Abstract
Dactylis glomerata L. is a widespread perennial grass species, which has been reported to be adapted to shaded conditions. Its populations thrive in a variety of environments. However, little information is available concerning the comparative response of its natural populations from contrasting environments under the reduced light intensity conditions that exist in silvopastoral systems. The objective of the present study was to estimate the comparative ability of three populations of D. glomerata from northern, central and southern Greece to grow under full sun, 60 % shade and 90 % shade in terms of their growth characteristics, phenotypic plasticity, dry matter production and nutritive value. Shade reduced tillering and dry matter production, increased tiller height and modified leaf characteristics. Under shade fewer leaves were grown simultaneously on the same tiller, but these were longer and thinner compared to full sun. Differentiation in response to shade among the populations examined of D. glomerata was observed mainly for leaf characteristics. The population from Pertouli (central Greece) responded better, particularly to moderate shade as it had a higher leaf area, longer leaf and higher dry matter production, compared to the others. Evidence for adaptive phenotypic plasticity to moderate shade was suggested only for this population. Additionally, Pertouli had higher nutritive value compared to Taxiarchis (northern Greece) and Crete (southern Greece) under shade. The divergent responses of natural populations of D. glomerata could justify breeding germplasm with enhanced shade tolerance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Light interception in silvopastoral systems affects the growth and development of understorey herbaceous vegetation in various ways (Dodd et al. 2005). Generally, herbage production decreases as light intensity decreases, due to reduced photosynthesis and modification of leaf and tiller anatomy (Devkota and Kemp 1999). Shaded plants usually have higher specific leaf area, thinner but wider leaves and lower leaf area ratio, than unshaded plants (Ciavarella et al. 2000a; Wahl et al. 2001; Zhai et al. 2006). Furthermore, shade affects the rate of leaf development and tiller appearance (Devkota et al. 1998; Monaco and Briske 2001; Wherley et al. 2005; Lemaire et al. 2009) with a consequent effect on herbage production.
The modification of leaf morphology is a type of phenotypic plasticity by which individual plants in natural populations cope with shade (Pigliucci 2001). The phenotypic response to environmental stress of plants could be the consequence of passive reductions in growth due to resource limitation (Dorn et al. 2000; van Kleunen et al. 2000). However, in many cases, the phenotypic responses increase plant function and fitness under specific environmental conditions; this can be defined as adaptive phenotypic plasticity (Sultan 1995). The adaptive significance of plasticity is generally accepted only for a few plant traits. Leaf elongation in response to shade is one of these (Dudley and Schmitt 1995; Schmitt et al. 1995). However, differentiation in plasticity to shade among populations has been reported for several herbaceous species (Winn and Evans 1991; Donohue et al. 2001; Avramov et al. 2007). This differentiation could be the result of differences in plastic response to shade (Dudley and Schmitt 1995) resulting from differences in genetic variation between populations and/or of environmental differences in the habitat of origin (Donohue et al. 2000).
Introduction of shade tolerant grass species in silvopastoral systems in many cases increases herbage production and its nutritive value (Munoz and Weaver 1999). Moreover, the use of the most shade tolerant cultivars of selected species (Feldhake and Belesky 2005) will be also important for successful silvopastoral management. From this point of view, natural populations of the species are an important source of germplasm for cultivar development. Differences in the response of grass species to shade in terms of plant growth, morphology and nutritive value have been reported in many studies. However, there is limited information about differences in response to shade among populations of the same plant species that grow in contrasting environments.
Dactylis glomerata L. is a widespread perennial grass species, which has been reported as well adapted to shaded conditions (Lin et al. 1999; Peri et al. 2001; Devkota et al. 2009). Peri et al. (2007) reported it as one of the most tolerant grass species in New Zealand agroforestry systems, while studies from the Mediterranean region have also suggested it as a suitable species for introduction in silvopastoral systems (Koukoura and Kyriazopoulos 2007; Kyriazopoulos et al. 2013). Although its populations thrive in a variety of environments, little information is available concerning their plastic response under the reduced light intensity conditions existing in silvopastoral systems. However, the understanding of growth characteristics, dry matter production and nutritive value is important for sustainable utilization of D. glomerata swards, and it may also help forage breeders in selecting superior genotypes with improved performance under shading.
The primary objective of this paper was to study natural populations of D. glomerata in relation to various shading treatments. The specific objectives were to estimate the comparative ability of the studied populations to grow under shade in terms of their growth characteristics, phenotypic plasticity, dry matter production and nutritive value. The following questions are proposed: (1) Do populations of D. glomerata from different habitats respond differently under shade? (2) Are there differences in phenotypic plasticity to shade between the studied populations? (3) Do the populations differ in their nutritive value under shade?
