Abstract
The plasticity of radial growth and earlywood anatomy to climate has not been properly assessed. To solve this, we evaluated how growth and earlywood anatomy changed in a Mediterranean ring-porous oak (Quercus faginea) across a climatic gradient. We hypothesized that the anatomical variables will be the most sensitive to climatic stress, particularly to water deficit in late winter and early spring when earlywood is formed. Testing these hypotheses allows evaluating whether such plasticity in growth and earlywood anatomy affects the projected responses of trees to climate forecasts. The gradient included high- (H), mid- (M), and low-elevation (L) sites and a xeric site (X). We used dendrochronology to quantify changes in growth and anatomy and to relate them to recent and twenty-first-century forecasted climatic data. The highest interannual variance was observed for the latewood width in the xeric site, where the earlywood vessel area reached the lowest values. Tree-ring width correlated positively with spring precipitation, whereas the earlywood vessel area correlated negatively with winter temperature. The predicted drier and warmer climate caused a 10 % reduction in tree-ring width and earlywood vessel area. Earlywood anatomical traits were less sensitive to climatic variability than radial-growth ones since the percentage of their variance explained by climate varied from 12 to 49 % in the former variables and from 15 to 57 % in the latter ones. The plastic xylem responses to climatic variability across time, mainly in the xeric site, indicate that this plasticity must be explicitly considered when forecasting changes in growth and persistence of trees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Xylem captures relevant and diverse ecological information in several of its anatomical features (e.g. area, density), usually measured across the transversal plane, which are assumed to be mainly related to water availability during the period when the conduits were formed (Carlquist 1975; Baas and Schweingruber 1987). The search for links between such wood-anatomical characteristics and climate may provide a functional proxy of tree performance over time (Wimmer 2002; Fonti et al. 2010). In angiosperm tree species, some studies have found climatic responses of the earliest-formed vessels in diffuse-porous (Sass and Eckstein 1995) and semi-ring-porous to ring-porous wood types (Pumijumnong and Park 1999; Fonti et al. 2006; Giantomasi et al. 2008; Campelo et al. 2010). For example, the area of earlywood vessels of a temperate oak decreased in response to reduced precipitation in the early growing season (García-González and Eckstein 2003).
In ring-porous and winter-deciduous trees from continental areas, freezing-induced embolism of vessels during winter, which is proportional to the transversal conduit area, leads to a pronounced loss of hydraulic conductivity (Hacke and Sperry 2001). Therefore, the tree must form new functional earlywood vessels every spring (Ellmore and Ewers 1986). Growth in these tree species resumes when buds start to break, which is a process shortly preceded by the differentiation of the first earlywood vessels formed by overwintering cambial derivatives (Suzuki et al. 1996; Frankenstein et al. 2005). Thus, the transversal vessel area (VA) or lumen size of those vessels should be mainly driven by climatic conditions at the time of cell enlargement (Tardif and Conciatori 2006). However, it may also depend on climatic constrains affecting carbohydrate synthesis and storage during the winter season preceding ring formation (Fonti and García-González 2004). For instance, narrow vessel lumens are expected to be formed in response to low water availability at the time of cambial resumption which may be induced by warm conditions during late winter and early spring in drought-prone areas (Woodcock 1989). However, it has also been observed that earlywood VA increased in response to warm conditions during spring, particularly in sites with reduced water deficit (WD), suggesting an earlier cambial resumption and enhanced vessel enlargement in response to mild climatic conditions (Tardif and Conciatori 2006). These contrasting findings have pushed the development of multiple studies to build continuous vessel chronologies and to quantify the responses of earlywood anatomical features to climate, mainly in ring-porous tree species (Fonti et al. 2010). A tenet of these research efforts is that measuring these features, which is usually time-consuming, might provide more relevant information on tree performance than obtaining more easily gathered dendrochronological variables such as tree-ring width (but see Tardif and Conciatori 2006).
In the case of winter-deciduous oaks from Mediterranean continental areas, where xylem development is constrained by low temperatures in winter and drought in summer, radial growth is usually enhanced by late winter–early spring rainfall (Tessier et al. 1994). The winter quiescence of these oak species forces them to perform most of their growth in spring before soil water availability decreases (Montserrat-Martí et al. 2009). Thus, the number and total area of earlywood vessels formed by these species and the latewood width might respond to the spring precipitation amount. In addition, the recently formed earlywood vessels are functionally very relevant to ring-porous oaks since they may account for ca. 95 % of the total hydraulic conductivity although latewood formation is still important for radial growth (Corcuera et al. 2006). However, little is known on the relative effects of late winter and early spring climatic conditions on earlywood anatomy and radial growth in Mediterranean ring-porous oaks from continental drought-prone areas subjected to this double climatic stress (Camarero et al. 2010). In the western Mediterranean Basin, both winter temperatures and summer drought may become more relevant for the future growth and persistence of these species since rising winter temperatures and decreasing spring precipitation have been observed and forecasted there (IPCC 2007). Furthermore, increased interannual variability is another component of climatic change whose effects on growth and earlywood anatomy have been rarely assessed. For instance, unusually severe spring droughts can induce a reduction in earlywood conductive area and in turn the production of rings without latewood (Corcuera et al. 2004; Eilmann et al. 2009). The lack of latewood formation in the outermost ring can have profound functional consequences when preceded by low temperatures which induce the cavitation of vulnerable earlywood vessels because in rings with few functional earlywood conduits latewood vessels may act as alternative paths for water transport (Granier et al. 1994).
