Abstract
Waste from cattle production contains protozoa, such as Cryptosporidium spp. and Giardia, which can be transmitted to humans. People residing in areas of high cattle density may be at increased risk for protozoan infections. The objective of this study was to assess spatial and temporal associations between cattle density and hospitalizations for protozoan infections in the U.S. elderly. Data on protozoan infections were abstracted from Centers for Medicare and Medicaid Services datasets for a 14-year period (1991–2004). Cattle inventory data were abstracted from the 2002 U.S. Census of Agriculture. Counties were classified into one of five exposure categories based on both cattle density and human density. Our analyses considered differences in rates, trends, and variations in seasonal patterns based on exposure categories. Cryptosporidiosis demonstrated a trend of increasing annual rates related to increased potential exposure to cattle. Both cryptosporidiosis and giardiasis demonstrated significant seasonal patterns peaking during the fourth week of October in areas of high cattle/low population density and the second week of September in counties with low cattle/low human density, respectively. Counties with low human population density (regardless of cattle density) had the highest rate of all protozoan infections, peaking in the summer. These results demonstrate the elderly population is at increased risk of protozoan infections in areas of high cattle density, particularly cryptosporidiosis. The seasonal patterns and higher annual rates seen in rural areas suggest time-variant environmental exposures, which may be affected with geographical and temporal targeting of agricultural policies and interventions to improve public health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Waste from intensive livestock production may contain microorganisms of zoonotic origin, such as Salmonella (Hald and Andersen, 2001; Cox et al., 2005), Campylobacter (Jacobs-Reitsma et al., 1995; Cox et al., 2005), Cryptosporidium spp. and Giardia (Caccio et al., 2005; Hunter and Thompson 2005). These pathogens can be spread to humans directly via contaminated water or food or indirectly, transmitted through application of animal manure onto land. Runoff from agricultural fields after heavy rainfall or irrigation, potentially carrying pathogens, often ends up in streams and surface waters, which may be sources for drinking water; therefore, the waterborne route of transmission for these protozoa is of increasing concern. Depending on the microorganism, environmental conditions, such as manure treatment, season, vegetation, moisture and temperature of the soil, and water will affect their survival (Bicudo and Goyal, 2003). Likewise, the potential for disease transmission to humans will be affected by factors, such as pathogen type, minimum infective dose, seasonality of pathogen occurrence in manure, and effect of manure treatment (Bicudo and Goyal, 2003).
Waterborne protozoa, particularly Cryptosporidium spp. and Giardia, are associated with zoonotic transmission (Hunter and Thompson, 2005). Currently, there are at least 16 recognized species of Cryptosporidium of which two most affect humans, C. hominis and C. parvum (Sopwith et al., 2005; Cama et al., 2008). The host reservoir for C. hominis is humans and reservoirs for C. parvum include cattle, other livestock, and humans (Hunter and Thompson, 2005). Cryptosporidium oocysts are shed in extremely high numbers, up to 106 oocysts per gram of feces, by livestock younger than 6 months of age (Garber et al., 1994; Xiao and Herd, 1994). Only a very low infective dose of Cryptosporidium oocysts are needed to initiate infection in humans (Griffiths, 1998). Currently, there are six recognized species of Giardia of which only one is known to affect humans, Giardia duodenalis, also known as Giardia intestinalis or Giardia lamblia (Thompson, 2004; Caccio and Ryan, 2008). Giardia duodenalis cysts are commonly found in domestic wildlife, ruminants, and humans (Appelbee et al., 2003). Giardiasis also is highly infectious requiring only 10–100 cysts for an infectious dose (Rendtorff, 1954). To compound the infectivity of these diseases, the (oo)cysts are immediately infective when excreted and are easily transmitted via person-to-person contact and animal-to-person contact (Thompson, 2004).
Both Cryptosporidium oocysts and Giardia cysts can survive for weeks to months in the environment (Smith et al., 2006) and specifically in manure (Svoboda et al., 1997). Both protozoa also have prolonged survival times in water and are resistant to chemical and physical agents typically used in water treatment (Brandonisio, 2006). One study demonstrated that movement of Cryptosporidium ooycsts could occur in runoff for at least 21 days, and up to 70 days, after introduction in the environment (Mawdsley et al., 1996). High numbers of Cryptosporidium oocysts and Giardia cysts have been detected in river waters that pass through areas of extensive animal husbandry (Rose et al., 1988). Studies have shown that oocyst concentration in streams depends on the quality of farm and manure management practices and the intensity of rain.
