Abstract
To advance our understanding of the effects of inoculation with ectomycorrhizal fungi (EMF) on seedling colonization in mine wastelands, we conducted a field experiment in a copper tailing. Six-month-old seedlings of Japanese red pine (Pinus densiflora) and oak (Quercus variabilis) separately inoculated with three EMF species (Pisolithus sp., Cenococcum geophilum, Laccaria laccata) were transplanted to the copper tailing. The survival rates of tree seedlings were monitored monthly, and growth (biomass and height), contents of nutrients and heavy metals (K, P, Ca, Mg, Cu, Zn), and mycorrhizal infection rates of seedlings were determined 6 months after planting. Oak seedlings exhibited higher survival rates than pine seedlings after 6 months of growth on the tailing. EMF inoculations of pine seedlings significantly enhanced their survival, growth, and nutrient uptake. In contrast, EMF inoculations of oak seedlings improved growth only in terms of biomass. Additionally, EMF inoculation caused pine seedlings to accumulate more Cu and Zn in roots compared to non-inoculated seedlings, whereas inoculation inhibited the accumulation of heavy metals in shoots. However, similar results were not observed in oak seedlings. Observations of roots indicated that the rates of mycorrhizal infection of both tree species had dramatically declined at harvest time. In conclusion, ectomycorrhizal symbioses can improve the survival and performance of pine seedlings in mine tailings. The present study provided direct evidence of the importance of EMF inoculation of seedlings to the reforestation of mine wastelands.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mine tailings produced by mining activities often act as sources of heavy metal pollution by wind dispersion or water erosion, and are detrimental to human health and ecological systems in surrounding regions (Li 2006). Phytoremediation using plants to remove or stabilize various pollutants in heavy metal-contaminated soils is an environmentally friendly and low-cost technology for restoring these heavy metal-polluted sites (Singh et al. 2003). Within the pool of plant species that can be used, trees exhibit high efficiency in stabilizing heavy metals in polluted soils due to their high biomass and large root systems, which trap metals and ameliorate soil conditions (Roy et al. 2007). However, mine tailing substrates are unsuitable for tree seedling colonization, not only because of the high concentration of heavy metals, but also because of the unfavorable soil conditions, i.e., poor nutrients, imbalanced pH levels, sandy composition, and low soil ventilation (Pulford and Watson 2003).
In natural forests, ectomycorrhizal fungi (EMF) perform mutually beneficial symbiosis with host trees. This symbiosis improves tree growth by enhancing nutrient and water uptake from the soil, and thus EMF play an essential role in the colonization, growth, and fitness of trees under harsh conditions, such as heavy metal-polluted soils (Arocena and Glowa 2000; Jentschke and Godbold 2000). Previous field studies have found that some trees (e.g., pine, oak, spruce) whose roots were colonized with an assemblage of EMF species could grow well in mine wastelands (Gebhardt et al. 2007; Huang et al. 2012, 2014). Greenhouse experiments have also confirmed that tree seedlings inoculated with specific EMF perform better in terms of biomass growth, nutrient uptake, and lower accumulations of heavy metals when exposed to high concentrations (Adriaensen et al. 2006; Krznaric et al. 2009). These studies suggest that EMF may support tree seedling survival and growth in metal-polluted sites, which holds great potential for the progress of phytoremediation. However, field experiments to date have provided only limited evidence of such benefits.
The present study examined whether ectomycorrhizal symbiosis would improve tree seedling colonization in mine tailings by transplanting EMF-inoculated and non-inoculated seedlings of Pinus densiflora and Quercus variabilis into a copper tailing. Seedlings were inoculated with three EMF species: Pisolithus sp., Cenococcum geophilum, and Laccaria laccata. After transplanting, seedling survival was monitored monthly. Seedling growth in terms of biomass and shoot height, contents of nutrients and heavy metals, and mycorrhizal inoculation rates in roots were determined 6 months after transplantation.
