Abstract
Little information is available on the sleeping cluster pattern and retiring behavior of Sichuan snub-nosed monkeys (Rhinopithecus roxellana). Here, we provide observational data on a provisioned free-ranging band in the Qinling Mountains, central China. The results suggest that winter night activity of R. roxellana is a compromise between antipredator and thermoregulatory strategies and an adaptation to ecological conditions of their temperate habitat. Monkeys retired between 1804 and 1858 h in winter. In support of the antipredation hypothesis, all monkeys slept in trees at night, whereas 18.8% of individuals slept on the ground during the day. Also, the study band was more spatially cohesive at night than in daytime, with shorter distances between one-male units. Keeping warm is critical for survival in freezing temperatures. Monkeys often slept in the lower stratum of the tree canopy, avoiding the upper canopy where it is cold and windy. They formed larger sleeping clusters at night than in daytime. The most common types of night-sleeping clusters were adult females and juveniles, followed by adult females with other adult females. These accounted for 60.2% of the total records. The frequency of female–male clusters is two times greater, and that of adult male–juvenile clusters was four times less at night than during the day. The variations in composition of sleeping clusters suggest affiliative patterns at night-sleeping sites differ from those during the day.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sleeping clusters, in which two or more individuals maintain body contact for extended periods, are characteristic of most primate species and represent an important part of social behavior (Anderson 2000). Studies on sleeping clusters and night-retiring behaviors have been done in chimpanzees, bonobos, gibbons, baboons, and New World monkeys (e.g., Lophocebus albigena, Chalmers 1968; Papio ursinus, Stolz and Saayman 1970; Macaca radiata, Koyama 1973; M. mulatta, Vessey 1973; Miopithecus talapoin, Gautier-Hion 1973; Papio papio, Anderson and McGrew 1984; P. hamadryas, Kummer et al. 1985; Hylobates lar, Reichard 1998; M. fuscata, Zhang and Watanabe 2007; Pan troglodytes, Ogawa et al. 2007; Pan paniscus, Mulavwa et al. 2010), but little is known about colobine species, especially those taxa living in areas characterized by cold night temperatures (Anderson 2000).
From socioecological perspectives, there are several hypotheses to explain the formation of sleeping clusters. The thermoregulation hypothesis suggests that a primary function of sleeping clusters is the conservation of heat during cold temperatures (Takahashi 1997). In Guinea baboons, pigtail macaques (M. nemestrina), and rhesus macaques, individuals are most often seen in sleeping clusters in cold weather, on windy nights, or during heavy rain (e.g. Southwick et al. 1965; Bernstein 1972; Anderson and McGrew 1984). Sleeping cluster sizes of Japanese macaques are reported to be larger on colder nights than on warmer nights (Takahashi 1997). Along with thermoregulation, safety from predators is an important principle underlying the formation of sleeping clusters in primates. Many predators of primates, e.g., felids and snakes, are mainly crepuscular or nocturnal hunters (Meddis 1979). The antipredation hypothesis suggests that increased cohesion and large sleeping congregations might facilitate predator detection and enhance group defense (Anderson 1984; Ogawa et al. 2007). In Ethiopia, hamadryas baboons often aggregate at the same sleeping site: several hundred individuals sleep on cliffs, presumably to avoid predators; conversely, hamadryas baboons in areas of Saudi Arabia, where predators are scare, sleep with much smaller numbers of individuals on easily accessible slopes (Kummer et al. 1985). Sleeping groups of rhesus macaques at night occupied less than half of the area in which they were dispersed during the day (Vessey 1973). Moreover, formation of sleeping clusters can be influenced by other environmental factors, such as availability of suitable sleeping sites. Neyman (1978) found that cotton-top tamarin (Saguinus oedipus) social groups usually sleep in a single cluster, but members sometimes split into two clusters, possibly in response to limited space on branches. Davidge (1978) considered that chacma baboon sleeping clusters might be shaped by physical characteristics of the sleeping site, including size, strength, and location of suitable branches and substrates.
Night-sleeping clusters can reveal information about social dynamics, as patterns of association may differ from those during the day. Adult females were the most regular members of night-sleeping clusters in Japanese monkeys, rhesus monkeys, Guinea baboons, and talapoin monkeys (Gautier-Hion 1973; Vessey 1973; Dunbar 1992; Takahashi 1997). In these species, social partners who are tolerant of each other (based either on kinship or affiliative relationships) frequently huddled together during the night, whereas less closely associated individuals were often rejected when attempting to join existing clusters (Anderson 1998). Interestingly, male Japanese monkeys sometimes huddled together during winter nights; conceivably, cold temperatures increased their mutual tolerance, facilitating the formation of sleeping clusters (Wada and Tokida 1985).