In this study, we quantify how radial growth and earlywood anatomy in a Mediterranean ring-porous winter-deciduous oak (Quercus faginea) respond to climatic conditions before and during the year of tree-ring formation. We compare these responses in oak populations subjected to different climatic stressors which are considered as spatial analogues representing some of the expected future climatic conditions. This analogy was used to critically evaluate predictions on the potential distribution of tree species under future climates because these forecasts ignore the growth plasticity of trees (Camarero et al. 2010). We hypothesize that: (1) earlywood anatomical variables will be more sensitive to changes in precipitation than radial-growth variables, and (2) earlywood anatomy will respond more to changes in water availability during the previous winter and the current spring than to temperature variability, particularly in xeric sites. Testing the aforementioned two hypotheses will allow evaluating whether xylem plasticity modifies the forecasted potential distributions of tree species based on climate projections.
Materials and methods
Study species and sites
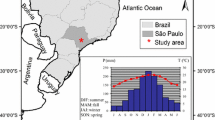
Quercus faginea Lam. is a deciduous ring-porous oak tree distributed in Mediterranean areas of the Iberian Peninsula and North Africa with basic soils (Loidi and Herrera 1990). To avoid confusion with coexisting oak hybrids, we only selected those individuals which presented clear leaf characteristics of Q. faginea (Amaral Franco 1990). We selected four study sites with such contrasting climatic conditions that they represent most of the climatic variability currently experienced by Iberian Q. faginea forests (Jiménez et al. 1998). The study sites were located in two different mountain ranges from Aragón, in north eastern Spain: the Pre-Pyrenees and the Sierra de Alcubierre, located at about 53 km southwards from the first range. In the Pre-Pyrenees, three sites along an altitudinal gradient were selected, the extreme sites being 8 km distant: Pico del Águila (high-elevation cold site, hereafter abbreviated as site H), Arguis (mid-elevation mesic site, hereafter abbreviated as site M) and Nueno (low-elevation dry site, hereafter abbreviated as site L) (Table 1; Fig. 1). In the Sierra de Alcubierre, a fourth low-elevation xeric site (hereafter abbreviated as site X) was selected. In all sites, the climate is Mediterranean continental with a summer drought and a cold winter (Fig. 1; see also Supporting Information, Table S1). The study sites shared similar geological substrate (limestone) and aspects but presented contrasting vegetation types (Table 1).
Field sampling and dendrochronological methods
In November 2007, 10 Q. faginea individuals were selected and tagged in each site, and their size was measured (Dbh, diameter at breast height, at 1.3 m; total height). Based on ring counts in basal samples, all sampled individuals were older than 30 years, which we regarded as an adequate age for avoiding ontogenetic effects on vessel features. At site X, seventeen additional individuals were sampled yielding a total of 27 trees at that site. From each individual, the main trunk was cored at 1.3 m using an increment borer and two cores from opposite directions were taken. All wood samples were prepared following standard dendrochronological methods and visually cross-dated (Fritts 1976). To perform further anatomical analyses, the cores were carefully cut until getting a level wood surface using a sledge microtome (G.S.L.1 microtome, Gärtner and Schweingruber, WSL, Birmensdorf, Switzerland). Earlywood (EW), latewood (LW) and tree-ring width (RW) were then measured at a precision of 0.001 mm using a LINTAB measuring system (Rinntech, Heidelberg, Germany). Visual cross-dating was evaluated using the COFECHA program, which calculates cross-correlations between individual series and a reference mean chronology (Holmes 1983).
Analyses of earlywood anatomical variables
To maximize the climatic signal of wood-anatomical variables, we focused on earlywood vessels. Prior to image acquisition, each cross-dated core was cleaned with pressurized air. Then, the cores were coloured black using a marker pen and rubbed with white chalk to fill the vessel lumina with chalk powder to improve the contrast of vessels. The measurement of vessel features was performed for the period 1985–2007 on images of the transversal wood surface along a tangential area 4.5-mm wide since it provides a representative selection of vessels for investigating their climatic response (García-González and Eckstein 2003; García-González and Fonti 2008). Two complementary methods were used. First, two cores from three trees per site were captured with a 4,800-dpi resolution scanner (Epson Perfection V750 Pro, Seiko Epson Corporation, Japan). Second, images of all wood samples of each site (ten trees, twenty cores) were captured at 2,048 × 1,536 resolution and 16–20× magnification using a stereomicroscope (MZ12.5 Leica, Heerbrugg, Switzerland) attached to a digital camera (Leica DFC290). To compare both methods, cores were analysed using scanned and stereomicroscope-acquired images. The estimates of VA based on scanned samples were usually greater than those derived from stereomicroscope measurements (Supporting Information, Figs. S1, S2). Thus, we only considered stereomicroscope-acquired images.
A total of 1,443 tree rings were captured (H, 384 rings; M, 365 rings; L, 357 rings; X, 337 rings), and 29,228 earlywood vessels were measured. Each image of each ring width was analysed using the WinCell PRO (version 2010a) software (Régents Instruments Inc., Québec, Canada). To define earlywood vessels, the minimum and maximum vessel lumen cross-sectional areas were set to 0.001 and 0.1 mm2, respectively, based on previous studies on Q. faginea (Villar-Salvador et al. 1997; Corcuera et al. 2004). These thresholds included most earlywood vessels (Supporting Information, Fig. S3).