Materials and methods
Selection of plant material
Three populations of D. glomerata from north, central and south Greece growing in different habitats were used (Fig. 1; Table 1). Vegetative tillers were randomly selected from individual plants in each site. One tiller from each plant was transplanted into a plastic pot filled with organic matter in autumn 2007. These were placed in a greenhouse for 2 months. A study of morphological characteristics of the above populations was carried out (Madesis et al. 2013). Based on this study, 18 plants with average morphological characteristics for each population i.e. a total of 54 plants, were selected and transplanted to 54 plastic pots (30 cm diameter, 30 cm depth) filled with surface soil (0–20 cm) from the Aristotle University farm.. The plants which were intended for use in the shading treatments were subjected to shade a month before the onset of the experiment.
Establishment of the experiment
The experiment was conducted at the farm of the Aristotle University of Thessaloniki, Northern Greece (40°32′N, 22°59′E), at an altitude of 10 m above sea-level. The climate according to the bioclimatogram of Emberger (1942) is characterized as Mediterranean semiarid with cold winters. The mean annual precipitation is 443 mm and the mean annual temperature is 15.5 °C. The mean temperature was 17 °C for both the experimental periods of 2009 and 2010, while mean precipitation was 5 and 7 mm for the experimental period in 2009 and 2010 respectively (Fig. 2).
Different shading levels 0 % (full sun), 60 % (moderate shade) and 90 % (heavy shade) were applied. The different shading levels were provided by shade cloths placed over a greenhouse frame in the field. Average light intensity as measured by a quantum sensor (Li 190 SB, LiCor Biosciences, Lincoln, NE, USA) was 1200 μmol m−2 s−1 (100 % of the total radiation), 480 μmol m−2 s−1 (40 % of the total radiation) and 120 μmol m−2 s−1 (10 % of the total radiation), respectively. Of the 54 pots, eighteen (six pots from each of the three population) were placed under the above shading levels at the end of February 2009. All pots were randomized within each shading treatment biweekly and were irrigated to field capacity once a week. Plants were maintained in an open area after the end of the experimental period until February 2010, when they were subjected to the same treatments as in 2009.
Measurements
In each pot, two tillers with four leaves, located at the edge of each pot were randomly selected and marked with a plastic ring. The number of tillers per pot, the length of tillers from the base to the tip, and the length of the leaf lamina between the ligule of the youngest fully expanded to the tip of the emerging leaf were measured weekly in each of the marked tillers, during the growing season (April–May) for two consecutive years (2009 and 2010). The following parameters were calculated: Stem elongation rate (SER) = (T2 − T1)/t, leaf elongation rate (LER) = (L2 − L1)/t, where T1 and L1 are the tiller and leaf length at the beginning and T2 and L2 are the tiller and leaf length respectively at the end of a time (T), leaf appearance rate (LAR) = 1/Ph, where Ph is the time (days) between the appearance of two successive leaves, leaf elongation duration (LED) = (a)*Ph, where (a) is the number of leaves growing simultaneously on the same tiller and full leaf length (FLL) = (a)*(LER/LAR) (Lemaire and Agnusdei 2000).
Leaf area index (LAI) was measured using an area measurement system (Delta-T Devices, Cambridge, UK) (Norman and Campbell 1991); the same sample was then oven dried at 70 °C for 48 h in order to determine dry weight. Specific leaf area (SLA) was calculated as the ratio of leaf area to leaf dry weight.
The relative distances plasticity index (RDPI) (Valladares et al. 2006) was used as an indicator of comparison of phenotypic plasticity among the studied populations. This index is based on phenotypic distances among individuals of a given population exposed to different environments and was calculated for each population as: RDPI = ∑(dij → i′j′/(xi′j′ + xij))/n, where dij → i′j′ is the absolute value of the difference (xi′j′ − xij), n is the total number of distances, xij is the trait value of a given individual j (j = 1,…6) subjected to light treatment i (i = 1…3) and xi′j′ is the trait value of a given individual j′ (j = 1,…6) subjected to light treatment i′ (i = 1…3).
Dry matter (DM) production by each plant was clipped at 1 cm above ground level at the end of the growing season in both years of the experiment (June 2009, June 2010), oven-dried at 60 °C for 48 h and weighed. All the plant samples in 2009 were ground through a 1 mm screen. The individuals of each population in each shading level were mixed and two samples of each treatment were analyzed for neutral detergent fiber (NDF), acid detergent fiber (ADF), acid detergent lignin (ADL) (Van Soest et al. 1991) and N using the Kjeldahl procedure (AOAC 1990). Crude protein was then calculated by multiplying the N content by 6.25.