The Sichuan snub-nosed monkey (Rhinopithecus roxellana) is an Asian colobine that inhabits temperate forests in the mountainous highlands of central and southwestern China at altitudes between 1,500 and 3,400 m amsl (Chen et al. 1989; Hu et al. 1989; Li et al. 2000). The fundamental social unit of this species is the one-male, multifemale unit (OMU), with several OMUs sharing the same home range and forming a large band (Zhang et al. 2006b). Bad size commonly ranges between 100 and 300 individuals (Ren et al. 2000; Zhang et al. 2008a). Affiliative interactions, such as huddling, sitting in contact, sitting in proximity, and grooming, usually occur among members of the same OMU, whereas interunit affiliative interactions are rare (Xi et al. 2008). Copulations occur mostly within OMUs (Qi et al. 2008). Approaches to within 2 m among adults from different OMUs often result in aggression or avoidance (Zhang et al. 2008b). In contrast to the abundance of information about daytime activities, little is known concerning nighttime activities of snub-nosed monkeys. Hu et al. (1989) noted that all members of a captive OMU often huddle in a single cluster on the ground during the day. They proposed that huddling relationships at night might resemble those during the day. Chen et al. (1989) argued that Sichuan snub-nosed monkeys in the wild sleep primarily in trees. They noted that monkeys usually slept from 1830 to 0600 h (the next morning) in winter and spring and from 2000 to 0600 h in summer and autumn. Previous studies were descriptive, with no focus on nighttime activities; therefore precise information regarding retiring behaviors is not yet available for this species.
In this paper, we present data on sleeping cluster patterns and retiring behaviors in a free-ranging band of R. roxellana living in the Qinling Mountains, China. We attempt to test the following hypotheses and predictions. Regarding the antipredation strategy hypothesis, we predicted that: (1) monkeys would sleep in places difficult for predators to access (e.g., in trees) and avoid sleeping on the ground at night; and (2) that distances between OMUs at night would be shorter than during the day, because increased cohesion and large sleeping congregations might facilitate predator detection and enhance group defense. Regarding the thermoregulation hypothesis, we predicted that: (1) sleeping cluster sizes would vary with temperature changes—that night-sleeping clusters would be larger than daytime clusters; (2) in winter, monkeys would choose warm sleeping sites, avoiding the high canopy where it is windy and cold. Additional comparisons of night and day clusters aimed to assess the flexibility of cluster formation and social relations.
Methods
Study sites and subjects
The study was carried out on the northern slope of the Qinling Mountains near Yuhuangmiao Village in Zhouzhi National Nature Reserve, Shaanxi, central China (33°48′68″N, 108°16′18″E, Fig. 1). The climate is considered to be semihumid mountainous, with snowfall common in winter between November and February (Li et al. 2000). The average annual temperature was 6.4°C, with a minimum of −12°C in January and maximum of 21.7°C in July in 2001 (Zhang et al. 2006a). The vegetation type is secondary deciduous broadleaf forest mixed with coniferous trees, ranging from 1,400 to 2,896 m amsl. Several potential predators including panthers (Panthera pardus), jackals (Cuon alpinus), lynxes (Lynx lynx), wolves (Canis lupus), and goshawks (Accipiter gentilis) were reported in the habitat (Shi et al. 1982; Hu et al. 1989; Zhang et al. 1999).
Two bands of Sichuan snub-nosed monkeys, East Ridge (ER) Band and West Ridge (WR) Band, live in the study area and are separated by the Nancha River. The WR band has a range of 9.2 km2 in winter, tending to migrate to lower elevations than in spring (Li et al. 2000). This band is habituated and has been observed since 1995 (Li et al. 2000). To achieve better observation conditions, provisioning commenced on 24 October 2001. Based on accessibility and the desire to avoid human disturbance, provisioning was restricted to the lower part of the Gongni Valley in an area where the band was normally observed to forage (Zhang et al. 2003). A total of 3.5 kg of corn grain and 20 kg of sliced apples and radishes were scattered across the valley at a fixed location at 0900, 1200, and 1500 h. The quantity of provisioned food was approximately 200 g per individual per day, which is a minor proportion of their daily food consumption (Qi 1989). The monkeys also commonly foraged for natural food items at the provisioning site. In order to reduce the influence of provisioning on their ecology, we limited provisioning to observation periods, which cover approximately 4 months of the year. Observations and provisioning mostly took place in winter when the foliage was less dense. We also avoided hand feeding or other physical contact with monkeys. No observers were present at other times.
