna: a crisis at hand. S Afr J Sci 95:8–12
Gollasch S, Nehring S (2006) National checklist for aquatic alien species in Germany. Aquat Invasions 1:245–269
Griffiths CL (2000) Overview of current problems and future risks. In: Preston G, Brown G, van Wyk E (eds) Best management practices for preventing and controlling invasive alien species. Working for Water Programme, Cape Town, pp 235–241
Griffiths CL, Branch GM (1991) The macrofauna of rocky shores in False Bay. Trans R Soc S Afr 47:575–594
Griffiths CL, Hockey PAR, Van Erkom Schurink C, Le Roux PJ (1992) Marine invasive aliens on South African shores—implications for community structure and trophic functioning. S Afr J Mar Sci 12:713–722
Griffiths CL, Robinson TB, Mead A (2009) The status and distribution of marine alien species in South Africa. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 393–408
Hampton S, Griffiths CL (2007) Why Carcinus maenas cannot get a grip on South Africa’s wave exposed coastline. Afr J Mar Sci 9:123–126
Hayden BJ, Inglis GJ, Schiel DR (2009) Marine invasions in New Zealand: a history of complex supply-side dynamics. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 409–424
Hewitt CL, Campbell ML, Thresher RE, Martin RB, Boyd S (2004) Introduced and cryptogenic species in Port Phillip Bay, Victoria, Australia. Mar Biol 144:183–202
Hewitt CL, Gollasch S, Minchin D (2009) The vessel as a vector—Biofouling, ballast water and sediments. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 117–132
Jenner HA, Rajagopal S, Van der Velde G, Duad MS (2003) Perforation of ABS pipes by boring bivalve Martesia striata: a case study. Int Biodeterior Biodegrad 52:229–232
Jensen K, Knudsen J (2005) A summary of alien marine benthic invertebrates in Danish waters. Oceanol Hydrobiol Stud 34:137–162
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16(4):199–204
Le Roux PJ, Branch GM, Joska MAP (1990) On the distribution, diet and possible impact of the invasive European shore crab Carcinus maenas (L.) along the South African coast. S Afr J Mar Sci 9:85–93
Leppakoski E, Olenin S (2000) Non-native species and rates of spread: lessons from the brackish Baltic Sea. Biol Invasions 2:151–163
Leppakoski E, Shiganova T, Alexandrov B (2009) European enclosed and semi-enclosed seas. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 529–547
Levings CD, Kieser D, Jamieson GS, Dudas S (2002) Marine and estuarine alien species in the Strait of Georgia, British Columbia. In: Claudia R, Nantel P, Muckle-Jeffs E (eds) Alien invaders in Canada’s waters, wetlands and forests. Natural Resources Canada, Canadian Forest Service, Ottawa, pp 111–131
Lombard AT (2004) Marine component of the National Spatial Biodiversity Assessment for the development of South Africa’s National Biodiversity Strategic and Action Plan. National Botanical Institute, p 101
Lonhart SI (2009) Natural and climate change mediated invasions. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 57–70
Mead A, Carlton JT, Griffiths CL, Rius M (2011) Introduced and cryptogenic marine and estuarine species in South Africa. J Nat Hist (in press)
Miller AW, Ruiz GM (2009) Differentiating successful and failed invaders: Species pools and the importance of defining vector, source and recipient regions. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 353–374
Minchin D, Gollasch S, Cohen AN, Hewitt CL, Olenin S (2009) Characterizing vectors of marine invasions. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 109–116
Molnar JL, Gamboa RL, Revenga C, Spalding MD (2008) Assessing the global threat of invasive species to marine biodiversity. Front Ecol Env 6:485–492
Nuñez MA, Pauchard A (2010) Biological invasions in developing and developed countries: does one model fit all? Biol Invasions 12:707–714
Occhipinti-Ambrogi A (2007) Global change and marine communities: alien species and climate change. Mar Pollut Bull 55:342–352
Occhipinti-Ambrogi A, Savini D (2003) Biological invasions as a component of global change in stressed marine ecosystems. Mar Pollut Bull 46:542–551
Olyarnik SV, Bracken MES, Byrnes JE, Randall Hughes A, Hultgren KM, Stachowicz JJ (2009) Ecological factors affecting community invasibility. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 215–240
Orensanz JM, Schwindt E, Pastorino G, Bortulus A, Casas G, Darrigran G et al (2002) No longer the pristine confines of the world’s ocean: a survey of exotic marine species in the southwestern Atlantic. Biol Invasions 4:115–143