Zoonotic transmission varies both spatially and temporally for these protozoan infections. Outbreaks of cryptosporidiosis infections in humans have been shown to be associated with drinking water contaminated by livestock (Meinhardt et al., 1996; Clark, 1999; Hunter and Thompson, 2005). Outbreaks of giardiasis in humans have been associated with contamination of surface waters from sewage (Levine et al., 1990) and intensive animal husbandry operations (Rose et al., 1988). A number of studies have assessed the difference in seasonal patterns of cryptosporidiosis in humans by species and found that C. parvum peaks in the late spring, which coincides with the spring lambing and calving season, whereas C. hominis peaks in the fall (McLauchlin et al., 2000; Learmonth et al., 2004; Sopwith et al., 2005). Studies also have reported differing geographical range for the two species (McLauchlin et al., 2000; Learmonth et al., 2001). As expected, zoonotic transmission is more commonly noted in rural areas (Feltus et al., 2006; Ng et al., 2008). Therefore, the proper handling and treatment of livestock manure is a public health concern.
In the United States, large-scale animal production facilities, such as confined animal feeding operations (AFOs) and concentrated animal feeding operations (CAFOs), are a primary concern because they generate large amounts of manure waste which can contaminate the surrounding environment. A study conducted in Sweden using surveillance data demonstrated a significant statistical association of cattle and farm density with human Escherichia coli infection (Kistemann et al., 2004). Studies conducted in North Carolina also demonstrated adverse environmental and health impacts for communities near livestock production facilities (Mirabelli et al., 2006a, b). Concentrated animal feedlot operations are increasing in number and size in the United States. In 2003, the U.S. EPA estimated that animal feeding operations in the United States produced 500 million tons of manure and that CAFOs account for more than half of that manure (Burkholder et al., 2007; Thorne, 2007). As of 2006, there are 18,800 CAFO facilities contributing more than 60% of the farm animal waste in the U.S. (EPA (2006). The intensification of large-scale animal feeding operations in the United States poses a public health threat in potential for increased rates of animal to human transmission of pathogens via waterborne routes.
Although outbreaks of waterborne diseases have been associated with water contaminated by livestock feces, a comprehensive analysis of protozoan infections in areas with large-scale cattle production has not been conducted. In this study we evaluate variations in rates, trends, and seasonal patterns between areas with high density of cattle, high exposure, and those with low density of cattle, low exposure. Understanding variations in seasonal patterns will allow for better assessment of the impact that intensive cattle farming has on the rates of waterborne protozoan disease in the United States. These results may be useful for policy making and will allow for improved geographical and temporal targeting of agricultural, environmental, and public health policies and intervention strategies.
Methods
We assessed spatial and temporal associations between rates of protozoan diseases in the elderly and cattle density. Each county in the United States was classified into one of five exposure categories based on both the density of cattle and human population density in that county, and analyses considered the differences in disease rates and seasonal patterns in the elderly by exposure category.
Outcome Data
For outcome data we utilized hospitalization records of elderly patients from the Centers for Medicare and Medicaid Services. This dataset is the only comprehensive, national level dataset for hospitalizations in the United States and covers 98% of the elderly population (Cohen and Naumova, 2007). Hospitalization records for persons aged 65 years or older were abstracted for each county for a 14-year period (January 1, 1991–December 31, 2004). The hospitalization dataset contains individual patient information including state of residence, gender, age at admission, dates of admission and discharge, and ten diagnosis codes, which are populated based on the ICD 9-CM system. We abstracted records with the following diagnoses in any of the ten diagnosis fields: cryptosporidiosis (ICD 007.2, 007.4) (Mor et al., 2009), giardiasis (ICD 007.1), other protozoa (ICD 007.8, 007.9), and all protozoa (ICD 007.1, 007.2, 007.4, 007.8, 007.9). Records were aggregated according to patient’s diagnosis code, county of residence, and date of admission. To facilitate temporal and spatial analysis, for each county we determined counts of each disease, estimated annual rates, and created weekly time-series of counts. To minimize spurious high rates caused by extremely low denominators, a spatial aggregation scheme was applied to incorporate counties with low elderly population into the adjacent counties until the total number of elderly exceeded 1,000, resulting in a total of 2755 counties for analysis (for details related to aggregation rules see Castronovo et al., 2009).
We supplemented our analysis with sociodemographic data from the 1990 and 2000 U.S. Census. These data include county size, population, population density, median household income, and average household size.