Data collection
Observations were conducted on 28 nights and 73 days from 11 November 2002 to 31 February 2003. The band comprised 65 individuals (eight adult males, 28 adult females, 21 juveniles, eight infants) distributed across eight OMUs. Members of OMUs were usually located in the same tree or in neighboring trees (Qi et al. 2004). No solitary males or all-male groups were observed during the study period. Due to the close observation distance (range 2–50 m), we could identify all adults and most adolescent monkeys in the band based on sex, body size, pelage color, and any physical anomalies. Monkeys were categorized into four age/sex classes following Zhang et al. (2003): adult males and females (estimated to be 5 years or older), juveniles (estimated to be from 2 to 5 years old), and infants (estimated to be <2 years old). Scan sampling (Altmann 1974) was used to collect data on cluster size and composition. To exclude the influence of provisioning, data were not collected within the hour following provisioning. We scanned the study group four times per day when monkeys were sleeping: at 1100 and 1300 h for daytime data, and 1900 and 2000 h for nighttime data (sunset was between 1750 and 1843 h during the study period). We quantified cluster size as the number of adults and juveniles in a cluster; infants were not counted because they were often carried by their mothers or other females and were thus difficult to observe. We also recorded the presence and number of individuals remaining outside the clusters. We recorded weather conditions, cluster location, size, and composition, and activities. As identifying unit members was difficult at night, the distance among OMUs was estimated by measuring distances among resident males in OMUs. We located the sleeping place of each resident male on a topographic map and measured the estimated distances among males (e.g., 28 dyads for eight resident males in each scan). A total of 784 dyads of male–male distances at night and 2,030 dyads in the day were recorded (one resident male was not found for 2 days during the study). We measured air temperature at approximately 1.5 m above the ground immediately following each scan. Presleeping and nocturnal behaviors were observed using a night viewer (Yukon Nvmt 2 × 24, plus an extra lens, Yukon 29053 Nvmt 4 × 50 mm). Spotlights were occasionally used to aid visibility; these did not appear to influence the monkeys’ behavior. The research and care of the study monkeys abides by the rules of Zhouzhi National Reserve, China, and by the national and institutional guidelines for the care and management of primates established by the Primate Research Institute, Kyoto University, Japan.
Data analysis
We calculated mean cluster size and age–sex composition of sleeping clusters during the day and night, and analyzed their relationships with temperature. To avoid resampling bias, we analyzed only one scan per day and night, respectively – those containing at least 90% of group members. In total, data from 28 night scans and 73 day scans were analyzed. We used chi-square tests and independent samples t tests to examine differences in cluster sizes, numbers of adults in each cluster, and distances between OMUs in daytime versus nighttime. We used Kendall’s rank correlation coefficient to analyze the association between temperature and cluster formations. All statistical tests were two-tailed, with α < 0.05.
Results
Retiring activities and spatial location of sleeping sites
Monkeys in the study band arrived at the sleeping site by 1828 h ± 14 min (n = 28, range 1804–1858 h). The retiring time was 26 ± 16 min later than sunset (1757 h ± 14 min). Monkeys were observed foraging in trees at sunset and then moving to the sleeping site while vocalizing. Sleeping sites were not fixed but were commonly within 500 m of the study band’s last feeding site. There was no indication that OMUs competed for sleeping trees or for favored areas within sleeping trees. Of a total of 392 trees used for sleeping, 241 (61.5%) were estimated to be taller than 15 m. Of a total of 1,788 individual nighttime records, 1,582 individuals (88.5%) were observed sleeping in the lower canopy (no more than two thirds of the height of the vegetation), whereas 206 individuals (11.5%) were observed in the upper canopy. No monkeys were found to sleep on the ground at night. During the day, by contrast, monkeys slept on the ground and in the lower and higher canopies, accounting for 879 (18.8%), 2,760 (59.0%), and 1,039 (22.2%) records, respectively (total 4,678). The proportion of individuals sleeping in trees at night was significantly higher than in daytime (χ2 = 33.2, p < 0.001), as was the proportion of individuals sleeping in the lower canopies (χ2 = 24.5, p < 0.001).