Materials and methods
Study site
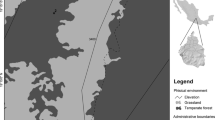
The transplant experiment was conducted at the Donggua copper mine tailing, Tongling, Anhui Province, China (N30°54′, E117°53′, Fig. 1a) in 2009. This region experiences a semi-tropical wet monsoon climate, with average annual temperature of 16.2–16.6 °C and average precipitation of 1390 mm. In the period after transplantation, 85.2 days and up to 1068.3 mm of rainfall occurred at the experimental field according to climate data. The Donggua tailing dam (top surface area of approximately 0.54 km2) was operated from 1966 to 1990, and stores about 13 million tons of processed residues. The tailing substrates were primarily composed of sand and silt, and lacked macronutrients and organic matter. The surface often experiences drought (Zhou et al. 2010). Over the 20 years of abandonment, most areas of the tailing surface were still completely bare, except for a few naturally established herbaceous plant species (Fig. 1a).
Soils were collected from the tailing at the time of transplantation. Soil analyses revealed that the tailing soils were slightly alkaline [pH 8.02 ± 0.33 (mean ± standard error)] and contained remarkably high levels of heavy metals [total metals: Cu, 1701.6 ± 486.1 mg kg−1; Zn, 505.6 ± 87.5 mg kg−1; Diethylene triamine pentaacetic acid (DTPA)-extractable metals: Cu, 92.5 ± 27.5 mg kg−1; Zn, 41.7 ± 14.7 mg kg−1], accompanied by low levels of nutrients (N, 141.4 ± 31.7 mg kg−1; P, 282.6 ± 44.6 mg kg−1; K, 12.8 ± 4.2 g kg−1). In particular, soil Cu concentrations were threefold higher than values provided by GB 15618-1995 grade III (Chinese soil environmental quality standards, Cu 500 mg kg−1).
Preparation of ectomycorrhizal seedlings
The cultivation of ectomycorrhizal seedlings was conducted in a greenhouse at the Asian Natural Environmental Science Center, University of Tokyo, Japan. Three EMF [Pisolithus sp. (Ps; Chen et al. 2015), C. geophilum (Cg; Nara 2006), and L. laccata (Ll; Nara 2006)] and two tree species (P. densiflora, Q. variabilis) were used. The three EMF are commonly observed in a wide range of habitats worldwide. The EMF-infected seedlings were prepared using the method reported by Nara (2006). Briefly, the seedlings germinated from seeds were infected by the EMF-infected seedlings (mother seedling) previously planted in the same pot. The seeds of P. densiflora and Q. variabilis were surface-sterilized in a 30 % H2O2 solution for 10 min, and then placed on an autoclaved (121 °C, 180 min) mixture of Shibanome soil (Tachikawa Heiwa Nouen Co., Ltd., Tokyo, Japan) and nursery soil (2:1 vol) in a plastic pot (60 × 25 × 15 cm), in which four mother seedlings, each inoculated with EMF, were also planted. At the same time, non-inoculated seedlings [CK] were prepared for each tree species. In total, eight combinations of seedling treatments were used in the present study. Seedlings were grown in the greenhouse with 16:8 h (day/night) illumination at 25/23 °C (day/night). After 6 months of growth, the mycorrhizal status of seedlings was determined under a stereomicroscope (S8 AP0, Leica Microsystems, Wetzlar, Germany). Seedlings with a rate of mycorrhizal infection greater than 90 % were chosen for transplantation. Before planting, height and fresh weight was determined for all seedlings. In order to monitor each seedling after transplantation, all seedlings were labeled using plastic tags.
Transplanting and experimental design
Transplanting was conducted at the Dongguashan copper mine tailing, at which ten plots (2 × 2 m) were randomly established. Plots were set apart at distances greater than 50 m (Fig. 1b). The three seedlings of each combination (each tree species inoculated or not inoculated with one of the EMF species) were planted randomly at each intersection point (hole size: 10 × 10 × 10 cm) on a 50 × 50-cm grid (Fig. 1c, d). Each combination was replicated three times. Totally, the number of seedlings of each combination planted was 90. To enhance the survival of seedlings, the soils in each hole were excavated and mixed with an equal volume of autoclaved nursery soil, and the mixture was then added into each hole. Before transplanting the seedlings, the surface soil in each plot was collected for the determination of soil characteristics. After planting, seedlings were watered weekly for the first month.
