Abstract
Chronic exposure to solar radiation is the primary cause of photoaging and benign and malignant skin tumors. A conditioned serum-free medium (SFM) was prepared from umbilical cord mesenchymal stem cells (UC-MSCs) and its anti-photoaging effect, following chronic UV irradiation in vitro and in vivo, was evaluated. UC-MSC SFM had a stimulatory effect on human dermal fibroblast proliferation and reduced UVA-induced cell death. In addition, UC-MSC SFM blocked UVA inhibition of superoxide dismutase activity. Topical application of UC-MSC SFM to mouse skin prior to UV irradiation blocked the inhibition of superoxide dismutase and glutathione peroxidase activities, and prevented the upregulation of malonaldehyde. UC-MSC SFM thus protects against photoaging induced by UVA and UVB radiation and is a promising candidate for skin anti-photoaging treatments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Skin is exposed to ambient UV irradiation which risks photo-oxidative damage and thus long-term detrimental effects of photoaging. Epidemiological, clinical and biological studies indicate that solar UV radiation is the major etiological agent of skin cancers (Nichols and Katiyar 2010). The solar UV spectrum is classified into three bands based on wavelength: short wave (UVC, 200–290 nm), medium wave (UVB, 290–320 nm), and long wave (UVA, 320–400 nm). UVA penetrates deepest into the epidermis and dermis and is the largest component of solar UV radiation (90–95 %).
Stem cells have been used for skin regeneration with promising results (Kim et al. 2009a, 2011; Mizuno et al. 2012). Mesenchymal stem cells (MSCs) isolated from Wharton’s jelly of the umbilical cord (UC-MSCs) display fewer multipotent properties than embryonic stem cells but more than adult stem cells, and have a higher proliferation rate and self-renewal capacity than MSCs from other origins (Kestendjieva et al. 2008). One concern regarding stem cell therapies is that they may lead to tumorigenesis and promote metastasis (Karnoub et al. 2007). However, we hypothesized that secretions from short-term stem cell cultures should contain chemicals with beneficial effects on the proliferation of the skin cells. Stem cell-derived materials, such as stem cell conditioned medium, have previously been explored for disease treatment (Chen et al. 2008; Lee et al. 2012).
MSCs display multilineage developmental plasticity and secrete growth factors for paracrine signaling (Hocking and Gibran 2010), including insulin-like growth factor (IGF), transforming growth factor-beta 1 (TGF-β1), and other proteins (Chen et al. 2008; Yoon et al. 2010). Adipose-derived mesenchymal stem cells (AD-MSCs) and AD-MSC-conditioned media can increase hair growth (Yoo et al. 2010), promote wound healing (Doorn et al. 2012; Kim et al. 2007), and have antioxidant (Kim et al. 2008a), antiwrinkle (Kim et al. 2009a), and whitening (Kim et al. 2008b) properties. In this study, we investigated the protective effects of UC-MSC conditioned serum-free medium (SFM) on skin photoaging.
Materials and methods
Isolation and culture of umbilical cord mesenchymal stem cells (UC-MSC) and human dermal fibroblasts (HDF)
Human umbilical cord tissue was obtained from full-term healthy babies with the consent of each mother. All the procedures were conducted according to the guidelines of the Medical Ethics Committee of the Health Bureau of Guangdong Province, China. The fresh umbilical cord was dissected into 2–3 cm pieces and vessels (one vein and two arteries) were stripped manually from the cord segments. Approx. 1 mm3 segments were cultured in Dulbecco’s modified Eagle’s medium (DMEM)/F12 containing 10 % (v/v) fetal bovine serum (FBS). When confluency reached 80 %, cells were passaged using 0.25 % trypsin. Passage 3–6 UC-MSCs were used in all experiments.
Human prepuce tissues were acquired from the circumcision of healthy males with informed consent, with approval from the relevant institutional review boards. Tissues were digested in 0.25 % trypsin at 4 °C overnight, and the epidermis was separated from the adipose layer. Each dermis was cut into 1 mm3 pieces and cultured as described above. After approx. 10 days, HDFs were removed and cultured in growth medium. Passage 3–6 HDFs were used in subsequent experiments.