Age–sex composition of night and day sleeping clusters
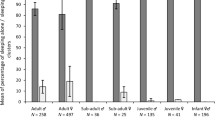
The age–sex composition of 418 of 549 night-sleeping clusters (76%) and of all 1,243 daytime clusters was identified (Table 1). At night, the most common cluster consisted of adult females and juveniles (FJ), followed by female–female clusters (FF); these accounted for 60.2% of all clusters. Adult male–female (MF) and adult male–female–juvenile (MFJ) clusters were also commonly observed. During the day, by contrast, adult males and females huddled together less frequently; MF clusters accounted for 14.3% of night clusters but only 9.1% of daytime clusters (χ2 = 1.3, p > 0.05). MFJ clusters were considerably less frequent during daytime than at night (4.1 vs. 13.4%: χ2 = 5.46, p < 0.05, Table 1). All-juvenile (JJ) and juvenile–male (JM) clusters were more than twice as frequent in daytime than at night (JJ cluster 26 vs. 10%, χ2 = 8.9, p < 0.01; MJ cluster 8 vs. 2%, χ2 = 4, p < 0.05). In addition, sleeping clusters formed more frequently at night than during the day [1,642/1,788 individual night records (91.8%), 3,582/4,678 day records (76.6%), χ2 = 4.06, p < 0.05].
Night and day sleeping cluster sizes and the number of adults
Night clusters were larger than daytime clusters (t = −3.04, df = 1,791, p < 0.001). Also, the number of adults in clusters was greater at night (t = −7.82, df = 1,348, p < 0.01). The average size of night clusters was 2.99 ± 0.8 (range 2–5), with 89.4% of clusters including adults. The mean number of adults per cluster was 1.7 ± 0.6 (range 1–3) (Table 2). In contrast, the average size of daytime clusters was 2.8 ± 1.1 (range 2–8), with 74.1% including adults. The mean number of adults per daytime cluster was 1.4 ± 0.6 (range 1–5). Clusters of more than five individuals were occasionally observed on the ground.
Distance between OMUs during sleep at night and in the daytime
Inter-OMU distances in the study band were significantly shorter at night than in the daytime (Table 3, t = −23, df = 2,813, p < 0.001). The mean distance at night was 17.5 ± 16.8 m (range approximately 5–50 m) (n = 784 dyads) compared with 40 ± 26.8 m (range 5–300 m) (n = 2,030 dyads) during the day.
Influence of ambient temperature on sleeping clusters
The air temperature and the average size of sleeping clusters were significantly negatively correlated during the day (τ = −0.83, n = 20, p < 0.001, Fig. 2). There was also a significant negative correlation between air temperature and number of adults in sleeping clusters (τ = −0.80, n = 20, p < 0.001). On cold or windy days, monkeys often formed clusters on the ground. Large clusters were formed by all OMU members huddling together on the ground. At night, all monkeys slept in trees. Although nighttime temperatures (range −13 to 1°C) were lower than in daytime (ranges −13 to 1°C vs. −6 to 16°C) (z = −3.8, p < 0.001), the correlations between temperature and average sleeping cluster size (τ = −0.19, n = 14, p > 0.05) and temperature and number of adults per cluster (τ = −0.03, n = 14, p > 0.05) were not significant.
Discussion
Retiring behaviors and antipredator strategies of R. roxellana
R. roxellana sleep in trees at night. In contrast to the many observed cases of sleeping in clusters on the ground during the day, we never observed sleeping clusters on the ground at night. It seems likely that nighttime arboreal sleeping sites are used to minimize predation risk. For example, trees are generally difficult for potential nocturnal terrestrial predators such as jackals and wolves to access (Chen et al. 1989). Furthermore, sleeping in higher locations allows a good view of the surroundings, which may facilitate early predator detection (Anderson 2000; Ogawa et al. 2007). Consistent with this idea, snub-nosed monkeys frequently monitored the surroundings immediately before retiring for the night (Chen et al. 1989). Goshawks were reported to prey on young R. roxellana playing in the canopy during the day (Zhang et al. 1999). At night, however, young monkeys may reduce their predation risk by joining sleeping clusters with adults or other individuals.