In all sites, selected vessel features (lumen cross-sectional area, number of vessels) were obtained in each ring for the common period 1985–2007. Vessel density (VD) was calculated as the ratio between the number of vessels and the total analysed area. The conductive area (CA) was calculated as the ratio between the total lumen area and the total analysed area. In addition to the mean cross-sectional VA of each tree ring, we also calculated the mean area of those vessels with areas above the 3rd quartile of lumen size (VA3q) and the mean area of the vessels located in the first half of the earlywood (VA1h), i.e., those located in the early earlywood (Supporting Information, Fig. S4). Previous studies suggested the selection of vessels according to their area or to their position within the ring for maximizing the climatic signal of vessel chronologies (García-González and Fonti 2006; Tardif and Conciatori 2006). Vessels were analysed for their frequency in 0.01 mm2 area classes and for the relative contribution of each area class to the total predicted hydraulic conductance (Kh; mm4), i.e., the sum of all the conduits diameters raised to the fourth power. Assuming that the vessel cross-sectional shape was circular, the predicted hydraulic conductance was calculated following the Hagen-Poiseuille law (Sperry et al. 1994).
To preserve the high-frequency variability in radial growth and earlywood anatomical features, tree-ring width and wood-anatomical series were standardized applying a two-step detrending using ARSTAN (Cook 1985). First, a negative exponential function was fitted to the measured values, and the second, detrending was based on a spline function with a 50 % frequency response of 20 years. Radial-growth and wood-anatomical indices were obtained by dividing the observed values by the expected ones estimated by the spline function. Then, autoregressive modelling was performed to remove the first-order temporal autocorrelation. Finally, a robust biweight mean was applied to average the standardized series and produce local residual chronologies of radial-growth (earlywood, latewood and tree-ring widths) and wood-anatomical (earlywood VA, CA and VD) variables. The quality of these chronologies was evaluated for the period 1985–2007 using the following dendrochronological statistics (Briffa and Cook 1990): the expressed population signal (EPS) which measures the tree-to-tree common growth variance and it is an indicator of chronology reliability as compared with a theoretical population’s chronology (Wigley et al. 1984); the mean correlation between trees (Rbt); the mean sensitivity (MSx) or the average relative difference between successive rings (Fritts 1976); the first-order autocorrelation coefficient (AR) based on raw data; the variance accounted for the first principal component (PC1); and the percentage of growth or wood anatomy variance explained by climate based on multiple linear regressions (R 2 climate).
Climatic data
Data from nearby meteorological stations were averaged for the period 1960–2007 to study growth–climate relationships (Supporting Information, Table S1). We built regional climatic time series due to the lack of local stations with complete records and to account for the spatial variability of climate across mountainous areas. Two regional series were built: one was used for the sites located along the altitudinal gradient (H, M, and L sites), and a second series was used for the xeric site (X). To estimate the missing data for each station and to combine them, we used the program MET from the Dendrochronology Program Library (Holmes 1992). For each station, monthly variables were transformed into normalized standard deviation to give each station the same weight in calculating the average values for each month and year. Regional climatic series were checked against spatially interpolated data for the Iberian Peninsula (Ninyerola et al. 2005). The temperature lapse rate (temperature = −0.06 elevation + 16.30, r = 0.88, P = 0.02) for the sites located in the altitudinal gradient was estimated using local climatic data. The precipitation amount increased upwards but the elevation–precipitation relationship was not linear. See additional details on climatic data in Alla et al. (2011).
To estimate the yearly water balance in each site, we used a modified Thornthwaite water-budget procedure (Willmott et al. 1985). Soil water balance was modelled by calculating soil–water withdrawal (actual evapotranspiration), recharge and surplus. Actual evapotranspiration was related to the percentage of the current soil at the available water capacity, and to potential evapotranspiration, which was estimated from monthly mean temperature data for the period 1960–2007. We calculated a site cumulative WD from previous September up to September of the year of tree ring formation, i.e., for the period when the studied species forms most of its tree ring and including previous months affecting growth the following year (Corcuera et al. 2004). Positive and negative WDs corresponded to dry and wet conditions, respectively. Calculations were done by using the AET software available at http://geography.uoregon.edu/envchange/pbl/software.html.
Simulated future climate
To assess the potential effects of future climatic conditions on growth and wood anatomy, we used projected monthly climatic data (mean temperature, total precipitation). We used climate predictions from two general circulation models (ECHAM4 and CGCM2) under two greenhouse gas emission scenarios covering half the range of the IPCC scenarios in terms of greenhouse gas (SRES scenarios A2 and B2). The IPCC (2007) recommends using a range of climate projections and emission scenarios to quantify uncertainty in predictive biological models. These are the emission scenarios and downscaled general circulation models currently available for the Iberian Peninsula (Brunet et al. 2007). These scenarios were also previously used to model the future distribution of Q. faginea in the Iberian Peninsula based on bioclimate models (Benito Garzón et al. 2008). Scenarios A2 and B2 correspond to a global intensive economy with rapid population increase and to local environmentally sustainable economies with lower population increase, respectively. The A2 and B2 scenarios would cause projected increases of 3–9 and 2–6 °C of the mean summer temperature over the study area during the late twenty-first century (Brunet et al. 2007). Predicted climate data were obtained for the late twenty-first century (period 2071–2100), and they were downscaled using an analogue method and provided by the Spanish National Meteorological Agency (AEMET). Simulated climatic data for the twenty-first century were compared with modelled data of the second half of the twentieth century (1961–1990) and with regional climatic series based on local climatic data (period 1960–2000) to check for seasonal and monthly consistencies (Supporting Information, Fig. S5).