Exposure Data
Data on cattle were abstracted from the 2002 U.S. Census of Agriculture, which is conducted by the U.S. Department of Agriculture every 5 years (USDA, 2007a). We abstracted data on the inventory of calves and cattle and number of cattle and calf farms per county. The Census of Agriculture reports inventory of cattle in most counties in the United States; only 32 counties did not report any cattle inventory. We calculated the density measure, number of cattle per 10 square miles, for each county as our primary exposure measure. We also calculated the number of cattle per farm for each county.
Exposure Classification
Preliminary analysis of the exposure parameter, density of cattle by county, demonstrated that this variable has and extremely skewed distributions, which would require complex transformations to achieve normality. Therefore, we categorized the exposure and assessed associations by exposure category. Each county in the United States was classified based on both cattle density and human population density to account for different modes of disease transmission, animal-to-human and human-to-human. Counties were categorized into one of five categories representing increasing exposure to protozoan diseases: limited exposure (0); low cattle/low human density (I); low cattle/high human density (II); high cattle/low human density (III); or high cattle/high human density (IV). Counties which did not report any cattle were assigned to the limited exposure category (0). The remaining counties were divided into two equal categories (high and low) based on the median number of cattle per 10 square miles. Both of these groups were then further divided into two equal groups based on the median human population density (high and low) of the county.
Analyses
Descriptive Analysis
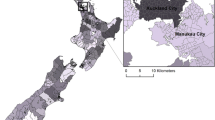
Descriptive statistics of sociodemographic and cattle density parameters (including population density, density of elderly population, percent elderly in the county, median household income, average household size, cattle per 10 square miles, and cattle per farm) were calculated for each of the five exposure categories and analysis of variance (ANOVA) was used to test difference of the mean by exposure category. The density of cattle per 10 square miles and exposure category by county were mapped using ArcGIS 9.2 (ESRI, Redlands, CA) (Figs. 1, 2).
Because the outcomes of interest had low counts, we calculated the proportion of counties with no cases for each outcome. To assess significant differences between categories, we compared proportions of counties within each exposure category without cases using the Marascuillo procedure for comparison of multiple proportions (Sheskin, 2003). We also tested whether the proportion of counties with no records of hospitalizations within each exposure category was significantly different from the national proportion (all counties) using the χ2 test.
Trend Analysis
Average annual rates for each outcome were calculated for each county using linearly interpolated elderly population for 1997 (midpoint of the 14-year, 1990–2004, data timeframe) from 1990 to 2000 U.S. Census data as the denominator. Mean and standard deviations of annual outcome rates were calculated for all counties in each exposure category. To assess whether annual outcome rates demonstrate a significant increasing trend with an increase in exposure, we ran a simple linear regression predicting outcome rates by exposure category (0–IV) for each outcome.
Seasonality Analysis
Weekly time series of counts were created for each exposure category for each of the four disease outcomes: cryptosporidiosis, giardiasis, other protozoa, and all protozoa. The time series of counts were recalculated as a time series of rates using the total sum of the elderly population in 1997 in the counties in each exposure category as a constant denominator (Figs. 3–6). Average weekly rates are provided as reference for scale for the weekly times series data. We estimated seasonal parameters for each category for each disease outcome using harmonic regression. Seasonality is characterized as systematic, periodic fluctuations within the course of a year and is assessed by several parameters: 1) the time when the seasonal curve reaches its maximum; 2) annual maximum value (peak); and 3) annual minimum value (nadir) (Naumova et al., 2007; Naumova and MacNeill, 2007). These seasonal parameters are calculated based on values predicted by the harmonic regression:
Average weekly rate (per 10,000,000 elderly) and seasonal pattern for cryptosporidiosis reported by CMS (ICD 007.2, 007.4) during a 14 year-period (1991–2004) by exposure categories based on cattle and human density. Seasonal parameters that are significant (P < 0.05) are denoted with an asterisk (*).
Average weekly rate (per 10,000,000 elderly) and seasonal pattern for all protozoa reported by CMS (ICD 007.1, 007.2, 007.4, 007.8, 007.9) during a 14 year-period (1991–2004) by exposure categories based on cattle and human density. Seasonal parameters that are significant (P < 0.05) are denoted with an asterisk.
—for which t is time, ω is frequency, Y(t) is a time-series of a specific outcome, β 0 is intercept, β 1 and β 2 are parameters and ε is the error term. The regression parameters β 1 and β 2 are used to estimate the peak timing, 95% confidence interval around the peak timing and the relative intensity. The details of these calculations can be found in the Supplemental material. All analyses were conducted using SPSS 13 (Chicago, IL) and SPlus 7.0 (Seattle, WA).