In addition to selecting safe sleeping trees, the study band was more spatially cohesive during the night than during the day, with shorter distances between OMUs. The increased nighttime cohesiveness may also reflect an antipredator strategy. Spatial aggregations of individuals at night have been documented in many primate species, e.g., western black-and-white colobus (Colobus polykomos) and red colobus (C. badius, Booth 1956), Hanuman langurs (Presbytis entellus, Jay 1965), long-tailed macaques (Bernstein 1972), bonnet macaques (Rahaman and Parthasarathy 1969), and baboons (Papio spp. Hamilton 1982). However, members of different R. roxellana OMUs rarely interact affiliatively during the day (Qi et al. 2004); close proximity can lead to social conflict. Resident males in different OMUs typically remained at least 5 m apart, with the average nighttime distance between OMUs being about 20 m. During the day when the threat of predation is weaker, the distance between OMUs increased. These variations suggest that OMUs form spatial aggregations at night as an antipredator adaptation, although individuals avoid clustering with members of different OMUs.
Although common in R. roxellana, sleeping cluster sizes are relatively small. This species lives in large bands of up to 600 individuals (Hu et al. 1989). It is therefore theoretically possible for them to form very large sleeping clusters, similar to the large groups of >100 individuals huddling together in Japanese macaques (Zhang and Watanabe 2007). Our data show that R. roxellana form larger clusters at night than in daytime; however, unlike Japanese macaques (trees, rocks, ground), they sleep exclusively in trees at night. The size of their night sleeping clusters is restricted by the limited space available on branches. By contrast, in daytime, the monkeys are often on the ground, where they form larger clusters when it was cold. Sleeping cluster size is also restricted by subgrouping patterns. Our study band consisted of eight OMUs ranging from five to 11 members (including infants). On cold days, monkeys formed clusters on the ground of up to eight individuals, comprising most members of an OMU. Cluster formation of R. roxellana is thus influenced by environmental factors such as spatial constraints and temperature, and by social factors such as OMU size.
Compromise between thermoregulatory and antipredator strategies
Although sleeping high in the canopy may decrease predation risk (Anderson 2000), R. roxellana did not do so in this study. Instead, they slept mostly in the lower strata. These observations suggest a tradeoff between predator avoidance and thermoregulation; keeping warm is also critically important for survival during winter. R. roxellana inhabits the most northerly range of all colobines, with temperatures frequently below freezing during winter nights. Japanese macaques, found at latitudes similar to those of R. roxellana, also frequently form clusters on low branches where they are better sheltered from wind and snow. These macaques prefer to sleep on lower branches of coniferous trees, where ambient temperatures are approximately 1–4°C higher than in the higher canopies of deciduous oak trees (Wada and Tokida 1985). Sleeping-site selection by R. roxellana may thus represent a compromise between the opposing needs to both avoid terrestrial predators and adequately thermoregulate in their temperate habitat.
Compromise between foraging requirements and anti-predation strategy
To avoid crepuscular or nocturnal predators, many diurnal primates typically enter their sleeping sites before sunset (Anderson 2000), as observed in chacma baboons (Davidge 1978), hamadryas baboons (Sigg and Stolba 1981), rhesus monkeys (Makwana 1978), green monkeys (Cercopithecus sabaeus, Harrison 1983), chimpanzees (Ogawa et al. 2007), and bonobos (Mulavwa et al. 2010). Agile gibbons (Hylobates agilis) retired approximately 3 h before sunset during the wet season (Gittins 1982). In contrast, R. roxellana continued foraging after sunset and did not arrive at their sleeping sites until later. This observation is consistent with a previous study of R. roxellana in Wolong Reserve (Hu et al. 1989); this pattern may thus be widespread in this species during winter. The behavior is unlikely to have been caused by artificial provisioning, as the monkeys left the provisioning site soon after the last provisioning at 1500 h. An alternative possibility is that it may be necessary for these monkeys to extend their foraging activities beyond sunset. In winter, they mainly consume lichen, which is evenly distributed but more difficult to consume than fruits (Li et al. 2003). The study band was previously documented to forage over an area of approximately 9.2 km2 in winter (Li et al. 2000). As sunset in winter is 1 or 2 h earlier than in summer (summer sunset is around 2015 h, authors’ ad libitum records) at the study site, the monkeys might not have enough daylight to acquire sufficient food, forcing them to continue foraging after sunset. However, because moonlight is reflected by snow on the ground and trees, the area is typically not very dark after sunset, which may facilitate nocturnal activities. Monkeys tended to retire later on moonlit evenings than on evenings with no moonlight or with cloud cover (Hu et al. 1989). Furthermore, the monkeys’ dense coats may reduce the costs of foraging in cold weather (Wada K, personal communication). We propose that the late retiring habit of R. roxellana may be a response to ecological conditions in winter, which both necessitate and facilitate the extension of foraging activities. Future investigations should compare activity patterns between winter, as studied here, and summer nights.
