Abstract
Purpose
To investigate the risk factors for primary open-angle glaucoma (POAG) in the Namil study population.
Methods
A cross-sectional, population-based epidemiological study of residents aged ≥40 years from Namil-myon, South Korea, was conducted. Fifty-five subjects with POAG and 1,409 controls were enrolled in this study. Univariate and multivariate logistic regression analyses were performed to identify ocular and systemic factors associated with POAG.
Results
Multivariate logistic regression analysis demonstrated that older age, a history of thyroid disease and higher IOP were associated with an increased risk of POAG. Subgroup analysis showed that older age (OR 1.033, 95 % CI 1.003–1.063 per year), a history of thyroid disease (OR 7.373, 95 % CI 1.407–38.636) and higher IOP (OR 1.132, 95 % CI 1.011–1.268 per mmHg) were risk factors for normal tension glaucoma (NTG, POAG with IOP ≤21 mmHg).
Conclusions
In the Namil study, higher IOP, older age and a history of thyroid disease were significant risk factors for POAG.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glaucoma is a significant cause of irreversible blindness worldwide [1]. Primary open-angle glaucoma (POAG) is the most common type, particularly in populations of European and African ancestry [2], and those of East Asia, including Korea [3] and Japan [4].
A number of epidemiologic studies sought to identify risk factors for POAG. Elevated intraocular pressure (IOP), older age [5, 6] and African ancestry [2] have been shown to be strongly associated with POAG. A number of other risk factors, including myopia [6, 7], a family history of POAG [8, 9], a thinner central cornea [9, 10], smoking [11] and a history of thyroid disease [12, 13], are suggested to be risk factors. However, the studies are not consistent in their findings. Moreover, few studies have been performed to evaluate the risk factors for normal tension glaucoma (NTG) in a population.
A recent population-based epidemiologic study in Korea (the Namil study) demonstrates that 77 % of POAG patients (prevalence of POAG, 3.5 %) had an IOP ≤21 mmHg at screening [3]. A high proportion of NTG has also been previously demonstrated in Japan. The Tajimi study shows that 92 % of patients with POAG (prevalence of POAG, 3.9 %) had an IOP ≤21 mmHg [6]. These data together suggest that NTG is a major health problem in these regions.
The purpose of the present study was to investigate the ocular and systemic risk factors for POAG, as revealed by the Namil study. In addition, we performed subgroup analysis to determine the risk factors for NTG, identified as the most common form of glaucoma in Korea.
Materials and methods
The detailed protocol of the Namil study is reported elsewhere [3]. The study was a population-based cross-sectional survey conducted at Namil-myon, a 47.14-km2 area in central South Korea. The study was carried out in accordance with the Declaration of Helsinki and the municipal laws of the Province of Chungcheongnam-do, and was approved by the Institutional Review Board of Chungnam National University Hospital.
Local residents aged 40 years or older (n = 2,027), were selected for the study. Of these, 20 died during the screening period, and 79 moved from the area or could not be located in Namil-myon at the end of the survey (28 February 2008). Of the remaining 1,928 eligible subjects, 1,532 (887 women and 645 men) received eye examinations, corresponding to an overall response rate of 79.5 %.
After informed written consent was obtained, each participant was interviewed and completed a questionnaire exploring the occurrence of systemic diseases such as hypertension, diabetes mellitus, thyroid disease and stroke. Subjective symptoms such as migraine and cold extremities, any family history of glaucoma and smoking history were also explored in the questionnaire. After interviews, ophthalmic examinations were conducted by trained technicians. Each examination included measurement of visual acuity, refractive status, anterior segment dimensions using IOL Master® (Carl Zeiss Meditec, Oberkochen, Germany) and central corneal thickness (CCT) employing IOPac® (Heidelberg Engineering, Heidelberg, Germany). Slit-lamp biomicroscopic examination, IOP measurement by Goldmann applanation tonometry and gonioscopy were performed by a glaucoma specialist. A screening visual field test using frequency doubling technology (N30-1 screening, Humphrey Matrix; Carl Zeiss Meditec Inc., Dublin, CA, USA) and fundus photography (TRC-NW200; Topcon, Tokyo, Japan) were also performed.