Results
Descriptive Analysis
We assessed spatial and temporal trends in rates of protozoan diseases in the elderly based on exposure categories defined by both cattle density and population density. The categories display a distinct gradient between low and high human population density categories (I, III and II, IV) and low and high cattle density categories (I, II and III, IV) (Table 1). As expected, the counties in the limited exposure category (0) are significantly more densely populated than all other counties. Counties with lower population density also demonstrate a lower density of elderly population (I and III). Counties with the highest cattle density are primarily in the central Plains states (Fig. 1). The exposure categories demonstrate distinct clusters with high cattle/high human density areas in the Appalachian region and high cattle/low human density categories in the Plains states (Fig. 2).
Higher annual rates of giardiasis and outcome of all protozoa were seen in areas with low population density (I and III) (Table 2). For cryptosporidiosis, the lowest proportion of counties with no records of hospitalizations was seen in the limited exposure (0) category. For giardiasis and all protozoa, the lowest proportion of counties with no records of hospitalization was seen in the low cattle/high human density (II) category (Table 2).
Trend Analysis
Only cryptosporidiosis demonstrated a trend with increased exposure; the other outcomes did not demonstrate a trend with increased potential exposure to cattle. There was a significant increasing trend in mean disease rate for cryptosporidiosis with increased exposure to protozoan diseases (category 0 to IV; β = 0.036, P < 0.0001).
Seasonality Analysis
Figures 3–6 demonstrate the seasonal patterns for each exposure category for the outcomes of cryptosporidiosis, giardiasis, other protozoa, and all protozoa, respectively. For cryptosporidiosis, the highest weekly rate was seen in the category with high cattle/low human density (III; Fig. 3). Significant seasonal patterns were seen in the low cattle/high human density (II) and high cattle/low human density (III) categories with the peak week estimated approximately during the third week of March (week 12.71) and the fourth week of October (week 43.72), respectively. For giardiasis, the highest weekly rates are seen in categories with low human population density (I and III; Fig. 4). The low cattle/low human density category (I) is the only category that demonstrated a significant seasonal pattern peaking during the second week of September (week 37.01). The highest weekly rate for the outcome of other protozoa is seen in the high cattle/low human density (III) category (Fig. 5). This outcome demonstrates significant seasonal patterns in all categories except the limited exposure (0) and high cattle/high human density (IV) categories. The estimated peaks range from the second week of April (week 16.08) to the fourth week of May (week 22.88). The highest weekly outcome rate for all protozoa is seen in the low human population density categories (I and III). A significant seasonal pattern is only seen in the low cattle/low human density category (I) with a peak estimated during the third week of August (week 34.44) for the outcome of all protozoa. The relative intensity fluctuated from 1.03 to 3.77 across all disease outcomes. Values for relative intensity close to 1 indicate an absence of seasonal pattern regardless of the calculated peak timing. High values for relative intensity indicate a strong seasonal pattern or an unusual event such as an outbreak of the disease.
Discussion
Increased livestock production and the concentration of livestock production can pose a public health threat through direct contamination of food and water, or indirectly by improper management of animal waste. In this study, we developed exposure categories based on both density of cattle and human population density to account for both animal-to-human and human-to-human disease transmission. We assessed whether rates and seasonal patterns of protozoan diseases varied by these exposure categories.
Because cattle are host reservoirs for the protozoa Cryptosporidium ssp. and Giardia, we expected to see high rates of cryptosporidiosis and giardiasis in areas with high cattle density. The annual rate of cryptosporidiosis did demonstrate an increasing trend by category, suggesting that increased spatial exposure to cattle poses a risk for this infection. High weekly rates and significant seasonal patterns for cryptosporidiosis were seen in areas with low cattle/low human density (II) and areas of high cattle/low human density (III) peaking during the third week of March and the fourth weeks of October, respectively. Although studies conducted in the United Kingdom demonstrate two seasonal peaks for cryptosporidiosis based on species genotype (McLauchlin et al., 2000; Sopwith et al., 2005), the seasonal pattern in the low cattle/low human density (II) category is driven by the large spring outbreak in 1993 (Fig. 3). Upon further investigation, these counts are all from Milwaukee County, which experienced a large outbreak of cryptosporidiosis in March 1993 (Morris et al., 1998; Naumova et al., 2003). This outbreak is clearly seen in the time series for the low cattle/high human density category (II) for the outcome of all protozoa (Fig. 6). For giardiasis, higher rates and significant seasonal patterns were seen in areas with low cattle/low human density (I). For all protozoa, counties with low human population density (regardless of cattle density) had the highest rate of hospitalization and a significant seasonal pattern was only seen in the low cattle/low human density (I) category peaking during the third week of August.
