Abstract
Chinese privet (Ligustrum sinense Lour.), an invasive shrub from Asia, is well established in the southeastern United States where it dominates many floodplain forests. We used flight intercept traps to sample beetles at three heights (0.5, 5 and 15 m) in ~2 ha plots in which L. sinense had (by chainsaws or mulching machine) or had not been removed from forests in northeastern Georgia. Removing L. sinense, particularly by machine, increased the richness and diversity of beetles and affected composition near the ground (0.5 m) but not in the forest canopy (15 m). There were no differences among treatments above the L. sinense canopy (5 m) aside from Xylosandrus crassiusculus, an exotic ambrosia beetle from Asia, dominating the beetle community at that height in control plots. Removing the L. sinense layer greatly increased beetle richness near the ground, resulting in vertical distribution patterns more similar to those observed in areas of forest devoid of L. sinense. We suspect that even organisms in the canopy will benefit from privet removal in the long term given that tree regeneration is nearly impossible in forests in which L. sinense has become well established.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since its introduction in the mid nineteenth century, Chinese privet (Ligustrum sinense Lour.), an evergreen shrub native to Asia, has become one of the most abundant non-native invasive plants in the southeastern United States. The species currently occupies about 3.5% of the total land area covered by forests in the region (Rudis et al. 2006) and dominates many riparian areas. For example, 59% of the Upper Oconee River floodplain in north Georgia was covered by L. sinense by 1999 (Ward 2002). The expansion of L. sinense into new areas can be largely attributed to seed dispersal by animals (Miller 2003) and floodwaters (Ward 2002) and rapid establishment is aided by high growth rates, vegetative reproduction, shade tolerance, and prolific seed production (Langeland and Burkes 1998). Once established, L. sinense grows in dense thickets up to 9 m in height (Miller 2003). The species competes strongly with native plants for light and nutrients (Fig. 1), displacing many species from the understory and suppressing tree regeneration (Miller 1997; Brown and Pezeshki 2000; Kittell 2001; Morris et al. 2002; Merriam and Feil 2002; Wilcox and Beck 2007; Hanula et al. 2009). Although L. sinense may provide suitable forage for some generalist vertebrate herbivores (Stromayer et al. 1998; Kittell 2001), it is doubtful that many species have benefited from its introduction. Forest floors lacking native plants will likely lack other species as well, including many insects. Insects active above the level of L. sinense may also be affected if L. sinense disrupts their seasonal (e.g., Schaefer 1991) or daily (e.g., Costa and Crossley 1991) movements between the forest canopy and ground. Previous studies have shown that invasive plants can affect the abundance, diversity and composition of native insect communities (e.g., Greenwood et al. 2004; de Groot et al. 2007; Wiezik et al. 2007; Remsburg et al. 2008). However, to our knowledge, nothing is known about how insect faunas are affected above the reach of invasive plants, for example, in the forest canopy. The objective of this study was to determine how removing L. sinense would affect beetle diversity and composition at three heights in southeastern U.S. floodplain forests.
Methods
Study area
Study sites were located within the Oconee River watershed in northeast Georgia (Fig. 2). The four sites (i.e., blocks) selected were Sandy Creek Nature Center, Clarke Co.; Georgia State Botanical Gardens, Clarke Co.; Scull Shoals Experimental Forest, Oconee National Forest, Oglethorpe Co.; and Watson Springs Forest, University of Georgia, Greene Co.
Experimental design
Three ~2 ha plots were established in floodplain forests dominated by L. sinense at each of the four sites. Each plot at each site received one of the following treatments in October/November 2005: (1) Mulch, mechanical removal of all L. sinense using a machine (Klepac et al. 2007), leaving a layer of mulch on the soil surface; (2) Chainsaw, hand removal of all L. sinense <1.27 cm using gas-powered saws and machetes, leaving piles of cut L. sinense <1 m high; and (3) Control, no removal of L. sinense. Treatments were not randomly assigned to plots for logistical reasons; the most accessible plot at each location received the mulch treatment. Following removal, all L. sinense stumps were treated with either 30% triclopyr (Garlon®4) or 30% glyphosate (Foresters’®).