Statistical analyses
All variables were checked for normality using the Shapiro-Wilks test prior to performing statistical analyses. The following variables were log-transformed: tree-ring and latewood widths, total VA and CA. Comparisons between variables were assessed by hierarchical linear mixed models, based on Restricted Maximum Likelihood methods, considering trees (random factors) nested within sites (fixed factors) (Littell et al. 2006). Differences between site means were assessed by Bonferroni tests when variances were equal or Dunnett’s T3 tests otherwise. The relative contribution of the inter-annual variability and the factor “trees” to the overall variance of indexed radial-growth wood-anatomical variables was also estimated considering the period 1990–2007 when the maximum sample size (number of cores) was reached in all study sites. We also assessed differences in the distribution frequencies of earlywood vessels according to their area using the G-test. All these analyses were performed using the MIXED procedure in SPSS 19.0 (SPSS, Chicago, USA).
The Pearson correlation coefficient (r) was used: (1) to assess the relationships between radial-growth and wood-anatomical indexed variables, and (2) to quantify the associations between growth and wood-anatomical indices and monthly (mean temperature, total precipitation) climatic data for the period 1985–2007. These analyses were performed considering the year of ring formation (t) and the previous (t − 1) and subsequent years (t + 1) (Tardif and Conciatori 2006). In the case of growth–climate correlations, we used a temporal window of 13 months from of the previous (year t − 1) to the current September (year t). We also calculated bootstrapped response functions among growth indices and climatic variables using the program PRECON (version 5.16) (Fritts et al. 1991). To perform a reliable growth–climate analysis for earlywood anatomical variables, usually characterized by a generally low common signal (low correlation between trees, Rbt) (e.g., Fonti and García-González 2004), only those variables with Rbt > 0.05 were included. Finally, we selected the climatic variables more tightly related to growth and anatomical data to assess the changes in these variables for the late twenty-first century as a function of forecasted climatic conditions.
Results
Comparisons between growth and earlywood anatomical variables
The tree-ring, earlywood and latewood widths differed significantly among sites with the highest values observed in sites H and M and the lowest ones in sites L and X (Table 2). The mean area of earlywood vessels showed the lowest values in the X (0.020 mm2) and H (0.027 mm2) sites, whereas VD showed the highest value in the X site (ca. 8 vessels mm−2). The percentage of earlywood CA reached the lowest and highest value in sites X (15.6 %) and M (19.5 %), respectively, with intermediate values in sites H and L (17.5 %).
Considering all the study sites, the vessels with smallest area (lower than 0.01 mm2) corresponded to 36 % of all measured earlywood vessels (Fig. 2), but they only contributed to about 1 % of the total predicted hydraulic conductance (Kh). These smallest area conduits comprised 44 % of all vessels in the X and L sites but only 26 % in the M and H sites (Fig. 2). Wide earlywood vessels with cross-sectional areas >0.05 mm2 were 15 % (accounting for 57 % of Kh) of all vessels in the H and M sites versus 6 % (accounting for 26 % of Kh) in the L and X sites, respectively. The distributions of the earlywood vessels according to their lumen area differed significantly among the four study sites (G = 32.5, P = 0.05).
Distribution of earlywood vessels by lumen cross-sectional area for Quercus faginea compared with their relative contribution to the total predicted hydraulic conductance (Kh). Data are presented for each study site (H, L, M, X) and for the combined data set (All) indicating the number of measured earlywood vessels (sample size, n)
Growth variables showed higher year-to-year variance than earlywood anatomical features which showed high tree-to-tree variance (Fig. 3). Indeed, the highest year-to-year variance was observed for the latewood width (46 %), whereas VA showed the highest tree-to-tree variance (46 %). CA and VD showed the lowest percentages of common year-to-year variance, being 7 and 8 %, respectively.
The mean between-trees correlation was higher in the radial-growth than in the wood-anatomical variables, reaching a maximum value of 0.66 for latewood width in site X (Table 3). The mean sensitivity of all measured variables was always higher in the X site than elsewhere, and it reached a maximum value of 0.63 for latewood width in that site (Table 3). The variance amount accounted for the PC1 was also the highest in the case of latewood width in site X (82 %), while in the case of VA, this variance was highest in sites H and M (64–69 %). The highest percentages of growth variance explained by climate corresponded to latewood and ring widths in sites X, L and H (42–57 %), whereas the highest percentages for earlywood anatomical variables were found for VA and CA in these same sites (39–48 %). The maximum correlation among radial-growth site chronologies was obtained between sites H and M for latewood width (r = 0.69, P = 0.01), but in the case of wood-anatomical features, the strongest association was found between L and X sites for CA (r = 0.43, P = 0.04).
Considering all sampled trees (n = 57), the mean VA for the 1985–2007 period was positively related to tree diameter (r = 0.37, P = 0.04) and also with total height (r = 0.27, P = 0.045). The latewood width of the year of tree-ring formation was positively associated with the earlywood width of the following year in the H and M sites. The earlywood CA and the tree-ring or latewood widths were negatively related in all sites excepting for site X (Table 4). The mean area of all earlywood vessels was more tightly related to the mean area of the third-quartile earlywood vessels (VA3q) than to the mean area of the first half of earlywood vessels (VA1h) (Supporting Information, Fig. S4). The CA was positively and significantly related to mean VA and density only in the sites L and X. In site X, VA was also related to tree-ring or latewood width of the preceding year, whereas VD was also associated with previous radial growth in the site H.
Relationships among climate, growth and earlywood anatomy
All sites showed either increases or decreases in radial growth in response to wet (e.g. 1988 and 1997) or dry conditions in spring or early summer (e.g. 1995 and 2005), respectively. However, such changes in growth were always more pronounced in the X site than elsewhere (Fig. 4). In fact, tree-ring and latewood widths were positively related to current-year June precipitation in the H and M sites but also to May or April precipitation in the L and X sites (Figs. 5, 6). Current spring rainfall (e.g. in April) was also positively related to earlywood width in the X site, but earlywood formation also depended on October precipitation of the year before tree-ring formation in the H site. The earlywood width was negatively related to current May temperature, whereas the latewood and tree-ring widths responded negatively to June and July temperatures. In the X site, high March temperatures were associated with a low production of earlywood and latewood.