Social relations in night sleeping clusters
The composition of sleeping clusters varied between night and day, suggesting differences in affiliative patterns. JJ clusters were common in daytime, but rare at night. Adult MJ clusters were four times more frequent during the day than at night. With increasing independence, or a premigratory phase (Zhang et al. 2006a), juveniles spend increasing amounts of time away from their mothers during the day but return to them at night to sleep. A similar pattern was observed in gelada baboons (Theropithecus gelada), in which immature individuals formed senior groups during the day but descended the sleeping cliff to join their respective natal OMUs in the evening (Mori 1979). In this study, adult females almost always formed clusters for sleeping. This indicates a core role for females in maintaining social relations among members of OMUs (Zhang et al. 2008a). Our observations suggest some affiliative relations are common to both day and night, which is reflected by proximity between unit members, especially females with juveniles or other females. Others appear particular to nighttime, and include the frequent return of juveniles to their mothers or other related females, increased clustering between resident males and their unit females, and reduced MJ clustering.
The sleeping habits of R. roxellana appear to differ from those of other primate species with similar multilevel societies, such as hamadryas and gelada baboons (Kummer 1971; Mori 1979; Dunbar 1983; Kawai et al. 1983). Both hamadryas and gelada baboons sleep on cliffs at night, and OMUs tend to form sleeping aggregations if space is available (Swedell 2002; Grueter and Zinner 2004). Sleeping sites were recognized as limiting resources that influence ranging behavior, band size, and expression of the multilevel social system in these species (Altmann 1980). Hamilton and Watt (1970) considered the social organizations of these species as a refuging system, as the groups usually aggregate at fixed sleeping sites but fragment in the day for efficient foraging. However, the studied R. roxellana band did not show such regular aggregation and fragmentation. At night, OMU members usually slept together, with a distance of 5–50 m between neighboring OMUs. These OMUs traveled together during the day. Stable associations of OMUs were also observed in wild populations in Shennongjia Mountains (Ren et al. 2000). Composition and social organization of R. roxellana bands, therefore, appear less flexible than those of hamadryas baboons and gelada baboons. The variability in sleeping habits of these species indicates diversity in the social dynamics of primate multilevel social systems in response to their specific ecological conditions.
References
Altmann J (1974) Observational study of behavior: sampling methods. Behaviour 49:227–265
Altmann J (1980) Baboon mothers and infants. Harvard University Press, Cambridge
Anderson JR (1984) Ethology and ecology of sleep in monkeys and apes. Adv Stud Behav 14:165–229
Anderson JR (1998) Sleep, sleeping sites, and sleep-related activities: awakening to their significance. Am J Primatol 46:63–75
Anderson JR (2000) Sleep-related behavioural adaptations in free-ranging anthropoid primates. Sleep Med Rev 4:355–373
Anderson JR, McGrew WC (1984) Guinea baboons (Papio papio) at a sleeping site. Am J Primatol 6:1–18
Bernstein IS (1972) Daily activity cycles and weather influences on a pigtail monkey group. Folia Primatol 16:390–415
Booth AH (1956) The distribution of primates in the Gold Coast. J West Afr Sci Assoc 2:122–133
Chalmers NR (1968) Group composition, ecology and daily activities of free living mangabeys in Uganda. Folia Primatol 8:247–262
Chen FG, Min ZL, Gan QF, Luo SY, Xie WZ (1989) Resources and protection of the golden monkey (Rhinopithcus roxellana) in Qinling Mountains. In: Chen FG (ed) Progress in the studies of golden monkey. Northwest University Press, Xi’an, pp 279–283