Subjects with suspected glaucoma were referred for definitive examination. Visual field testing was performed using the Humphrey Field Analyzer SITA Standard 30-2 program (HFA II 720i; Carl Zeiss Meditec Inc.). When an abnormal result was found or when the data were of poor reliability, retinal nerve fiber layer analysis was performed using either optical coherence tomography (Stratus OCT; Carl Zeiss Meditec) or scanning laser polarimetry (GDxVCC; Carl Zeiss Meditec). Any definitive test yielding suspicious or abnormal results was repeated on either the same or the following day.
Two separate evaluation committees, each composed of four glaucoma specialists from four different institutes, made the diagnosis of glaucoma based on IOP levels, anterior chamber depth, gonioscopic results, appearance of the optic disc and retinal nerve fiber layer, and perimetric results, in accordance with the criteria of the International Society for Geographical and Epidemiological Ophthalmology [14]. The decisions of the two committees were re-evaluated by a third committee that discussed any discrepancies between the first two results and then made the final diagnosis [3].
Ophthalmologic and systemic data on all patients diagnosed with POAG in the Namil study were analyzed in the present work. Nonglaucomatous eyes of the Namil study were used as a control group. Such eyes were defined by excluding eyes with any type of glaucoma or that were glaucoma-suspect, but not those with ocular hypertension (IOP >21 mmHg). When both eyes were eligible, one eye was randomly selected and used for the analyses.
Data analysis
The demographic and systemic parameters included in the analysis were age, gender, history or presence of any of the following: diabetes mellitus, hypertension, thyroid disease or stroke, migraine or cold extremities, smoking habit and any family history of glaucoma. Smoking status was evaluated as non-smoker, ex-smoker or current smoker. Of the ocular parameters, IOP, spherical equivalent (SE), CCT, axial length (AL) and anterior chamber depth (ACD) were included in the analysis. When ocular parameters such as SE, ACD and AL were analyzed, pseudophakic or aphakic eyes were excluded, because cataract surgery could change these parameters [6, 7, 15]. When the analysis involved IOP, eyes with a history of glaucoma surgery or under current medication for glaucoma were excluded.
Univariate and multivariate logistic regression analyses were performed to identify risk factors for POAG. In the multivariate analysis, variables that retained significance at P < 0.2 in the univariate analysis were included. Subgroup analysis was performed to determine the risk factors for NTG, defined as POAG with IOP ≤21 mmHg. Statistical significance in logistic regression analysis was defined as a probability value <5 %.
Results
In the Namil study, 55 subjects were diagnosed with POAG. Of the 55 eyes of these subjects, 43 eyes had IOP ≤21 mmHg [3]. A total of 1,409 eyes, including normal eyes (1,400 eyes) and eyes with ocular hypertension (9 eyes), were used as a control group. Four eyes of four glaucoma patients who either received antiglaucoma medication or underwent glaucoma surgery were excluded from the analysis involving IOP. In addition, a total of 141 eyes, which were either pseudophakic or aphakic, were also excluded from the analysis whenever it involved SE, AL or ACD.
Comparisons of ocular and systemic parameters between the POAG and control subjects are summarized in Table 1. No statistically significant difference was evident between the POAG patients and the controls in regard to smoking history, history of stroke, symptoms including migraine and cold extremities, CCT, SE, AL or ACD, (P = 0.04). The history or presence of systemic hypertension (P = 0.026), and IOP (P < 0.001) differed significantly between POAG patients and controls. The history or presence of diabetes mellitus and thyroid disease were of marginal significance.
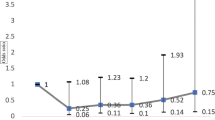
Multivariate logistic regression analysis demonstrated that older age [odds ratio (OR) 1.047, 95 % confidence interval (CI) 1.020–1.074], a history of thyroid disease (OR 8.386, 95 % CI 1.539–45.685) and higher IOP (OR 1.286, 95 % CI 1.182–1.398) were associated with an increased risk of POAG (Table 2).
