Abstract
Native vessels–grafts biomechanical mismatch (BM) is related to graft failure. The BM could be reduced using human cryopreserved/defrosted arteries (cryografts), but post-thaw cryografts’ recovery could be associated with an impaired biomechanical behavior. In vitro, we demonstrated that our cryopreservation methods do not affect arteries’ biomechanics, but only post-implant studies would allow determining the cryografts’ biomechanical performance. Aim To characterize the biomechanical properties of implanted cryografts, and to compare them with cryografts pre-implant, recipients’ native arteries, and arteries from subjects with characteristics similar to those of the recipients and multiorgan donors (MOD) whose arteries were cryopreserved. Methods Native femoral arteries anastomosed to cryografts, implanted cryografts, and arteries from subjects, recipient-like and MOD-like, were studied. In vitro (pre-implant cryografts) and in vivo non-invasive studies were performed. Arterial pressure, diameter, and wall thickness were obtained to quantify local and regional biomechanical parameters, and to evaluate the arterial remodeling. Conclusion Implanted cryografts were remodeled, with an increased wall thickness, wall-to-lumen ratio, and wall cross-sectional area. The proximal–distal gradual transition in stiffness remained unchanged. Implanted cryografts were stiffer than MOD-like arteries, but more compliant than recipients’ arteries. The cryografts–native arteries biomechanical differences were lesser than those described for venous grafts or expanded polytetrafluoroethylene.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The biomechanical mismatch (BM) between the recipients’ native arteries and the vascular grafts has been related to the graft failure.16, 37 An alternative to reduce the BM would be the use of autologous arteries as vascular grafts. However, except in certain limited surgeries (i.e., coronary bypass), it is difficult to obtain autologous arteries of adequate length and size without the impairment of an essential tissue. In the lack of suitable autologous materials, cryopreserved/defrosted arteries (cryografts) could be used.1, 30 However, the post-thaw functional recovery of cryografts could be associated with an altered biomechanical performance.1, 5, 33 Related with this, we recently demonstrated, in in vitro studies, that the techniques of cryopreservation used in our national tissue bank do not significantly affect the biomechanical behavior of human veins and elastic and muscular arteries in any detrimental way.9, 10, 12 In addition, we demonstrated, in pre-implant studies, that cryopreserved arteries would allow reducing the BM with native arteries from potential recipients.13 However, only in vivo post-implant studies could give information about the cryograft’s functional capability once implanted and about its adaptation to the recipient vascular system. In addition, post-implant biomechanical studies could contribute to understanding the causes of the graft failure.
In this context, this study addresses the following main questions: (a) considering that cryografts could undergo post-implant structural changes: Are there differences in the biomechanical behavior of pre-implanted and implanted human cryografts? Are there biomechanical differences between implanted cryografts and arteries from subjects with characteristics similar to those of the multi-organ donors (MOD) whose arteries were cryopreserved? (b) Considering that ideally the implanted cryograft should mimic the biomechanical properties of the native vessels: In biomechanical terms, is the cryograft behavior similar to that of the recipient’s native arteries?
Thus, this work aims (1) to characterize local and regional biomechanical properties of implanted human cryografts and (2) to compare them with pre-implanted cryografts, adjacent native arteries, and arteries from subjects with characteristics similar to those observed in the recipients and in the MOD whose arteries were procured and cryopreserved.
This work contributes to the biomedical field not only due to the specific biological results shown but also due to the approach used to evaluate clinically the wall dynamics of implanted arterial homografts and their adaptation to the recipient vascular system.
Methodological Approach
Non-Invasive Biomechanical Studies
The study was approved by the ethics committee of the Universidad de la República (Uruguay). All subjects gave informed consent.
Groups
Nine implanted cryografts (length: 51 ± 21 cm; cryopreservation time: 98 ± 154 days; implant time at the study date: 557 ± 297 days) were studied (implanted cryografts group). In addition, in the recipients (n = 5; age: 66 ± 5 years; weight: 76 ± 9 kg; height: 1.7 ± 0.1 m; and body mass index: 26.2 ± 4.0 kg/m2), the femoral arteries (n = 9) anastomosed to the cryografts were studied (native arteries group). Cryografts were implanted as limb-salvage procedures, when there were no suitable autologous veins and polytetrafluoroethylene (PTFE) prosthesis were not recommended. Implanted cryografts, recipients, and MOD characteristics are summarized in Table 1.
Femoral arteries from 15 subjects (12 males), with characteristics (gender, age, race, and anthropometry) and hemodynamic conditions similar to those of the recipients (recipient-like group) but without bypass, arterial reconstructions, or peripheral arterial disease, were studied. In addition, 15 subjects (10 males), with characteristics (gender, age, race, and anthropometry) and hemodynamic conditions similar to those of the MOD whose arteries were procured (MOD-like group), were also studied.
The non-invasive biomechanical evaluations were the same for all the subjects. Before the biomechanical studies were carried out, the recipients were submitted to a clinical examination of the limb circulation and to a standard non-invasive ultrasound evaluation, performed by a trained physician, to confirm the cryograft’s permeability. Color Doppler imaging paired with duplex ultrasound was used.17, 36 The color Doppler imaging was used to inspect the cryografts, the proximal and distal anastomosis, and 4–5 cm of the inflow and outflow native arteries, allowing to localize possible lesions. Duplex ultrasound was then used to obtain Doppler waveforms of blood flow, especially at the sites of possible abnormal blood flow patterns. In case of lesions (i.e., stenosis), they were graded using information obtained from the Doppler waveforms (i.e., peak-systolic velocity, PSV; peak-systolic velocity ratio, V r). For instance, the V r (ratio between the PSV in the “altered” and in the “normal” segment) was used to quantify the severity of stenotic lesions. A V r ≥ 2 indicated a stenosis ≥50%, and a V r ≥ 4 indicated a stenosis ≥70–75%.17, 36 The absence of flow signals would indicate total occlusions with a high level of accuracy.
