Abstract
The propagation of most aquatic macrophytes occurs vegetatively, via fragments. After dispersal, the fragments may exhibit two survival strategies: colonization (the ability to develop roots) and regeneration (the ability to develop new propagules that can disperse). Invasive species, for example, Hydrilla verticillata, are known to have a high potential for dispersal and growth. We experimentally evaluated the colonization and regeneration abilities and growth rates of this species in the early stage of development and compared these properties to the corresponding characteristics of two native species of Hydrocharitaceae (Egeria densa and Egeria najas). The following hypotheses were tested: (1) fragments with apical tips have greater colonization and regeneration abilities and higher growth rates than fragments without apical tips and (2) H. verticillata has greater colonization and regeneration abilities and higher growth rates than other native species of Hydrocharitaceae. Our results showed that both types of fragments had the same ability to colonize, whereas fragments without tips had a higher ability to regenerate. However, fragments with apical tips showed higher growth rates. It is probable that these higher growth rates resulted from apical dominance. H. verticillata had the greatest propagation potential. Its colonization and regeneration abilities and growth rates were greater than those of the native species. These differences can produce competitive advantages for the exotic species in the early stages of development in new habitats and may contribute to understanding the success of the exotic compared to the native species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Submerged aquatic species are especially vulnerable to physical and biological disturbances associated, for example, with the water regime (Barrat-Segretain and Cellot 2007) and herbivory (Li et al. 2010). These disturbances help to propagate the plants by causing the release of a variety of vegetative plant parts, including entire plants, rhizomes, stolons, tubers, turions, and, more frequently, fragments (Riis and Sand-Jensen 2006). Fragments, which are mainly produced by the mechanical breakage of the plants, disperse from the original colonies and settle in the sediment (Madsen and Smith 1999). These fragments can exhibit two survival strategies: the development of roots and rapid establishment in the sediment (colonization) and the development of new propagules that can be dispersed (regeneration) (Southwood 1988; Barrat-Segretain and Bornette 2000).
Particular species may show a greater ability to colonize and regenerate in new environments. In certain cases, it is probable that these species will expand beyond their natural range and population density, thus becoming invasive (Richardson et al. 2000) and often causing ecological and economic damages (Vitousek et al. 1997; Pimentel et al. 2001). Hydrilla verticillata (L.f.) Royle, for example, has been one of the most problematic invasive species worldwide. This species is a submerged aquatic macrophyte, native to Asia and Australia (Cook and Lüond 1982) and considered “the perfect aquatic weed” (Langeland 1996) because it has many traits commonly attributed to invasive species, including rapid growth and reproductive and physiological characteristics that allow it to become a dominant species in a broad range of freshwater ecosystems (Cook and Lüönd 1982; Langeland 1996; Chadwell and Engelhardt 2008; Bianchini et al. 2010; Sousa 2011). In the Upper Paraná River Floodplain System—UPRFS, H. verticillata was first recorded in 2005 (Sousa et al. 2009). This record is most likely the first for South America. Previous studies have shown that this species is highly competitive (Van et al. 1999; Mony et al. 2007; Wang et al. 2008) and can compete with the native species Egeria densa Planchon and Egeria najas Planchon in the UPRFS (Sousa et al. 2010). These three species have very similar morphology (Fig. 1) and ecological features, and these similarities can cause intense competitive interactions among the species (Sousa et al. 2009). Observations in the habitats of the UPRFS indicate that these species begin to establish from small fragments.
A study similar to ours showed that fragments of H. verticillata have higher colonization and regeneration abilities than fragments of the native species after desiccation stress (Silveira et al. 2009). However, it is unknown whether fragments without apical tips, which are also frequently dispersed by flowing water and by birds, have the same colonization and regeneration abilities as those with tips. To better understand how the attributes of one exotic species can contribute to their invasiveness compared with native species, we conducted experiments with plant fragments to compare the colonization and regeneration abilities and growth rates of H. verticillata with the corresponding characteristics of two phylogenetically related native species, E. densa and E. najas. Based on the studies performed on other continents and in South America (Mony et al. 2007; Riis et al. 2009; Sousa et al. 2010), we tested the following hypotheses: (1) fragments with apical tips have greater colonization and regeneration abilities and higher growth rates than fragments without apical tips and (2) H. verticillata has greater colonization and regeneration abilities and higher growth rates than other native species of Hydrocharitaceae.