Risk factors for normal tension glaucoma
Table 3 shows the results of the subgroup, univariate and multivariate logistic regression analyses where only subjects with IOP ≤21 mmHg were included among the POAG patients. Univariate analysis revealed that older age, a history or presence of diabetes mellitus, systemic hypertension or thyroid disease, and higher IOP were all significantly associated with NTG. Of these parameters, older age (OR 1.033, 95 % CI 1.003–1.063), a history or presence of thyroid disease (OR 7.373, 95 % CI 1.407–38.636) and higher IOP (OR 1.132, 95 % CI 1.011–1.268) remained significant upon multivariate analysis.
Discussion
The Namil study was the first population-based survey to examine the prevalence of and risk factors for glaucoma in Korea. As with the Tajimi study in Japan [4], the most common type of glaucoma was POAG, and the majority of the patients (77 %) diagnosed with POAG had IOP ≤21 mmHg [3].
As in most other epidemiologic surveys, the present study identified higher IOP as a risk factor for POAG [5, 6, 9, 10, 15]. It is notable that this was also true in the subgroup analysis in which only NTG patients were included. This is consistent with the results of the Tajimi study, where IOP was identified as a risk factor for POAG, despite the fact that most (92 %) of the POAG patients had an IOP ≤21 mmHg [6]. These data together suggest that the IOP plays an important role in the pathogenesis of glaucoma even when it is within the normal range. In addition, older age was also identified as a risk factor for POAG, consistent with the results of previous reports [6, 9, 15].
A history of thyroid disease was a risk factor for the overall POAG and NTG. This is consistent with a finding of the Blue Mountains study, which showed an association between thyroid disease and POAG [13]. The cited work also found more lower than high-tension glaucoma patients among POAG patients with thyroid disease. The association of thyroid disease and glaucoma may be explained immunologically. Thyroid disease has an immunological basis, and it is acknowledged that the development of autoimmunity may also be linked to glaucoma pathogenesis [16, 17]. It is possible that an autoimmune response to a sensitizing antigen may damage either the retinal ganglion cells or the optic nerve vasculature, leading to increased optic nerve head susceptibility to glaucomatous damage.
Myopia was not a risk factor for POAG in the present study. This is contradictory to some of the other reports [6, 7, 18]. Although we do not have a definitive explanation for this observation, it is possible that the age of the population in this study affected our results. In any analysis of myopia prevalence, pseudophakic or aphakic eyes should be excluded, because cataract surgery changes the refractive error. In the present work, the mean patient age was 64.1 ± 11.5 years, older than that of the subjects of other epidemiologic studies, and 9.7 % of the eyes were excluded from SE analysis because of a history of cataract surgery. This relatively high exclusion rate may have affected the outcome.
A thin cornea was not a significant risk factor in the present work, unlike that found in the Ocular Hypertension Treatment Study (OHTS), where a low CCT was a significant risk factor for conversion to POAG [19]. The Barbados study also found that a thinner CCT was associated with POAG [9, 20]. In the Tajimi study, however, as in the present work, CCT was not a significant risk factor for POAG [6]. We speculate that ethnic differences may partly explain these discrepancies. In addition, IOP differences among reports may be relevant. The IOPs of our subjects and of those in the Tajimi study were much lower than those of the subjects in the OHTS and Barbados surveys. This finding suggests that the relevance of CCT to the risk of developing POAG may differ depending on the IOP levels.
Some known risk factors, including a family history of glaucoma, migraine and cold hands, were not identified by the Namil study. The absence of such factors should be interpreted with caution because the relevant information here was obtained solely by interview.
There were several limitations to the present study. First, as mentioned above, the information on family history and systemic diseases was obtained by interview without a biochemical examination. A future, population-based study where systemic and ophthalmologic parameters are more deliberately selected and estimated is warranted. Second, about 10 % of the subjects were excluded from the analysis involving IOP or biometric parameters because of a history of either glaucoma treatment or cataract surgery. This should be considered in interpreting the data of the IOP and biometric parameters. Third, because of the relatively small number of patients with high-tension POAG, subgroup analysis for these patients was not performed. Further work with a larger study population is needed.
In conclusion, the Namil study found that higher IOP, older age and the presence of thyroid disease were significant risk factors for POAG.