Measurements, Data Collection, and Analysis
Before being studied, the subjects were rested for 15 min in supine position. Then, heart rate and blood pressure from the left brachial artery (automated blood pressure monitor, Dynamap Compact TS, Johnson & Johnson Medical, Newport, UK) were recorded. Non-invasive vascular studies were done in accordance with the international recommendations.22 Each implanted cryograft was analyzed by means of its local (segments’ properties) and regional or whole biomechanical behavior (Fig. 1).22 To evaluate the local properties, measurements were taken at each 3–6 cm, starting at the cryografts’ proximal anastomoses. In this way, the local properties were “mapped” all along the cryograft (Fig. 1).
Left: Arteriography showing the cryograft implanted in the recipient lower limb. Anastomoses are indicated by arrows. Right-Top: Patient’s lower limbs. Right-Bottom: Schema of the cryografts and native arteries “local” and “regional” biomechanical analysis. Dashed lines indicate sites in which pressure, diameter, and wall thickness were “locally” recorded in the native vessel (#1) and in the cryografts (#2–10). Simultaneous pressure waveforms recording in segments 2 and 10 were used to quantify the cryograft pulse wave velocity (PWV)
The biomechanical properties of femoral arteries from recipient-like and MOD-like subjects, as well as the biomechanical behavior of the recipients’ femoral arteries (in close proximity to the cryograft), were analyzed (Fig. 1).
Local Biomechanical Properties
To evaluate the local properties, instantaneous pressure and diameter signals were obtained at the same site.4, 13, 22 Echographic studies were performed with a real-time B-mode ultrasound imager (30 images per second, 7.5-MHz probe; Portable Ultrasound System, model: Aloka SSD210, Aloka Co., Ltd, Tokyo, Japan).4 Fixed images at end-diastole and sequences of images were acquired to determine the intima-media arterial thickness (IMT) and the instantaneous diameter waveform, respectively (Fig. 2).19 Signals were analyzed off-line using an automated step-by-step algorithm applied to each digitized image.19 From each echographic arterial recording, two arterial sites (left and right) were analyzed.
(a) Proximal and distal pressure recordings used to quantify pulse wave velocity (PWV FF) in an implanted cryograft. Δt: time difference between the “foot” of the proximal and distal pressure waveforms. (b) Pressure and diameter waveforms obtained from a segment of an implanted cryograft. (c) Pressure–diameter relationship obtained from the pressure and diameter signals shown in (b)
Instantaneous pressure waveforms were recorded non-invasively with a strain gauge mechano-transducer (Motorola MPX 2050, Motorola Inc., Schaumburg, IL) (Fig. 2).4 The mean and the diastolic values of the brachial pressure were used to calibrate the pressure waveform.4, 22 Pressure and diameter waveforms were interpolated in time to obtain the same number of data points, obtaining an averaged cardiac cycle.4
To evaluate the arterial elastic behavior, different parameters were calculated which gave complementary information and allow comparing our data with reported results.26
The pressure–diameter elastic index (E pd) was calculated as the slope of a linear regression fit to the diastolic phase of the pressure–diameter relationship.8 To ensure an isobaric comparison with respect to the implanted cryografts, in arteries from the MOD-like group, the E pd was calculated at the recipients’ prevailing mean diastolic pressure.11 The E pd is determined by the arterial geometrical properties and the intrinsic stiffness of the arterial wall. Since it is calculated from the diastolic phase, which corresponds to the purely elastic pressure–diameter relationship, the E pd is a strong indicator of the arterial elastic properties (without considering the viscous behavior).
The incremental elastic modulus (E INC) was calculated as the slope of a linear regression fit to the diastolic phase of the instantaneous circumferential stress–strain relationship.8 It provides information about the arterial wall intrinsic properties regardless of its geometry and/or size. Thus, the E INC is used to determine changes in the arterial wall components. To quantify the E INC, the arterial strain (ε) and circumferential stress (σ) were calculated as
where R D is the diastolic midwall radius, R is the midwall radius, r e is the measured external radius, and r i is the internal radius. The arterial σ–ε relationship was constructed, and the E INC was calculated as
(considering the diastolic stress–strain purely elastic relationship).13
In addition, the arterial wall stiffness was expressed in terms of the pressure–strain or Peterson modulus (E P) and the stiffness index (β), two indexes commonly used in the clinical practice, since they can be obtained just using systolic (or maximal) and diastolic (or minimal) pressure and diameter values.
They were calculated as
where SP and DP are the systolic and diastolic arterial pressure levels, respectively, and SD and DD are the systolic and diastolic arterial external diameters, respectively.
where ln is the natural logarithm.
Both parameters (E P and β) depend on the arterial geometry and intrinsic wall properties. However, while the E P assumes a linear pressure–diameter relationship, β considers that the relationship can be fitted by a logarithmic function.