Harvest and analysis
Seedling surviving rates were observed monthly after transplantation into the field. After 6 months, all surviving seedlings were carefully collected. The numbers of surviving seedlings were as follows: P. densiflora, Ps-48, Cg-45, Ll-39, and CK-6; O. variabilis, Ps-75, Cg-60, Ll-75, and CK-57, respectively. Seedling height was measured, and ectomycorrhizal infection in seedling roots was quantified under a stereomicroscope. Because it was difficult to count root tips of the whole root system, ectomycorrhizal infection was determined in roots of 5 cm intercepted randomly; the total number of root tips in a seedling reached from 358 to 512. The roots and shoots of each seedling were then washed in tap water, rinsed with deionized water (DW), and oven-dried at 105 °C for 5 min and at 60 °C for 48 h. The dry weight of seedlings was measured using an ultra-microbalance (UMT2; Mettler-Toledo International, Greifensee, Switzerland), and seedling tissues were then milled (Hung Chuan Machinery Enterprise Co. Ltd., Tokyo, Japan). Soil pH values were determined using a HM-30 G pH meter (DKK-TOA Corporation, Tokyo, Japan) after mixing with DW (1:2 w/v). Electrical conductivity (EC) was measured with a B-173 compact conductivity meter (Horiba, Ltd., Tokyo, Japan) using soil suspensions (soil/DW = 1:5). After digestion by sulfuric acid and perchloric acid, total nitrogen of soils was determined by the indophenol blue method (Olsen and Sommers 1982) using U-2010 spectrophotometer equipped with an AS-1000 auto sampler (Hitachi Instruments, Inc., Tokyo, Japan). The total amounts of potassium (K), copper (Cu), lead (Pb), cadmium (Cd), and zinc (Zn) in soil and seedlings were determined using a Z6100 A polarized Zeeman atomic absorption spectrometer (Hitachi High-Technologies Corp., Tokyo, Japan) after wet digestion with a mixture of nitric (HNO3) and perchloric acids (HClO4) (4:1, v/v for soil; 7:1 for leaves). Concentrations of soil-available metals (Cu, Pb, Zn, Cd) were also analyzed by extracting metal ions from soil using a DTPA solution (5 mM DTPA, 10 mM CaCl2 and 100 mM triethanolamine at pH 7.3) (Lindsay and Norvell 1978). Soil samples were air-dried, ground, and passed through a 2-mm mesh screen.
The study was conducted by the Collaborated Lab. of Plant Molecular Ecology between College of Life Sciences of Nanjing Agricultural University and Asian Natural Environmental Science Center of University of Tokyo, Nanjing Agricultural University. All experimental materials imported into Japan had the permission of Yokohama Plant Protection Station and were autoclaved after use.
Statistical analysis
One-way ANOVA analyses followed by Tukey–Kramer test at a 5 % significance level were performed to compare seedling growth, seedling mycorrhizal infection rate, and soil and plant nutrient data among the different EMF treatments. All statistical analyses were conducted using SPSS version 11.5 software (SPSS Inc., Chicago, IL, USA).
Results
Seedling survival
In general, the survival rates of pine seedlings decreased over time after transplantation, but EMF inoculation significantly improved the survival of P. densiflora seedlings (Fig. 2a). One month after transplanting, many non-inoculated pine seedlings had died, and only 16.7 ± 6.7 % of seedlings survived to the end of the month. In contrast, 86.7 ± 13.7, 76.6 ± 16.4, and 83.3 ± 14.5 % of pine seedlings inoculated with Ps, Cg, and Ll survived, respectively, which were significantly higher survival rates than those of non-inoculated seedlings (P < 0.01). After 6 months, the survival rates of EMF-inoculated P. densiflora seedlings (Ps, 53.3 ± 17.2 %; Cg, 50.0 ± 17.6 %; Ll, 43.3 ± 16.1 %) were still significantly higher than those of non-inoculated seedlings (CK, 6.7 ± 14.1 %) (P < 0.01). No differences in survival rates among the three types of EMF-inoculated pine seedlings were observed 1, 2, or 6 months after transplanting. However, the survival rates of Ps-inoculated pine seedlings were significantly higher than those of Cg- and Ll-inoculated seedlings 3–5 months after transplanting.