Immunophenotyping and UC-MSC differentiation
UC-MSCs were characterized by flow cytometry. Cells were harvested using 0.25 % trypsin, washed with phosphate buffered saline (PBS; pH 7.4), and incubated for 30 min at room temperature in the dark with the following human anti-CD antibodies: CD28, CD29, CD31, CD34, CD44, CD45, CD59, CD90 and CD105. The antibodies were made fluorescent with PE or FITC tags. After washing with PBS, cells were analyzed by flow cytometry. Binding of each antibody was analyzed in 20,000 cells.
UC-MSC differentiation into adipogenic and osteogenic cells was assessed using passage 3–6 cells according to a previously published reference (Kestendjieva et al. 2008). In brief, to induce adipogenic differentiation, the cells were treated with adipogenic medium consisting of DMEM supplemented with 1 μM dexamethasone, 10 μg bovine insulin/ml, 0.5 μM 3-isobutyl-1-methyl-xanthine, 200 μM indomethacin and 10 % fetal calf serum (FCS) for 3 weeks, and then stained with fresh 0.6 % oil red O to show the lipid droplets. To differentiate into osteogenic cells, the cells were cultured in DMEM supplemented with 10 % (v/v) FCS, 0.1 μM dexamethasone, 10 μM β-glycerophosphate and 50 μM ascorbic acid for 3 weeks. Osteogenic differentiation was evaluated by calcium deposition staining using the von Kossa method.
Preparation of conditioned UC-MSC medium
When UC-MSCs reached 80 % confluency, they were washed three times with PBS and cultured in DMEM/F12 SFM for 72 h. Conditioned UC-MSC SFM was collected after 72 h, centrifuged at 300×g for 5 min, filtered through a 0.22 μm filter, and concentrated (approx. 10 times) using a dialysis cassette (Sartorius) with a 3 kDa cut-off according to the manufacturer’s instructions (Kim et al. 2007, 2008a; Sze et al. 2007). The protein concentration in the UC-MSC-SFM was analyzed using the Bradford method, and then diluted to different concentration with non-conditioned medium (DMEM/F12): UC-MSC SFM was at 20 μg/ml usually equal to 20 % UC-MSC SFM after a 10-fold concentration.
Evaluation of the effect of UC-MSC SFM on HDF proliferation
HDFs (104 cells/well) were seeded into 96-well culture plates in DMEM/F12 containing 1 % FBS and cultured for 24 h. Medium was then replaced by UC-MSC SFM at various protein concentrations (5 μg/ml, 10 μg/ml, or 20 μg/ml) supplemented with 1 % FBS, and incubated for 72 h. DMEM/F12 medium containing 1 % FBS was used for controls. Cell proliferation was analyzed using an MTT assay. Briefly, after 72 h, 10 μl MTT at 0.5 mg/ml in DMEM was added to each well, plates were incubated at 37 °C for 4 h, and then 100 μl dimethyl sulfoxide was added to each well and mixed thoroughly. Plates were incubated at 37 °C for 10 min and the absorbance at 570 nm was measured using a microplate reader.
UC-MSC SFM protection assay against HDF UVA damage
Cell viability assay
HDFs (5 × 105 cells) in DMEM/F12 medium containing 10 % FBS were seeded into each culture flask and incubated overnight to reach 70 % confluency. Culture medium was then replaced with DMEM/F12 medium containing 1 % FBS and supplemented with (1) UC-MSC SFM at 10 μg/ml or 20 μg/ml, (2) 100 μg ascorbic acid/ml (as a positive control), or (3) DMEM/F12 (non-conditioned medium; negative control). After incubation for 12 h, cells were irradiated with 0.2 mW/cm2 UVA and 0.01 mW/cm2 UVB for 12 h, and allowed to recover for 36 h. Unirradiated cells were controls. The MTT assay was used to quantify living metabolically active cells. Cell viability was proportional to the absorbance measured.
Superoxide dismutase (SOD) activity assay for HDFs
HDFs were seeded and UVA-irradiated as described above. After trypsinization, cells were centrifuged at ~1,500×g for 10 min at 4 °C. Cell pellets were sonicated in ice-cold buffer (50 mM Tris–HCl, pH 7.5, 5 mM EDTA, and 1 mM DTT [Sigma-Aldrich]) and lysates were clarified at 10,000×g for 10 min at 4 °C. Lysates were assayed using a SOD assay kit according to the manufacturer’s instructions.