This analysis demonstrates that seasonal patterns do vary based on overall cattle density. However, the analysis cannot differentiate between the types of cattle production, such as dairy and beef, which utilize different manure management practices and may therefore lead to differing seasonal patterns. Our ability to detect differences relating to beef or dairy cattle is limited by the relatively low number of protozoan diseases reported in the CMS dataset. Variation in seasonal patterns based on type of cattle production can be assessed in a future study using local surveillance data or site specific data which can provide the needed power and sufficient level of detail.
This study uses secondary data sources, such as the Centers for Medical Services and Census of Agriculture, to assess relationships between human health and livestock. These secondary data repositories are particularly useful for better understanding the spatiotemporal pattern of diseases on a national scale, especially for diseases with zoonotic transmission and a strong environmental link. Although these data are not collected specifically for public health analyses, their importance should not be underestimated. These large databases are not collected specifically for public health investigations and, therefore, do not contain detailed information on pathogen genotype or patient’s exposure or route of transmission. However, hospitalization records are collected uniformly and comprehensively on a national scale and provide a general understanding of magnitude and complexity of the examined relationship. In the United States, diarrheal diseases with zoonotic potential, such as cryptosporidiosis and giardiasis, are rarely tested for, particularly in the elderly population. When protozoan pathogens are tested for and found, the data are typically aggregated at a coarse spatial level (i.e., by state) and temporal levels (i.e., by month). These factors make a detailed analysis at fine temporal and spatial scales (i.e., by week and by county) difficult.
The analysis undertaken in this paper demonstrated that a future, more detailed national level evaluation of the effects of livestock on human health will be complex and should ideally incorporate a number of factors. Pathogen transmission can be affected by climatic conditions and extreme meteorological events (Naumova et al., 2005; Jagai et al., 2007; Jagai et al., 2009); therefore, it would be valuable to explore such effects on the detected associations. The probability of exposure to pathogen of interest and the dominant routes of transmissions may be better described by measures other than those we have used and those measures, as well as adequate adjustments for marked differences in county size and adjacency, need to be examined Furthermore, the designed exposure categories, as demonstrated in Table 1, varied by socioeconomic status, age of infrastructure, and other factors that can potentially affect health care and access to healthcare; these factors should be assessed on a more refined spatial scale.
In estimating annual and weekly disease rates, we applied a standard approach of using a single constant for county-specific population as a denominator. Whereas the population estimate was centered at the midpoint for the study time period, such estimation can be further improved by using county-specific annual elderly population data. Our investigation on the accuracy of rate estimation using Census data and Medicare Denominator Files has demonstrated that these two denominators produce very similar results (Cohen et al., 2008) and it is unlikely that our approach introduced a systematic bias. Obviously, a comparison of disease trends in rural and urban environments, especially in vulnerable populations, requires attention to the accurate estimation of population at risk with the most appropriate information.
Data for cattle inventory were gathered from the 2002 U.S. Census of Agriculture (USDA, 2007a). Although this is a comprehensive data source for livestock in the United States, it has several complex aspects that limit this analysis. The Census is conducted every 5 years at the end of the calendar year and farmers are asked to report their current inventory (USDA, 2007a). Therefore, data collected are inventory of cattle on the farm at the time of survey and may not be representative of the annual production and seasonal variation. For example, there may be seasonal variations in the number and type of cattle on a farm that are not accounted for in this year-end inventory. The density of cattle, and therefore, manure and waste on the land, is the primary concern when assessing public health risk, and the Census data do not provide this directly. The Census of Agriculture also censors counties with only one farm for a particular type of livestock for privacy purposes (USDA, 2007a; Sneeringer, 2009). If a county has only one farm of a certain type, it is possible that it is a large farm and may pose an increased threat to human health with increased waste to manage. For cattle production only 32 counties did not report any cattle, therefore, censoring may not be an issue. However, the map of cattle density by county demonstrates that some of the counties that do not report cattle neighbor counties with high density of cattle (Fig. 1). Therefore, it is possible that these counties actually have no cattle production or they may have one farm for which the data are censored.
