Abstract
Invasive species that strongly modify their physical habitat are a particular management concern. Theoretical models predict that habitat modification could speed spread rates or allow invasion of sites that would otherwise resist invasion. There are few empirical tests of this hypothesis, however. We tested whether habitat modification by invading Spartina alterniflora populations facilitates conspecific seedling recruitment and spatial spread in Willapa Bay, WA, USA. Established S. alterniflora individuals strongly modified their local physical environment. Hydrologic flow, porewater salinity, and light availability were decreased while sediment NH4 + increased with increasing S. alterniflora stem density. The S. alterniflora seed bank was greater and spring seedlings were denser within meadows of S. alterniflora than on unvegetated tideflats. However, almost all seedling recruitment after 1 year occurred on tideflats or on meadow edge plots where the above ground S. alterniflora biomass had been removed. Instead of facilitating invasive spread, ecosystem engineering in this system appears to create conditions that inhibit local seedling recruitment. These results suggest that the influence of ecosystem engineering on invasive spread is highly contingent on the relative spatial scales of habitat modification, environmental heterogeneity, and propagule availability. Control activities could change these spatial relationships, however, inadvertently promoting invasive recruitment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Where recruitment occurs relative to established individuals is a fundamental demographic trait controlling the spatial spread of invasive populations (Kot et al. 1996; Hastings et al. 2005). A number of conceptual models of plant recruitment describe seed or seedling survival as negatively density (or distance) dependent due to mechanisms such as predator aggregation or intraspecific competition (see reviews in McCanny 1985; Nathan and Casagrandi 2004). However, it is possible that conspecific individuals could facilitate seedling recruitment. One mechanism for this facilitation is physical habitat modification by organisms. The process of physical habitat modification by organisms has been termed ecosystem engineering (Jones et al. 1994, 1997), and there is increasing evidence that it is an important driver of population and community dynamics in a wide variety of systems (Wright and Jones 2006). A number of studies have assessed propagule establishment success within and outside of conspecific beds of sessile organisms and report intraspecific facilitation mediated through the amelioration of physical stress (Smith 1984; Callaway and D’Antonio 1991; Bertness and Yeh 1994; Bertness and Leonard 1997; Bertness et al. 1999; Weltzin and McPherson 1999; Santangelo and Abbiati 2001; Bruno et al. 2003). Yet, we still have a poor understanding of the degree to which ecosystem engineering facilitates recruitment and spatial spread in invading populations.
Using a spatially implicit model, Cuddington and Hastings (2004) demonstrated that habitat modifying invaders can have significantly faster population growth rates in sub-optimal habitats than invaders that do not modify their environment. However, we have scant empirical evidence documenting recruitment facilitation in actual invasive populations (although see Reinhart et al. 2006). Invasive spread is often disproportionately driven by recruitment in areas where the parent plant is absent or rare (Moody and Mack 1988). In addition, local intraspecific competition could obviate any positive feedbacks on recruitment in cases where the strength of engineering is proportional to local plant density.
The relative balance of the negative and positive feedbacks produced by organisms can vary spatially. There is empirical evidence that wetland zonation patterns are driven by the relative balance of positive (often engineering driven) and negative (often competition driven) feedbacks that vary spatially with factors such as levels of resource supply and local adult density (Callaway and Walker 1997). The relative spatial scales over which positive and negative population feedbacks operate can also vary. Banded vegetation patterns and similar patterns in mussel beds reflect conspecific facilitation operating at small scales and intraspecific competition operating at larger scales (Rietkerk et al. 2002; Gascoigne et al. 2005). Other studies have demonstrated the opposite scale relationship between competition and facilitation. Beds of the intertidal grass Spartina alterniflora facilitate seedling recruitment of a number of marsh species in immediately adjacent rocky shoreline habitats by ameliorating tidal energy (Bruno and Kennedy 2000). At the same New England sites, S. alterniflora inhibits recruitment of marsh species within beds due to the intense shading cast by the adult canopy (Van de Koppel et al. 2006).
Spartina alterniflora is a strong ecosystem engineer that has drastically altered physical conditions where it has invaded Pacific estuaries (Dahler and Strong 1996). In its native range, seedling recruitment is strongly inhibited within large established populations. Consequently, population dynamics are driven primarily by the vegetative growth of clonal ramets. However, in its introduced range and in large restoration projects in the native range seedling recruitment contributes significantly to spatial spread and population growth. The long distance spread of nascent populations along the Pacific coast has been driven primarily by seedling recruitment and to a lesser extent by the dispersal of vegetative fragments (Sayce et al. 1997; Ayres et al. 2004). Seedling recruitment also contributes significantly to local scale population growth. Following initial colonization by tidally born seed, invasive and re-colonizing populations grow both vegetatively and through continued seedling recruitment (Daehler 1998; Feist and Simenstad 2000; Proffitt et al. 2003; Davis et al. 2004a). Previous work in Willapa Bay, WA has shown that variation in seed production can profoundly influence S. alterniflora invasion dynamics. Strong pollen limitation causes clonal seed production to be positively density dependent (Davis et al. 2004a, b). Estimates from demographic models suggest that the resulting Allee effect has slowed invasive spread by almost 2/3 in Willapa Bay (Taylor et al. 2004).