Experimental design and statistical analysis
Data for DM production, LAI, SLA, LER, LAR, LED and FLL of the three populations, were analyzed in each year as partly nested split-plot design with populations nested in three shading treatments (Quinn and Keough 2002) with six replications. The nutritive value parameters of mixed samples were similarly analysed. Data for the number of tillers, length of tillers and SER were analysed as partly nested split–split-plot design in time with populations nested in three shading treatments (Quinn and Keough 2002). The general linear models procedure (SPSS 18 for Windows) was used for ANOVA. The LSD at the 0.05 probability level was used to detect differences among means (Steel and Torrie 1980). The RDPI was calculated for those phenotypic traits for which significant interaction between populations and shading treatments was observed and subjected to analysis of variance (SPSS 18 for Windows). Additionally, the adaptive value of the above phenotypic traits revealing population differentiation under shade was estimated by regression analysis. The relationship between the phenotypic traits and aboveground DM production under shade was evaluated as a proxy for plant fitness across populations (Gianoli and Gonzalez-Teuber 2005).
Results
Dry matter production and plant growth characteristics
Shading significantly affected DM production, LAI and SLA in both years of the experiment (Table 2), while the number of tillers, SH and SER were only affected in 2010. Significant differences in DM production, number of tillers, LAI and SLA were recorded among the tested populations (across shading treatments) in both years, but only for SH and SER in 2009. Significant interaction was observed between the shading treatments and the populations only for DM production and LAI in 2010 (Table 2), indicating that the populations differed in shading responses. The date of measurement significantly affected the number of tillers, which progressively increased from the first to the last measurement date (data not shown).
DM production across populations was significantly reduced by 21 and 60 % in 2009 and 2010 respectively under 60 % shade (Table 3), compared to DM production under full sun. However, this reduction was even higher under 90 % shade (61 and 74 % respectively). The population from Crete had the lowest DM production (across shading treatments) in both years of the experiment (Table 4). Concerning the other two populations, there were no significant differences in 2009, while Pertouli had significantly higher DM production compared to Taxiarchis in 2010.
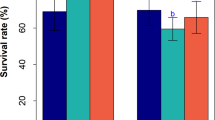
The effect of shade varied between the tested populations in 2010. Specifically, Pertouli had significantly higher DM production than Taxiarchis under both 60 and 90 % shade (Fig. 3a), but they did not significantly differ under full sun. Additionally, DM production of Taxiarchis and Crete decreased significantly between the 60 and 90 % shade treatments (Fig. 3a), while of Pertouli did not significantly differ between them.
The number of tillers significantly decreased whereas SH and SER increased from sun to shade in 2010 (Table 3). Pertouli had the highest and Crete the lowest number of tillers in 2009, while Pertouli and Crete had the highest and Taxiarchis the lowest number of tillers in 2010 (Table 4). Additionally, Taxiarchis had the highest SH and SER and Crete the lowest in 2009, whereas there were no significant differences between populations in 2010.
Overall, LAI (across populations) was greatest under 60 % shade (Table 3) and was lowest (across shading treatments) for Crete in both years (Table 4). LAI of Pertouli was significantly higher than in Taxiarchis under 60 and 90 % shade level (Fig. 3b) in 2010. Shade levels of 60 and 90 % significantly increased SLA by 51 and 64 % respectively in 2009, while this increase was much higher in 2010, reaching 130 and 134 % respectively (Table 3). SLA was highest for the population from Crete in both years (Table 4).
Leaf growth characteristics
Shading effects on LAR, (a), LER and FLL were significant for both experimental years, but was not significant for LED (Table 2). Significant differences were detected among the tested populations only for LER and FLL in both years (Table 2). There was no significant interaction for LED, LAR and (a) for both years, indicating that the shading effect on the populations was consistent. Significant interaction between shade and population was detected for FLL for both years and for LER in 2010 (Table 2).
LAR (across populations) was highest under 0 % shade during both years, while no significant differences were detected between the other two shading treatments (Table 3). Shading significantly reduced the number of leaves present simultaneously on the same tiller (a), while 60 % shade level significantly increased LER and FLL for both years of experiment. Generally, FLL did not significantly differ between full sun and 90 % shade, while LER was significantly higher in full sun compared to 90 % shade.
LER and FLL (across the shading treatments) were lowest and highest in Crete and Pertouli respectively, during both years of the experiment (Table 4). The populations responded differently to the shading treatments for LER (Fig. 3c) and FLL during 2010 (Fig. 3d). Particularly, while the populations examined had similar LER and FLL under full sun, Pertouli had significantly higher LER and FLL values than Taxiarchis and Crete under shade in 2010 (Fig. 3c, d).