Finally, the pulse wave velocity was calculated using the Moens–Korteweg’s equation (PWV MK):
where h is the mean wall thickness and ρ a is the wall density (assumed equal to 1.06 g cm−3).13, 29
To evaluate the arterial remodeling, four parameters were quantified for each femoral segment analyzed: the mean internal diameter, the wall thickness, the wall cross-sectional area (CSA, CSA = 3.1416 * Wall thickness * (Wall thickness + mean internal diameter), and the wall-to-lumen ratio (wall thickness/mean internal diameter).15, 27, 34
Regional Biomechanical Properties: The Cryograft as a Whole
The foot-to-foot pulse-wave velocity (PWV FF) was used to analyze the regional cryograft’s properties. It was calculated as the quotient between the proximal–distal pulse transit time difference and the distance between the sites (Fig. 1). To measure the pulse transit time, the pressure waveforms were simultaneously recorded in the cryograft’s proximal and distal segments, using high-fidelity strain gauges mechano-transducers (Motorola MPX 2050, Motorola Inc., Schaumburg, IL) (Fig. 2). Distance was measured on the body surface using a tape. In the recipients and MOD-like subjects, the femoral artery-dorsalis pedis or post-tibial artery PWV FF was measured.6
Cryopreservation and In Vitro Biomechanical Evaluation of Cryografts Pre-Implant
Donor Criteria Selection
All procedures took into account ethical and safety concerns for therapeutic use, including consent documentation according to No. 14005 and No. 17668 legal rules (Uruguay). The general and particular exclusion criteria for vascular tissue procurement agreed with the International Atomic Energy Agency (IAEA, International Standards for Tissue Banks) and the American (AATB) and European (EATB) Association for Tissue Banking.
Tissue Procurement
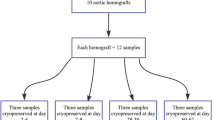
Segments (5 cm in length) from both right and left femoral arteries were harvested from MOD (n = 12), selected in order to match the characteristics (gender, age, race, and anthropometry) of MOD whose arteries were cryopreserved and implanted. The arterial segment of 5 cm length was accurately measured with a caliper and marked with two suture stitches. After that, the arteries were excised at the level of the suture stitches and, after vascular removal and cryopreservation, the segments were mounted (in the in vitro setup) preserving the in vivo length.4, 9, 10 The techniques used to procure the arteries were similar to those used in the implanted cryografts. After harvesting, the segments were washed with saline solution (NaCl 0.9 g%) and stored at 4 °C in a saline solution with gentamicin (16 mg%), cefuroxime (300 mg%), penicillin G (400.000 IU%), and fluconazol (8 mg%). The warm ischemia time was 53–67 min (Mean = 61 min), and the cold ischemia time was 24–48 h (Mean = 34 h). After 24–48 h the segments were cryopreserved.
Cryopreservation
After the incubation in saline-antibiotic solution, the samples were placed in a sterile bag (volume: 350 cc) containing 85 cc of cryopreservant solution: Culture medium (RPMI 1640): 85%; human albumin solution (20%): 5%; and dimethylsulfoxide (DMSO, cryoprotectant): 10%. The bag was sealed hermetically at vacuum (Mod. 011342, Joisten and Kettenbaum, Bereisch Gladbach, Germany) in a laminar flow cabinet (Microflow, Laminar Flow Work Station, MDH Ltd, Andover Hants, England, SP.10.5.AA) and was equilibrated for 30 min at 20 °C. Then, a programmed cryopreservation procedure was carried out in a controlled rate freezing system (Model 9000, Gordinier Electronics, Inc., Roseville, MI).
For the cooling process, we chose a protocol based on Pegg et al. 31 It comprised three operative time steps. First, a slow programmed cooling rate (2 °C/min) until −40 °C. Second, a slow programmed cooling rate (5 °C/min) until −90 °C. Third, a rapid cooling rate, obtained by transferring the bag to the gaseous phase of the liquid nitrogen compartment (−142 °C). Then, the segments were stored for 30 days at −142 °C (Mark III, Temperature and Liquid Level Controller, Taylor-Wharton, Theodore, AL). After the storage, arteries were thawed. The warming protocol comprised a two-stage process, also taking into account of the Pegg et al.’s work, in which the importance of the thawing rate in the avoidance of the elastic fibers fractures was analyzed.31 Our first thawing step comprised a slow process achieved by transferring the bag from the nitrogen gaseous phase to the room temperature (20 °C) during 30 min. After that, the bag was rapidly transferred to a 40 °C water bath until the segments were completely thawed (second warming step). To reduce the cell damage determined by osmotic changes (i.e., cell dehydration or intracellular ice formation), after thawing, the cryoprotectant solution was gradually removed in four steps (10 min each) by immersion in tapered concentrations (10, 5, 2.5, and 0% of DMSO). Finally, the arterial segments were sent, in saline solution, to be tested biomechanically.
In Vitro Biomechanical Evaluation of the Cryografts
Each segment was non-traumatically mounted (at in vivo length) on specifically designed cannulas of a flow circuit loop (circulation mock). A full description of the device has been published.4, 12 Once mounted, the segment remained immersed and perfused with thermally regulated (37 °C), oxygenated Tyrode’s solution, with pH = 7.4. To measure pressure, the segment was instrumented with a pressure micro transducer (1200 Hz frequency response, Konigsberg Instruments, Inc., Pasadena, CA). To measure the segment’s external diameter, a pair of ultrasonic crystals (5 MHz, 2 mm diameter) was sutured to the adventitia. The transit time of the ultrasonic signal (1580 m/s) was converted into distance by means of a sonomicrometer (1000 Hz frequency response, Triton Technology Inc. San Diego, CA). Optimal positioning of the dimensional gauges was assessed by an oscilloscope (model 465B; Tektronix 3). Before data collection, the segments were allowed to equilibrate for 10 min under steady flow and pressure conditions.