The average size of livestock farms has grown consistently in the past 50 years as the industry has consolidated into fewer farms while producing the same number of animals (Sneeringer, 2009). This has lead to livestock production being one of the primary polluters of rivers and streams and sources of agricultural runoff (Burkholder et al., 2007; EPA, 2009), which is a cause for concern for the surrounding area. To address these concerns, the Environmental Protection Agency revised the Clean Water Act, permitting requirements and effluent limitations for CAFOs in 2003 because they were considered point source polluters. However, in response to the decision in the case of Waterkeeper Alliance et al. v EPA, the EPA has changed these requirements (EPA, 2008). Under the new rule only those CAFO facilities that discharge or propose to discharge waste are required to apply for National Pollutant Discharge Elimination System (NPDES) permits. Assessment of discharge is determined by the owner or operator of the CAFO, with no independent assessment of the potential for environmental contamination through the discharge of waste. Permitted CAFOs that apply for NPDES permits are required to present Nutrient Management Plans with their applications (EPA, 2008). The changes in the CAFO rule apply only to designated CAFO facilities, which are determined based on a minimum threshold of the number of animals on the farm, and not to those facilities with livestock numbers just below the threshold (Burkholder et al., 2007; Thorne, 2007). It can be argued that facilities just below the threshold can pose the same risk to the surrounding community and are not required to adhere to Nutrient Management Plans. The changes to the CAFO rule took effect in December 2008, and it remains to be seen whether they will have an impact on the public health of surrounding communities.
This study demonstrates that the density of cattle is a risk factor for protozoan infections, particularly cryptosporidiosis, in the U.S. elderly. Overall, rural communities are at a greater risk for protozoan infections, regardless of cattle density. The strong seasonal patterns and higher annual rates seen in rural areas also suggest a strong effect of environmental exposure. The areas of low population density are, by definition, rural and tend to use self-supplied water sources or public water sources with older water treatment infrastructure that communities find difficult and costly to maintain (USDA, 2007b). Therefore, counties with small populations may not offer the same degree of protection in their water treatment as more densely populated areas. The seasonal nature of livestock production also may affect the seasonal patterns seen in outcome rates. This association may be driven by the time varying seasonal parameters of water quality, such as snow melt and runoff due to precipitation. The fact that we demonstrated well-defined seasonal patterns in certain exposure categories and not in all is suggestive of time-variant environmental contamination. This provides the basis for further investigation on patterns of environmental contamination, infrastructure, and human exposure. Our findings offer insight about the potential impact of climate change, which may result in more variable precipitation and increased flooding leading to increased runoff and pathogens in soil and water, on gastrointestinal infections.
References
EPA (2006) Fact sheet: concentrated animal feeding operations proposed rule making. Available: http://www.epa.gov/npdes/regulations/cafo_revisedrule_factsheet.pdf (Accessed 10 Nov 2007)
EPA (2008) Concentrated Animal Feeding Operations (CAFO) Final rule. Available: http://cfpub.epa.gov/npdes/afo/cafofinalrule.cfm (Accessed 20 July 2009)
EPA (2009) National water quality inventory: report to Congress. Washington DC: Environmental Protection Agency
Appelbee AJ, Frederick LM, Heitman TL, Olson ME (2003) Prevalence and genotyping of Giardia duodenalis from beef calves in Alberta, Canada. Veterinary Parasitology 112:289-294
Bicudo JR, Goyal SM (2003) Pathogens and manure management systems: a review. Environmental Technology 24:115-130
Brandonisio O (2006) Waterborne transmission of Giardia and Cryptosporidium. Parassitologia 48:91-94
Burkholder J, Libra B, Weyer P, Heathcote S, Kolpin D, Thorne PS, et al. (2007). Impacts of waste from concentrated animal feeding operations on water quality. Environmental Health Perspectives 115:308-312