If seedling recruitment is also positively density dependent it could be an important contributor to the observed Allee effect and consequent time lag in invasive spread. However, no studies have tested the degree to which habitat modification by S. alterniflora influences its own recruitment patterns. In this study we test the hypothesis that invading populations of S. alterniflora facilitate conspecific seedling recruitment along the edge of the invasion front. We decompose the recruitment process into seed availability, seed survival, and seedling establishment stages, and then test the degree to which these stages vary between established beds, the edge of the invasion front, and uninvaded tideflat. We assess the degree to which established S. alterniflora individuals competitively inhibit seedling establishment along this gradient in adult density by removing the above ground canopy and following the survival of transplanted seedlings. We also test whether the relative balance of positive and negative influences on recruitment vary across sites that differ in their relative physical stress.
Materials and methods
Study sites
This study was conducted in Willapa Bay, Washington USA (46°40′N, 124°02′W). Spartina alterniflora was likely introduced here roughly 115 years ago from the east coast of North America (Civille et al. 2005). Populations currently occupy about 75 km2 of tidal flats in the bay (Murphy 2003).
Four sites were selected in March 2003 to span a range of hydrologic energy and sediment characteristics. Sites were chosen based on published data on potential wave energy patterns and sediment type (Feist 1999). Decreasing sediment sand also corresponds with increasing sediment N content at these sites (Tyler et al. in press). All sites had been colonized by S. alterniflora for at least 15 years, and contained extensive high-density S. alterniflora meadows. Spartina meadows were bordered on their landward margins either by upland vegetation or by high elevation native marsh, and on their bayward margins by gently sloping tidal flats. At all four sites tideflats were occupied by two species of eelgrass (Zostera japonica and Z. marina). Natural tideflat S. alterniflora seedling densities were greater at Palix and Tarlatt than at Leadbetter and Pickernell (Lambrinos, unpublished data).
Abiotic patterns
Ambient hydrologic energy and sediment texture were measured on the unvegetated tideflat at each site. Relative hydrologic energy was estimated using the dissolution rate of zinc anodes held in contact with copper cathodes. The dissolution rate of zinc anodes in this setup is directly proportional to water flow velocity and can be used as a measure of relative hydrologic energy (McGehee 1998). Zincs (n = 10 per site) were deployed in June 2003 and retrieved after 5 months. Dissolution was assessed gravimetrically. Sediment was sampled with 10 cm depth × 3 cm diameter cores (n = 3 per site). Sediment grain size was analyzed using the suspension hydrometer method (Sheldrick and Wang 1993).
Local abiotic conditions were assessed in relationship to local S. alterniflora stem density and above ground biomass at each site. Sampling was stratified along three parallel 50 m long belt transects positioned, respectively, (a) within the S. alterniflora meadow 10 m from the bayward meadow edge; (b) along the meadow edge; (c) on the tideflat 10 m from the meadow edge. Three types of abiotic parameters were evaluated.
Light
Photosynthetically active radiation (PAR) was measured at the sediment surface and 2 m above the surface (above the S. alterniflora canopy) using a spherical light sensor (LiCor LI-93). The two values were used to calculate the percentage of PAR reaching the sediment surface. Readings were taken within ten 0.0625 m2 quadrats spaced 5 m apart along each transect at each site. Spartina alterniflora stem density and height of the longest leaf blade was measured in each quadrat. Spartina stem density and height were converted to estimates of above ground biomass using an allometric regression (Tyler et al. in press).
Water flow
We used anode dissolution to assess the relative hydrographic differences among sites over an entire season. To test the finer scale effect of Spartina density on relative water flow within a site we used the dissolution rate of plaster chalk blocks (Jokiel and Morrissey 1993). This method provides an integrative measure of flow exposure that incorporates both current velocity and inundation time. Direct observations indicated that the difference in exposure time for the chalk blocks along each transect was <30 min. for each tidal cycle. Chalk blocks were formed from dental grade gypsum plaster (Castone® Type III Dental Stone) in 85 mm diameter Petri dishes. Blocks were air dried for 48 h, weighed, then attached in the field to horizontally oriented pieces of PVC pipe positioned 30 cm above the sediment surface. Eight blocks spaced 10 m apart were deployed along each transect at each site. Stem density and height were recorded within 0.0625 m2 quadrats centered on each block. Blocks were retrieved after 72 h, air dried for 48 h, and weighed. The percentage dissolution (weight) of each block over the 72 h deployment was calculated.
Porewater characteristics
Porewater from the rooting zone of seedlings (5 cm) was sampled in June 2003 using suction lysimeters (Tyler and Ziemann 1999). Ten porewater samples were taken 5 m apart along each transect at each site. Salinity was measured with a temperature compensating refractometer and NH4 + concentration was analyzed using the phenolhypochlorite method (Solorzano 1969).