Data Acquisition and Analysis
Diameter and pressure waves were measured under dynamic conditions, displayed in real time, digitized every 5 ms, and stored for off-line analysis. Approximately, 20–30 consecutive beats were sampled and analyzed. Pressure and stretching rate levels similar to those observed in the recipients were chosen. In all cases, the pump and tubing resistance were regulated so as to generate adequate pressure waveforms.12, 13
At the end of the experiments, the cryograft was weighted for volume calculation. The wall thickness (h) was calculated as
where r e and r i are the external and internal arterial radius, respectively. The r i was calculated as
where L is the in vivo length and V is the arterial volume, calculated by dividing the segment weight measurement by an assumed density of 1.06 g cm−3. Assuming an unchangeable arterial wall volume, r i and h can be instantaneously calculated.13
A computerized procedure was used to determine the pressure–diameter and circumferential stress–strain loops, and to calculate the biomechanical parameters described above: E pd, E INC, E P, β, and PWV MK.4, 11, 13 Like in the in vivo studies, in order to evaluate the arterial wall remodeling, the mean internal diameter, wall thickness, wall cross-sectional area, and wall-to-lumen ratio were determined.15, 27, 34
Statistical Analysis
Values were expressed as mean value ± standard deviation (MV ± SD). All comparisons with respect to the implanted cryografts group were done using two-tailed unpaired Student t-test. Linear regression analysis was done to determine the relationship between implanted cryografts’ local (E pd) and regional (PWV FF) biomechanical parameters, and between implanted cryografts’ local or regional biomechanical parameters and the parameters used to characterize the arterial remodeling. A p < 0.05 was considered significant.
Results
All the cryografts, anastomoses, and native arteries were permeable and without significant stenotic sites.
Differences in the Biomechanical Behavior of Pre-Implanted and Implanted Human Cryografts
Studied at similar pressure and heart rate levels (isobaric and isofrequency comparisons), the implanted and pre-implanted cryografts showed a similar mean internal diameter, indicating that there was neither significant luminal dilatation nor constriction after the implant. However, compared with the pre-implanted cryografts, the implanted ones showed higher levels of wall thickness (70%), CSA (82%), and wall-to-lumen ratio (68%) (p < 0.05), indicating post-implant non-inward and non-outward (no changes in luminal diameter), hypertrophic (increased CSA) remodeling (Table 2).
The remodeling was associated with a reduction in the maximal or systolic circumferential stress that supports the implanted cryografts’ wall (p < 0.05) (Table 2). In addition, despite the post-implant wall remodeling, when the wall stiffness was analyzed (with independence on the biomechanical parameter considered), its levels were lesser in the implanted cryografts than in the pre-implanted cryografts (p < 0.05) (Table 2).
In the implanted cryografts, the proximal–distal gradual transition in the arterial wall properties was preserved with an increase in wall stiffness and a reduction in mean internal diameter toward the cryografts’ distal end (Fig. 3).
Arterial elasticity (evaluated using the elastic pressure–diameter index) and mean diameter for the native arteries, anastomoses, and implanted cryograft from the patient shown in Fig. 1. Note that native femoral arteries were stiffer than the cryograft and long after the implantation, the physiological gradual transition of the arterial diameter (tapering) and biomechanical properties remained unchanged among central and peripheral segments of the remodeled cryograft. From each mode-B echographic arterial recording (n = 10), two arterial sites (left and right) were analyzed. Thus, 20 arterial segments were characterized
Biomechanical Differences Between Implanted Cryografts and Arteries from Subjects Similar to Donors
When the implanted cryografts were compared with arteries from MOD-like subjects, we found no significant differences in mean internal diameter, but the implanted cryografts showed larger wall thickness (53%), wall-to-lumen ratio (60%), and wall CSA (52%) (p < 0.05) (Table 2). These results also support a post-implant remodeling of the cryograft (Table 2). Despite the maximal circumferential stress tended to decrease in the remodeled grafts, the values remained higher (26%) in the implanted cryografts than in MOD-like arteries. This could be related with the higher blood pressure levels of the recipients’ vascular system (hypertensive patients). Briefly, despite the post-implant changes in the cryografts, the wall circumferential stress levels did not reach normotensive values.
On the other hand, when the arterial (E pd, E P, PWV MK, and β) and the wall intrinsic (E INC) stiffness were evaluated, the implanted cryografts showed higher stiffness with respect to MOD-like subjects (p < 0.05). Since isobaric and isofrequency comparisons were performed, the higher stiffness levels found in the implanted cryografts could not be attributed to pressure or heart rate differences.
As can be seen in Fig. 4, in the implanted cryografts, the parameters related with the wall remodeling process were associated with the local (E pd) and regional (PWV FF) stiffness levels. The larger the cryograft’s remodeling, the higher the stiffness levels (Fig. 4).
Relationship between implanted cryografts’ remodeling parameters and local (E pd) and regional (PWV FF) stiffness levels. In the implanted cryografts, higher levels of wall thickness, wall-to-lumen ratio, and CSA, and lesser levels of mean internal diameter, were positively associated with higher levels of E pd and PWV FF (p < 0.05)
In the implanted cryografts, the PWV FF was 15 ± 2 m/s and in the recipients’ lower limb, in which the cryograft was implanted, it was 16 ± 2 m/s. Both values were higher than the PWV FF found in the MOD-like subjects (9 ± 2 m/s) (p < 0.05). In addition, it is to note that the increase in the local stiffness of the implanted cryograft’s femoral segment was directly associated with the regional or global increase in stiffness (Fig. 5).
Biomechanical Matching Between Cryografts and Native Arteries
There were no significant differences in mean internal diameter, wall thickness, wall-to-lumen ratio, and wall CSA between implanted cryografts and recipients’ native arteries, indicating an adequate geometrical matching between native arteries and implanted cryografts (Table 2). In addition, the implanted cryografts and recipients’ arteries showed similar maximal circumferential stress levels (Table 2).