Trends of earlywood and latewood widths in the four study sites (H high-elevation site, M mid-elevation site, L low-elevation site, X xeric site) as related to June precipitation. Note the high year-to-year variability in the series of the xeric site (X). The right-side axis in the upper graph describes the yearly sample size (number of analysed radii) whereas June precipitation, the climatic variable more related with latewood width, is represented in the lower graph as bars (different fills correspond to the two regional climatic series for sites H, M and L, and for site X, respectively)
Correlation coefficients calculated between earlywood, latewood and tree-ring width indices and monthly climatic data (T mean temperature, black bars; P total precipitation, white bars). Pearson correlations were calculated considering climatic data of the year of tree-ring formation (t, months abbreviated by capital letters) and the preceding year (t − 1, months abbreviated by minor letters) in the four study sites. The dashed lines correspond to the significance (P ≤ 0.05) thresholds for correlations and the asterisks indicate the significant bootstrapped regression coefficients
Predicted (twenty-first century) tree-ring width and earlywood vessel area in Q. faginea sites H (high-elevation site) and X (xeric site) based on the current (late twentieth century) associations between selected climatic variables and the mentioned growth and wood-anatomical variables. The associations were quantified using the displayed linear regressions. The graphs show current (empty box plots and symbols) and forecasted (filled box plots) climatic conditions (June precipitation, April temperature) for the last twenty-first century and corresponding tree-ring width and vessel-area indices. In the box plots, the thin and thick lines correspond to the median and the mean, respectively. The asterisks indicate significant (P < 0.001) differences between observed and predicted growth and anatomical variables for the twentieth and twenty-first centuries, respectively
Cumulative WD was negatively related to tree-ring (mean r for all sites was = −0.50, P < 0.05 in all cases) and latewood widths (mean r = −0.51, P < 0.05) in all sites, whereas considering anatomical variables, WD was only significantly related in a negative way to the mean area of the biggest vessels (3rd quartile of lumen size) in site X (r = −0.50, P = 0.03).
The chronologies of the widest earlywood vessels (VA3q) or those of the mean VA showed negative relationships with current February temperature in sites H, L and X (Table 5). The VA was also negatively related to previous December temperature in the site L and to current April temperature in the site X (Fig. 6). Nonetheless, the area of those vessels located in the first half of the earlywood (VA1h) was positively associated with February precipitation in the site X. The CA was negatively related to winter temperature and precipitation in site L, whereas winter and spring precipitation negatively affected CA and VD in site X.
We found that tree-ring width in site H and earlywood VA in site X during the late twentieth century were significantly (ANOVA, tree-ring width F = 31.2, earlywood VA, F = 75.5; P < 0.001 in both cases) higher than those predicted for the late twenty-first century (Fig. 6). The decrease in growth and VA was a response to the forecasted drier June and warmer April conditions in the late twenty-first century.
Discussion
Most published studies on climate–vessel relationships are based on correlative approaches and not on mechanistic models. Such findings must be discussed taking into account vessel ontogenesis or at least the vessel position within the ring as has been done for tree species with ring-porous (Fonti and García-González 2004) and semi-ring-porous or diffuse-porous wood types (Giantomasi et al. 2008). For instance, poplar trees formed smaller vessel cells in response to WD in early summer while no response was detected in vessels formed later (Arend and Fromm 2007). In ring-porous species such as Quercus robur, the earlywood vessel lumen area was positively related to late winter–early spring rainfall (February–April) (García-González and Eckstein 2003). The aforementioned studies show the adjustment of the earlier vessels to water availability in late winter and early spring before tree growth resumes. Accordingly, the lumen area of earlywood vessels of Q. faginea responded to February precipitation in the xeric site. In addition, the earlywood width was positively related to April and May precipitation in sites with the highest WD where both earlywood and latewood widths were lower than elsewhere. The plasticity of the growth–climate response was also detected in latewood and tree-ring widths which responded positively to June precipitation in the high-elevation site and to May precipitation in the low-elevation site. Such discrepancies may be explained by an earlier cambial reactivation in Q. faginea stands at low elevation where the first earlywood vessels may be formed between late March and early April when radial growth resumes (Alla, personal observation). This is also in agreement with the rapid cell enlargement of earlywood cells in oaks which may last less than 10 days (Zasada and Zahner 1969). The plastic response of Q. faginea growth to climatic variability along the studied climatic gradient does not support the forecasts based on bioclimatic envelope models predicting the local extinction of Q. faginea forests in xeric sites for the late twenty-first century (Benito Garzón et al. 2008).
The ring and wood-anatomical growth responses to climatic variability found in the xeric site, where January water availability determined earlywood CA and VA, were those expected for a drought-prone site. In addition, the lumen area of the first-formed earlywood vessels was also positively associated with February rainfall there, whereas the mean area of earlywood vessels was the lowest among all sites and consequently VD was the highest. As expected, the winter growth quiescence and deciduousness of Q. faginea and the Mediterranean summer drought confined most secondary growth to spring when climatic stress is low and consequently earlywood vessel area responded positively to late winter rainfall (Montserrat-Martí et al. 2009). However, the response of earlywood vessel area to winter precipitation was detected at annual (cumulative WD) and monthly scales in the more drought-stressed sites, whereas late winter temperatures were negatively related to earlywood VA in most sites as has been found before in other tree species with ring-porous wood (Tardif and Conciatori 2006; Fonti et al. 2006). Furthermore, at an annual scale, cumulative WD affected tree-ring and latewood growth in all sites. Such discrepancies indicate that the growth and earlywood anatomical responses to climate depend on the analysed time scale.