Seed availability
In March 2003 the density of S. alterniflora viable seeds in the seed bank at each site was assessed using sediment cores. This method provided a temporally integrated and direct assessment of viable seed availability as a function of Spartina density. We chose this method because measures of seed dispersal such as those provided by seed traps provide only an indirect measure of seed availability. Seed trap samples are necessarily a temporal snapshot of seed abundance, and consequently often provide poor estimates of actual seed distribution particularly in dynamic environments such as wetlands.
Twenty 70 mm (diameter) by 50 mm (depth) cores spaced 2.5 m apart were taken along each transect at each site. Cores were washed through a 1 mm sieve and S. alterniflora florets counted and removed. Recovered florets were placed in Petri dishes with deionized water and incubated at 20°C for 30 days. Dishes were scored daily for germinations. The numbers of viable seeds per core were expressed as densities relative to the area of sediment surface sampled by the core (38.48 cm2). Natural germinating S. alterniflora seedling abundance was estimated in April 2003. The abundance of newly emerged seedlings was measured in forty 1 m2 quadrats spaced 5 m apart along a 200 m belt transect positioned adjacent to the 50 m transect in each of the microhabitat zones (meadow, edge, tideflat) at each site.
Seed survival
The influence of microhabitat on S. alterniflora seed germination was assessed using seed bags tethered to the sediment surface. In September 2003 S. alterniflora seed was collected from a single field population at a site distinct from the four sites used in this study. Seed was collected from 10 presumably distinct clones within a continuous meadow and stored at 5°C. Seed bags were constructed of bridal veil fabric. Five hundred seeds from the thoroughly mixed collection were placed in each 15 × 15 cm bag and the edges sewn closed. In October 2003 10 bags spaced 5 m apart were deployed along each of the three transects at each site. Bags were secured to the sediment surface using landscaping staples. Bags were recovered in May 2004, immediately washed and florets scored for germination.
Seedling survival
The influence of microhabitat and adult S. alterniflora cover on seedling survival was assessed using seedling transplants. In May 2003 naturally occurring seedlings at each site were harvested and transplanted into experimental plots. Seedlings were approximately 2 months old, and consisted of a single stem 9–12 cm tall. Twenty 1 m2 plots were spaced 1 m apart along 20 m sub-sections of the three parallel 50 m transects at each site. Plots were randomly assigned to either a removal treatment or control (n = 10 per treatment). Removal plots had any pre-existing above ground S. alterniflora biomass removed by hand clipping. Removal plots were maintained free of S. alterniflora re-sprouts monthly during the duration of the experiment. The upper 1 cm of sediment in the control plots was turbated by hand to mimic the sediment disturbance associated with clipping in the removal plots. Six seedlings were transplanted into each plot, and spaced 25 cm apart in two parallel rows. Seedlings were marked with PVC pins. Any seedlings dying within the first week of transplantation were replaced. From June 2003 to October 2003 seedlings were assessed monthly for survivorship and growth. Monthly censuses resumed April 2004–June 2004. The percentage of the six seedlings surviving was calculated for each plot. Mean percent survivorship was calculated for each treatment × microhabitat × site combination. On a sunny mid-summer day at the Leadbetter site we measured midday (12:00–1:00 PM) light availability at the sediment surface within the experimental plots using a spherical light sensor (LiCor LI-93).
Statistical analyses
We used regression to assess the relationship between S. alterniflora abundance (stem density and biomass) and the abiotic variables. Stem density explained more of the variation in the abiotic variables than biomass, and we therefore used stem density for the rest of the analyses. We tested whether the relationships between Spartina stem density and the abiotic variables were consistent across the four sites using a one-way ANCOVA. Within each site we tested whether seed bank density and natural seedling abundance varied between microhabitats (meadow, edge, tideflat) using a non-parametric Kruskal–Wallis test. Within sites we tested how seed germination varied among the microhabitat groupings using one-way ANOVA. Germination data were arcsine transformed to satisfy the normality requirement of ANOVA. We assessed how microhabitat and experimental removal of above ground S. alterniflora biomass influenced transplant survival, light availability at the sediment surface, and porewater ammonia concentration using two-way ANOVA with microhabitat and removal treatment as factors. Survival data were log + 1 transformed to satisfy the normality requirement of ANOVA.
Results
Abiotic patterns
Two sites (Pickernell and Leadbetter) experienced relatively high hydrodynamic energy and were dominated by coarse grained sediment; two sites (Palix and Tarlatt) experienced relatively low hydrodynamic energy and were dominated by relatively fine grained sediments (Table 1).