The arterial and wall intrinsic stiffness were lesser in the implanted cryografts than in the recipients’ native arteries (Table 2). The differences in the biomechanical properties between native arteries and cryografts determine a “biomechanical mismatch” in the anastomotic region. However, the biomechanical mismatch would be lesser than those found when venous grafts or synthetic prosthesis are used.
When implanted cryografts were compared to femoral arteries from subjects with characteristics similar to those of the recipients, but without bypass, arterial reconstructions, or peripheral arterial disease, the cryografts showed a lesser internal diameter (18%), but higher wall thickness (23%) and wall-to-lumen ratio (51%), indicating that the remodeling found in the cryografts could not be explained only by the hypertensive conditions found in the recipients (p < 0.05) (Table 2).
In addition, in the implanted cryografts, the wall stiffness and circumferential stress were lesser than those found in the arteries from recipient-like subjects (p < 0.05) (Table 2). At the light of these results, it could be said that the pattern of wall remodeling in the implanted cryografts is not equal to that found in hypertensive subjects.
Discussion
Post-Implant Changes in Cryografts’ Biomechanics
Compared to pre-implant cryografts, the implanted grafts showed higher wall thickness, wall-to-lumen ratio, and wall CSA, and a lesser wall stiffness (Table 2). We previously demonstrated, in human arteries, that after excision there is an increase in wall stiffness.3, 4 Then, the higher stiffness found in the pre-implanted cryografts was, in some way, expected. However, our results showed, for the first time, that after implantation there was a remodeling of the cryograft, and a reduction in its stiffness, reaching levels more alike (but not identical) to those found in the MOD-like group. The observed increase in wall thickness, wall-to-lumen ratio, and arterial wall CSA agrees with previous works that described an increase in vascular grafts’ wall thickness early after implantation.7, 28
Taking into account that the increase in the implanted cryograft’s wall thickness was generalized, the PWVFF measured in the implanted cryografts was higher than in MOD-like subjects and higher than that expected in healthy subjects (approximately 10.1 m/s),6 and that the local and regional increases in the cryografts’ stiffness were positively associated (Fig. 5), the cryografts’ remodeling could not be considered mainly as a local process related with the anastomosis confection and/or with the cryograft–native artery biomechanical mismatch. In addition, it should be noted that the level of remodeling—evaluated by means of the wall thickness, the wall-to-lumen ratio, and the wall CSA—was directly and positively associated with the local and regional stiffness’ levels found in the cryografts (Fig. 4).
The global changes in the implanted cryografts could be part of an adaptative and/or a pathological response. For instance, it is known that arteries’ remodeling could be a homeostatic response that contributes to keep reduced the circumferential and shear stress levels (i.e., during changes in pressure and/or flow velocities).20 Thus, the implanted cryografts’ remodeling could be considered as a response to the new hemodynamic (hypertensive) conditions, which allowed reducing the maximal circumferential stress. Related with this, it is to note that the maximal circumferential stress was larger in the cryografts pre-implant than in the implanted ones, which, in turn, had a circumferential stress similar to that of the recipients’ arteries (Table 2).
However, note that the implanted cryografts’ maximal circumferential stress was lesser than that of the recipient-like arteries, as a consequence of the great increase in wall thickness observed in the implanted cryografts. Related with this, it is to note that the wall thickness and the wall-to-lumen ratio were higher in the implanted cryografts than in arteries from recipient-like subjects despite the recipient-like subjects also had arteries with an increased wall thickness (Table 2).3 The same could be said for the recipients’ native arteries. Then, the increase in wall thickness found in implanted cryografts was higher than that expected if only hemodynamic factors were the determinants of the cryograft remodeling. Furthermore, the differences between the intima-media thickness of the femoral portion of the implanted cryografts (1.00 ± 0.26 mm) and that of femoral arteries from recipient-like subjects (0.81 ± 0.07 mm) and MOD-like subjects (0.66 ± 0.04 mm) were similar to those reported by Cheng et al. for femoral arteries from subjects with (1.05 ± 0.39 mm) and without (0.69 ± 0.34 mm) peripheral vascular disease.18 Thus, the changes in the implanted cryografts’ wall could not be only ascribed to changes in the hemodynamic conditions, but factors related with the recipients’ vascular disease should be also considered. Related with this, it is known that an increased arterial wall thickness is an early morphological evidence of blood vessels and vascular grafts disease.2, 14, 26, 32, 35 Thus, the remodeling found in the implanted cryografts could represent the initial phase of the vascular disease in the cryograft (“graft vasculopathy”).26, 32 Anyway, to determine the mechanisms responsible for the cryograft remodeling was beyond the scope of our work.
Post-Implant Cryografts’ Proximal–Distal Transition in the Biomechanical Properties
As is widely known in the healthy arterial system, there is a gradual change in size and biomechanical properties, with an increase in the arterial impedance and stiffness toward the periphery, but without great differences between consecutive segments.23, 29 Hence, in physiological conditions, the impedance and wall stiffness matching between consecutive arterial segments are high.23 The gradual change in the arterial properties and the maintenance of adequate levels in each particular segment are important for a particular arterial pathway and for the entire cardiovascular system since it minimizes: (a) the left ventricle afterload and the arterial wall mechanical erosion, (b) the generation of wave reflection sites near the heart, keeping the ventricle afterload reduced, and (c) the generation of local vascular disturbances that have been associated with the development of vascular alterations (i.e., luminal obstruction by intimal hyperplasia).16, 23
Our results showed that in the implanted cryografts, after a mean time of approximately 550 days, the gradual proximal–distal transition in diameter and biomechanical properties and thus, their associated benefits (reduced local BM, pulse wave amplification, and nonphysiological wave reflection) were ensured (Fig. 3).16, 37
Implanted Cryograft–Recipient Arterial System Coupling
Our results demonstrated that after the implantation there were no significant differences in the geometrical properties of the implanted cryografts and the recipients’ native arteries, indicating an adequate geometrical matching between native arteries and implanted cryografts. This is an important finding since geometrical differences between the vascular grafts and the native arteries could determine alterations in the blood flow pattern, and consequently hemodynamic disturbances in the anastomotic region.