In vivo UV protection assays
Five-week-old female Kunming mice were provided by the Experimental Animal Medical Center (Guangdong Province, China). The study was approved by the local ethics committee for animal experimentation.
All mice were housed in climate-controlled quarters (25 ± 1 °C) and allowed free access to water and food before and after all treatments. An area of approx. 2 cm × 2 cm on the back of each mouse was shaved for UV exposure. Mice were randomly divided into five groups of 10. For each group, a different preparation was swabbed onto the shaved area each day before UV exposure: group 1, unirradiated; group 2, treated with 20 μg/ml UC-MSC SFM; group 3, treated with 10 μg UC-MSC SFM/ml; group 4, treated with 20 μg ascorbic acid/ml; and group 5, treated with DMEM-F12. Different solutions were swabbed onto the naked back of mice in Groups 2–5 before they were subjected to UV-irradiation from the same UV source everyday.
UV exposure
A Philips TL12/60W fluorescent lamp with a UV spectrum of 290–320 nm centered at 300 nm was used as the UVB source. Six fluorescent tubes (Philips TL09/60 W) with a UV spectrum of 320–400 nm centered at 365 nm were used as the UVA source. Radiation intensity was measured by a UV meter: 6 mW/cm2 for UVA and 0.3 mW/cm2 for UVB. The duration of UV irradiation was varied to ensure that the same total dose was applied to each mouse. An initial dose of 24 J/cm2 UVA in weeks 1–2 was increased to 48 J/cm2 in weeks 3–4, 72 J/cm2 in weeks 5–6, and 96 J/cm2 in weeks 7–8. The UVB dose was set at 1.2 J/cm2 in weeks 1–2, and increased to 2.4 J/cm2 in weeks 3–4, 3.6 J/cm2 in weeks 5–6, and 4.8 J/cm2 in weeks 7–8.
In vivo assays for SOD activity, glutathione peroxidase (GPx) activity, and malonaldehyde content
Irradiated and control mice were exsanguinated at week 8, and serum was prepared centrifuging blood at 2,500×g for 5 min at 4 °C. SOD activity, GPx activity, and malonaldehyde (MDA) content were determined using diagnostic kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions.
Statistical analysis
The SPSS statistical program was used for all analyses. One-way ANOVA was used to assess the statistical significance of differences between experimental groups. P values of <0.05 or <0.01 were considered statistically significant.
Results
Confirmation of UC-MSC phenotype
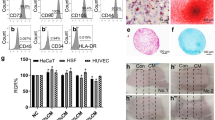
UC-MSCs prepared in our investigation expanded easily in vitro and exhibited a fibroblast-like morphology similar to that of BM-MSCs. Flow cytometry analysis showed that UC-MSCs were negative for CD28, CD31, CD34, and CD45 and positive for CD44, CD29, and CD59; cells were also positive for CD90 and CD105 which are generally considered as markers of MSCs and therefore different from HDFs (Supplementary Fig. 1; Supplementary Table 1), thus confirming that homogeneous MSC cultures had been isolated. To verify multipotency, UC-MSCs were cultured in either adipogenic differentiation or osteogenic differentiation medium. Adipogenic differentiation was identified by Oil Red O staining of intracellular lipid droplets (Supplementary Fig. 2a) and osteogenic differentiation by alkaline phosphatase staining using the modified Gomori calcium-cobalt method (Supplementary Fig. 2c), while HDF could not differentiate into adipogenic (Supplementary Fig. 2b) or osteogenic (Supplementary Fig. 2d) cells.
UC-MSC SFM stimulates HDF proliferation
UC-MSC SFM enhanced HDF proliferation in a dose-dependent manner, and the growth rate of the HDFs cultured for 3 days in UC-MSC SFM at either 5, 10 or 20 μg/ml was significantly higher than in control medium (Fig. 1).