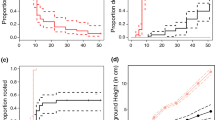
All the measured abiotic variables varied with changing S. alterniflora stem density (Fig. 1). Chalk block dissolution declined linearly with increasing Spartina density. The amount of light transmitted to the sediment surface declined exponentially with stem density (y = 98.6*e−0.02*x; r 2 = 0.95). Porewater salinity decreased and NH4 + concentration increased linearly with increasing Spartina stem density. The strong dependence of flow and light transmittance on S. alterniflora stem density is reflected in marked decreases in these parameters across the S. alterniflora invasion front in Willapa Bay (Table 2). While porewater salinity and NH4 + were less strongly dependent on S. alterniflora stem density these parameters also varied across the invasion front.
The influence of Spartina alterniflora stems on (A) % weight loss of gypsum dissolution blocks (linear regression: F 1,94 = 158.01, P < 0.001, r 2 = 0.62) (B) % light transmittance to the sediment surface (y = 98.6*e−0.02*x; df = 119, r 2 = 0.95) (C) Pore water salinity (linear regression: F 1,58 = 26.35, P < 0.001, r 2 = 0.23) (D) Pore water ammonium concentration (linear regression: F 1,58 = 12.10, P < 0.001, r 2 = 0.16). Data are pooled across all sites
The influence of Spartina stem density on the abiotic variables was consistent across the four sites (no significant stem density × site interaction in an ANCOVA, P > 0.2). The concomitant variation in abiotic variables across the three microhabitats was also consistent among sites (no significant site × microhabitat interaction in ANOVA, P < 0.05). However, site did have a significant overall effect on block dissolution and NH4 + concentration. Chalk blocks dissolved less at the Tarlatt site than at the other three sites (ANCOVA: site F 3,88 = 5.12, P = 0.003; Table 1). NH4 + concentrations were significantly greater at Tarlatt and Palix (ANCOVA: site F 3,52 = 4.46, P = 0.007; Table 1).
Seed availability
The density of viable S. alterniflora seeds in the sediment was highest along meadow transects and declined sharply on the edge and tideflat transects (Fig. 2). The effect of microhabitat on seed bank density was significant at three of the four sites using a non-parametric Kruskal–Wallis test within sites (Leadbetter: H = 23.23, P < 0.001; Pickernell: H = 0.58, P = 0.75; palix: H = 28.88, P < 0.001; Tarlatt: H = 29.13, P < 0.001). At the three sites where microhabitat had a significant influence on seed bank density no viable seeds were recovered from tideflat cores and meadow seed banks were 27–30 times larger than edge seed banks.
The distribution of emerging S. alterniflora seedlings paralleled the observed patterns in the viable seed bank. There were significantly more S. alterniflora seedlings emerging within established meadows than along the edge or on the tideflat at three of the four sites (Fig. 3). At Pickernell overall seedling abundance was low; here seedling densities on the edge and in the meadow were similar and higher than on the tideflat. The effect of microhabitat on seedling density was significant at all four sites using a non-parametric Kruskal–Wallis test within sites (Leadbetter: H = 82.93, P < 0.001; Pickernell: H = 9.71, P = 0.008; Palix: H = 50.18, P < 0.001; Tarlatt: H = 49.20, P < 0.001). Six to 26 times more seedlings emerged within meadows than on tideflats across the four sites. At Palix no seedlings were observed along the tideflat sample transects, although seedlings were observed at low density elsewhere on the tideflat.
Seed germination
The influence of microhabitat on seed germination in the experimentally tethered seed bags was inconsistent across sites (Fig. 4). Germination in the meadow was 2-fold greater than on the tideflat at Leadbetter and Palix. There was a statistically significant effect of microhabitat at these two sites using one-way ANOVA within site (Leadbetter: F 2,27 = 8.61, P = 0.001; Palix: F 2,27 = 3.31, P = 0.05). There was also a significant microhabitat effect on germination at Pickernell (one-way ANOVA; F 2,27 = 7.43, P = 0.003), but here germination rate was five times greater on the tideflat than within the meadow. There was no statistically significant microhabitat effect on germination at Tarlatt (one-way ANOVA; F 2,27 = 1.09, P = 0.35).
Seedling survival
Clipping above ground S. alterniflora biomass significantly increased the amount of light reaching the sediment surface to levels comparable to that on the unvegetated mudflat (Fig. 5). Clipping also increased porewater ammonia concentration within the meadow and edge removal plots (Fig. 5). The 1-year survival of experimental transplants was strongly dependent on site, microhabitat, and the presence of existing S. alterniflora (Fig. 6). Across all sites only a single seedling out of the 480 planted into meadow plots survived the year. At the two low energy sites (Palix and Tarlatt) seedling survival was markedly higher on the tideflat treatments than within or on the edge of the meadow. Survivorship was more than three times greater on edge removal plots than on edge control plots at these two sites. At Leadbetter and Pickernell the only seedlings surviving a year were on edge removal plots.