Materials and methods
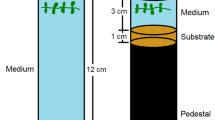
Samples of E. densa, E. najas, and H. verticillata were collected in the Itaipu Reservoir in April 2010. Healthy fragments were stored in open plastic containers with water collected at the same site and immediately transported to the laboratory. The fragments were washed in running tap water to remove material attached to the surface. In contrast to the material used by Silveira et al. (2009), we used two types of 10-cm vegetative fragments from each species: fragments with an apex (apical stem) and fragments without an apex (mid-stem). For each fragment type, we selected 15 replicates. In total, we analyzed 90 samples. Each fragment was individually placed in 0.8-L plastic containers (15.5 cm in height and 10 cm in diameter) containing tap water, which remained without changes throughout the experiment, and 3 cm of substrate (horticultural peat). The substrate used in this experiment was chosen because it did not limit plant growth, as shown by a pilot experiment. The fragments were individually incubated to avoid interspecific competition. They were not planted in the substrate but were simply dropped into the containers. As in the nature, the fragments sink to bottom and began to root after a few days (Langeland and Sutton 1980). The position of the plastic containers was randomized in the greenhouse. The physical and chemical parameters of the water were measured weekly. The plants were grown at a temperature of 17.86 ± 0.50 °C, a dissolved oxygen concentration of 10.15 ± 0.23 mg L−1, a pH of 8.78 ± 0.03 and a conductivity of 510.75 ± 18.74 μS cm−1 (mean ± standard deviation).
By 38 days after the beginning of the experiment, most fragments had rooted in the sediment and grown to the surface of the microcosm. We then terminated the experiment. We measured the number of roots, the dry weight (DW) of the roots, and the root ratio (calculated as the DW of the roots divided by the DW of the entire individual) to evaluate the colonization ability of the plant fragments. To assess the regeneration ability of the plant fragments, we measured the number of shoots (including any secondary shoots that emerged during the experiment), the DW of the shoots, and the shoot ratio (calculated as the DW of the secondary shoots divided by the DW of the entire individual). Root ratio and shoot ratio were used as the traits of growth in biomass accumulation and allocation. The plants were oven-dried at 60 °C until they reached a constant weight.
Relative growth rates (RGR) were calculated for the fragments alone (considering only the growth of the original fragments and excluding any shoots and roots that appeared during the experiment) and for the total biomass (considered as the total investment in the growth and production of new biomass, that is, the growth of the fragment plus new shoots and roots): RGR = (ln W f −ln W i )/tdays, where W f = final DW and W i = initial DW. The initial DW of the fragments was estimated from 15 fragments of each species with a size similar to those used in the experiment.
We used a two-way ANOVA to test the effect of the species and fragment type as well as the effect of the interaction between these factors on the colonization ability of the fragments (the number and the DW of the roots and the root ratio), the regeneration ability of the fragments (the number and the DW of the shoots and the shoot ratio), and fragments and total RGR. When ANOVA showed no significant interaction between species and fragment type, we just tested for the main effect and multiple comparisons of the attributes were performed using Fisher’s test. The level of significance of the tests was set at 0.05.
Results
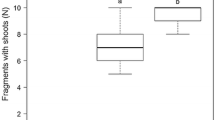
The number of shoots was significantly affected by the species and fragment type (Table 1). In general, the mid-stem fragments had a greater shoot number than the apical stems. H. verticillata had the highest number of shoots for both fragment types (Fig. 2a). The number of roots was affected only by the species (Table 1), and again, H. verticillata exhibited the highest number of roots for both fragment types, followed by E. najas (Fig. 2b).