UC-MSC conditioned medium inhibits of UVA-induced cell death
Following UVA and UVB irradiation, the viability of HDFs incubated with UC-MSC SFM at 10 μg/ml or 20 μg/ml was 75 ± 3.4 % and 86 ± 1.1 % of unirradiated cells, respectively (Fig. 2). This represents an increased viability compared to DMEM/F12-treated cells (65.3 ± 5.5 %), but is lower compared to cells treated with vitamin C (77.2 ± 8.8 %). Therefore, UC-MSC SFM and ascorbic acid protected acid HDFs against UV-induced cell death, and the 20 μg UC-MSC SFM/ml was significantly different compared to the ascorbic acid-treated group.
UC-MSC serum-free medium (SFM) improves human dermal fibroblast (HDF) viability after UV exposure. HDFs were analyzed by flow cytometry after UVA and UVB irradiation. Unirradiated cells were used as controls, ascorbic acid-treated cells were positive controls, and DMEM/F12-treated cells were negative controls. **P < 0.01, *P < 0.05
UC-MSC conditioned medium inhibits UV downregulation of SOD activity in HDFs
Intracellular SOD activity in UC-MSC SFM-treated HDFs was significantly higher than in DMEM/F12-treated controls (Fig. 3). Furthermore, the effect of UC-MSC SFM on SOD activity was dose dependent, although it did not reach the level of activity in vitamin C-treated controls.
UC-MSC serum-free medium (SFM) inhibits downregulation of intracellular SOD activity in human dermal fibroblast (HDF) after UVA and UVB irradiation. Unirradiated cells were used as controls, cells treated with 20 μg ascorbic acid/ml before UVA irradiation were positive controls, and cells treated with DMEM/F12 were negative controls. **P < 0.01, *P < 0.05
UC-MSC conditioned medium protects against photoaging in vivo
Chronic exposure of saline-treated mice to UVA and UVB radiation for 8 weeks led to a significant decrease in both SOD and GPx activity and a significant increase in MDA expression (Supplementary Table 2). In contrast, topical UC-MSC SFM treatment blocked both the reduction in SOD and GPx activity and the increase MDA content. However, GPx activity in irradiated skin following treatment with UC-MSC SFM at 20 μg/ml or 10 μg/ml was significantly higher than in DMEM/F12-treated controls. GPx activity in irradiated skin treated with ascorbic acid was lower than in irradiated skin treated with 20 μg UC-MSC SFM/ml. The malonaldehyde content in irradiated skin treated with DMEM/F12 was markedly higher than in unirradiated skin samples, and at least twice that of irradiated skin treated with UC-MSC SFM at 10 or 20 μg/ml. Again, ascorbic acid was less effective than 10–20 μg UC-MSC SFM/ml at blocking UV upregulation of MDA.
Discussion
MSCs are effective in repairing damaged tissues (Chung et al. 2009; Lau et al. 2009) although the mechanisms are not well understood and uncontrolled growth and differentiation of transplanted MSCs may cause damage to recipient tissues (Cyranoski 2010; Prantl et al. 2010). We aimed to use the beneficial features of MSCs (e.g., tissue repair and body protection) while avoid any harmful effects (e.g., carcinogenicity). Thus, we investigated the anti-photoaging properties of stem cell conditioned medium rather than of stem cells.
Two hypotheses have been proposed to explain how stem cells function in the tissue repair. The differentiation hypothesis states that immature stem cells migrate to the injured area, differentiate into cells with a phenotype corresponding to the injured tissue, repopulate, and subsequently repair the tissue. In contrast, the paracrine hypothesis states that only factors secreted from stem cell participate in tissue repair after stem cell mobilization into the injured tissue. Current data suggest that the contribution of MSC differentiation to tissue repair is limited due to poor engraftment and cell survival after transplantation (Uemura et al. 2006). In addition, much of the functional improvement following injury provided by stem cells can be achieved with cell-free conditioned media derived from stem cell cultures (Chen et al. 2008; Yoon et al. 2010). MSCs produce and secrete a wide variety of chemokines, cytokines, and growth factors (Caplan and Dennis 2006; Kovacs et al. 2009) that may be involved in anti-photoaging (Yaar and Gilchrest 2007). We explored the effect of UC-MSC conditioned medium on anti-photoaging both in vitro and in vivo.