Caccio SM, Ryan U (2008) Molecular epidemiology of giardiasis. Molecular and Biochemical Parasitology 160:75-80
Caccio SM, Thompson RCA, McLauchlin J, Smith HV (2005) Unravelling Cryptosporidium and Giardia epidemiology. Trends in Parasitology 21:430-437
Cama VA, Bern C, Roberts J, Cabrera L, Sterling CR, Ortega Y, et al. (2008). Cryptosporidium species and subtypes and clinical manifestations in children, Peru. Emerging Infectious Diseases 14:1567-1574
Castronovo DA, Chui KKH, Naumova EN (2009) Dynamic maps: a visual-analytic methodology for exploring spatio-temporal disease patterns. Environmental Health 8:61
Clark DP (1999) New insights into human cryptosporidiosis. Clinical Microbiology Reviews 12:554-563
Cohen SA, Naumova EN (2007) Population dynamics in the elderly: the need for age-adjustment in national biosurveillance systems. In: Intelligence and Security Informatics: Biosurveillance: Second NSF Workshop, BioSurveillance 2007, New Brunswick, NJ, USA, May 22, 2007, Zeng D, Gotham I, Komatsu K, Lynch C, Thurmond M, Madigan D, Lober B, Kvach J, Chen H (editors), Springer, pp 47-58
Cohen SA, Oomer IA, Naumova EN (2008) Regional Differences in the Estimation of Influenza Burden in the Elderly: Does Choice of Population Denominator Matter ? Population Association of America Annual Meeting, New Orleans, Louisiana
Cox NA, Bailey JS, Richardson LJ, Buhr RJ, Cosby DE, Wilson JL, et al. (2005) Presence of naturally occurring Campylobacter and Salmonella in the mature and immature ovarian follicles of late-life broiler breeder hens. Avian Diseases 49:285-287
Feltus DC, Giddings CW, Schneck BL, Monson T, Warshauer D, McEvoy JM (2006) Evidence supporting zoonotic transmission of Cryptosporidium spp. in Wisconsin. Journal of Clinical Microbiology 44:4303-4308
Garber LP, Salman MD, Hurd HS, Keefe T, Schlater JL (1994) Potential risk factors for Cryptosporidium infection in dairy calves. Journal of the American Veterinary Medical Association 205:86-91
Griffiths JK (1998) Human cryptosporidiosis: epidemiology, transmission, clinical disease, treatment, and diagnosis. Advances in Parasitology 40:37-85
Hald T, Andersen JS (2001) Trends and seasonal variations in the occurrence of Salmonella in pigs, pork and humans in Denmark, 1995-2000. Berliner und Munchener Tierarztliche Wochenschrift 114:346-349
Hunter PR, Thompson RCA (2005) The zoonotic transmission of Giardia and Cryptosporidium. International Journal for Parasitology 35:1181-1190
Jacobs-Reitsma WF, van de Giessen AW, Bolder NM, Mulder RW (1995) Epidemiology of Campylobacter spp. at two Dutch broiler farms. Epidemiology & Infection 114:413-421
Jagai JS, Castronovo DA, Monchak J, Naumova EN (2009) Seasonality of cryptosporidiosis: a meta-analysis approach. Environmental Research 109:465-478
Jagai JS, Castronovo DA, Naumova EN (2007) The Use of Köppen Climate Classification System for Public Health Research. International Society for Environmental Epidemiology, Mexico City, Mexico
Kistemann T, Zimmer S, Vagsholm I, Andersson Y (2004) GIS-supported investigation of human EHEC and cattle VTEC O157 infections in Sweden: geographical distribution, spatial variation and possible risk factors. Epidemiology & Infection 132:495-505
Learmonth J, Ionas G, Pita A, Cowie R (2001) Seasonal shift in Cryptosporidium parvum transmission cycles in New Zealand. Journal of Eukaryotic Microbiology (Suppl):34S–35S
Learmonth JJ, Ionas G, Ebbett KA, Kwan ES (2004) Genetic characterization and transmission cycles of Cryptosporidium species isolated from humans in New Zealand. Applied & Environmental Microbiology 70:3973-3978
Levine WC, Stephenson WT, Craun GF (1990) Waterborne disease outbreaks, 1986-1988. MMWR CDC Surveillance Summary 39:1-13
Mawdsley JL, Brooks AE, Merry RJ, Pain BF (1996) Use of novel soil tilting table apparatus to demonstrate the horizontal and verticle movement of the protozoan pathogen Cryptosporidium spp. in soil. Biology and Fertility of Soils 23:215-220
McLauchlin J, Amar C, Pedraza-Diaz S, Nichols GL (2000) Molecular epidemiological analysis of Cryptosporidium spp. in the United Kingdom: results of genotyping Cryptosporidium spp. in 1,705 fecal samples from humans and 105 fecal samples from livestock animals. Journal of Clinical Microbiology 38:3984-3990
Meinhardt PL, Casemore DP, Miller KB (1996) Epidemiologic aspects of human cryptosporidiosis and the role of waterborne transmission. Epidemiologic Reviews 18:118-136