Mean (± SE) shoot number (a), root number (b) shoot DW (c), root DW (d), shoot ratio (e), root ratio (f) fragment RGR (g), and total RGR (h) for species according to the type of fragment: apical stem and mid-stem. Different letters above columns indicate the significant differences between treatments at P < 0.05, following Fisher’s test. Black bar E. densa, White bar E. najas, and Gray bar, H. verticillata
The shoot DW was significantly affected by the species and fragment type (Table 1). The mid-stem fragments exhibited higher values of shoot DW, and E. densa had the highest values of shoot DW for both fragment types (Fig. 2c). The root DW was significantly affected only by species (Table 1). E. densa and E. najas had the highest values of root DW for both types of fragments, whereas H. verticillata had the lowest values (Fig. 2d).
The shoot ratio was significantly affected by the species and the fragment type (Table 1). The highest shoot ratios were observed in the mid-stem fragments. H. verticillata generally had the highest shoot ratios (Fig. 2e). The root ratio was affected only by the species (Table 1), and H. verticillata again showed higher values than the other two species (Fig. 2f).
Both the fragment RGR and the total RGR were significantly affected by the interaction between species and fragment type (Table 1). In general, higher values were obtained in the apical stems than in the mid-stem fragments. E. densa exhibited the lowest RGR for both fragments and its mid-stem fragment RGR differed significantly from the other species (Fig. 2g, h). The significant interaction indicates that the values for both the fragment RGR and the total RGR were lower in the mid-stem fragments of E. densa and E. najas but higher in H. verticillata (Figs. 2g, h).
Discussion
The results of this experiment only partially support the hypothesis that apical fragments have greater regeneration and colonization abilities and higher growth rates than mid-stem fragments. In fact, the apical stems exhibited higher growth rates than the mid-stems. However, there was no significant difference in colonization ability between fragment types because both types had, on average, the same root number, the same root DW, and the same root ratio. Moreover, mid-stem fragments exhibited a greater ability to regenerate than apical stems because they allocated more biomass to produce secondary shoots. These secondary shoots contributed to produce the highest values of shoot DW and shoot ratio. Similar results were found in previous studies with several species of aquatic macrophytes (Wu et al. 2007; Jiang et al. 2009, Riis et al. 2009). Unlike mid-stem fragments, apical fragments invested more in growth and presented higher values of fragment RGR, supporting the concept of apical dominance (Cline 1997), defined as the control exerted by the shoot apex over the outgrowth of the lateral buds in association with the investment made only in growth by the apex.
The results of the study indicated that although the species of Hydrocharitaceae have similar architecture, they differ in efficiency based on their colonization and regeneration ability. The exotic H. verticillata was more efficient compared with native species because it had higher numbers of shoots and roots for both fragment types, an additional factor that indicates its advantages over the native species and contributes to its ability to disperse and establish in novel sites. Despite the lower values of shoot and root DW shown by the exotic species, the proportions (shoot and root ratio) of these structures in relation to total biomass (including fragments) were more representative. Its adventitious roots, which are fine and filiform (Blackburn et al. 1969), gave it the lowest DW, but this morphology would not represent a disadvantage because fine elongated roots may be more efficient at nutrient uptake than thicker roots (Xie and Yu 2003; Xie et al. 2006). According to P’yankov and Ivanov (2000), the individual contribution of each plant part in relation to its total biomass can identify the primary strategy of the species. In this case, such individual contributions shown by the exotic species would indicate its investment in both survival strategies: colonization and regeneration. The native E. densa and E. najas had a lower number of sprouts and roots and lower proportions of these structures in relation to total weight but had higher values of DW. These differences are simply the consequences of the structure of these species, which have short internodes, large leaves, and thick roots (Blackburn et al. 1969).
