Abstract
Purpose
Sphincter injury is a common cause of anal incontinence. Surgical repair remains the operation of choice; however, the outcome often is poor. We investigated the ability of injected bone marrow-derived mesenchymal stem cells to enhance sphincter healing after injury and primary repair in a preclinical model.
Methods
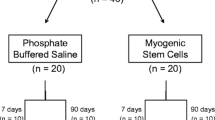
Twenty-four inbred Wistar Furth rats were divided into three groups. As a control, Group A underwent sham operation. Group B had sphincterotomy and repair of both anal sphincters plus saline injections. The study group (Group C) underwent sphincterotomy and repair followed by intrasphincteric injections of syngenic bone marrow-derived mesenchymal stem cells. A further group (Group D) of outbred Wistar rats treated with mesenchymal stem cells and immunosuppressive therapy also was evaluated. At 30 days, histologic and morphometric analysis and in vitro contractility testing was performed.
Results
A significant decrease of muscle tissue was observed at the site of repair after sphincter injury. However, in Groups C and D, histologic examination demonstrated new muscle fibers and morphometric analysis revealed a significantly greater muscle area fraction than in Group B (P < 0.05). Moreover, mesenchymal stem cells injection improved contractility of sphincters strips compared with Group B (P < 0.05). No significant differences were found between Groups C and D.
Conclusions
In our experimental model, bone marrow-derived mesenchymal stem cells injection improved muscle regeneration and increased contractile function of anal sphincters after injury and repair. Therefore, mesenchymal stem cells may represent an attractive tool for treating anal sphincter lesions in humans. Investigations into the biologic basis of this phenomenon should increase our knowledge on underlying mechanisms involved in sphincter repair.






Similar content being viewed by others
References
Rothbarth J, Bemelman WA, Meijerink WJ, et al. What is the impact of fecal incontinence on quality of life? Dis Colon Rectum 2001;44:67–71.
Whitehead WE, Wald A, Norton NJ. Treatment options for fecal incontinence. Dis Colon Rectum 2001;44:131–44.
Oliveira L, Wexner SD. Anal incontinence. In: Beck DE, Wexner SD, eds. Fundamentals of anorectal surgery. London: WB Saunders Company Ltd., 1998:115–52.
Sultan AH, Kamm MA, Hudson CN, Thomas JM, Bartram CI. Anal-sphincter distruption during vaginal delivery. N Engl J Med 1993;329:1905–11.
Varma A, Gunn J, Gardiner A, Lindow SW, Duthie GS. Obstetric anal sphincter injury: prospective evaluation of incidence. Dis Colon Rectum 1999;42:1537–43.
Oberwalder M, Dinnewitzer A, Baig MK, et al. The association between late-onset fecal incontinence and obstetric anal sphincter defects. Arch Surg 2004;139:429–32.
Lindsey I, Jones OM, Smilgin-Humphreys MM, Cunningham C, Mortensen NJ. Patterns of fecal incontinence after anal surgery. Dis Colon Rectum 2004;47:1643–9.
Casillas S, Hull TL, Zutshi M, Trzcinski R, Bast JF, Xu M. Incontinence after a lateral internal sphincterotomy: are we underestimating it? Dis Colon Rectum 2005;48:1193–9.
Karoui S, Leroi AM, Koning E, Menard JF, Michot F, Denis P. Results of sphincteroplasty in 86 patients with anal incontinence. Dis Colon Rectum 2000;43:813–20.
Malouf AJ, Norton CS, Engel AF, Nicholls RJ, Kamm MA. Long-term results of overlapping anterior anal-sphincter repair for obstetric trauma. Lancet 2000;355:260–5.
Halverson AL, Hull TL. Long-term outcome of overlapping anal sphincter repair. Dis Colon Rectum 2002;45:345–8.
Bravo Gutierrez A, Madoff RD, Lowry AC, Parker SC, Buie WD, Baxter NN. Long-term results of anterior sphincteroplasty. Dis Colon Rectum 2004;47:727–32.
Vaizey CJ, Norton C, Thornton MJ, Nicholls RJ, Kamm MA. Long-term results of repeat anterior anal sphincter repair. Dis Colon Rectum 2004;47:858–63.
Zorcolo L, Covotta L, Bartolo DC. Outcome of anterior sphincter repair for obstetric injury: comparison of early and late results. Dis Colon Rectum 2005;48:524–31.
Rosenthal N. Prometheus’s vulture and the stem-cell promise. N Engl J Med 2003;349:267–74.
Herzog EL, Chai L, Krause DS. Plasticity of marrow-derived stem cells. Blood 2003;102:3483–93.
LaBarge MA, Blau HM. Biological progression from adult bone marrow to mononucleate muscle stem cell to multinucleate muscle fiber in response to injury. Cell 2002;111:589–601.
Muguruma Y, Reyes M, Nakamura Y, et al. In vivo and in vitro differentiation of myocytes from human bone marrow-derived multipotent progenitor cells. Exp Hematol 2003;31:1323–30.
Bossolasco P, Corti S, Strazzer S, et al. Skeletal muscle differentiation potential of human adult bone marrow cells. Exp Cell Res 2004;295:66–78.
Santa María L, Rojas CV, Minguell JJ. Signals from damaged but not undamaged skeletal muscle induce myogenic differentiation of rat bone–marrow-derived mesenchymal stem cells. Exp Cell Res 2004;300:418–26.