Light availability at the sediment surface (A) and porewater ammonia concentration (B) within experimental plots. Values are means across all sites (n = 30) ± SE. Shaded bars are control un-manipulated experimental blocks; open bars are blocks with all above ground S. alterniflora biomass removed. Same letters indicate no significant difference (P > 0.05) using planned comparisons in a two-way ANOVA with location and clipping treatment as factors
Survivorship after 12 months of experimental Spartina alterniflora seedlings across microhabitat at four sites. Control = un-manipulated experimental blocks. Removal = clipped above ground S. alterniflora biomass (excluding transplants). Values are means (n = 10) ± SE of percent survival within experimental plots (n = 6 seedlings per plot). See text for details of experimental design
Discussion
Influence of engineering on seed availability and survival
Spartina alterniflora invasion creates marked spatial heterogeneity in physical conditions. Abiotic conditions varied dramatically across the S. alterniflora invasion front. Light, hydrologic flow and salinity decreased while NH4+ concentration increased as a function of S. alterniflora density. Other studies have documented a similar influence of Spartina on these abiotic variables, both in native and introduced contexts (Leonard et al. 2002; Neira et al. 2005; Proffitt et al. 2005).
This habitat modification differentially influences recruitment components. Although S. alterniflora seeds and inflorescences are buoyant and thus have the capacity to disperse long distances, the distribution of the seed bank suggests that at the local scale a significant proportion of seeds are retained within meadows. The same physical mechanisms that reduce hydrological flow and increase sedimentation within meadows likely also increase seed deposition and retention. In river systems seed and sediment deposition are closely related (Goodson et al. 2003). Evidence from New England tidal wetlands also suggest that the seeds of many wetland plants have limited dispersal and are retained close to their site of origin (Rand 2000).
Seed germination was significantly greater within conspecific beds at only two of the four sites, although there was a non-significant trend toward greater seed germination within beds at the Tarlatt site. Other studies have shown that physical conditions associated with S. alterniflora beds are favorable for conspecific seed and seedling survival. Spartina alterniflora seedling growth and survival increase with decreasing porewater salinity (Shumway and Bertness 1992; Lewis and Weber 2002). Moderation of wave energy also significantly increases S. alterniflora seedling survival (Bergen et al. 2000). In this study, the equivocal relationship between S. alterniflora beds and seed germination contrasts with the strong spatial association between beds and seed availability in the seed bank. This suggests that the high abundance of early spring seedlings we observed within beds is mostly a result of the high seed availability here.
Influence of engineering on seedling survival
Light levels at the sediment surface within meadows are high early in the spring. As the above ground biomass of established individuals increases over the course of the growing season, light conditions within beds become increasingly unfavorable for seedling establishment. In New England marshes S. alterniflora seedlings successfully recruit only in disturbance created canopy gaps, and recruitment success increases with increasing gap size (Metcalfe et al. 1986). The experimental removal of the S. alterniflora canopy in New England marshes led to a large emergence of annual forb species (Van de Koppel et al. 2006). In this study, removal of the S. alterniflora canopy only increased seedling establishment along the edge of beds, however. The failure of canopy removal to boost seedling survivorship within beds was likely caused by the increase in porewater ammonia to levels known to be phytotoxic that we observed within removal plots. Data from another long-term experiment at this site indicates that the elevated porewater ammonia in Spartina removal plots gradually returns to ambient levels over a period of at least 3 years (Tyler et al. unpublished data). Elevated ammonia levels following Spartina removal have not been reported in the native range.
Seedling recruitment likely does occur within established S. alterniflora meadows following large disturbances such as the deposition of wrack, and this recruitment may significantly influence the genetic composition of stands (Richards et al. 2004; Travis et al. 2004). For the most part, however, population dynamics within S. alterniflora meadows appear largely governed by the vegetative growth of established individuals. This is likely not the case for species with less pronounced vegetative growth or more open canopy structures. In these cases habitat modification that facilitates seedling establishment near the parent plant may have a more pronounced influence on local densities and consequently the local impact of the invasive population.
Interaction between life history and engineering
At the local scale in this study, the aspects of habitat modification that tend to favor successful recruitment such as reduced hydrologic flow and decreased porewater salinity are spatially closely associated with the aspects that tend to inhibit recruitment namely reduced light availability. The results of the seedling survival experiment indicate that the net outcome is strong inhibition of recruitment rather than facilitation.
Isolated Spartina individuals in Willapa Bay are strongly pollen limited (Davis et al. 2004a, b). This pollen limitation is overcome as clones grow vegetatively and reach an adult density that also exerts a strong competitive inhibition on seedling survival. The results of the present study suggests that positive density dependence acting on seed production in this system is partly counteracted by strongly negative density dependence acting on seedling recruitment. It seems likely that the strength of conflicts between positive density dependence acting on reproductive success and negative density dependence acting on seedling survival depend to a large degree on the relative spatial scale of the two processes.