Hydrilla verticillata deserves attention because it showed, in general, higher values of fragment RGR and total RGR. These findings clearly indicate that this species invests in the growth of its own vegetative fragments and in new structures (shoots and roots). The significant interaction between species and fragment type highlights the lower growth rates of the mid-stem of the native plants compared with H. verticillata (see Figs. 2g, h). Thus, our results supported our hypothesis that this exotic species has greater colonization and regeneration abilities and higher growth rates than the native species.
The simultaneous allocation of resources for colonization and regeneration and the greater regeneration ability of H. verticillata from mid-stem fragments compared with native plants may offer a competitive advantage to the exotic species because the fragmentation of stems certainly creates a greater number of mid-stem fragments than apices. Previous studies demonstrated a competitive advantage for H. verticillata over E. densa (Mony et al. 2007) and E. najas (Sousa et al. 2010) as well as other aquatic macrophytes with lower relative growth rates (Wang et al. 2008). Recent studies comparing only apical tips also demonstrated that fragments of H. verticillata regenerate more rapidly from desiccation stress than the native E. densa and E. najas (Silveira et al. 2009). These strategies allow this species to establish more rapidly than native species after disturbances. In fact, Sousa et al. (2010) found that after a very large flood in the Paraná River, E. najas and H. verticillata were able to regenerate at nearly the same time. However, the former species grew more rapidly than the latter.
Our experiment highlights species differences in colonization and regeneration ability. These results may be useful for better understanding the dynamics of establishment and the effects of exotic plants on native species and may also assist in management and ecological restoration. In this regard, the substantial regeneration ability of H. verticillata, regardless of fragment type, indicates that mechanical control techniques that result in the formation of fragments may in fact facilitate the invasion of novel sites by this species. The native plants showed poorer colonization and regeneration abilities. This result indicates that the control techniques that produce fragments would facilitate more the success of this exotic compared with the native species. However, studies in situ are needed to better determine the colonization and regeneration abilities of native and exotic species in natural ecosystems.
References
Barrat-Segretain MH, Bornette G (2000) Regeneration and colonization abilities of aquatic plant fragments: effect of disturbance seasonality. Hydrobiologia 421:31–39
Barrat-Segretain MH, Cellot B (2007) Response of invasive macrophyte species to drawdown: the case of Elodea sp. Aquat Bot 87:255–261
Bianchini I Jr, Cunha-Santino MB, Milan JAM, Rodrigues CJ, Dias JHP (2010) Growth of Hydrilla verticillata (L. f.) Royle under controlled conditions. Hydrobiologia 644:301–312
Blackburn RD, Weldon LW, Yeo RR, Taylor TM (1969) Identification and distribution of certain similar-appearing submersed aquatic weed in Florida. Hyacinth Control J 8:17–23
Chadwell TB, Engelhardt KAM (2008) Effects of pre-existing submersed vegetation and propagule pressure on the invasion success of Hydrilla verticillata. J Appl Ecol 45:515–523
Cline MG (1997) Apical dominance. Bot Rev 57:318–358
Cook CDK, Lüönd R (1982) A revision of the genus Hydrilla (Hydrocharitaceae). Aquat Bot 13:485–504
Jiang J, An S, Zhou C, Guan B, Sun Z, Cai Y, Liu F (2009) Fragment propagation and colonization ability enhanced and varied at node level after escaping from apical dominance in submerged macrophytes. J Integr Plant Biol 51(3):308–315
Langeland KA (1996) Hydrilla verticillata (L.F.) Royle (Hydrocharitaceae), “the perfect aquatic weed”. Castanea 61(3):293–304
Langeland KA, Sutton DL (1980) Regrowth of hydrilla from axillary buds. J Aquat Plant Manag 18:27–29