Sampling
In total, 72 flight intercept traps identical to those used by Ulyshen and Hanula (2007) were used to sample beetles at three heights (0.5, 5 and 15 m) above the forest floor. Because L. sinense reached 4–5 m tall on average, the traps at 5 m in control plots were usually situated just above the L. sinense canopy. However, L. sinense canopies are not even surfaces with some individuals exceeding 10 m in height (personal obs.). The traps at 15 m were generally above the lowest leaf bearing branches of the tallest trees and were therefore considered to be within the lower forest canopy. The lower (0.5 m) traps were suspended from metal poles (Ulyshen and Hanula 2007). The middle and upper traps were suspended above the lower traps from ropes pulled over limbs in the canopy (Ulyshen and Hanula 2007). These traps were attached to rectangular hangers made from 1.6 cm diam PVC tubing and rope. To construct each hanger, two 46 cm lengths of PVC tubing were attached from their ends by rope to create a rectangular frame 1 m long. Each trap was suspended from the middle of the upper section of PVC tubing. Sections of rope cut to length were attached to the upper and lower sections of tubing to complete the assembly. Trap assemblies were installed at two locations near the center of each plot. When in use, a 1% formaldehyde and saturated salt solution with a small amount of dish soap was added to the traps to kill and preserve the catch. We sampled on the following dates in 2006: 7–14 March, 4–11 April, 2–9 May, 2–9 June, 13–20 July and 17–24 August. The two traps at each height in each plot were combined before analysis.
Statistical analysis
The complete dataset, consisting of 550 morphospecies and 10,412 individuals, was used to perform rarefaction using PAST (Hammer et al. 2001) to compare the number of beetle morphospecies among treatments and trap heights. After excluding several sampling periods (March, July and August) and locations (botanical garden control 5 m and sandy creek control 0.5 m) due to missing samples, the dataset used for all other analyses (but further modified for PC-ORD and ANOSIM, see below) consisted of 427 morphospecies and 7,237 individuals. Separate ANOVAs for fixed effects were performed on morphospecies richness, Shannon’s diversity and evenness (i.e., the response variables) using SAS. The blocking variable, location, was removed from the model after preliminary analyses found it to be insignificant for all response variables. Nonmetric multidimensional scaling (NMS) was performed on a dataset consisting of 191 morphospecies captured in ≥3 samples using PC-ORD (McCune and Mefford 2006). The same modified data set was used in PAST (Hammer et al. 2001) to perform ANOSIM with 10,000 permutations using a Bray–Curtis distance measure to quantitatively compare beetle community similarity among treatments at each trap height.
Results
Rarefaction
For a given number of individuals, there were more morphospecies at 15 and 5 than at 0.5 m above the ground and there were fewer morphospecies in control plots than mulch and chainsaw plots (Fig. 3).
Richness
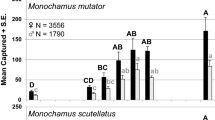
Richness varied significantly among treatments (F 2,25 = 8.78, P < 0.01) and there was a significant interaction between treatment and trap height (F 4,25 = 9.60, P < 0.0001). Richness was similar among treatments for traps suspended 5 and 15 m, but varied greatly 0.5 m above the ground (Fig. 4). On average, there were nearly twice as many morphospecies near the ground in mulch plots than in control plots and about 11 more morphospecies near the ground in mulch plots than in chainsaw plots (Fig. 4).
Evenness
Evenness varied significantly among both treatments (F 2,25 = 3.39, P = 0.05) and trap heights (F 2,25 = 6.19, P < 0.01) and there was a significant interaction between treatment and trap height (F 4,25 = 3.93, P < 0.05). Evenness was similar among treatments for traps suspended 0.5 and 15 m, but varied greatly 5 m above the ground (Fig. 5). Evenness was considerably lower in control plots at 5 m because >51% of the beetles collected there belonged to a single species, Xylosandrus crassiusculus. When X. crassiusculus was removed from the dataset there were no differences in evenness among treatments or trap heights (Fig. 5).