Otani M (2004) Introduced marine organisms in Japanese coastal waters and processes involved in their entry. Jpn J Bentho 59:45–57 (note English translation available from author)
Rilov G, Galil B (2009) Marine bioinvasions in the Mediterranean Sea—history, distribution and ecology. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 549–576
Rius M, McQuaid CD (2009) Facilitation and competition between invasive and indigenous mussels over a gradient of physical stress. Basic Appl Ecol 10:607–613
Robinson TA, Griffiths CL, McQuaid CD, Rius M (2005) Marine alien species of South Africa—status and impacts. Afr J Mar Sci 27:297–306
Ruiz GM, Hewitt CL (2002) Toward understanding patterns of coastal marine invasions: a prospectus. In: Leppäkoski E, Gollasch S, Olenin S (eds) Invasive aquatic species of Europe. Distribution, impacts, and management. Kluwer, Dordrecht, pp 529–547
Ruiz GM, Carlton JT, Grosholz ED, Hines AH (1997) Global invasions of marine and estuarine habitats by non-indigenous species: mechanisms, extent and consequences. Am Zool 37:621–632
Ruiz GM, Fofonoff P, Hines AH, Grosholz ED (1999) Non-inidgenous species as stressors in estuarine and marine communities: assessing invasion impacts and interactions. Limnol Oceanogr 44:950–972
Ruiz GM, Fofonoff PW, Carlton JT, Wonham MJ, Hines AJ (2000) Invasion of coastal marine communities in North America: apparent patterns, processes and biases. Annu Rev Ecol Syst 31:481–531
Sala OE, Chapin FS, Armesto JJ, Berlow E et al (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774
Seo KS, Lee Y (2009) A first assessment of invasive marine species on Chinese and Korean coasts. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 577–585
Sink KJ, Branch GM (2005) Biogeographic patterns in rocky intertidal communities in KwaZulu-Natal, South Africa. Afr J Mar Sci 27:81–96
Sliwa C, Migus S, McEnnulty F, Hayes KR (2009) Marine bioinvasions in Australia. In: Rilov G, Crooks JA (eds) Biological invasions in marine ecosystems. Springer, Berlin, pp 425–437
Solow AR, Costello CJ (2004) Estimating the rate of species introductions from the discovery record. Ecology 85:1822–1825
Stegenga H, Bolton JJ, Anderson RJ (1997) Seaweeds of the South African west coast. Contrib Bolus Herb 18:3–637
Wells MJ, Poynton RJ, Balsinhas AA, Musil KJ, Joffe H, van Hoepen E, Abbot SK (1986) The history of the introduction of invasive alien plants to Southern Africa. In: Macdonald IAW, Kruger FJ, Ferrar AA (eds) The ecology and management of biological invasions in southern Africa. Oxford University Press, Cape Town, pp 21–35
Wilson JRU, Dormontt EE, Prentis PJ, Lowe AJ, Richardson DM (2009) Something in the way you move: dispersal pathways affect invasion success. Trends Ecol Evol 24:136–144
Winston JE (1982) Drift plastic: an expanding niche for a marine invertebrate? Mar Pollut Bull 13:348–351
Wonham MJ, Carlton JT (2005) Trends in marine biological invasions at local and regional scales: the Northeast Pacific Ocean as a model system. Biol Invasions 7:369–392
Wonham MJ, Pachepsky E (2006) A null model of temporal trends in biological invasion records. Ecol Lett 9(6):663–672
Wonham MJ, Carlton JT, Ruiz GM, Smith LD (2000) Fish and ships: relating dispersal frequency to success in biological invasions. Mar Biol 136:1111–1121
Zar JH (2004) Biostatistical analysis, 4th edn. Prentice Hall, Upper Saddle River, p 663
Acknowledgments
We are grateful to a large number of colleagues (cited in Mead et al. 2011) who generously provided unpublished records, museum data, systematic expertise, identified specimens or gave valuable advice. This research was conducted with support from History of the Near-shore (HNS) and the South African Environmental Observation Node (SAEON) to AM, and a grant from the Department of Science and Technology/National Research Foundation Centre of Excellence for Invasion Biology to CLG, MR and JTC. MR was supported by the ‘Agencia Española de Cooperación Internacional para el Desarrolló from the Spanish ‘Ministerio de Asuntos Exteriores y de Cooperación’.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mead, A., Carlton, J.T., Griffiths, C.L. et al. Revealing the scale of marine bioinvasions in developing regions: a South African re-assessment. Biol Invasions 13, 1991–2008 (2011). https://doi.org/10.1007/s10530-011-0016-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-011-0016-9