In addition, our results demonstrated that after the implant, the cryograft–native arteries BM was lesser than that expected if synthetic prosthesis (i.e., ePTFE) or venous grafts were implanted.4 Previously, we demonstrated, in pre-implant studies, that the cryograft–native vessel BM was lesser than that found for vascular substitutes currently available.9, 13 In this work we demonstrated that after approximately 19 months from the implant, in geometrical and biomechanical terms, the remodeled cryografts remained advantageous, with respect to ePTFE prosthesis and venous grafts.
Clinical Relevance
At least two issues should be emphasized taking into account their meaning in the biomedical field. First, nowadays, a good-quality autologous vein provides the best results for infra-inguinal bypass. However, in a considerable number of patients, with critical ischemia, veins are not available, and since the use of a prosthetic graft (i.e., expanded polytetrafluoroethylene, ePTFE, prosthesis) in the environment of chronic leg ulcerations is associated with a high risk of infection, alternative vascular substitutes are necessary.21, 25 In this context, this work results add advances in this area, evidencing that implanted arterial cryografts ensure an acceptable biomechanical behavior after months or years post-implant, and that cryografts–native arteries BM levels would be lesser than those found when synthetic prosthesis (i.e., ePTFE) or venous grafts (i.e., saphenous veins) are used.13 Despite the cryografts’ wall remodeling, it could represent an early phase of the cryograft vasculopathy26; our results evidenced that months or years after the implant the cryografts showed wall stiffness levels that would allow ensuring an adequate functional capability in the recipient arterial system.
Second, the periodic surveillance of implanted vascular grafts is considered important since early identification of failing grafts could avert impending graft failure and/or improve the secondary bypass or vascular access graft patency rate. In the surgical field, the measurement of blood flow is the method of choice for the evaluation of the functional status of lower limb bypass and/or vascular accesses used in hemodialysis. Since the ultrasound measurements are non-invasive, provide rapid access, give accurate data, and have relatively low costs, they have been advocated as the primary screening techniques in the surveillance of vascular grafts and in the identification of bypass or vascular access complications. Ultrasound studies allow to evaluate the grafts’ blood flow conduit capability and to diagnose lesions and/or stenoses in the native vessels or vascular substitutes,24 but they do not allow evaluating the graft biomechanical behavior and/or its biomechanical adaptation to the recipient cardiovascular system. Moreover, despite homograft’s biomechanical behavior could be considered as a predictor of its functional adaptation and that biomechanical factors are recognized as main determinants of vascular graft failures, in our knowledge, there are no works evaluating the post-implant cryografts’ biomechanical performance and/or adaptation to the recipient cardiovascular system. In this context, and taking into account of our results, we think that an adequate post-implant evaluation of the vascular grafts should include not only the analysis of their permeability but also the study of the grafts and native vessels biomechanical and functional characteristics, which considering our methodological approach is feasible.
Conclusion
In this work, for the first time, the wall remodeling and the local and regional biomechanical properties of implanted human arterial cryografts were non-invasively analyzed.
After the implant, the cryografts remodeled with an increase in the wall thickness, wall-to-lumen ratio, and wall CSA, being the wall remodeling in each cryograft directly and positively associated with the local and regional stiffness levels.
After the implant, the cryografts showed a lesser stiffness. In addition, even after isobaric analyses, implanted cryografts were stiffer than arteries from the MOD-like group. Thus, the implantation would determine a reduction in the graft stiffness, but without reaching the values expected in vivo (in the MOD cardiovascular system).
The implanted cryografts were more compliant than the recipients’ native arteries, despite their similitude in wall thickness, wall-to-lumen ratio, arterial wall CSA, and mean internal diameter. The biomechanical differences between cryografts and native vessels were lesser than those described for venous grafts or ePTFE prosthesis.
Long after the implantation, the physiological proximal–distal gradual transition in the biomechanical properties remained unchanged in the remodeled cryograft, ensuring an adequate biomechanical function of the graft.
Abbreviations
- BM:
-
Biomechanical mismatch
- CSA:
-
Wall cross-sectional area
- E INC :
-
Incremental elastic modulus
- IMT:
-
Intima-media thickness
- MOD:
-
Multi-organ donors
- PSV:
-
Peak-systolic velocity
- V r :
-
Peak-systolic velocity ratio
- E P :
-
Peterson modulus
- E pd :
-
Pressure–diameter elastic index
- PWV FF :
-
Pulse wave velocity calculated using the foot-to-foot method
- PWV MK :
-
Pulse wave velocity calculated using the Moens–Korteweg’s equation
- β :
-
Stiffness index
- ε :
-
Strain
References
1. Albertini, J., X. Barral, A. Branchereau, J. Favre, H. Guidicelli, J. Magne, P. Magnan. 2000. Long-term results of arterial allograft below-knee bypass grafts for limb salvage: a retrospective multicenter study. J Vasc Surg 31: 426-35. doi:10.1016/S0741-5214(00)90301-X
Allan, P.L., P.I. Mowbray, A.J. Lee, F.G. Fowkes. 1997. Relationship between carotid intima-media thickness and symptomatic and asymptomatic peripheral arterial disease. The Edinburgh Artery Study. Stroke 28: 348-53.