Survival rates of Pinus densiflora and Quercus variabilis seedlings at each month after transplanting and mycorrhizal rates at the time of harvest. Ps, Cg, and Ll indicate that seedlings were inoculated with Pisolithus sp., Cenococcum geophilum, and Laccaria laccata, respectively; CK indicates no inoculation by EMF. The numbers of seedlings used for mycorrhizal rate statistics were as follows: P. densiflora, Ps-48, Cg-45, Ll-39, and CK-6; O. variabilis, Ps-75, Cg-60, Ll-75, and CK-57. Different small letters above columns indicate significant differences among EMF inoculation types within the same tree species (P < 0.05). Bars indicate the standard deviation
In contrast, EMF inoculation did not affect oak seedling survival after transplanting, and no differences in seedling survival rates were observed among the three types of EMF-inoculated seedlings during any month after transplanting. At harvest, survival rates of oak seedlings (Ps, 83.3 ± 17.6 %; Cg, 66.7 ± 31.4 %; Ll, 83.3 ± 23.6 %; CK, 63.3 ± 39.9 %) were higher than those of pine seedlings.
Dynamics of mycorrhizal infection rates in seedling roots
Mycorrhizal infection rates in the roots of both species of tree seedlings (initially >90 % inoculation rate by each EMF type) declined dramatically after planting (Fig. 2b). Six months after transplantation, the mycorrhizal infection rates of Ps-inoculated pine seedlings (15.9 ± 4.9 %) were significantly higher than those of Cg- and Ll-inoculated seedlings (Cg, 6.5 ± 2.4 %; Ll 5.4 ± 1.8 %) (P < 0.05), whereas Cg colonization in oak seedlings (11.5 ± 3.8 %) was significantly higher than that in Ps and Ll (Ps, 4.0 ± 1.7 %; Ll 3.3 ± 1.5 %) (P < 0.05). In addition, all mycorrhizal morphotypes found in seedling roots corresponded to the initial inoculation types, indicating that no infections by other EMF types occurred during the 6 months of growth in the field.
Seedling growth
Before transplanting, fresh weight measurements of pine seedlings were noted as follows: Pt, 0.54 ± 0.24 g; Cg, 0.48 ± 0.33 g; Ll, 0.42 ± 0.12 g; and CK, 0.35 ± 0.08 g. The fresh weight values for oak seedlings were: Pt, 7.41 ± 5.02 g; Cg, 4.83 ± 2.39 g; Ll, 5.90 ± 2.49 g; CK, and 5.24 ± 2.68 g. Higher biomass was observed in oak seedlings compared to pine seedlings. For the latter, the increase in biomass of inoculated seedlings was significantly higher than that of control seedlings at harvest (Fig. 3a). Among the three types of EMF seedling, Ps seedlings exhibited the highest biomass (Ps, 1.15 ± 0.74 g), followed by Cg (0.86 ± 0.68 g), Ll (0.34 ± 0.30 g), and CK (0.19 ± 0.21 g) (one-way ANOVA, P < 0.05). The increases in seedling height in Ps- and Cg-inoculated seedlings were significantly larger than those of control seedlings at harvest (Fig. 3b). A Tukey–Kramer test revealed no significant differences in seedling height increments between Ps- and Cg-inoculated seedlings (Ps, 3.16 ± 1.23 cm; Cg, 3.23 ± 1.67 cm), but both types were taller than Ll-inoculated seedlings (Ll, 0.34 ± 0.30 cm; CK, 0.93 ± 1.09 cm).
Biomass and height increment of Pinus densiflora and Quercus variabilis seedlings at harvest time. Ps, Cg, and Ll indicate that seedlings were inoculated by Pisolithus sp., Cenococcum geophilum, and Laccaria laccata, respectively; CK was not inoculated by EMF. The numbers of seedlings used for statistics were as follows: P. densiflora, Ps-48, Cg-45, Ll-39, and CK-6; O. variabilis, Ps-75, Cg-60, Ll-75, and CK-57. Different small letters above columns indicate significant differences among EMF inoculation types within the same tree species (P < 0.05). Bars indicate the standard deviation
For oak seedlings, there were no significant differences between inoculated and non-inoculated seedlings with respect to shoot height increments. There was no significant difference in the increment of seedling biomass among the different EMF-inoculated oak seedlings, but the biomass increment of Cg- and Ll-inoculated seedlings was higher than that of non-inoculated seedlings (Ps, 1.69 ± 1.13 g; Cg, 2.16 ± 1.05 g; Ll, 1.98 ± 1.18 g; CK, 1.53 ± 0.82 g; P < 0.05).