Diversity
For the complete dataset, diversity also varied significantly among both treatments (F 2,25 = 6.44, P ≤ 0.01) and trap heights (F 2,25 = 7.06, P < 0.01) and there was a significant interaction between treatment and trap height (F 4,25 = 2.90, P < 0.05). However, after removing X. crassiusculus from the dataset, diversity differed only among treatments (F 2,25 = 3.66, P ≤ 0.05), being lowest in control plots (Fig. 5).
Composition
The ordination from NMS (final stress 16.1) shows that the beetle faunas at 0.5 and 15 m were distinctly different (i.e., the points representing the trap locations at the two heights are widely separated) and that the fauna at 5 m was intermediate in composition (Fig. 6). Although treatments did not form obvious groupings at 0.5 m or at 15 m, control plots formed a distinct grouping at 5 m (Fig. 6). The results from ANOSIM support these conclusions: there were no significant differences in community similarity among treatments at 0.5 or 15 m but the fauna in control plots was significantly dissimilar from that in chainsaw or mulch plots at 5 m (Table 1). However, when X. crassiusculus was removed from the dataset, there were significant differences among treatments at 0.5 m but not at 5 or 15 m (Table 1).
Discussion
Removing L. sinense, particularly by machine, increased the richness and diversity of beetles and affected composition near the ground (0.5 m) but not in the forest canopy (15 m). More species were captured at 0.5 m in mulch plots than in chainsaw plots, possibly because the piles of debris left in chainsaw plots reduced the numbers of insects reaching the traps. There were no differences among treatments above the L. sinense canopy (5 m) aside from X. crassiusculus, an exotic ambrosia beetle from Asia, dominating the beetle community there in control plots. X. crassiusculus is not known to attack L. sinense, however it seems possible given the wide host range of the species and the fact that both species are originally from Asia.
Previous studies on the vertical distribution patterns of beetles in temperate deciduous forests have shown either no differences between the ground and canopy, or higher richness or diversity near the ground (Nielsen 1987; Preisser et al. 1998; Ulyshen and Hanula 2007). For example, Ulyshen and Hanula (2007) sampled beetles at 0.5 and ≥15 m in the same Scull Shoals forest used in this project, only slightly higher on the floodplain where L. sinense grew sparsely or not at all. In that study, richness was similar between the two heights and diversity was significantly higher near the ground. In this study, there were significantly fewer beetle species near the ground in control plots (i.e., beneath a canopy of L. sinense) than at 5 or 15 m. Removing the L. sinense layer greatly increased beetle richness near the ground, resulting in vertical distribution patterns more similar to those observed in areas of forest devoid of L. sinense.
The results of this study speak only to the effects of L. sinense removal on beetles within the first year. Understanding the long-term effects will require further research. Because tree regeneration is nearly impossible beneath L. sinense, even organisms in the canopy will be at risk as canopies become increasingly sparse with each dying tree.