The low responsiveness of the mesic mid-elevation site to climatic variability was observed in both ring and anatomical variables. Such low sensitivity, the response of VA to early summer climatic conditions (e.g., June rainfall) and the highest mean VA values observed for this mesic site suggest a longer earlywood growth period there than in the other sites. Additionally, these results indicate that the climatic stress in the mid-elevation site is the lowest of all compared sites for the radial growth of Q. faginea.
In Q. faginea, radial-growth variables such as tree-ring and latewood widths were more valuable than wood-anatomical variables. First, obtaining wood-anatomical variables was much more time-consuming than taking width-related measures. Second, the latter variables (e.g. latewood width) showed a higher mean correlation between trees, a greater variance accounted for the PC1, and a higher year-to-year variability (mean sensitivity) than vessel-related variables (Figs. 2, 4; Table 3). Third, latewood-width chronologies were more sensitive to climatic variability than vessel-area chronologies (Table 3). Thus, we concur with Tardif and Conciatori (2006) on the limited use of vessel-area chronologies as dendroclimatic records as compared with width-related measures. However, earlywood VA contained unique climatic information related to late winter temperature and precipitation, and the earlywood climatic sensitivity increased in the cold and more drought-stressed sites. Thus, the dendroclimatic potential of wood-anatomical studies in Mediterranean oaks is high in continental and xeric sites where climatic stress constrains growth through low winter temperatures and summer drought. Such anatomy–climate correlations present some weaknesses since (1) these relationships may not hold under future climatic regimes and (2) earlywood VA is a proxy of mechanistic variables as hydraulic conductivity. To amend these shortcomings, we advise (1) combining wood-anatomical studies with phenological measures (e.g. Eilmann et al. 2009) and (2) quantifying how unstable anatomy–climate associations are using series covering at least the past century.
The lower common signal in wood-anatomical features may seem to be a minute reward for the time-consuming task which involves generating well-replicated vessel series. Furthermore, if the climatic signal is weak, then the apparent new climatic information provided by wood anatomy may not be relevant for robust paleoclimatic reconstructions. Such limitation is probably derived from the lack of consensus and uniform procedures to select and define the most “climatically relevant” vessels which may contain different climatic information depending on vessel ontogenesis and location within the ring (Fonti and García-González 2004; García-González and Fonti 2006). Appropriate wood-anatomical information should be derived from the measurement of contemporaneous vessels with similar transversal area, if the ontogenetic origin of vessels may be properly discerned. In ring-porous wood, contemporaneous vessels are not perfectly arranged in tangential rows. The lack of a clear climatic signal in the Q. faginea vessel chronologies may be related to their shortness or to the fact that climate series were built using data from local stations situated along an altitudinal gradient. However, such methodological caveats do not explain our results since the common signal strength statistics, and the climatic responses of Q. faginea vessel chronologies were similar to those found in other study (Corcuera et al. 2004).
The high variability of vessel cross-sectional area found among Q. faginea stands located in sites with contrasting climatic conditions indicates that wood-anatomical features may be an additional and valuable source of information to assess how plastic growth and phenology responses are to local climatic variability. Moreover, the anatomical response of earlywood features to late winter temperatures could be used as a growth indicator of spring growth resumption in combination with phenological data such as bud burst and leaf unfolding (Čufar et al. 2008).
The presented results emphasize the site-to-site variability in the wood-anatomical response to climate. To improve the climatic information provided by the earlywood vessels, it has been suggested to select vessels according to their transversal area (García-González and Fonti 2006). Our results support this suggestion because most relationships between vessel area and climate in Q. faginea were more evident when using the widest earlywood vessels. Exclusively, the lumen area of the first-formed earlywood vessels responded positively to late winter rainfall in the xeric site. The reasons for the observed differences in our results compared with some previous works can probably be attributed to the dissimilarity between areas of study, the different species examined, the diverse different growing conditions and the contrasting internal controls of earlywood formation (Woodcock 1989).
In the case of Q. faginea, water shortage was the most important factor influencing xylem anatomy as compared with the co-occurring Q. ilex which is better adapted to summer drought (Villar-Salvador et al. 1997). However, we also found that the spring resumption of secondary growth in Q. faginea was also affected by conditions during the previous cold season since winter temperatures were negatively related to earlywood conductivity in the low-elevation site. We also uncovered a negative relationship between February temperature and earlywood vessel lumen area not only in high-elevation cold sites, but also in the dry low-elevation and xeric sites. Low temperature seems to be the main climatic stress in the uppermost site where Q. faginea grows close to its upper distribution limit. A possible explanation for the temperature-constraint found in the low-elevation site can be linked to the frequent thermal inversions and persistent fogs observed there where winter minimum temperatures are episodically lower than in upper sites (Alla et al. 2011). Late winter and early spring temperatures influence the physiological processes involved in spring xylogenesis such as vessel enlargement and differentiation, affecting the final lumen area of earlywood vessels in species with ring-porous wood (Fonti et al. 2006). The negative relationships between late winter temperatures before bud break and earlywood vessel lumen area may be due to an earlier differentiation of overwintering cambial derivatives (Aloni 1991). Warmer winter temperatures may cause cambium reactivation leading to the formation of narrower earlywood vessels with low theoretical hydraulic conductance.
