Abstract
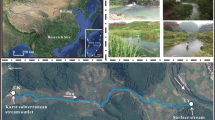
Diurnal variations of hydrochemistry were monitored at a spring and two pools in a travertine-depositing stream at Baishuitai, Yunnan, SW China. Water temperature, pH and specific conductivity were measured in intervals of 5 and 30 min for periods of 1 to 2 days. From these data the concentrations of Ca2+, HCO −3 , calcite saturation index, and CO2 partial pressure were derived. The measurements in the spring of the stream did not show any diurnal variations in the chemical composition of the water. Diurnal variations, however, were observed in the water of the two travertine pools downstream. In one of them, a rise in temperature (thus more CO2 degassing) during day time and consumption of CO2 due to photosynthesis of submerged aquatic plants accelerated deposition of calcite, whereas in the other pool, where aquatic plants flourished and grew out of the water (so photosynthesis was taking place in the atmosphere), the authors suggest that temperature-dependent root respiration underwater took place, which dominated until noon. Consequently, due to the release of CO2 by the root respiration into water, which dominated CO2 production by degassing induced by temperature increase, the increased dissolution of calcite was observed. This is the first time anywhere at least in China that the effect of root respiration on diurnal hydrochemical variations has been observed. The finding has implications for sampling strategy within travertine-depositing streams and other similar environments with stagnant water bodies such as estuaries, lakes, reservoirs, pools and wetlands, where aquatic plants may flourish and grow out of water.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
O. K. Atkin E. J. Edwards B. R. Loverys (2000) ArticleTitleResponse of root respiration to changes in temperature and its relevance to global warming New Phytol 147 141–154 Occurrence Handle10.1046/j.1469-8137.2000.00683.x
W. Dreybrodt D. Buhmann J. Michaelis E. Usdoswski (1992) ArticleTitleGeochemically controlled calcite precipitation by CO2 outgassing: Field measurements of precipitation rates to theoretical predictions Chem. Geol. 97 287–296 Occurrence Handle10.1016/0009-2541(92)90082-G
W. Dreybrodt L. Eisenlohr B. Madry S. Ringer (1997) ArticleTitlePrecipitation kinetics of calcite in the system CaCO3–H2O–CO2: the conversion to CO2 by the slow process H++HCO −3 → CO2+H2O as a rate limiting step Geochimica et Cosmochimica Acta 61 3897–3904 Occurrence Handle10.1016/S0016-7037(97)00201-9
R. Drysdale M. Taylor C. Ihlenfeld (2002) ArticleTitleFactors controlling the chemical evolution of travertine-depositing rivers of the Barkly karst, northern Australia Hydrol. Processes 16 2941–2962 Occurrence Handle10.1002/hyp.1078
R. Drysdale S. Lucas K. Carthew (2003) ArticleTitleThe influence of diurnal temperatures on the hydrochemistry of a tufa-depositing stream Hydrol. Processes 17 3421–3441 Occurrence Handle10.1002/hyp.1301
D. C. Ford P. W. Williams (1989) Karst Hydrology and Geomorphology Unwin Hyman London
T. D. Ford H. M. Pedley (1996) ArticleTitleA review of tufa and travertine deposits of the world Earth Sci. Rev. 41 117–175 Occurrence Handle10.1016/S0012-8252(96)00030-X
J. S. Herman M. M. Lorah (1987) ArticleTitleCO2 outgassing and calcite precipitation in Falling Spring Creak, USA Chem. Geol. 62 251–262 Occurrence Handle10.1016/0009-2541(87)90090-8
Z. Liu U. Svensson W. Dreybrodt D. Yuan D. Buhmann (1995) ArticleTitleHydrodynamic control of inorganic calcite precipitation in Huanglong Ravine, China: Field measurements and theoretical prediction of deposition rates Geochim. Cosmochim. Acta. 59 3087–3097 Occurrence Handle10.1016/0016-7037(95)00198-9
Z. Liu M. Zhang Q. Li S. You (2003) ArticleTitleHydrochemical and isotope characteristics of spring water and travertine in the Baishuitai area (SW China) and their meaning for paleoenvironmental reconstruction Environ. Geol. 44 698–704 Occurrence Handle10.1007/s00254-003-0811-4
G. Lu C. Zheng R. J. Donahoe W. B. Lyons (2000) ArticleTitleControlling processes in a CaCO3 precipitating stream in Huanglong Natural Scenic District,Sichuan, China J. Hydrol. 230 34–54 Occurrence Handle10.1016/S0022-1694(00)00171-2
H. M. Pedley (2000) Ambient temperature freshwater microbial tufas R. E. Riding S. M. Awramik (Eds) Microbial Sediments Springer-Verlag Berlin 179–186
A. Pentecost (1995) ArticleTitleThe Quaternary Travertine Deposits of Europe and Asia Minor Quatern. Sci. Rev. 14 1005–1028 Occurrence Handle10.1016/0277-3791(95)00101-8
A. Pentecost (1998) ArticleTitleThe significance of calcite (travertine) formation by algae in a moss-dominated travertine from Matlock Bath, England Arch. fur Hydrobiol. 143 487–509
L. N. Plummer E. Busenberg (1982) ArticleTitleThe solubilities of calcite, aragonite, and vaterite in CO2–H2O solutions between 0 and 90°C and an evaluation of the aqueous model for the system CaCO3–CO2–H2O Geochim. Cosmochim. Acta 46 1011–1040 Occurrence Handle10.1016/0016-7037(82)90056-4
J. Swoerbel (1987) Handbook of Limnology Ellis Horwood Ltd Chichester
W. Stumm J. J. Morgan (1981) Aquatic Chemistry EditionNumber2 Wiley-Interscience New York
T. M. L. Wigley (1977) ArticleTitleWATSPEC. A computer program for determining the equilibrium of aqueous solutions Br. Geomorphol. Res. Group Tech. Bull. 20 1–46
F. Winde (2002) ArticleTitleUranium contamination of fluvial systems-mechanisms and processes, PIII: Diurnal and event-related fluctuations of stream chemistry-Pitfalls from mining affected streams in South Africa, Germany and Australia Cuadernos de Investiacion Geografica 28 75–100
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Z., Li, Q., Sun, H. et al. Diurnal Variations of Hydrochemistry in a Travertine-depositing Stream at Baishuitai, Yunnan, SW China. Aquat Geochem 12, 103–121 (2006). https://doi.org/10.1007/s10498-005-2962-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10498-005-2962-2