Davidge C (1978) Activity patterns of chacma baboons (Papio ursinus) at Cape point. Afr Zool 13:143–155
Dunbar RIM (1983) Structure of gelada baboon reproductive units II. Social relationships between reproductive females. Anim Behav 31:556–564
Dunbar RIM (1992) A model of the gelada socio-ecological system. Primates 33:69–83
Gautier-Hion A (1973) Social and ecological features of talapoin monkey-comparison with sympatric Cercopithecines. In: Michael RP, Cook JH (eds) Comparative ecology and behavior of primates. Academic Press, New York, pp 147–170
Gittins SP (1982) Feeding and ranging in the agile gibbon. Folia Primatol 38:38–71
Grueter GC, Zinner D (2004) Nested societies. Convergent adaptations of baboons and snub-nosed monkeys? Primate Rep 70:1–98
Hamilton WJ III (1982) Baboon sleeping site preferences and relationships to primate grouping patterns. Am J Primatol 3:41–53
Hamilton EJ III, Watt KEF (1970) Refuging. Annu Rev Ecol Syst 1:263–286
Harrison MJS (1983) Pattern of range use by the green monkey, Cercopithecus sabaeus, at Mt Assirik, Senegal. Behav Ecol Sociobiol 12:85–94
Hu JC, Deng QX, Yu ZW, Zhou SD, Tian ZX (1989) Research on the ecology and biology of the giant panda, golden monkey and other rare animals. In: Chen FG (ed) Progress in the studies of golden monkey. Northwest University Press, Xi’an, pp 207–215
Jay P (1965) The common langur of north India. In: DeVore I (ed) Primate behavior: field studies of monkeys and apes. Holt, New York, pp 197–249
Kawai M, Dunbar R, Ohsawa H, Mori U (1983) Social organization of gelada baboons: social units and definitions. Primates 24:13–24
Koyama N (1973) Dominance, grooming, and clasped-sleeping relationships among bonnet monkeys in India. Primates 14:225–244
Kummer H (1971) Primate societies: group techniques of ecological adaptation. Aldine, Chicago
Kummer H, Banaja AA, Abo-Khatwa AN, Ghandour AM (1985) Differences in social behavior between Ethiopian and Arabian hamadryas baboons. Folia Primatol 45:1–8
Li BG, Chen C, Ji WH, Ren BP (2000) Seasonal home range changes of the Sichuan Snub-nosed monkey (Rhinopithecus roxellana) in the Qinling Mountains of China. Folia Primatol 71:375–386
Li BG, Zhang P, Watanabe K, Tan CL, Fukuda F, Wada K (2003) A dietary shift in Sichuan Snub-nosed monkeys. Acta Theriol Sin 23:258–260
Makwana SC (1978) Field ecology and behavior of the rhesus macaques (Macaca mulatta) I: group composition, home range, roosting sites, and foraging routes in the Asarori Forest. Primates 19:483–492
Meddis R (1979) The evolution of sleep. In: Hayes A (ed) Sleep functions and mechanisms in animals and man. Van Nostrand, New York
Mori U (1979) Social structure of gelada baboons. In: Kawai M (ed) Ecological and sociological studies of gelada baboons. Kodansha Press, Tokyo, pp 243–247
Mulavwa MN, Yangozene K, Yamba-Yamba M, Motema-Salo B, Mwanza NN, Furuichi T (2010) Nest groups of wild bonobos at Wamba: selection of vegetation and tree species and relationships between nest group size and party size. Am J Primatol 72:575–586
Neyman PF (1978) Aspects of the ecology and social organization of free-ranging cotton-top tamarins (Saguinus oedipus) and the conservation status of the species. In: Kleiman DG (ed) The biology and conservation of the Callitrichidae. Smithsonian Institution Press, Washington, DC, pp 39–71
Ogawa H, Idani G, Moore J, Pintea L, Hernandez-Aguilar A (2007) Sleeping parties and nest distribution of chimpanzees in the savanna woodland, Ugalla, Tanzania. Int J Primatol 28:1397–1412
Qi JF (1989) The feed and reproduction of the golden monkey. In: Chen FG (ed) Progress in the study of golden monkey. Northwest University Press, Xi’an, pp 287–292
Qi XG, Li BG, Tan CL, Gao YF (2004) Spatial structure in a Sichuan golden snub-nosed monkey Rhinopithecus roxellana group in Qinling Mountains while being in no-locomotion (in Chinese with English abstract). Acta Zool Sin 50:697–705