Based on wood-anatomical and isotopic-discrimination data, Ponton et al. (2001) suggested that trees with a smaller earlywood VA might compensate the loss in hydraulic conductance through an increase in water-use efficiency. If the predicted climate warming for the Mediterranean Basin leads to warmer winters (IPCC 2007), the mean area of the widest earlywood vessels will decrease leading to a loss in total predicted hydraulic conductance which will negatively affect the development of the new canopy foliage in spring. Such loss in hydraulic conductance will be expected in Q. faginea stands similar to the cold high-elevation and xeric low-elevation study sites. However, if climate warming causes an increase in spring evapotranspiration, the earlywood CA will increase in drought-prone sites similar to the xeric sites. Therefore, winter temperatures can be the main climatic driver of the theoretical hydraulic conductance in Mediterranean deciduous oaks as Q. faginea.
Our results do not support the local disappearance of the Iberian Q. faginea populations near their xeric distribution limit in response to future warmer and drier conditions. The evidenced growth plasticity of these “rear-edge” populations from xeric sites in response to recent climatic variability suggests that they will persist during the twenty-first century unless thresholds even greater than those linked to climatic projections are surpassed. Climate-driven predictive models should introduce some measure of growth plasticity and variability to evaluate the uncertainty in their long-term forecasts of persistence of tree populations. The plastic responses of Q. faginea to climate among sites and across time indicate that when predicting changes in growth and wood anatomy of trees, the variance matters more than the mean.
References
Alla AQ, Camarero JJ, Rivera P, Montserrat-Martí G (2011) Variant allometric scaling relationships between bud size and secondary shoot growth in Quercus faginea: implications for the climatic modulation of canopy growth. Ann For Sci 68:1245–1254
Aloni R (1991) Wood formation in deciduous hardwood trees. In: Raghavendra AS (ed) Physiology of Trees. Wiley, New York, pp 75–197
Amaral Franco J (1990) Quercus. In: Castroviejo S, Laínz M, López González G, Montserrat P, Muñoz Garmendia F, Paiva J, Villar L (eds) Flora Iberica. Real Jardín Botánico, CSIC, Madrid, pp 15–36
Arend M, Fromm J (2007) Seasonal change in the drought response of wood cell development in poplar. Tree Physiol 27:985–992
Baas P, Schweingruber FH (1987) Ecological trends in the wood anatomy of trees, shrubs and climbers from Europe. IAWA Bull 8:245–274
Benito Garzón M, Sánchez de Dios R, Sainz Ollero H (2008) Effects of climate change on the distributions of Iberian forest. Appl Veg Sci 11:169–178
Briffa K, Cook ER (1990) Methods of response function analysis. In: Cook ER, Kairiuktsis LA (eds) Methods of dendrochronology. Kluwer, Dordrecht, pp 240–247
Brunet M, Casado MJ, Castro M et al (2007) Generación de Escenarios de Cambio Climático Para España. Ministerio de Medio Ambiente, Madrid
Camarero JJ, Olano JM, Parras A (2010) Plastic bimodal xylogenesis in conifers from continental Mediterranean climates. New Phytol 185:471–480
Campelo F, Nabais C, Gutiérrez E, Freitas H, García-González I (2010) Vessel features of Quercus ilex L. growing under Mediterranean climate have a better climatic signal than tree-ring width. Trees 24:463–470
Carlquist S (1975) Ecological strategies of xylem evolution. University of California Press, Berkeley
Cook ER (1985) A time series analysis approach to tree-ring standardization. University of Arizona, Tucson
Corcuera L, Camarero JJ, Gil-Pelegrin E (2004) Effects of a severe drought on growth and wood anatomical properties of Quercus faginea. IAWA J 25:185–204
Corcuera L, Camarero JJ, Sisó S, Gil-Pelegrin E (2006) Radial-growth and wood-anatomical changes in overaged Quercus pyrenaica coppice stands: functional responses in a new Mediterranean landscape. Trees 20:91–98
Čufar K, Prislan P, de Luis M, Gričar J (2008) Tree-ring variation, wood formation and phenology of beech (Fagus sylvatica) from a representative site in Slovenia, SE Central Europe. Trees 22:749–758
Eilmann B, Zweifel R, Buchmann N, Fonti P, Rigling A (2009) Drought-induced adaptation of the xylem in Scots pine and pubescent oak. Tree Physiol 29:1011–1020
Ellmore GS, Ewers FW (1986) Fluid flow in the outermost xylem increment of a ring-porous tree, Ulmus americana. Am J Bot 73:1771–1774
Fonti P, García-González I (2004) Suitability of chestnut earlywood vessel chronologies for ecological studies. New Phytol 163:77–86
Fonti P, Solomonoff N, García-González I (2006) Earlywood vessels size of Castanea sativa records temperature before their formation. New Phytol 173:562–570
Fonti P, von Arx G, García-González I, Eilmann B, Sass-Klaassen U, Gärtner H, Eckstein D (2010) Studying global change through investigation of the plastic responses of xylem anatomy in tree rings. New Phytol 185:42–53
Frankenstein C, Eckstein D, Schmitt U (2005) The onset of cambium activity—a matter of agreement? Dendrochronologia 23:57–62
Fritts HC (1976) Tree rings and climate. Academic Press, New York
Fritts HC, Vaganov EA, Sviderskaya IV, Shashkin AV (1991) Climatic variation and tree-ring structure in conifers: empirical and mechanistic models of tree-ring width, number of cells, cell-size, cell-wall thickness and wood density. Clim Res 1:97–116