Seedling nutrient uptake
The effects of EMF inoculation on nutrient uptake by seedlings differed markedly between pine and oak. For oak seedlings, no significant difference in nutrient concentrations in roots and shoots was observed across treatments (P > 0.05; Table 1). For pine seedlings, the concentrations of K in roots and shoots of EMF-inoculated seedlings were significantly higher than those in non-inoculated seedlings (P < 0.05), although no significant differences were found in Mg concentrations (Table 1). In Ps-inoculated seedlings, the shoots and roots contained significantly higher P and Ca concentrations than those of non-inoculated seedlings. For Cg-inoculated seedlings, the P concentration in roots and Ca concentration in shoots were significantly higher than those of non-inoculated seedlings (Table 1).
The effects of EMF inoculation on K, P, and Ca uptake varied among the three EMF species (Table 1). For K, concentrations in roots of Ps-inoculated seedlings were higher than those in roots of Cg- and Ll-inoculated seedlings (Ps, 10.4 ± 1.1 g kg−1; Cg, 8.8 ± 0.9 g kg−1, Ll, 9.2 ± 1.0 g kg−1), but no significant differences were observed in shoots. For P, Ps-inoculated seedlings accumulated more P in roots and shoots than did Cg and Ll seedlings (shoots: Ps, 1.7 ± 0.2 g kg−1; Cg, 1.4 ± 0.2 g kg−1, Ll, 1.3 ± 0.3 g kg−1; roots: Ps, 2.1 ± 0.4 g kg−1; Cg, 1.7 ± 0.2 g kg−1, Ll, 1.4 ± 0.3 g kg−1). For Ca, concentrations in shoots did not differ between Ps- and Cg-inoculated seedlings (Ps, 3.7 ± 0.2 mg kg−1; Cg, 3.6 ± 0.1 mg kg−1), but values for both types were higher than those in Ll-inoculated seedlings (3.2 ± 0.2 mg kg−1) (P < 0.05).
Heavy metal accumulation in seedlings
For pine seedlings, Ps- and Cg-inoculated seedlings accumulated significantly more Cu (Ps, 140.5 ± 54.2 mg kg−1; Cg, 123.4 ± 47.3 mg kg−1) in roots than did non-inoculated seedlings (64.7 ± 34.6 mg kg−1) (Table 1). In contrast, Cu accumulation in shoots was generally lower than that in roots, and differences were observed across the four treatments. Among the three types of EMF inoculation, Cu concentrations in shoots of Ps seedlings (10.3 ± 5.6 mg kg−1) were lower than those of Cg, Ll, and non-inoculated seedlings (Cg, 18.7 ± 7.7 mg kg−1; Ll, 22.5 ± 11.4 mg kg−1; CK, 23.6 ± 12.9 mg kg−1). Notably, we found no significant differences in Cu or Zn concentrations between shoots and roots or among different treatments for oak seedlings (Table 1).
Discussion
To our knowledge, this study is the first to experimentally determine the contribution of EMF inoculation to the survival and growth of P. densiflora and Q. variabilis in a mine tailing in the field. The Cu mine tailing exhibited high concentrations of Cu, low N content, and high evaporation, thus providing realistic conditions for examining the effects of EMF inoculation on seedling survival and performance.