The use of conditioned SFM from MSCs to promote tissue repair has been the subject of numerous recent studies. For example, Kim et al. (2007, 2008a) reported that factors secreted by adipose-derived stem cells prevent skin damage by free radicals under various conditions, including photoaging and wound healing. Similarly, we explored the anti-photoaging properties of UC-MSCs, from the umbilical cord, which are discarded in clinical situations and rich in MSCs. Similar to other groups, we concentrated and desalinated SFM from UC-MSC cultures before use. Consequently, the protein concentration of UC-MSC SFM could be quantified to allow a systematic evaluation of its protective effects against skin photoaging.
UV irradiation generates reactive oxygen species that deplete and damage non-enzymatic and enzymatic antioxidant defense systems in the skin, thought to be involved in aging, cancer, and various inflammatory disorders (Steenvoorden and van Henegouwen 1997). The human body counteracts oxidants using enzymatic scavengers such as SOD and GPx. SOD enzymes effectively remove free radicals, for example peroxides, thereby preventing or reducing premature aging. GPx is an abundant scavenger of H2O2, large amounts of which are produced by UV radiation. GPx can therefore preserve the integrity of cell membranes and maintain cellular functions. SOD and GPx activities are impaired after cellular exposure to UVA, UVB, or combinations thereof, leading to reduced superoxide anion-scavenging activity and oxidative damage to the cell (Merwald et al. 2005).
We found that SOD and GPx activity increased in HDFs treated with UC-MSC SFM in vitro and in murine skin samples both before and after UV irradiation. High-dose (20 μg/ml) UC-MSC SFM treatment was comparable to 20 μg vitamin C/ml in preventing photoaging. Malonaldehyde (MDA) is a relatively simple aldehyde produced when free radicals attack cellular macromolecules and is the most widely used index of lipid peroxidation(Hou et al. 2009). An increase in MDA has been reported in numerous studies that have involved acute and chronic UV exposure (Peres et al. 2011). This is one of the parameters that indicates lipid peroxidation cell damage. Increased MDA production following UV irradiation indicates that increased peroxidative damage correlates with photoaging. Our results demonstrate that UC-MSC SFM reduces MDA overexpression following UV irradiation.
Ascorbic acid has antiaging properties and is widely used as a positive control when assessing new antiaging reagents (Farris 2005; Peres et al. 2011). Although it was effective in reducing UV damage both in vitro and in vivo, our data showed that the UC-MSC SFM had better anti-photoaging properties than ascorbic acid in some assays. UC-MSC SFM contains a mixture of proteins, we cannot currently determine which compound(s) are responsible for its anti-photoaging effects.
MSCs secrete copious amounts of cytokines for paracrine signaling into the conditioned medium when cultured (Hocking and Gibran 2010), including vascular endothelial growth factor (VEGF), IGF, transforming growth factor-beta 1 (TGF-β1), basic fibroblast growth factor (bFGF), epidermal growth factor (EGF), keratinocyte growth factor (KGF) and hepatocyte growth factor (HGF). These cytokines are proposed to be the active constituent in MSC-conditioned medium (Horwitz and Prather 2009). Many of these cytokines have effects on the skin and are widely used in cosmetics. bFGF and EGF suppressed the photoaging of skin by inhibiting the transcription of nuclear factor-kappa B (Hong et al. 2008; Ono 2002). The MSC conditioned medium increases the secretion of type I collagen by HDFs and regulates the expression of extracellular matrix proteins in HDF including collagen I, collagen III, fibronectin and MMP1 (Kim et al. 2008a). We surmise that UCMSC conditioned medium may prevent photoaging via the latter via improving the extracellular matrix proteins.
Both the UC-MSC SFM and ascorbic acid have anti-photoaging properties, but the conditioned medium have multiple fuctions including wound-healing (Doorn et al. 2012), antiwrinkle properties (Kim et al. 2009b), skin whitening (Kim et al. 2008b), anti-oxidant properties (Kim et al. 2008a) and the promotion of hair growth (Won et al. 2010). The conditioned medium will have potentially broad applications.