The regression analysis revealed a positive relationship of dry matter yield in shade with LAI (60 %: r = 0.8, r2 = 0.6, P ≤ 0.05; 90 %: r = 0.8, r2 = 0.7, P ≤ 0.05), with LER (60 %: r = 0.5, r2 = 0.3, P ≤ 0.05; 90 %: r = 0.6, r2 = 0.4, P ≤ 0.05) and with FLL (60 %: ns, P ≤ 0.05; 90 %: r = 0.6, r2 = 0.4, P ≤ 0.05).
Phenotypic plasticity index
The RDPI phenotypic plasticity index for LAI was significantly higher for Taxiarchis compared to Pertouli and Crete between full sun and 90 % shade and between 60 and 90 % shade, but did not differ significantly among populations between full sun and 60 % shade (Fig. 4). Pertouli and Taxiarchis had the highest and lowest RDPI respectively for LER and FLL between full sun and 60 % shade and between full sun and 90 % shade. Moreover, the RDPI for LER did not significantly differ among the studied populations between 60 and 90 % shade, Pertouli had significantly the lowest FLL value.
Nutritive value
Shading significantly affected CP, NDF, ADF and ADL content (Table 5). Additionally, CP, NDF and ADF differed significantly between populations, but ADL was not. Significant interaction between the shading treatments and populations was observed for CP, NDF, ADF and ADL content (Table 2), indicating that the shading effect was not consistent.
CP content across populations (Table 5) was significantly higher under 90 % shade than under 60 % shade and full sun. CP content across shading treatments was significantly higher in Pertouli than in the other two populations examined (Table 6). The CP concentration of the populations was differed between shading treatments. Under the 60 % shade Pertouli had the highest CP content, while under 90 % shade no significant differences among populations were detected (Fig. 5a). On the contrary, under full sun Taxiarchis had the highest CP content (Fig. 5a).
Conversely, NDF content across populations (Table 5) was significantly lower under 90 % shade than under 60 % shade and full sun. ADF content across populations was significantly lower under 60 % shade with than under 90 % shade and full sun. NDF and ADF content across shading treatments were significantly higher in the Taxiarchis population than in the other two populations (Table 6). NDF content of Pertouli and Crete was generally lower under shade compared to full sun, while that of Taxiarchis was higher (Fig. 5b). Shading did not affect the ADF content of Pertouli, whereas the corresponding value was reduced under 60 % shade in Crete and under both shade levels in Taxiarchis (Fig. 5c).
Both shading treatments significantly reduced ADL content across populations (Table 5). No statistical differences were detected for ADL content between populations but ADL content was significantly reduced when shading was applied for all the populations except Pertouli (Fig. 5d). Under full sun, Pertouli had the lowest ADL content, while under both shading treatments Crete had the lowest ADL content (Fig. 5d).
Discussion
Dry matter production and growth characteristics
Shading reduced herbage production in all of studied populations of D. glomerata. Joshi et al. (1999) and Peri et al. (2007) also reported a significant reduction of herbage production of irrigated D. glomerata under shaded conditions. In contrast a recent study in the Mediterranean region under rainfed conditions, Kyriazopoulos et al. (2013) reported that herbage production of D. glomerata was not affected by shading. This inconsistency in the results could be attributable to the differences in water availability between rainfed and irrigated conditions, especially in the Mediterranean region. The limited water availability under rainfed conditions favours the plant growth in shade as evapotranspiration is reduced (Douglas et al. 2001), and soil moisture is elevated (Rao et al. 1997). In contrast, photosynthetically active radiation is a greater constraint on dry-matter production than soil moisture when water is available (Burner and Belesky 2008). This is further supported by the fact that the differences in herbage production between full sun and shade were increased during the second year of the experiment when precipitation was higher than in the first year (Fig. 2).
Modification of tiller and leaf morphology provides the phenotypic plasticity by which monocotyledonous plants cope with shade. Specifically, reduced tillering and increases in tiller height were observed under shade. This effect is attributed in part to the preferential allocation of current assimilates to existing tillers rather than to new ones (Robson et al. 1988; Kephart et al. 1992). Regarding leaf characteristics, LAI was increased under moderate shade and was maintained at the same level under full sun or decreased under heavy shade, while SLA gradually increased from full sun to heavy shade. Additionally, shade decreased LAR, but increased LER and FLL. This means that under shade fewer leaves were grown on the same tiller simultaneously, and were longer and thinner than those produced under full sun. The results reported in the present study are in agreement with those reported in many studies for D. glomerata (Devkota et al. 2000; Lin et al. 2001; Belesky 2005; Peri et al. 2007).
Population significantly affected on most of the evaluated traits but only LAI, LER, and FLL differed between populations under shade. Differences in phenotypic plasticity among populations of several species from different habitats have been reported for both stem and leaf morphology under shade (Petit and Thomson 1997; Donohue et al. 2001; Avramov et al. 2007; Bell and Galloway 2008). However, differentiation among the studied populations of D. glomerata was observed only for leaf characteristics. In particular, the population from Pertouli (central Greece) had higher LAI, LER and FLL than Crete (south) and Taxiarchis (north) under both moderate and heavy shade.