Influence of engineering on recruitment is likely context dependent
In New England cobble beaches, the presence of fronting S. alterniflora stands reduces flow related physical stress, facilitating the establishment and persistence of a suite of plant species immediately behind stands (Bruno and Kennedy 2000). This zone of facilitation is limited to a few meters corresponding to a tidal elevation range of 1.0–1.5 m above MLW (Kennedy and Bruno 2000). Contrastingly, S. alterniflora populations in Willapa Bay first established at relatively high tidal elevations. Subsequent recruitment has occurred in the form of scattered isolated individuals in front of these founding populations, and as long distance colonists to previously unoccupied regions in the bay (Civille et al. 2005). This colonization pattern coupled with the strong density dependence of reproductive output suggests that established beds are probably not facilitating spatial spread at larger scales in this system. This situation may be different elsewhere in the potential Pacific range of S. alterniflora, however. In San Francisco Bay S. alterniflora has hybridized with the native S. foliosa creating some genotypes that exhibit high rates of self-fertilization. Isolated selfing hybrids could produce abundant localized seed shadows. In these low density situations localized habitat modification by individual clones could facilitate seedling establishment. Indeed, the lee sides of established clones appear to provide favorable microsites for recruitment in San Francisco Bay (Sloop and Ayres unpublished data). In addition, in Mediterranean climate and desert regions high tidal elevations are characterized by hypersaline conditions and lower elevations are more favorable for initial S. foliosa recruitment (Zedler et al. 1999). In these cases, founding mid and low elevation invasive Spartina populations could ameliorate recruitment conditions at adjacent high elevation sites. This suggests that the degree to which conspecific facilitation influences invasive spread is highly contingent on the spatial distribution of founding individuals relative to that of dispersal and the pertinent environmental heterogeneity.
The weak facilitation relative to the strong inhibition of seedling recruitment within and immediately adjacent to established meadows was consistent across the abiotic stress gradient in this study. This result contrasts with other experimental work in tidal wetlands demonstrating that the balance of interspecific interactions shifts from negative to positive along increasing stress gradients (Bertness and Shumway 1993; Bertness and Hacker 1994; Crain and Bertness 2005). Willapa Bay may have relatively muted stress gradients relative to the New England locations of these studies. Most of Willapa Bay is a protected relatively low energy system with little variability in salinity among sites. However, tideflat seedlings only successfully recruited at the two low energy sites in this study suggesting that relative stress levels do affect seedling establishment success in this system. These conflicting results illustrate the difficulty in developing common metrics of stress that are easily comparable across systems or taxa. The results also suggest that the relative balance between competition and facilitation is highly contingent on site and system specific conditions.
Conclusion
This study found no evidence that established S. alterniflora populations are facilitating their own sexual recruitment in Willapa Bay. On the contrary, the edge and interior of established meadows are poor places for seedling establishment. At the same time, local seed distribution is spatially associated with meadows, preventing most of this seed from contributing to population growth and spread. An interaction between habitat modification and dispersal is a potential mechanism by which ecosystem engineers could slow their own spatial expansion. Seed trapped within conspecific beds could limit the amount of reproductive output that contributes to spatial spread. The degree to which inhibition or facilitation occurs will depend on the relative spatial scaling of engineering, environmental heterogeneity, and demography.
Control activities could potentially change these spatial relationships. In addition, the way in which control activities influence the temporal persistence of engineered habitat could impact future recruitment dynamics. In Willapa Bay, the large spikes in porewater ammonium that inhibit recruitment following Spartina removal are short-lived compared to the highly recalcitrant raised sediment elevation and root mass that promote seedling retention and recruitment (Lambrinos 2007). Control consequently creates habitat patches that are potentially more susceptible to re-invasion than the original tideflat. Eradication sites are susceptible not only to Spartina re-colonization but also colonization by other plant species typical of higher elevation salt marsh (Lambrinos 2007). An explicit understanding of how control activities alter the relationship between engineered habitat and recruitment patterns will be required for the effective management of invasive ecosystem engineers and the eventual restoration of invaded communities.