Li K, Liu Z, Gu B (2010) Compensatory growth of a submerged macrophyte (Vallisneria spiralis) in response to partial leaf removal: effects of sediment nutrient levels. Aquat Ecol 44:701–707
Madsen JD, Smith DH (1999) Vegetative spread of dioecious Hydrilla colonies of experimental ponds. J Aquat Plant Manag 37:25–29
Mony C, Koschnick TJ, Haller WT, Muller S (2007) Competition between two invasive Hydrocharitaceae (Hydrilla verticillata (L.f.) (Royle) and Egeria densa (Planch)) as influenced by sediment fertility and season. Aquat Bot 86:236–242
P’yankov VI, Ivanov LA (2000) Biomass allocation in boreal plants with different ecological strategies. Russ J Ecol 31(1):1–7
Pimentel D, McNair S, Janecka J et al (2001) Economic and environmental threats of alien plant, animal, and microbe invasions. Agric Ecosyst Environ 84:1–20
Richardson DM, Pyšek P, Rejmánek M, Barbour MG, Panetta D, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107
Riis T, Sand-Jensen K (2006) Dispersal of plant fragments in small streams. Freshw Biol 51:274–286
Riis T, Madsen TV, Sennels RSH (2009) Regeneration, colonization and growth rates of allofragments in four common stream plants. Aquat Bot 90:209–212
Silveira MJ, Thomaz SM, Mormul RP, Camacho FP (2009) Effects of desiccation and sediment type on early regeneration of plant fragments of three species of aquatic macrophytes. Int Rev Hydrobiol 94:169–178
Sousa WTZ (2011) Hydrilla verticillata (Hydrocharitaceae), a recent invader threatening Brazil’s freshwater environments: a review of the extent of the problem. Hydrobiologia 669:1–20
Sousa WTZ, Thomaz SM, Murphy KJ, Silveira MJ, Mormul RP (2009) Environmental predictors of the occurrence of exotic Hydrilla verticillata (L.f.) Royle and native Egeria najas Planch. in a sub-tropical river floodplain: the Upper River Paraná, Brazil. Hydrobiologia 632:65–78
Sousa WTZ, Thomaz SM, Murphy KJ (2010) Response of native E. najas Planch. and invasive Hydrilla verticillata (L. f.) Royle to altered hydroecological regime in a subtropical river. Aquat Bot 92:40–48
Southwood TRE (1988) Tactics, strategies and templets. Oikos 52:3–18
Van TK, Wheeler GS, Center TD (1999) Competition between Hydrilla verticillata and Vallisneria americana as influenced by soil fertility. Aquat Bot 62:225–233
Vitousek PM, D’Antonio CM, Loope LL, Rejmánek M, Westbrooks R (1997) Introduced species: a significant component of human-caused global change. N Z J Ecol 21(1):1–16
Wang JW, Yu D, Xiong W, Han YQ (2008) Above- and belowground competition between two submerged macrophytes. Hydrobiologia 608:113–122
Wu Z, Zuo J, Ma J, Wu J, Cheng S, Liang W (2007) Establishing submersed macrophytes via sinking and colonization of shoot fragments clipped off manually. Wuhan Univ J Nat Sci 12(3):553–557
Xie Y, Yu D (2003) The significance of lateral roots in phosphorus (P) acquisition of water hyacinth (Eichhornia crassipes). Aquat Bot 75:311–321
Xie Y, An S, Wu B, Wang W (2006) Density-dependent root morphology and root distribution in the submerged plant Vallisneria natans. Environ Exp Bot 57:195–200
Acknowledgments
C. A. Umetsu and H. B. A. Evangelista acknowledge the support of the Brazilian Council of Research (CNPq). We also thank Dr. Liesbeth Bakker and two anonymous reviewers that contributed substantially to improve our manuscript. S. M. Thomaz is especially thankful to CNPq for continuous funding through a Research Productivity Grant. This research was funded by CNPq/MCT through the Long-Term Ecological Research Program, site number 6, and Itaipu Binacional.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Liesbeth Bakker
Rights and permissions
About this article
Cite this article
Umetsu, C.A., Evangelista, H.B.A. & Thomaz, S.M. The colonization, regeneration, and growth rates of macrophytes from fragments: a comparison between exotic and native submerged aquatic species. Aquat Ecol 46, 443–449 (2012). https://doi.org/10.1007/s10452-012-9413-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-012-9413-0