Pertouli and Taxiarchis had similar herbage production under full sun but Pertouli had higher DM production under shade, which could be partially attributed to its longer leaves and higher leaf area compared to Taxiarchis. In contrast, the population from Crete (south) had narrower leaves and was less productive compared to the others under both full sun and shade. It has been reported that plants growing in harsh environments such as the arid conditions of Crete Island grow more slowly than those growing under more equitable conditions and maintain this characteristic even under favorable growing conditions (Lambers and Poorter 1992).
Phenotypic plasticity and population responses to shade
The phenotypic plasticity index (RDPI) for LAI, LER and FLL also differed among populations and shading levels. Pertouli showed higher phenotypic plasticity for LER and FLL than the other populations, mainly between full sun and moderate shade. Conversely, Taxiarchis exhibited higher phenotypic plasticity for LAI between moderate and heavy shade. However, the observed phenotypic plasticity is not necessarily adaptive (Schmitt 1997; Donohue et al. 2000) i.e. for the plastic changes of the traits under shade to be associated with increased resource acquisition and plant fitness. According to the results, LAI, LER and FLL were positively associated with aboveground biomass (as a proxy for plant fitness) under shade. The increased LER and FLL of Pertouli between full sun and moderate shade implied better resource acquisition and plant fitness than the other populations. From this point of view, plasticity of LER and FLL for Pertouli could be considered adaptive. Conversely, the high plasticity index of LAI for Taxiarchis was the result of leaf size reduction between moderate and heavy shade. In this case, phenotypic plasticity was also induced but without the plastic changes being associated with increased resource acquisition and plant fitness, resulting in plasticity being non-adaptive or maladaptive (Weinig 2000; Poulton and Winn 2002). The above results indicate a better response to shade due to leaf modification for the Pertouli population than the other populations examined.
The divergent response to shade among the populations examined may be the result of differences in the extent of genetic variation and/or of environmental differences that the populations experience in their habitat of origin (Donohue et al. 2000). The Pertouli area is a heterogeneous landscape that comprises fir forest and open grasslands. In contrast, Taxiarchis population comes from the understorey of a coppice oak forest and Crete from open grassland. According to theoretical expectation, plasticity is favoured in heterogeneous environments (Alpert and Simms 2002). Additionally, where the heterogeneous environments increase the phenotypic plasticity, genetic variation at the population level is also promoted (Balaguer et al. 2001). There are indications that Pertouli population exhibits higher phenotypic and genotypic diversity, as revealed by SSR and ISSR molecular markers, compared to Taxiarchis (Madesis et al. 2013).
Nutritive value
The forage CP content of shade- grown cool season grasses, is generally higher than in unshaded plants (Burner 2003; Burner and Belesky 2004). Thus, increased CP content under shading has been reported for many grass species (Burner and Brauer 2003; Buergler et al. 2006; Parissi and Koukoura 2009). This was also the case in the present study. This increase could be associated with changes in phenology under shade, as the CP content of herbaceous plants decreases as they reach maturity (Kilcher 1981). Plants under shade delay their maturity compared to plants under full light intensity (Blair et al. 1983; Koukoura and Nastis 1989; Belesky et al. 2006). Furthermore, Wilson and Ludlow (1991) stated that a reduction in light causes a decrease in photosynthates, with a consequent rise in N concentration. In addition, Peri et al. (2004) reported that reduced light intensity increased soil N content by conserving soil moisture that increased N soil mineralization and subsequently N availability to herbage (Wilson et al. 1990).
The nutritive value of D. glomerata under shade based only on CP content could be considered excellent. However, utilization efficiency of CP in ruminants depends on readily fermentable carbohydrates (Hoover and Stokes 1990; Ciavarella et al. 2000b) and to relevant cell wall content. NDF, ADF and ADL contents were generally decreased under shade. Conversely, Lin et al. (2001) reported that the NDF and ADF contents of shaded grass species including D. glomerata were either unaffected or slightly increased. A similar decrease in ADL content under shade was reported by Iason and Hester (1993) for Calluna vulgaris in vegetative stage.
Differences between populations was observed for CP, NDF and ADF content under shade. Pertouli had constantly higher CP and lower NDF content than the other populations under shade, while its ADF content was unaffected by shade. This could be attributed to the higher leaf area that Pertouli maintained under shade compared to the other populations, as generally, leaves contain more protein and less fiber than stems (Gebrehiwot 1994). Additionally, the fact that Pertouli was the latest population in inflorescence appearance among several populations from Greece, including Taxiarchis and Crete (Parissi et al. 2007) could be attributed to its high CP content.