Armentano, R.L., J.G. Barra, D. Bia, F.M. Pessana, S. Graf, D. Craiem, L.M. Brandani, H.P. Baglivo, R.A. Sanchez. 2006. Smart damping modulation of carotid wall energetics in human hypertension: effects of angiotensin-converting enzyme inhibition. Hypertension. 47: 384-90. doi:10.1161/01.HYP.0000205915.15940.15
4. Armentano, R. L., J. G. Barra, F. M. Pessana, D. O. Craiem, S. Graf, D. B. Santana and R. A. Sanchez (2007) Smart smooth muscle spring-dampers. Smooth muscle smart filtering helps to more efficiently protect the arterial wall. IEEE Eng Med Biol Mag 26:62–70. doi:10.1109/MEMB.2007.289123
5. Arnaud, F. 2000. Endothelial and smooth muscle changes of the thoracic and abdominal aorta with various types of cryopreservation. J Surg Res 89: 147-151. doi:10.1006/jsre.2000.5816
Avolio, A.P., F.Q. Deng, W.Q. Li, Y.F. Luo, Z.D. Huang, L.F. Xing, M.F. O’Rourke. 1985. Effects of aging on arterial distensibility in populations with high and low prevalence of hypertension: comparison between urban and rural communities in China. Circulation. 71: 202-10.
7. Beard, J.D., J. Fairgrieve. 1986. Compliance changes in in situ femoropopliteal bypass vein grafts. Br J Surg. 73: 196-9. doi:10.1002/bjs.1800730313
Bia, D., R. Armentano, D. Craiem, J. C. Grignola, F. Ginés, A. Simon, J. Levenson. 2004. Smooth muscle role on pulmonary arterial function during acute pulmonary hypertension in sheep. Acta Physiol Scand. 181: 359-66. doi:10.1111/j.1365-201X.2004.01294.x
Bia, D., R. Armentano, Y. Zócalo, H. Pérez, E. Fischer, S. Graf, M. Saldías, W. Silva, I. Alvarez. 2007. Functional properties of fresh and cryopreserved carotid and femoral arteries, and venous and synthetic grafts: comparison with arteries from normotensive and hypertensive patients. Cell Tissue Bank. 8: 43-57. doi:10.1007/s10561-006-9000-5
Bia, D., F. Pessana, R. Armentano, H. Pérez, S. Graf, Y. Zócalo, M. Saldías, N. Pérez, O. Alvarez, W. Silva, D. Machin, P. Sueta, S. Ferrin, M. Acosta, I. Alvarez. 2006. Cryopreservation procedure does not modify human carotid homografts mechanical properties: an isobaric and dynamic analisys. Cell Tissue Bank. 7: 183-94. doi:10.1007/s10561-005-0655-0
Bia, D., Y. Zócalo, R. Armentano, J. Camus, E. de Forteza, E. Cabrera-Fischer. 2008. Increased reversal and oscillatory shear stress cause smooth muscle contraction-dependent changes in sheep aortic dynamics: role in aortic balloon pump circulatory support. Acta Physiol (Oxf). 192: 487-503. doi:10.1111/j.1748-1716.2007.01765.x
Bia, D., Y. Zocalo, F. Pessana, R. Armentano, H. Perez, M. Saldias, I. Alvarez. 2005. Femoral arteries energy dissipation and filtering function remain unchanged after cryopreservation procedure. Transpl. Int. 18: 1346-55. doi:10.1111/j.1432-2277.2005.00220.x
Bia, D., Y. Zócalo, F. Pessana, R. Armentano, H. Pérez, M. Saldías, I. Alvarez. 2007. Differential functional coupling between human saphenous cryoallografts and arteries: importance of the arterial type and the biomechanical parameter evaluated. Artif Organs 31: 809-18. doi:10.1111/j.1525-1594.2007.00467.x
Bots, M.L., A. Hofman, D.E. Grobbee. 1994. Common carotid intima-media thickness and lower extremity arterial atherosclerosis. The Rotterdam Study. Arterioscler. Thromb. 14:1885-1891.
Boutouyrie, P., C. Bussy, P. Lacolley, X. Girerd, B. Laloux, S. Laurent. 1999. Association between local pulse pressure, mean blood pressure, and large-artery remodeling. Circulation 100:1387-1393.
Cabrera, E.I., D. Bia, G.L. Cassanello, Y. Zócalo, E.V. Crawford, R.F. Casas, R.L. Armentano. 2005. Reduced elastic mismatch achieved by interposing vein cuff in expanded polytetrafluoroethylene femoral bypass decreases intimal hyperplasia. Artif Organs 29: 122-130. doi:10.1111/j.1525-1594.2005.29023.x
Casciani, T., M. A. Bettmann, A. S. Gomes, J. H. Grollman, S. R. Holtzman, J. F. Polak, D. Sacks, J. Schoepf, W. Stanford, M. Jaff, and G. L. Moneta. Expert panel on cardiovascular imaging. Follow-up of lower extremity arterial bypass surgery. [online publication]. Reston, VA: American College of Radiology (ACR), 2005, 6 pp.