The mycorrhizal infection rates in EMF-inoculated seedlings of P. densiflora and Q. variabilis declined dramatically, from >90 to <20 %, after 6 months of growth in the tailing. Similar results have been observed in in vitro studies conducted under controlled conditions of various stresses, such as high levels of heavy metals and poor soil nutrients (Chappelka et al. 1991; Hartley et al. 1997). Other researchers have also found lower mycorrhizal colonization rates in the field. Huang et al. (2012) observed similar mycorrhizal infection rates (25.9 ± 21.0 %) in the roots of Masson pine (Pinus massoniana, 8 years old) growing in a bare Pb–Zn tailing. Poor EMF colonization (3–36 %) was also observed in the roots of naturally established Salix caprea on a former ore site with high concentrations of Pb and Cu (Hrynkiewicz et al. 2008). Clearly, harsh conditions such as heavy metal pollution, poor nutrient properties, and adverse soil texture of mines inhibit ectomycorrhizal colonization of trees during the early stages of reforestation in mine wastelands. Although dramatic reductions in mycorrhizal infection rates and poor colonization were seen in all EMF-inoculated treatments of P. densiflora and Q. variabilis seedlings 6 months after planting, differences in mycorrhizal infection rates were observed among ectomycorrhizal fungal types and between hosts. For P. densiflora, seedlings inoculated with Pisolithus sp. exhibited higher mycorrhizal infection rates than did seedlings inoculated with the other fungal types. In contrast, for Q. variabilis, seedling roots exhibited higher infection rates with C. geophilum. These results indicate that different fungi–host combinations perform distinctive adaptive functions under heavy metal stresses. Previous in vitro experiments have also reported differences among hosts in terms of the EMF contribution to metal tolerance (Van Tichelen and Vanstraelen 1999; Adriaensen et al. 2005). Together with these studies, our results allow us to conclude that the severe stress of tailing conditions will likely disrupt the ectomycorrhizal root tips, but different EMF inoculation types may elicit different responses.
For EMF-inoculated seedlings, all three EMF inoculation types enhanced the survival of P. densiflora seedlings in the present study. In contrast, no such effect was observed for Q. variabilis. This differential effect of EMF species on host species has also been reported in previous studies (Adriaensen et al. 2006; Krznaric et al. 2009), suggesting that conifer seedlings in mine tailings may depend more heavily on ectomycorrhizal symbiosis than do broadleaf trees. In fact, many species of broadleaf trees used for phytoremediation (e.g., willow, poplar, and oak) appear to possess high tolerance to heavy metals (Burken and Schnoor 1997; Prasad and Freitas 2000; Pulford and Watson 2003). Our results, for example, demonstrated that survival rates of Q. variabilis seedlings were significantly higher than those of P. densiflora seedlings, suggesting that Q. variabilis was likely more tolerant to the Cu mine tailing. Research has suggested that these differences in tolerance to Cu mine tailings may result primarily from size differences. Quercus variabilis seedlings are significantly larger than P. densiflora (see "Results/Seedling growth"). The Cu mine tailing in the study site was sandy and maintained dry conditions during the study period, apart from 1 month of watering. The higher biomass of the oak species in comparison to the pine, therefore, would offer it an advantage because of its thick storage-like main roots, especially at the beginning of transplanting.
Previous studies have shown that EMF enhancement of host heavy metal tolerance is largely realized through improved nutrient uptake, physical protection of hyphae and mantle, and inhibition of absorption of heavy metals from roots (Donnelly and Fletcher 1994; Smith and Read 2010). Various molecular tolerance mechanisms have been well documented in several EMF species (Bellion et al. 2006; Gonzalez-Guerrero et al. 2010; Blaudez and Chalot 2011). The increased survival of P. densiflora seedlings inoculated by EMF in this study may have been influenced by several factors. First, EMF may have significantly enhanced nutrient uptake via ectomycorrhizal symbiosis. Our results indicated that EMF inoculation increased the uptake of K, P, and Ca in P. densiflora seedlings (Table 1). Second, EMF infection may have inhibited the translocation of heavy metals from roots to shoots. Cu and Zn accumulations were higher in roots but lower in shoots of EMF-inoculated seedlings of P. densiflora compared to those of non-inoculated seedlings (Table 1).
Several studies have documented that EMF infection affects heavy metal uptake and translocation in plant tissues, suggesting that certain EMF species directly reduce metal toxicity through functions such as compartmentalization, chelation, and intra- and extra-cellular complexation mechanisms (Blaudez et al. 2000; Bellion et al. 2006, 2007). In the present study, Cu and Zn content in P. densiflora shoots were significantly reduced with EMF inoculation, and inoculation with Pisolithus sp. was more effective than the other two fungal inoculations. At the same time, the roots of P. densiflora seedlings inoculated with C. geophilum and Pisolithus sp. exhibited higher metal accumulation than those infected with L. laccata, perhaps due to the larger external mycelium or the higher metal binding and ameliorative capacity of the first two EMF species.