Stem cells and stem cell conditioned medium containing secreted factors have been investigated in vitro and with animal models; however, clinical trials in humans are required to obtain conclusive results. Since we have demonstrated its topical anti-aging effects on mouse skin, UC-MSC SFM has the potential to be developed into an antiaging cosmetic.
In summary, UC-MSC SFM greatly enhances proliferation, increases viability, and increases SOD activity in human dermal fibroblasts. Furthermore, UC-MSC SFM promotes SOD and GPx activities and downregulates MDA after UV exposure in vivo. Thus UC-MSC SFM reduces oxidative damage to skin tissue and protects skin against UV-induced damage.
References
Caplan AI, Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98(5):1076–1084
Chen LW, Tredget EE, Wu PYG, Wu YJ (2008) Paracrine factors of mesenchymal stem cells recruit macrophages and endothelial lineage cells and enhance wound healing. PLoS One 3(4):12
Chung HM, Won CH, Sung JH (2009) Responses of adipose-derived stem cells during hypoxia: enhanced skin-regenerative potential. Expert Opin Biol Ther 9(12):1499–1508
Cyranoski D (2010) Strange lesions after stem-cell therapy. Nature 465(7301):997
Doorn J, Moll G, Le Blanc K, van Blitterswijk C, de Boer J (2012) Therapeutic applications of mesenchymal stromal cells: paracrine effects and potential improvements. Tissue Eng Part B-Rev 18(2):101–115
Farris PK (2005) Topical vitamin C: a useful agent for treating photoaging and other dermatologic conditions. Dermatol Surg 31(7):814–818
Hocking AM, Gibran NS (2010) Mesenchymal stem cells: paracrine signaling and differentiation during cutaneous wound repair. Exp Cell Res 316(14):2213–2219
Hong JP, Kim YW, Lee SK, Kim SH, Min KH (2008) The effect of continuous release of recombinant human epidermal growth factor (rh-EGF) in chitosan film on full thickness excisional porcine wounds. Ann Plast Surg 61(4):457–462
Horwitz EM, Prather WR (2009) Cytokines as the major mechanism of mesenchymal stem cell clinical activity: expanding the spectrum of cell therapy. Isr Med Assoc J 11(4):209–211
Hou H, Li BF, Zhao X, Zhuang YL, Ren GY, Yan MY, Cai YP, Zhang XK, Chen L (2009) The effect of Pacific cod (Gadus macrocephalus) skin gelatin polypeptides on UV radiation-induced skin photoaging in ICR mice. Food Chem 115(3):945–950
Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R, Weinberg RA (2007) Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449(7162):557–563
Kestendjieva S, Kyurkchiev D, Tsvetkova G, Mehandjiev T, Dimitrov A, Nikolov A, Kyurkchiev S (2008) Characterization of mesenchymal stem cells isolated from the human umbilical cord. Cell Biol Int 32(7):724–732
Kim WS, Park BS, Sung JH, Yang JM, Park SB, Kwak SJ, Park JS (2007) Wound heating effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci 48(1):15–24
Kim WS, Park BS, Kim HK, Park JS, Kim KJ, Choi JS, Chung SJ, Kim DD, Sung JH (2008a) Evidence supporting antioxidant action of adipose-derived stem cells: protection of human dermal fibroblasts from oxidative stress. J Dermatol Sci 49(2):133–142
Kim WS, Park SH, Ahn SJ, Kim HK, Park JS, Lee GY, Kim KJ, Whang KK, Kang SH, Park BS, Sung JH (2008b) Whitening effect of adipose-derived stem cells: a critical role of TGF-beta 1. Biol Pharm Bull 31(4):606–610
Kim WS, Park BS, Park SH, Kim HK, Sung JH (2009a) Antiwrinkle effect of adipose-derived stem cell: activation of dermal fibroblast by secretory factors. J Dermatol Sci 53(2):96–102
Kim WS, Park BS, Sung JH (2009b) Protective role of adipose-derived stem cells and their soluble factors in photoaging. Arch Dermatol Res 301(5):329–336