Conclusions
Shade reduced tillering and herbage production, increased tiller height and modified leaf characteristics in D. glomerata. Particularly under shade, fewer leaves were present simultaneously on individual tillers, but were longer and thinner than in full sun. Differentiation in response to shade among the populations of D. glomerata examined was observed mainly for leaf characteristics. Pertouli population from a heterogeneous landscape in central Greece responded better, particularly to moderate shade, than the others. Evidence for adaptive phenotypic plasticity, mainly to moderate shade, was suggested only for this population. Additionally, Pertouli had higher nutritive value than Taxiarchis and Crete under shade. The divergent response to shade of natural populations of D. glomerata could justify breeding germplasm with enhanced shade tolerance.
References
Alpert P, Simms EL (2002) The relative advantages of plasticity and fixity in different environments: when is it good for a plant to adjust? Evol Ecol 16:285–297
AOAC (1990) Official methods of analysis, 15th edn. Association of Official Analytical Chemists, Arlington
Avramov S, Pemac D, Tucić B (2007) Phenotypic plasticity in response to an irradiance gradient in Iris pumila: adaptive value and evolutionary constraints. Plant Ecol 190:275–290
Balaguer L, Martínez-Ferri E, Valladares F, Pérez-Corona ME, Baquedano FJ, Castillo FJ, Manrique E (2001) Population divergence in the plasticity of the response of Quercus coccifera to the light environment. Funct Ecol 15:124–135
Belesky DP (2005) Growth of Dactylis glomerata along a light gradient in the central Appalachian region of the eastern USA: II. Mechanisms of leaf dry matter production for plants establishing in spring or late summer. Agrofor Syst 65:91–98
Belesky DP, Neel JP, Chatterton NJ (2006) Dactylis glomerata growing along a light gradient in the Central Appalachian Region of the Eastern USA: III. Nonstructural carbohydrates and nutritive value. Agrofor Syst 67:51–61
Bell DL, Galloway LF (2008) Population differentiation for plasticity to light in an annual herb. Adaptation and cost. Am J Bot 95:59–65
Blair RM, Alcaniz R, Harrell A (1983) Shade intensity influences the nutrient quality and digestibility of southern deer browse leaves. J Range Manag 36:257–264
Buergler AL, Fike JH, Burger JA, Feldhake CM, McKenna JR, Teutsch CD (2006) Forage nutritive value in an emulated silvopasture. Agron J 98:1265–1273
Burner DM (2003) Influence of alley crop environment on Orchardgrass and Tall Fescue herbage. Agron. J 95:1163–1171
Burner DM, Belesky DP (2004) Diurnal effects on nutritive value of alley-cropped Orchardgrass herbage. Crop Sci 44:1776–1780
Burner DM, Belesky DP (2008) Relative effects of irrigation and intense shade on productivity of alley-cropped tall fescue herbage. Agrofor Syst 73:127–139
Burner DM, Brauer DK (2003) Herbage response to spacing of loblolly pine trees in a minimal management silvopasture in southeastern USA. Agrofor Syst 57:69–77
Ciavarella TA, Simpson RJ, Dove H, Leury BJ, Sims IM (2000a) Diurnal changes in the concentration of watersoluble carbohydrates in Phalaris aquatica L. pasture in spring, and the effect of short-term shading. Aust J Agric Res 51:749–756
Ciavarella TA, Dove H, Leary BJ, Simpson RJ (2000b) Diet selection by sheep grazing Phalaris aquatica L. pastures of differing water-soluble carbohydrate content. Aust J Agric Res 51:757–764
Devkota NR, Kemp PD (1999) Morphological aspects of pasture species in the shade in relation to various management practices under silvopastoral systems. J Inst Agric Anim Sci 19–20:1–26
Devkota NR, Kemp PD, Valentine I, Hodgson J (1998) Performance of perennial ryegrass and cocksfoot cultivars under tree shade. Proc Agron Soc NZ 28:129–135
Devkota NR, Kemp PD, Valentine I, Hodgson J (2000) Shade tolerance of pasture species in relation to deciduous tree, temperate silvopastoral systems. Agron NZ 30:101–107
Devkota NR, Kemp PD, Hodgson J, Valentine I, Jaya IKD (2009) Relationship between tree canopy height and the production of pasture species in a silvopastoral system based on alder trees. Agrofor Syst 76:363–374
Dodd MB, McGowan AW, Power IL, Thorrold BS (2005) Effects of variation in shade level, shade duration and light quality on perennial pastures. NZ J Agric Res 48:531–543