References
Hyman L, Wu SY, Connell AM, Schachat A, Nemesure B, Hennis A, et al. Prevalence and causes of visual impairment in The Barbados Eye Study. Ophthalmology. 2001;108:1751–6.
Tielsch JM, Sommer A, Katz J, Royall RM, Quigley HA, Javitt J. Racial variations in the prevalence of primary open-angle glaucoma. The Baltimore Eye Survey. JAMA. 1991;266:369–74.
Kim CS, Seong GJ, Lee NH, Song KC. Prevalence of primary open-angle glaucoma in central South Korea the Namil study. Ophthalmology. 2011;118:1024–30.
Iwase A, Suzuki Y, Araie M, Yamamoto T, Abe H, Shirato S, et al. The prevalence of primary open-angle glaucoma in Japanese: the Tajimi Study. Ophthalmology. 2004;111:1641–8.
Sommer A, Tielsch JM, Katz J, Quigley HA, Gottsch JD, Javitt J, et al. Relationship between intraocular pressure and primary open angle glaucoma among white and black Americans. The Baltimore Eye Survey. Arch Ophthalmol. 1991;109:1090–5.
Suzuki Y, Iwase A, Araie M, Yamamoto T, Abe H, Shirato S, et al. Risk factors for open-angle glaucoma in a Japanese population: the Tajimi Study. Ophthalmology. 2006;113:1613–7.
Kuzin AA, Varma R, Reddy HS, Torres M, Azen SP. Ocular biometry and open-angle glaucoma: the Los Angeles Latino Eye Study. Ophthalmology. 2010;117:1713–9.
Tielsch JM, Katz J, Sommer A, Quigley HA, Javitt JC. Family history and risk of primary open angle glaucoma. The Baltimore Eye Survey. Arch Ophthalmol. 1994;112:69–73.
Leske MC, Wu SY, Hennis A, Honkanen R, Nemesure B. Risk factors for incident open-angle glaucoma: the Barbados Eye Studies. Ophthalmology. 2008;115:85–93.
Francis BA, Varma R, Chopra V, Lai MY, Shtir C, Azen SP. Intraocular pressure, central corneal thickness, and prevalence of open-angle glaucoma: the Los Angeles Latino Eye Study. Am J Ophthalmol. 2008;146:741–6.
Zanon-Moreno V, Garcia-Medina JJ, Zanon-Viguer V, Moreno-Nadal MA, Pinazo-Duran MD. Smoking, an additional risk factor in elder women with primary open-angle glaucoma. Mol Vis. 2009;15:2953–9.
Lin HC, Kang JH, Jiang YD, Ho JD. Hypothyroidism and the risk of developing open-angle glaucoma: a five-year population-based follow-up study. Ophthalmology. 2010;117:1960–6.
Lee AJ, Rochtchina E, Wang JJ, Healey PR, Mitchell P. Open-angle glaucoma and systemic thyroid disease in an older population: The Blue Mountains Eye Study. Eye (Lond). 2004;18:600–8.
Foster PJ, Buhrmann R, Quigley HA, Johnson GJ. The definition and classification of glaucoma in prevalence surveys. Br J Ophthalmol. 2002;86:238–42.
Garudadri C, Senthil S, Khanna RC, Sannapaneni K, Rao HB. Prevalence and risk factors for primary glaucomas in adult urban and rural populations in the Andhra Pradesh Eye Disease Study. Ophthalmology. 2010;117:1352–9.
Cartwright MJ, Grajewski AL, Friedberg ML, Anderson DR, Richards DW. Immune-related disease and normal-tension glaucoma. A case–control study. Arch Ophthalmol. 1992;110:500–2.
Wax MB. Is there a role for the immune system in glaucomatous optic neuropathy? Curr Opin Ophthalmol. 2000;11:145–50.
Perera SA, Wong TY, Tay WT, Foster PJ, Saw SM, Aung T. Refractive error, axial dimensions, and primary open-angle glaucoma: the Singapore Malay Eye Study. Arch Ophthalmol. 2010;128:900–5.
Gordon MO, Beiser JA, Brandt JD, Heuer DK, Higginbotham EJ, Johnson CA, et al. The Ocular Hypertension Treatment Study: baseline factors that predict the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120:714–20. discussion 829–30.