Kim JH, Jung M, Kim HS, Kim YM, Choi EH (2011) Adipose-derived stem cells as a new therapeutic modality for ageing skin. Exp Dermatol 20(5):383–387
Kovacs D, Raffa S, Flori E, Aspite N, Briganti S, Cardinali G, Torrisi MR, Picardo M (2009) Keratinocyte growth factor down-regulates intracellular ROS production induced by UVB. J Dermatol Sci 54(2):106–113
Lau K, Paus R, Tiede S, Day P, Bayat A (2009) Exploring the role of stem cells in cutaneous wound healing. Exp Dermatol 18(11):921–933
Lee SH, Jin SY, Song JS, Seo KK, Cho KH (2012) Paracrine effects of adipose-derived stem cells on keratinocytes and dermal fibroblasts. Ann Dermatol 24(2):136–143
Merwald H, Klosner G, Kokesch C, Der-Petrossian M, Honigsmann H, Trautinger F (2005) UVA-induced oxidative damage and cytotoxicity depend on the mode of exposure. J Photochem Photobiol B-Biol 79(3):197–207
Mizuno H, Tobita M, Uysal AC (2012) Concise review: adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells 30(5):804–810
Nichols JA, Katiyar SK (2010) Skin photoprotection by natural polyphenols: anti-inflammatory, antioxidant and DNA repair mechanisms. Arch Dermatol Res 302(2):71–83
Ono I (2002) The effects of basic fibroblast growth factor (bFGF) on the breaking strength of acute incisional wounds. J Dermatol Sci 29(2):104–113
Peres PS, Terra VA, Guarnier FA, Cecchini R, Cecchini AL (2011) Photoaging and chronological aging profile: understanding oxidation of the skin. J Photochem Photobiol B-Biol 103(2):93–97
Prantl L, Muehlberg F, Navone NM, Song YH, Vykoukal J, Logothetis CJ, Alt EU (2010) Adipose tissue-derived stem cells promote prostate tumor growth. Prostate 70(15):1709–1715
Steenvoorden DPT, van Henegouwen G (1997) The use of endogenous antioxidants to improve photoprotection. J Photochem Photobiol B-Biol 41(1–2):1–10
Sze SK, de Kleijn DPV, Lai RC, Khia Way Tan E, Zhao H, Yeo KS, Low TY, Lian Q, Lee CN, Mitchell W, El Oakley RM, Lim S-K (2007) Elucidating the secretion proteome of human embryonic stem cell-derived mesenchymal stem cells. Mol Cell Proteomics 6(10):1680–1689
Uemura R, Xu M, Ahmad N, Ashraf M (2006) Bone marrow stem cells prevent left ventricular remodeling of ischemic heart through paracrine signaling. Circ Res 98(11):1414–1421
Won CH, Yoo HG, Kwon OS, Sung MY, Kang YJ, Chung JH, Park BS, Sung JH, Kim WS, Kim KH (2010) Hair growth promoting effects of adipose tissue-derived stem cells. J Dermatol Sci 57(2):134–137
Yaar M, Gilchrest BA (2007) Photoageing: mechanism, prevention and therapy. Br J Dermatol 157(5):874–887
Yoo BY, Shin YH, Yoon HH, Seo YK, Song KY, Park JK (2010) Application of mesenchymal stem cells derived from bone marrow and umbilical cord in human hair multiplication. J Dermatol Sci 60(2):74–83
Yoon BS, Moon JH, Jun EK, Kim J, Maeng I, Kim JS, Lee JH, Baik CS, Kim A, Cho KS, Lee HH, Whang KY, You S (2010) Secretory profiles and wound healing effects of human amniotic fluid-derived mesenchymal stem cells. Stem Cell Dev 19(6):887–902
Acknowledgments
This work was supported by grants from the Guangdong International Project (2010B050100022), the Guangdong University Scientific and Technological Achievements of Major Projects (cgzhzd0905) and the Fundamental Research Funds for the Central University (21612330) in R. P. China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Qiuying Liu and Zhenyu Luo contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Q., Luo, Z., He, S. et al. Conditioned serum-free medium from umbilical cord mesenchymal stem cells has anti-photoaging properties. Biotechnol Lett 35, 1707–1714 (2013). https://doi.org/10.1007/s10529-013-1242-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-013-1242-2