Donohue K, Messiqua D, Hammond Pyle E, Heschel SM, Schmitt J (2000) Evidence of adaptive divergence in plasticity: density- and site-dependent selection on shade avoidance responses in Impatiens capensis. Evolution 54:1956–1968
Donohue K, Hammond Pyle E, Messiqua D, Heschel SM, Schmitt J (2001) Adaptive divergence in plasticity in natural populations of Impatiens capensis and its consequences for performance in novel habitats. Evolution 55:692–702
Dorn LA, Pyle EH, Schmitt J (2000) Plasticity to light cues and resources in Arabidopsis thaliana: testing for adaptive value and costs. Evolution 54:1982–1994
Douglas GB, Walcroft AS, Wills BJ, Hurst SE, Foote AG, Trainor KD, Fung LE (2001) Resident pasture growth and the micro-environment beneath young, wide-spaced poplars in New Zealand. Proc NZ Grassl Assoc 63:131–138
Dudley SA, Schmitt J (1995) Genetic differentiation in morphological responses to simulated foliage shade between populations of Impatiens capensis from open and woodland sites. Funct Ecol 9:655–666
Emberger L (1942) Un projet d’une classification des climats du point de vue phytogéographique. Bull Soc Hist Nat Toulouse 77:97–124
Feldhake CM, Belesky DP (2005) Photosynthetically active radiation use efficiency of Dactylis glomerata in a hardwood silvopasture. North American Agroforestry Conference, CD-ROM, June 2005
Gebrehiwot L (1994) Productivity and Forage Quality of Selected Annual Legumes and Perennial Grasses in the Highlands of Ethiopia. PhD Dissertation, University of Missouri, Columbia
Gianoli E, Gonzalez-Teuber M (2005) Environmental heterogeneity and population differentiation in plasticity to drought in Convolvulus chilensis (Convolvulaceae). Evol Ecol 19:603–613
Hoover WH, Stokes SR (1990) Balancing carbohydrates and proteins for optimum rumen microbial yield. J Dairy Sci 36:36–44
Iason GR, Hester AJ (1993) The response of heather (Calluna vulgaris) to shade and nutrients–predictions of the carbon-nutrient balance hypothesis. J Ecol 81:75–80
Joshi MR, Lucas RJ, Sedcole JR, Pollock KM, Moot DJ, Mead DJ (1999) Shading effects of Pinus radiata on productivity and feeding value of orchardgrass pasture. Agrofor Forum 9:17–19
Kephart KD, Buxton DR, Taylor SE (1992) Growth of C3 and C4 perennial grasses to shade. Crop Sci 32:1033–1038
Kilcher MR (1981) Plant development, stage of maturity and nutrient composition. J Range Manag 34:363–364
Koukoura Z, Kyriazopoulos AP (2007) Adaptation of herbaceous plant species in the understorey of Pinus brutia. Agrofor Syst 70:11–16
Koukoura Z, Nastis AS (1989) Effect of shade on production and forage quality of herbaceous species. Geotech Sci Issues 1:17–25 (in Greek, English summary)
Kyriazopoulos AP, Abraham EM, Parissi ZM, Koukoura Z, Nastis AS (2013) Forage production and nutritive value of Dactylis glomerata and Trifolium subterraneum mixtures under different shading treatments. Grass Forage Sci 68:72–82
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 23:187–261
Lemaire G, Agnusdei M (2000) Leaf tissue turnover and efficiency of herbage utilization. In: Lemaire G, Hodgson J, de Moraes A, Nabinger C, de F Carvalho PC (eds) Grassland ecophysiology and grazing ecology. CAB International, Wallingford, pp 265–287
Lemaire G, Dasilva SC, Agnusdei M, Wade M, Hodgson J (2009) Interactions between leaf lifespan and defoliation frequency in temperate and tropical pastures: a review. Grass Forage Sci 64:341–353
Lin CH, McGraw RL, George MF, Garrett HE (1999) Shade effects on forage crops with potential in temperate agroforestry practices. Agrofor Syst 44:109–119
Lin CH, McGraw RL, George MF, Garrett HE (2001) Nutritive quality and morphological development under partial shade of some forage species with agroforestry potential. Agrofor Syst 53:269–281
Madesis P, Abraham EM, Kalivas A, Ganopoulos I, Tsaftaris A (2013) Genetic diversity and structure of natural Dactylis glomerata L. populations revealed by morphological and microsatellite-based (SSR/ISSR) markers, Genet Mol Res (under publication)
Monaco TA, Briske DD (2001) Contrasting shade avoidance responses in two perennial grasses: a field investigation in simulated sparse and dense canopies. Plant Ecol 156:173–182
Munoz AE, Weaver RW (1999) Competition between subterranean clover and ryegrass for uptake of 15 N-labelled fertilizer. Plant Soil 211:173–178
Norman JM, Campbell JS (1991) Canopy structure. In: Pearcy RW et al (eds) Plant physiological ecology. Field methods and instrumentation. Chapman and Hall, London, pp 301–325