García-González I, Eckstein D (2003) Climatic signal of earlywood vessels of oak on a maritime site. Tree Physiol 23:497–504
García-González I, Fonti P (2006) Selecting earlywood vessels to maximize their environmental signal. Tree Physiol 26:1289–1296
García-González I, Fonti P (2008) Ensuring a representative sample of earlywood vessels for dendroecological studies: an example from two ring-porous species. Trees 22:237–244
Giantomasi MA, Roig FA, Villagra PA, Srur AM (2008) Annual variation and influence of climate on the ring width and wood hydrosystem of Prosopis flexuosa DC trees using image analysis. Trees 23:117–126
Granier A, Anfodillo T, Sabatti M, Cochard H, Dreyer E, Tomasi M, Valentini R, Bréda N (1994) Axial and radial water-flow in the trunks of oak trees—a quantitative and qualitative analysis. Tree Physiol 14:1383–1396
Hacke UG, Sperry JS (2001) Functional and ecological xylem anatomy. Persp Plant Ecol Evol Syst 4:97–115
Holmes RL (1983) Computer-assisted quality control in tree ring dating and measurement. Tree Ring Bull 43:69–78
Holmes RL (1992) Dendrochronology program library. University of Arizona, Tucson
IPCC (2007) The physical science basis. Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. University Press, Cambridge
Jiménez P, Díaz-Fernández PM, Martín S, Gil L (1998) Regiones de Procedencia de Quercus pyrenaica W.; Quercus faginea Lam. y Quercus canariensis Willd. Dirección General de Conservación de la Naturaleza, Madrid
Littell RC, Milliken GA, Stroup WW, Wolfinger RD, Schabenberger O (2006) SAS for mixed models. SAS Institute, Cary
Loidi J, Herrera M (1990) The Quercus pubescens and Quercus faginea forests in the Basque Country (Spain): distribution and typology in relation to climatic factors. Vegetatio 90:81–92
Montserrat-Martí G, Camarero JJ, Palacio S, Pérez-Rontomé C, Milla R, Albuixech J, Maestro M (2009) Summer-drought constrains the phenology and growth of two co-existing Mediterranean oaks with contrasting leaf habit: implications for their persistence and reproduction. Trees 4:787–799
Ninyerola M, Pons X, Roure JM (2005) Atlas Climático Digital de la Península Ibérica. Metodología y aplicaciones en bioclimatología y geobotánica. Universidad Autónoma de Barcelona, Bellaterra
Ponton S, Dupouey JL, Bréda N, Feuillat F, Bodénès C, Dreyer E (2001) Carbon isotope discrimination and wood anatomy variations in mixed stands of Quercus robur and Quercus petraea. Plant, Cell Environ 24:861–868
Pumijumnong N, Park WK (1999) Vessel chronologies from teak in Northern Thailand and their climatic signal. IAWA J 20:285–294
Sass U, Eckstein D (1995) The variability of vessel size in beech (Fagus sylvatica L.) and its ecophysiological interpretation. Trees 9:247–252
Sperry JS, Nichols KL, Sullivan JEM, Eastlack SE (1994) Xylem embolism in ring-porous, diffuse-porous and coniferous trees of northern Utah and interior Alaska. Ecology 75:1736–1752
Suzuki M, Yoda K, Suzuki H (1996) Phenological comparison of the onset of vessel formation between ring-porous and diffuse-porous deciduous trees in a Japanese temperate forest. IAWA J 17:431–444
Tardif JC, Conciatori F (2006) Influence of climate on tree rings and vessel features in red oak and white oak growing near their northern distribution limit, southwestern Quebec, Canada. Can J For Res 36:2317–2330
Tessier L, Nola P, Serre-Bachet F (1994) Deciduous Quercus in the Mediterranean region: tree-ring/climate relationships. New Phytol 126:355–367
Villar-Salvador P, Castro-Díez P, Pérez Rontomé C, Montserrat-Martí G (1997) Stem xylem features in three Quercus (Fagaceae) species along a climatic gradient in NE Spain. Trees 12:90–96
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time-series, with applications in dendroclimatology and hydrometeorology. Int J Clim Appl Meteorol 23:201–213
Willmott CJ, Rowe CM, Mintz Y (1985) Climatology of the terrestrial seasonal water cycle. J Climatol 5:589–606
Wimmer R (2002) Wood anatomical features in tree-rings as indicators of environmental change. Dendrochronologia 20:21–36
Woodcock D (1989) Climate sensitivity of wood-anatomical features in a ring-porous oak (Quercus macrocarpa). Can J For Res 19:639–644
Zasada JC, Zahner R (1969) Vessel element development in earlywood of red oak (Quercus rubra). Can J Bot 47:1965–1971
Acknowledgments
A. Q. Alla thanks the support of CIHEAM-IAMZ and AECID grants. This study was supported by the projects CGL2007-66066-C04-02/BOS, CGL2008-04847-C02-01, RTA2005-00100-CO2-00 and SUM2006-00025-00-00 (Spanish Ministry of Science). JJC acknowledges funding by ARAID and collaborative effort within the Globimed network (http://www.globimed.net). We thank A.E. El-Kenawy for revising a previous version of the manuscript and J. Albuixech for his help in the field. We also thank the Spanish Meteorological Agency (AEMET) for providing climate data and predictions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Seifert.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alla, A.Q., Camarero, J.J. Contrasting responses of radial growth and wood anatomy to climate in a Mediterranean ring-porous oak: implications for its future persistence or why the variance matters more than the mean. Eur J Forest Res 131, 1537–1550 (2012). https://doi.org/10.1007/s10342-012-0621-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-012-0621-x