References
Ayres DR, Smith DL, Zaremba K, Klorh S, Strong DR (2004) Spread of exotic cordgrasses and hybrids (Spartina sp.) in the tidal marshes of San Francisco Bay, California, USA. Bio Inv 6:221–231
Bergen A, Alderson C, Bergfors R, Aquila C, Matsil MA (2000) Restoration of a Spartina alterniflora salt marsh following a fuel oil spill, New York City, NY. Wetlands Ecol Manage 8:185–195
Bertness MD, Hacker SD (1994) Physical stress and positive associations among marsh plants. Am Nat 144:363–372
Bertness MD, Leonard GH (1997) The role of positive interactions in communities: lessons from intertidal habitats. Ecology 78:1976–1989
Bertness MD, Shumway SW (1993) Competition and facilitation in marsh plants. Am Nat 142:718–724
Bertness MD, Yeh SM (1994) Cooperative and competitive interactions in the recruitment of marsh elders. Ecology 75:2416–2429
Bertness MD, Leonard GH, Levine JM, Bruno JF (1999) Climate-driven interactions among rocky intertidal organisms caught between a rock and a hot place. Oecologia 120:446–450
Bruno JF, Kennedy CW (2000) Patch size dependent habitat modification and facilitation on New England cobble beaches by Spartina alterniflora. Oecologia 122:98–108
Bruno JF, Stachowicz JJ, Bertness MD (2003) Inclusion of facilitation into ecological theory. Trends Ecol Evol 18:119–125
Callaway RM, D’Antonio CM (1991) Shrub facilitation of coast live oak establishment in central California. Madroño 38:158–169
Callaway RM, Walker LR (1997) Competition and facilitation: a synthetic approach to interactions in plant communities. Ecology 78:1958–1965
Civille JC, Sayce K, Smith SD, Strong DR (2005) Reconstructing a century of Spartina invasion with historical records and contemporary remote sensing. Écoscience 12:330–338
Crain CM, Bertness MD (2005) Community impacts of a tussock sedge: is ecosystem engineering important in benign habitats? Ecology 86:2696–2704
Cuddington K, Hastings A (2004) Invasive engineers. Ecol Model 178:335–347
Daehler CC (1998) Variation in self-fertility and the reproductive advantage of self-fertility for an invading plant (Spartina alterniflora). Evol Ecol 12:553–568
Dahler CC, Strong DR (1996) Status, prediction and prevention of introduced cordgrass Spartina spp. invasions in Pacific estuaries, U.S.A. Biol Conserv 78:51–58
Davis HG, Taylor CM, Civille JC, Strong DR (2004a) An Allee effect at the front of a plant invasion: Spartina in a Pacific estuary. J Ecol 92:321–327
Davis HG, Taylor CM, Lambrinos JG, Strong DR (2004b) Pollen limitation causes an Allee effect in a wind pollinated invasive grass (Spartina alterniflora). Proc Natl Acad Sci USA 101:13804–13807
Feist BE (1999) A spatio-temporal analysis of the environmental and climatic factors controlling the expansion of Spartina alterniflora in Willapa Bay, Washington. PhD dissertation, University of Washington, Seattle, Washington
Feist BE, Simenstad CA (2000) Expansion rates and recruitment frequency of exotic smooth cordgrass, Spartina alterniflora (Loisel), colonizing unvegetated littoral flats in Willapa Bay, Washington. Estuaries 23:267–274
Gascoigne JC, Beadman HA, Saurel C, Kaiser MJ (2005) Density dependence, spatial scale and patterning in sessile biota. Oecologia 145:371–381
Goodson JM, Gurnell AM, Angold PG, Morrissey IP (2003) Evidence for hydrochory and the deposition of viable seeds within winter flow-deposited sediments: the River Dove, Derbyshire, UK. River Res Appl 19:317–334
Hastings A, Cuddington K, Davies KF, Dugaw CJ, Elmendorf S, Freestone A, Harrison S, Holland M, Lambrinos J, Malvadkar U, Melbourne BA, Moore K, Taylor C, Thomson D (2005) The spatial spread of invasions: new developments in theory and evidence. Ecol Let 8:91–101
Jokiel PL, Morrissey JI (1993) Water motion on coral reefs; evaluation of the ‘chalk block’ technique. Mar Ecol-Prog Ser 93:175–181
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69:373–386
Jones CG, Lawton JH, Shachak M (1997) Positive and negative effects of organisms as ecosystem engineers. Ecology 78:1946–1957
Kennedy CW, Bruno JF (2000) Restriction of the upper distribution of New England cobble beach plants by wave-related disturbances. J Ecol 88:836–868
Kot M, Lewis MA, van den Driessche P (1996) Dispersal data and the spread of invading organisms. Ecology 77:2027–2042
Lambrinos JG (2007) Managing invasive ecosystem engineers: the case of Spartina in Pacific estuaries. In: Cuddington K, Byers J, Hastings A, Wilson W (eds) Ecosystem engineers: concepts, theory, and applications in ecology. Elsevier, London
Leonard LA, Wren PA, Beavers RL (2002) Flow dynamics and sedimentation in Spartina alterniflora and Phragmites australis marshes of the Chesapeake Bay. Wetlands 22:415–424
Lewis MA, Weber DE (2002) Effects of substrate salinity on early seedling survival and growth of Scirpus robustus and Spartina alterniflora Loisel. Ecotoxicology 11:19–26
McCanny SJ (1985) Alternatives in parent-offspring relationships in plants. Oikos 45, 148–149