Qi XG, Li BP, Ji WH (2008) Reproductive parameters of wild female Rhinopithecus roxellana. Am J Primatol 70:311–319
Rahaman H, Parthasarathy MD (1969) Studies on the social behavior of bonnet monkeys. Primates 10:149–162
Reichard U (1998) Sleeping sites, sleeping places and pre-sleeping behavior of gibbons (Hylobates lar). Am J Primatol 46:35–62
Ren RM, Yan KH, Su YJ, Zhou Y, Li JJ (2000) The society of golden monkey (Rhinopithecus roxellana). Beijing University Press, Beijing, pp 148–168
Shi DC, Li GH, Hu TQ (1982) Preliminary study on the ecology of golden haired monkey. Zool Res 3:105–110
Sigg H, Stolba A (1981) Home range and daily march in a hamadryas baboon troop. Folia Primatol 36:40–75
Southwick CH, Beg MA, Siddiqi MR (1965) Rhesus monkeys in north India. In: DeVore I (ed) Primate behavior: field studies of monkeys and apes. Holt, New York, pp 111–159
Stolz LP, Saayman GS (1970) Ecology and behavior of baboons in the northern Transvaal. Ann Transv Mus 26:99–143
Swedell L (2002) Ranging behavior, group size and behavioral flexibility in Ethiopian hamadryas baboons (Papio hamadryas hamadryas). Folia Primatol 73:95–103
Takahashi H (1997) Huddling relationships in night sleeping groups among wild Japanese macaques in Kinkasan Island during winter. Primates 38:57–68
Vessey SH (1973) Night observation of free-ranging rhesus macaques. Am J Phys Anthropol 38:613–620
Wada K, Tokida E (1985) Sleeping site cluster of Japanese macaques in Shiga A1 troop: about inter-individual relationships. In: Primate Research Institute (ed) The study of spatial distribution of Japanese macaque troops in Shiga Heights. Kyoto University Press, Kyoto, pp 2–10
Xi WZ, Li BG, Zhao DP, Ji WH, Zhang P (2008) Benefits to female helpers in wild Rhinopithecus roxellana. Int J Primatol 29:593–600
Zhang P, Watanabe K (2007) Extra large rest clusters of Japanese monkeys (Macaca fuscata) in Shodo Island. Am J Primatol 69:1119–1130
Zhang SY, Ren BP, Li BG (1999) A juvenile Sichuan golden monkey (Rhinopithecus roxellana) predated by a goshawk (Accipiter gentilis) in the Qinling Mountains. Folia Primatol 70:175–176
Zhang P, Li BG, Wada K, Tan CL, Watanabe K (2003) Social structure of Sichuan snub-nosed monkeys (Rhinopithecus roxellana) in the Qinling Mountains of China. Acta Zool Sin 49:727–735
Zhang P, Watanabe K, Li BG (2006a) Forest strata utilization of Sichuan snub-nosed monkeys (Rhinopithecus roxellana) in their natural habitat. Acta Zool Sin 52:429–436
Zhang P, Watanabe K, Li BG, Tan CL (2006b) Social Organization of the Sichuan snub-nosed monkeys (Rhinopithecus roxellana) in the Qinling Mountains of China. Primates 47:374–382
Zhang P, Watanabe K, Li BG (2008a) Female social dynamics in a provisioned free-ranging band of the Sichuan snub-nosed monkey (Rhinopithecus roxellana) in the Qinling Mountains, China. Am J Primatol 70:1–10
Zhang P, Watanabe K, Li BG, Qi XG (2008b) Dominance relationships among one-male units in a provisioned free-ranging band of the Sichuan snub-nosed monkeys (Rhinopithecus roxellana) in the Qinling Mountains, China. Am J Primatol 70:634–641
Acknowledgments
The study was supported by the National Natural Science Foundation of China (31000175, 30970444 and 30570312) and Fund-In-Aid of JSPS (P09103) in Japan. The authors are grateful to Zhouzhi Nature Reserve for giving us permission to carry out this research. We thank Dr. Amy L. Schreier and Andrew J. Macintosh for improving the language in the article. We thank all members of the Anthropology Department in Sun Yat-sen University China and those of the Social Ecology Department in the Primate Research Institute, Kyoto University, Japan, who gave us helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zhang, P., Li, Bg., Watanabe, K. et al. Sleeping cluster patterns and retiring behaviors during winter in a free-ranging band of the Sichuan snub-nosed monkey. Primates 52, 221–228 (2011). https://doi.org/10.1007/s10329-011-0241-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-011-0241-y