Cheng, K.S., A. Tiwari, C.R. Baker, R. Morris, G. Hamilton, A.M. Seifalian. 2002. Impaired carotid and femoral viscoelastic properties and elevated intima-media thickness in peripheral vascular disease. Atherosclerosis. 164: 113-20. doi:10.1016/S0021-9150(02)00042-4
Craiem, D., G. Chironi, J. Gariepy, J. Miranda-Lacet, J. Levenson, A. Simon A. 2007. New monitoring software for larger clinical application of brachial artery flow-mediated vasodilatation measurements. J Hypertens. 25: 133-140. doi:10.1097/HJH.0b013e3280109287
Dobrin, P.B. 1995. Mechanical factors associated with the development of intimal and medial thickening in vein grafts subjected to arterial pressure. A model of arteries exposed to hypertension. Hypertension. 26: 38-43.
Dosluoglu, H.H., J. Kittredge, G.S. Cherr. 2008. Use of cryopreserved femoral vein for in situ replacement of infected femorofemoral prosthetic artery bypass. Vasc Endovascular Surg. 42: 74-8. doi:10.1177/1538574407308204
Laurent, S., J. Cockcroft, L. Van Bortel, P. Boutouyrie, C. Giannattasio, D. Hayoz, B. Pannier, C. Vlachopoulos, I. Wilkinson, H. Struijker-Boudier. 2006. European Network for Non-invasive Investigation of Large Arteries. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 27: 2588-2605. doi:10.1093/eurheartj/ehl254
Li, J.K. 2005. The Arterial Circulation: Physical Principles and Clinical Applications. In: Li JK (Eds) Human Press Inc. Totowa, NJ
Lopot, F., B. Nejedlý, S. Sulková, J. Bláha. 2004. Comparison of different techniques of hemodialysis vascular access flow evaluation. J Vasc Access. 5: 25-32.
Matia, I., L. Janousek, T. Marada, M. Adamec. 2007. Cold-stored venous allografts in the treatment of critical limb ischaemia. Eur J Vasc Endovasc Surg. 34: 424-31. doi:10.1016/j.ejvs.2007.04.005
Mitchell, R.N., P. Libby. 2007. Vascular remodeling in transplant vasculopathy. Circ Res. 100: 967-78. doi:10.1161/01.RES.0000261982.76892.09
Mulvany, M.J. 1999. Vascular remodelling of resistance vessels: can we define this? Cardiovasc Res. 41: 9-13. doi:10.1016/S0008-6363(98)00289-2
Murphy, G.J., G.D. Angelini. 2004. Insights into the pathogenesis of vein graft disease: lessons from intravascular ultrasound. Cardiovasc Ultrasound. 21; 2-8.
Nichols, W. W. and M. F. O’Rourke (2005) McDonald’s Blood Flow in Arteries Theoretical, Experimental and Clinical Principles, 5th edn. London, UK: Edward Arnold, pp. 233–267
Pascual, G., F. Jurado, M. Rodríguez, C. Corrales, P. López-Hervás, J.M. Bellón, J. Buján. 2002. The use of ischaemic vessels as prostheses or tissue engineering scaffolds after cryopreservation. Eur J Vasc Endovasc Surg. 24: 23-30. doi:10.1053/ejvs.2002.1663
Pegg, D.E., M.C. Wusteman, S. Boylan. 1997. Fractures in cryopreserved elastic arteries. Cryobiology 34: 183-192. doi:10.1006/cryo.1996.1997
Rahmani, M., R.P. Cruz, D.J. Granville, B.M. McManus. 2006. Allograft vasculopathy versus atherosclerosis. Circ Res. 99: 801-815. doi:10.1161/01.RES.0000246086.93555.f3
Rosset, E., A. Friggi, G. Novakovitch, P.H. Rolland, R. Rieu, J.F. Pellissier, P.E. Magnan, A. Branchereau. 1996. Effects of cryopreservation on the viscoelastic properties of human arteries. Ann Vasc Surg. 10: 262-272. doi:10.1007/BF02001892
Schiffrin, E.L., D. Hayoz. 1997. How to assess vascular remodelling in small and medium-sized muscular arteries in humans. J Hypertens 15: 571-584. doi:10.1097/00004872-199715060-00002
Shi, C., W.S. Lee, Q. He, D. Zhang, D.L. Fletcher, J.B. Newell, E. Haber. 1996. Immunologic basis of transplant-associated arteriosclerosis. Proc Natl Acad Sci USA 93: 4051-4056. doi:10.1073/pnas.93.9.4051
Stone, P.A., P.A. Armstrong, D.F. Bandyk, W.B. Keeling, S.K. Flaherty, M.L. Shames, B.L. Johnson, M.R. Back. 2006. Duplex ultrasound criteria for femorofemoral bypass revision. J Vasc Surg. 44: 496-502. doi:10.1016/j.jvs.2006.06.002
Tai, N.R., H.J. Salacinski, A. Edwards, G. Hamilton, A.M. Seifalian. 2000. Compliance properties of conduits used in vascular reconstruction. Br J Surg 87: 1516-1524. doi:10.1046/j.1365-2168.2000.01566.x
Acknowledgments
This work was performed within a cooperation agreement between Universidad de la República (Uruguay) and Universidad Favaloro (Argentina). It was supported by research grants given to Dr. Armentano and Dr. Daniel Bia by the Development Program on Basic Sciences (PEDECIBA-Uruguay) and CSIC (Universidad de la República). The authors acknowledge the technical assistance of Ms. Paula Bia, Mr. Elbio Agote, Mr. Sebastián Lluberas, and the personnel of INDT.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bia, D., Zócalo, Y., Armentano, R. et al. Non-Invasive Biomechanical Evaluation of Implanted Human Cryopreserved Arterial Homografts. Ann Biomed Eng 37, 1273–1286 (2009). https://doi.org/10.1007/s10439-009-9693-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-009-9693-2