Leske MC, Connell AM, Wu SY, Hyman LG, Schachat AP. Risk factors for open-angle glaucoma. The Barbados Eye Study. Arch Ophthalmol. 1995;113:918–24.
Acknowledgments
The authors thank Soo Hee Kang, Medical Research Collaborating Center, Seoul National University Hospital, for assistance with statistical analysis. This study was supported by Alcon Korea, Merck Korea, Pfizer Korea, Taejoon Pharmaceutical, Zeiss Korea and the Korean Ophthalmological Society.
Author information
Authors and Affiliations
Corresponding author
Additional information
For the Namil Study Group, the Korean Glaucoma Society.
The members of Namil Study Group, the Korean Glaucoma Society, are given in the Appendix.
Appendix
Appendix
The Namil Study Group, Korean Glaucoma Society:
Byung-Heon Ahn, MD, PhD, Department of Ophthalmology, Samsung Medical Center, Sungkyunkwan University School of Medicine
Myung Douk Ahn, MD, PhD, Department of Ophthalmology, College of Medicine, The Catholic University of Korea
Nam Ho Baek, MD, PhD, Saevit Eye Hospital
Kyu-Ryong Choi, MD, PhD, Department of Ophthalmology, Ewha Womans University School of Medicine
Seung-Joo Ha, MD, Department of Ophthalmology, Soonchunhyang University, College of Medicine
Gyu-Heon Han, MD, PhD, Doctor Lee’s Eye Clinic
Young Jae Hong, MD, PhD, Nune Eye Hospital
Ja-Heon Kang, MD, PhD, Department of Ophthalmology, Kyung Hee University College of Medicine
Changwon Kee, MD, PhD, Department of Ophthalmology, Samsung Medical Center, Sungkyunkwan University School of Medicine
Hong-Seok Kee, MD, PhD, Leeyeon Eye Clinic
Chan Yun Kim, MD, PhD, Department of Ophthalmology, Yonsei University College of Medicine
Chang-Sik Kim, MD, PhD, Department of Ophthalmology, College of Medicine, Chungnam National University
Hwang-Ki Kim, MD, Department of Ophthalmology, Konyang University College of Medicine, Kim’s Eye Hospital
Joon Mo Kim, MD, Department of Ophthalmology, Sungkyunkwan University School of Medicine, Kangbuk Samsung Hospital
Seok-Hwan Kim, MD, PhD, Department of Ophthalmology, Seoul National University College of Medicine
Tae-Woo Kim, MD, PhD, Department of Ophthalmology, Seoul National University College of Medicine
Yong Yeon Kim, MD, PhD, Department of Ophthalmology, Korea University College of Medicine
Michael Scott Kook, MD, Department of Ophthalmology, University of Ulsan, College of Medicine, Asan Medical Center
Joo Hwa Lee, MD, PhD, Department of Ophthalmology, Sanggye-Paik Hospital, Inje University College of Medicine
Kyung-Wha Lee, MD, PhD, Department of Ophthalmology, Hallym University College of Medicine
Seung-Hyuck Lee, MD, PhD, Yonsei Plus Eye Center
Jung-Il Moon, MD, PhD, Department of Ophthalmology, College of Medicine, The Catholic University of Korea
Chan Kee Park, MD, PhD, Department of Ophthalmology, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea
Hyun Joon Park, MD, Merit Eye Clinic
Ki Ho Park, MD, PhD, Department of Ophthalmology, Seoul National University College of Medicine
Gong Je Seong, MD, PhD, Department of Ophthalmology, Yonsei University College of Medicine
Yong Ho Sohn, MD, PhD, Department of Ophthalmology, Konyang University College of Medicine, Kim’s Eye Hospital
Ki-Bang Uhm, MD, PhD, Department of Ophthalmology, Hanyang University College of Medicine
About this article
Cite this article
Kim, M., Kim, TW., Park, K.H. et al. Risk factors for primary open-angle glaucoma in South Korea: the Namil study. Jpn J Ophthalmol 56, 324–329 (2012). https://doi.org/10.1007/s10384-012-0153-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-012-0153-4