Parissi ZM, Koukoura Z (2009) Effect of fertilization and artificial shading on N and various mineral content of herbaceous species. Options Mediterr 85:159–164
Parissi ZM, Abraham EM, Karameri E, Kexagia A, Karatassiou M (2007) Comparable study of the nutritive value in Greek populations of Dactylis glomerata L. growing in contrasting environments in association to maturity. Grassland Sci Eur 12:371–374
Peri PL, Varella AC, Lucas RJ, Moot DJ (2001) Dactylis glomerata and lucerne productivity in a Pinus radiata silvopastoral system: a grazed comparison. Proc NZ Grassl Assoc 63:139–147
Peri PL, Lucas RJ, Moot DJ (2004) Dry matter production and nutritive value of Dactylis glomerata (Dactylis glomerata L.) grown under different light regimes. In: Mosquera-Losada MR, Mc Adam J, Rigueiro-Rodriguez A (eds) Silvopastoralism and sustainable land management. CABI Publishing, Oxford Shire, pp 172–173
Peri PL, Lucas RJ, Moot DJ (2007) Dry matter production, morphology and nutritive value of Dactylis glomerata growing under different light regimes. Agrofor Syst 70:63–79
Petit C, Thompson JD (1997) Variation in phenotypic response to light availability between diploid and tetraploid populations of the perennial grass Arrhenatherum elatius from open and woodland sites. J Ecol 85:657–667
Pigliucci M (2001) Phenotypic plasticity: beyond nature and nurture. Johns Hopkins University Press, Baltimore
Poulton J, Winn AA (2002) Costs of canalization and plasticity in response to neighbors in Brassica rapa. Plant Species Biol 17:109–118
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
Rao MR, Nair PKR, Ong CK (1997) Biophysical interactions in tropical agroforestry systems. Agrofor Syst 38:3–50
Robson MJ, Ryle GJA, Woledge J (1988) The grass plant—its form and function. In: Jones MB, Lazenby A (eds) The grass crop. The physiological basis for production. Chapman and Hall, London, pp 22–83
Schmitt J (1997) Is photomorphogenic shade avoidance adaptive? Perspectives from population biology. Plant Cell Environ 20:826–830
Schmitt J, McCormac AC, Smith H (1995) A test of the adaptive plasticity hypothesis using transgenic and mutant plants disabled in phytochrome-mediated elongation responses to neighbours. Am Nat 146:937–953
Steel RGD, Torrie JH (1980) Principles and procedures of statistics, 2nd edn. McGraw-Hill, New York
Sultan SE (1995) Phenotypic plasticity and plant adaptation. Acta Bot Neerl 44:363–383
Valladares F, Sanchez-Gomez D, Zavala MA (2006) Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications. J Ecol 94:1103–1116
van Kleunen M, Fischer M, Schmid B (2000) Clonal integration in Ranunculus reptans: by-product or adaptation? J Evol Biol 13:237–248
van Soest PJ, Robertson JB, Lewis BA (1991) Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 74:3583–3597
Wahl S, Ryser P, Edwards PJ (2001) Phenotypic plasticity of grass root anatomy in response to light intensity and nutrient supply. Ann Bot 88:1071–1078
Weinig C (2000) Plasticity versus canalization: population differences in the timing of shade-avoidance responses. Evolution 54:441–451
Wherley BG, Gardner DS, Metzger JD (2005) Tall fescue photomorphogenesis as influenced by changes in the spectral composition and light intensity. Crop Sci 45:562–568
Wilson JR, Ludlow MM (1991) The environment and potential growth of herbage under plantations. In: Shelton HM, Stur WW (eds) Forages for plantation crops. ACIAR, Canberra, pp 10–24
Wilson JR, Hill K, Cameron DM, Shelton HM (1990) The growth of Paspalum notatum under the shade of a Eucalyptus grandis plantation canopy or in full sun. Trop Grassl 24:24–28
Winn AA, Evans AS (1991) Variation among populations of Prunella vulgaris L. in plastic responses to light. Funct Ecol 5:562–571
Zhai T, Mohtar RH, Gillespie AR, von Kiparski GR, Johnson KD, Neary M (2006) Modeling forage growth in a Midwest USA silvopastoral system. Agrofor Syst 67:243–257
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abraham, E.M., Kyriazopoulos, A.P., Parissi, Z.M. et al. Growth, dry matter production, phenotypic plasticity, and nutritive value of three natural populations of Dactylis glomerata L. under various shading treatments. Agroforest Syst 88, 287–299 (2014). https://doi.org/10.1007/s10457-014-9682-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-014-9682-9