Mirabelli MC, Wing S, Marshall SW, Wilcosky TC (2006a) Asthma symptoms among adolescents who attend public schools that are located near confined swine feeding operations. Pediatrics 118:e66-75
Mirabelli MC, Wing S, Marshall SW, Wilcosky TC (2006b) Race, poverty, and potential exposure of middle-school students to air emissions from confined swine feeding operations. Environmental Health Perspectives 114:591-596
Mor SM, DeMaria A Jr, Griffiths JK, Naumova EN (2009) Cryptosporidiosis in the elderly population of the United States. Clinical Infectious Diseases 48:698-705
Morris RD, Naumova EN, Griffiths JK (1998) Did Milwaukee experience waterborne cryptosporidiosis before the large documented outbreak in 1993? Epidemiology 9:264-270
Naumova EN, Christodouleas J, Hunter PR, Syed Q (2005) Effect of precipitation on seasonal variability in cryptosporidiosis recorded by the North West England surveillance system in 1990-1999. Journal of Water & Health 3:185-196
Naumova EN, Egorov AI, Morris RD, Griffiths JK (2003) The elderly and waterborne Cryptosporidium infection: gastroenteritis hospitalizations before and during the 1993 Milwaukee outbreak. Emerging Infectious Diseases 9:418-425
Naumova EN, Jagai JS, Matyas B, DeMaria A Jr, MacNeill IB, Griffiths JK (2007) Seasonality in six enterically transmitted diseases and ambient temperature. Epidemiology & Infection 135:281-292
Naumova EN, MacNeill IB (2007) Seasonality assessment for biosurveillance. In: Auget JL, Balakrishnan N, Mesbah M, Molenberghs G (eds) Advances in Statistical Methods for the Health Sciences. Boston: Birkhauser, pp 437-450
Ng J, Eastwood K, Durrheim D, Massey P, Walker B, Armson A, et al. (2008) Evidence supporting zoonotic transmission of Cryptosporidium in rural New South Wales. Exp Parasitology 119:192-195
Rendtorff RC (1954) The experimental transmission of human intestinal protozoan parasites. II. Giardia lamblia cysts given in capsules. American Journal of Tropical Medicine & Hygiene 59:209-220
Rose JB, Darbin H, Gerba CP (1988) Correlations of the protozoa, Cryptosporidium and Giardia, with water quality variables in a watershed. Water Science and Technology 20:271-276
Sheskin DJ (2003) Handbook of Parametric and Nonparametric Statistical Procedures, 3rd edition. Boca Raton, FL: Chapman & Hall/CRC
Smith HV, Caccio SM, Tait A, McLauchlin J, Thompson RC (2006) Tools for investigating the environmental transmission of Cryptosporidium and Giardia infections in humans. Trends in Parasitology 22:160-167
Sneeringer S (2009) Does animal feeding operation pollution hurt public health ? A national longitudinal study of health externalities identified by geographic shifts in livestock production. American Journal of Agricultural Economics 91:124-137
Sopwith W, Osborn K, Chalmers R, Regan M (2005) The changing epidemiology of cryptosporidiosis in North West England. Epidemiology & Infection 133:785-793
Svoboda IF, Read I, Kemp JS, Wright SE, Coop RL, Mawdsley JL, et al. (1997) Cryptosporidium on cattle farms. In: Cryptosporidium in Water: The Challenge to Policy Makers and Water Managers. London, UK: The Chartered Institution of Water and Environmental Management
Thompson RC (2004) The zoonotic significance and molecular epidemiology of Giardia and giardiasis. Veterinary Parasitology 126:15-35
Thorne PS (2007) Environmental health impacts of concentrated animal feeding operations: anticipating hazards–searching for solutions. Environmental Health Perspectives 115:296-297
USDA (2007a) The Census of Agriculture. Available: http://www.agcensus.usda.gov/ (Accessed 10 Nov 2007)
USDA (2007b) Rural Development Strategies: Infrastructure. Available: http://www.ers.usda.gov/Briefing/RuralDevelopment/Infrastructure.htm (Accessed 10 March 2009)
Xiao L, Herd RP (1994) Infection pattern of Cryptosporidium and Giardia in calves. Veterinary Parasitology 55:257-262
Acknowledgments
The authors thank the support of funding agencies: the National Institute of Environmental Health Sciences (Gastroenteritis and Extreme Weather Events in Elderly (GEWEL) Project (NIEHS ES 013171)) and the Ruth L. Kirschstein NIH National Research Trainee Fellowship (T90 DK070117-05) administered by the Tufts University Water: Systems, Science, and Society Program.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jagai, J.S., Griffiths, J.K., Kirshen, P.H. et al. Patterns of Protozoan Infections: Spatiotemporal Associations with Cattle Density. EcoHealth 7, 33–46 (2010). https://doi.org/10.1007/s10393-010-0286-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10393-010-0286-1