Yokoyama T, Huard J, Pruchnic R, et al. Muscle-derived cell transplantation and differentiation into lower urinary tract smooth muscle. Urology 2001;57:826–31.
Yiou R, Dreyfus P, Chopin DK, Abbou CC, Lefaucheur JP. Muscle precursor cell autografting in a murine model of urethral sphincter injury. BJU Int 2002;89:298–302.
Cannon TW, Lee JY, Somogyi G, et al. Improved sphincter contractility after allogenic muscle-derived progenitor cell injection into the denervated rat urethra. Urology 2003;62:958–63.
Yiou R, Yoo JJ, Atala A. Restoration of functional motor units in a rat model of sphincter injury by muscle precursor cell autografts. Transplantation 2003;76:1053–60.
Peyromaure M, Sebe P, Praud C, et al. Fate of implanted syngenic muscle precursor cells in striated urethral sphincter of female rats: perspectives for treatment of urinary incontinence. Urology 2004;64:1037–41.
Kwon D, Minnery B, Kim Y, et al. Neurologic recovery and improved detrusor contractility using muscle-derived cells in rat model of unilateral pelvic nerve transaction. Urology 2005;65:1249–53.
Dobson KR, Reading L, Haberey M, Marine X, Scutt A. Centrifugal isolation of bone marrow from bone: an improved method for the recovery and quantitation of bone marrow osteoprogenitor cells from rat tibiae and femurae. Calcif Tissue Int 1999;65:411–3.
Brading AF, Sibley GN. A superfusion apparatus to study field stimulation of smooth muscle from mammalian urinary bladder. J Physiol 1983;334:11–2.
Young CJ, Mathur MN, Eyers AA, Solomon MJ. Successful overlapping anal sphincter repair: relationship to patient age, neuropathy, and colostomy formation. Dis Colon Rectum 1998;41:344–9.
Gilliland R, Altomare D, Moreira H Jr, Oliveira L, Gilliland JE, Wexner SD. Pudendal neuropathy is predictive of failure following anterior overlapping sphincteroplasty. Dis Colon Rectum 1998;41:1516–22.
Norderval S, Öian P, Revhaug A, Vonen B. Anal incontinence after obstetric sphincter tears: outcome of anatomic primary repairs. Dis Colon Rectum 2005;48:1055–61.
Fitzpatrick M, Behan M, O’Connell PR, O’Herlihy C. A randomized clinical trial comparing primary overlap with approximation repair of third-degree obstetric tears. Am J Obstet Gynecol 2000;183:1220–4.
Tjandra JJ, Han WR, Goh J, Carey M, Dwyer P. Direct repair vs. overlapping sphincter repair: a randomized, controlled trial. Dis Colon Rectum 2003;46:937–43.
Pinta TM, Kylänpää ML, Salmi TK, Teramo KA, Luukkonen PS. Primary sphincter repair: are the results of the operation good enough? Dis Colon Rectum 2004;47:18–23.
Kairaluoma MV, Raivio P, Aarnio MT, Kellokumpu IH. Immediate repair of obstetric anal sphincter rupture: medium-term outcome of the overlap technique. Dis Colon Rectum 2004;47:1358–63.
Garcia V, Rogers RG, Kim SS, Hall RJ, Kammerer-Doak DN. Primary repair of obstetric anal sphincter laceration: a randomized trial of two surgical techniques. Am J Obstet Gynecol 2005;192:1697–701.
Leroi AM, Kamm MA, Weber J, Denis P, Hawley PR. Internal anal sphincter repair. Int J Colorectal Dis 1997;12:243–5.
Morgan R, Patel B, Beynon J, Carr ND. Surgical management of anorectal incontinence due to internal anal sphincter deficiency. Br J Surg 1997;84:226–30.
Kratchmarova I, Blagoev B, Haack-Sorensen M, Kassem M, Mann M. Mechanism of divergent growth factor effects in mesenchymal stem cell differentiation. Science 2005;308:1472–7.
Togel F, Hu Z, Weiss K, Isaac J, Lange C, Westenfelder C. Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am J Physiol Renal Physiol 2005;289:F31–42.
Natsu K, Ochi M, Mochizuki Y, Hachisuka H, Yanada S, Yasunaga Y. Allogeneic bone marrow-derived mesenchymal stromal cells promote the regeneration of injured skeletal muscle without differentiation into myofibers. Tissue Eng 2004;10:1093–112.
Deasy BM, Jankowski RJ, Huard J. Muscle-derived stem cells: characterization and potential for cell-mediated therapy. Blood Cells Mol Dis 2001;27:924–33.
Cossu G, Mavilio F. Myogenic stem cells for the therapy of primary myopathies: wishful thinking or therapeutic perspective? J Clin Invest 2000;105:1669–74.
Acknowledgment
The authors thank Professor Alison F. Brading (University Department of Pharmacology, Oxford, United Kingdom) for critical discussion and review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by a grant from the University of Siena (PAR 2005).
Address of correspondence: Marco Lorenzi, M.D., Department of Surgery, University of Siena, Viale Bracci, 53100 Siena, Italy.
About this article
Cite this article
Lorenzi, B., Pessina, F., Lorenzoni, P. et al. Treatment of Experimental Injury of Anal Sphincters with Primary Surgical Repair and Injection of Bone Marrow-Derived Mesenchymal Stem Cells. Dis Colon Rectum 51, 411–420 (2008). https://doi.org/10.1007/s10350-007-9153-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10350-007-9153-8