References
Brown CE, Pezeshki SR (2000) A study on waterlogging as a potential tool to control Ligustrum sinense populations in western Tennessee. Wetlands 20:429–437
Costa JT III, Crossley DA Jr (1991) Diel patterns of canopy arthropods associated with three tree species. Environ Entomol 20:1542–1548
De Groot M, Kleijn D, Jogan N (2007) Species groups occupying different trophic levels respond differently to the invasion of semi-natural vegetation by Solidago canadensis. Biol Conserv 136:612–617
Greenwood H, O’Dowd DJ, Lake PS (2004) Willow (Salix x rubens) invasion of the riparian zone in south-eastern Australia: reduced abundance and altered composition of terrestrial arthropods. Divers Distrib 10:485–492
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4(1):9
Hanula JL, Horn S, Taylor JW (2009) Chinese privet (Ligustrum sinense) removal and its effect on native plant communities of riparian forests. Invasive Plant Science and Management (in press)
Kittell MM (2001) Relationships among invasive Chinese privet, plant diversity, and small mammal captures in southeastern deciduous forests. MS Thesis, Clemson University, 35 pp
Klepac J, Rummer RB, Hanula JL, Horn S (2007) Mechanical Removal of Chinese Privet. USDA Forest Service, Southern Research Station, Research Paper SRS-43
Langeland KA, Burkes KC (1998) Identification and biology of non-native plants in Florida’s natural areas. University of Florida, Gainesville, 165 pp
McCune B, Mefford MJ (2006) PC-ORD. multivariate analysis of ecological data. Version 5. MJM Software. Gleneden Bearch, Oregon
Merriam RW, Feil E (2002) The potential impact of an introduced shrub on native plant diversity and forest regeneration. Biol Invasions 4:369–373
Miller JH (1997) Exotic invasive plants in southeastern forests. In: Britton KO (ed) Proceedings of exotic pests of eastern forests. Tennessee Exotic Pest Plant Council, Nashville, TN, pp 97–106
Miller J (2003) Nonnative Invasive Plants of Southern Forests. USDA Forest Service, General Technical Report, SRS-62. Asheville, N.C.: USDA, Forest Service, Southern Research Station, p 93
Morris LL, Walck JL, Hidayati SN (2002) Growth and reproduction of the invasive Ligustrum sinense and native Forestiera ligustrina (Oleaceae): implications for the invasion and persistence of a nonnative shrub. Int J Plant Sci 163:1001–1010
Nielsen BO (1987) Vertical distribution of insect populations in the free air space of beech woodland. Entomol Meddelelser 54:169–178
Preisser E, Smith DC, Lowman MD (1998) Canopy and ground level insect distribution in a temperate forest. Selbyana 19:141–146
Remsburg AJ, Olson AC, Samways MJ (2008) Shade alone reduces adult dragonfly (Odonata: Liebellulidae) abundance. J Insect Behav 21:460–468
Rudis VA, Gray A, McWilliams W, O’Brien R, Olson C, Oswalt S, Schulz B (2006) Regional monitoring of nonnative plant invasions with the Forest Inventory and Analysis program. In: McRoberts RE, Reams GA, Van Duesen PC, McWilliams WH (eds) Proceedings of the sixth annual FIA Symposium 2004 Sept 21–24, pp 49–64. Denver, CO. Gen. Tech. Rep. WO-70 U.S. Department of Agriculture, Forest Service, Washington, DC
Schaefer M (1991) The animal community: diversity and resources. In: Röhrig E, Ulrich B (eds) Temperate deciduous forests. Ecosystems of the world. Elsevier, New York, pp 51–120
Stromayer KAK, Warren RJ, Harrington TB (1998) Managing Chinese privet for white-tailed deer. South J Appl For 22:227–230
Ulyshen MD, Hanula JL (2007) A comparison of the beetle (Coleoptera) fauna captured at two heights above the ground in a North American temperate deciduous forest. Am Midl Nat 158:260–278
Ward RW (2002) Extent and dispersal rates of Chinese privet (Ligustrum sinense) invasion on the upper Oconee River floodplain, North Georgia. Southeastern Geo 1:29–48
Wiezik M, Svitok M, Dovčiak M (2007) Conifer introductions decrease richness and alter composition of litter-dwelling beetles (Coleoptera) in Carpathian oak forests. Forest Ecol Manag 247:61–71
Wilcox J, Beck CW (2007) Effects of Ligustrum sinense Lour. (Chinese Privet) on abundance and diversity of songbirds and native plants in a southeastern nature preserve. Southeast Nat 6:535–550
Acknowledgments
We thank Stephanie Cahill for constructing the traps, Mike Cody for assisting with field work and Jared Swain for sorting samples. We also thank two anonymous reviewers for suggestions that greatly improved the manuscript. This research was supported by the USDA Forest Service Special Development Technology Program (Project Number R8-2005-01).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ulyshen, M.D., Horn, S. & Hanula, J.L. Response of beetles (Coleoptera) at three heights to the experimental removal of an invasive shrub, Chinese privet (Ligustrum sinense), from floodplain forests. Biol Invasions 12, 1573–1579 (2010). https://doi.org/10.1007/s10530-009-9569-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-009-9569-2