McGehee MA (1998) Comparisons of water motion in coral reefs by measuring corrosion rates of dissimilar metals. Caribbean J Sci 34:286–297
Metcalfe WS, Ellison AM, Bertness MD (1986) Survivorship and spatial development of Spartina alterniflora Loisel. (Gramineae) seedlings in a New England salt marsh. Ann Bot-London 58:249–258
Moody ME, Mack RN (1988) Controlling the spread of plant invasions the importance of nascent foci. J Appl Ecol 25:10009–10021
Murphy K (2003) Spartina report to the legislature. Washington State Department of Agriculture, Olympia, Washington
Nathan R, Casagrandi R (2004) A simple mechanistic model of seed dispersal, predation and plant establishment: Janzen-Connell and beyond. J Ecol 92:733–746
Neira C, Levin LA, Grosholz ED (2005) Benthic macrophaunal communities of three sites in San Francisco Bay invaded by hybrid Spartina, with comparison to uninvaded habitats. Mar Ecol-Prog Ser 292:111–126
Proffit CE, Travis SE, Edwards KR (2003) Genotype and elevation influence Spartina alterniflora colonization and growth in a created salt marsh. Ecol Appl 13:180–192
Proffit CE, Chiasson RL, Owens AB, Edwards KR, Travis SE (2005) Spartina alterniflora genotype influences facilitation and suppression of high marsh species colonizing an early successional slat marsh. J Ecol 93:404–416
Rand TA (2000) Seed dispersal, habitat suitability, and the distribution of halophytes across a salt marsh tidal gradient. J Ecol 88:608–621
Reinhart KO, Maestre FT, Callaway RM (2006) Facilitation and inhibition of seedlings of an invasive tree (Acer platanoides) by different tree species in a mountain ecosystem. Bio Inv 8:1573–1464
Richards CL, Hamrick JL, Donovan LA, Mauricio R (2004) Unexpectedly high clonal diversity of two salt marsh perennials across a severe environmental gradient. Ecol Lett 7:1155–1162
Rietkerk M, Boerlijst MC, Van Langevelde F, HilleRisLambers R, Van de Koppel J, Kumar L, Prins HHT, de Roos AM (2002) Self-organization of vegetation in arid ecosystems. Am Nat 160:524–530
Santangelo G, Abbiati M (2001) Red coral: conservation and management of an over-exploited Mediterranean species. Aquat Conserv 11:253–259
Sayce K, Dumbauld B, Hidy J (1997) Seed dispersal in drift of Spartina alterniflora. In: Proceedings of the second international Spartina conference, Olympia, WA. Washington State University-Cooperative Extension, Pullman, WA
Sheldrick BH, Wang C (1993) Particle-size distribution. In: Carter MR (ed) Soil sampling and methods of analysis. Canadian Society of Soil Science, Lewis Publishers, Ann Arbor, MI, pp 499–511
Shumway SW, Bertness MD (1992) Salt stress limitation of seedling recruitment in a salt marsh plant community. Oecologia 92:490–497
Smith AP (1984) Postdispersal parent-offspring conflict in plants: antecedent and hypothesis from the Andes. Am Nat 123:354–370
Solorzano L (1969) Determination of ammonia in natural waters by the phenolhypochlorate method. Limnol Oceanogr 14:799–801
Taylor CM, Davis HG, Civille JC, Grevstad FS, Hastings A (2004) Consequences of an Allee effect in the invasion of a Pacific estuary by Spartina alterniflora. Ecology 85:3254–3266
Travis SE, Proffitt CE, Ritland K (2004) Population structure and inbreeding vary with successional stage in created Spartina alterniflora marshes. Ecol Appl 14:1189–1202
Tyler AC, Ziemann JC (1999) Patterns of development in the creek bank region of a barrier island Spartina alterniflora marsh. Mar Ecol-Prog Ser 180:161–177
Tyler AC, Lambrinos JG, Grosholz ED Nitrogen inputs promote the spread of an invasive marsh grass. Ecol Appl (in press)
Van de Koppel J, Altieri AH, Silliman BR, Bruno JF, Bertness MD (2006) Scale-dependent interactions and community structure on cobble beaches. Ecol Let 9:45–50
Weltzin JF, McPherson GR (1999) Facilitation of conspecific seedling recruitment and shifts in temperate savanna ecotones. Ecol Mon 69:513–534
Wright JP, Jones CG (2006) The concept of organisms as ecosystem engineers ten years on: progress, limitations and challenges. Bioscience 56:203–209
Zedler JB, Callaway JC, Desmond JS, Vivian-Smith G, Williams GD, Sullivan G, Brewster AE, Bradshaw BK (1999) Californian salt marsh vegetation: an improved model of spatial pattern. Ecosystems 2:19–35
Acknowledgments
Christy Tyler, Ursula Mahl, Nicole Christiansen, Brian Couch, Cammie Couch, Richard Hall, and Don Strong provided much appreciated help with project design and fieldwork. We thank the Washington Department of Fish and Game and the Willapa Bay National Wildlife Refuge for access to field sites. Funding was provided by National Science Foundation Biocomplexity Grant # DEB0083583 (P.I. Alan Hastings). The experiments reported in this work comply with the current laws of the United States of America.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lambrinos, J.G., Bando, K.J. Habitat modification inhibits conspecific seedling recruitment in populations of an invasive ecosystem engineer. Biol Invasions 10, 729–741 (2008). https://doi.org/10.1007/s10530-007-9165-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-007-9165-2