Conclusions
We found that EMF inoculation significantly improved pine seedling survival and performance in a Cu bare mine tailing; however, no effects were observed for oak seedling survival. The enhanced tolerance of P. densiflora seedlings to the Cu bare mine tailing was primarily due to increased nutrient uptake and the inhibition of the translocation of heavy metals from roots to shoots. Moreover, different combinations of EMF and host species varied in their level of seedling colonization, suggesting that the use of specific EMF-host combinations may improve the efficiency of phytoremediation of mine wastelands.
References
Adriaensen K, Vrålstad T, Noben J-P, Vangronsveld J, Colpaert JV (2005) Copper-adapted Suillus luteus, a symbiotic solution for pines colonizing Cu mine spoils. Appl Environ Microbiol 71:7279–7284
Adriaensen K, Vangronsveld J, Colpaert Jan V (2006) Zinc-tolerant Suillus bovinus improves growth of Zn-exposed Pinus sylvestris seedlings. Mycorrhiza 16:553–558
Arocena JM, Glowa KR (2000) Mineral weathering in ectomycorrhizosphere of subalpine fir (Abies lasiocarpa (Hook.) Nutt.) as revealed by soil solution composition. For Ecol Manag 133:61–70
Bellion M, Courbot M, Jacob C, Blaudez D, Chalot M (2006) Extracellular and cellular mechanisms sustaining metal tolerance in ectomycorrhizal fungi. FEMS Microbiol Lett 254:173–181
Bellion M, Courbot M, Jacob C, Guinet F, Blaudez D, Chalot M (2007) Metal induction of a Paxillus involutus metallothionein and its heterologous expression in Hebeloma cylindrosporum. New Phytol 174:151–158
Blaudez D, Chalot M (2011) Characterization of the ER-located zinc transporter ZnT1 and identification of a vesicular zinc storage compartment in Hebeloma cylindrosporum. Fungal Genet Biol 48:496–503
Blaudez D, Jacob C, Turnau K, Colpaert JV, Ahonen-jonnarth U, Finlay R, Botton B, Chalot M (2000) Differential responses of ectomycorrhizal fungi to heavy metals in vitro. Mycol Res 104:1366–1371
Burken JG, Schnoor JL (1997) Uptake and metabolism of atrazine by poplar trees. Environ Sci Technol 31:1399–1406
Chappelka A, Kush J, Runion GB, Meier S, Kelley WD (1991) Effects of soil-applied lead on seedling growth and ectomycorrhizal colonization of loblolly pine. Environ Pollut 72:307–316
Chen Y, Nara K, Wen Z, Shi L, Xia Y, Shen Z, Lian C (2015) Growth and photosynthetic responses of ectomycorrhizal pine seedlings exposed to elevated Cu in soils. Mycorrhiza. doi:10.1007/s00572-015-0629-4
Donnelly PK, Fletcher JS (1994) Potential use of mycorrhizal fungi as bioremediation agents. ACS Symp Ser 563:93–99
Gebhardt S, Neubert K, Wöllecke J, Münzenberger B, Hüttl RF (2007) Ectomycorrhiza communities of red oak (Quercus rubra L.) of different age in the Lusatian lignite mining district, East Germany. Mycorrhiza 17:279–290
Gonzalez-Guerrero M, Oger E, Benabdellah K, Azcn-Aguilar C, Lanfranco L, Ferrol N (2010) Characterization of a CuZn superoxide dismutase gene in the arbuscular mycorrhizal fungus Glomus intraradices. Curr Genet 56:265–274
Hartley J, Cairney JWG, Sanders F, Meharg AA (1997) Toxic interactions of metal ions (Cd2+, Pb2+, Zn2+ and Sb3−) on in vitro biomass production of ectomycorrhizal fungi. New Phytol 137:551–562
Hrynkiewicz K, Baum C, Niedojadlo J, Dahm H (2008) Promotion of mycorrhiza formation and growth of willows by the bacterial strain Sphingomonas sp. 23L on fly ash. Biol Fertil Soils 45:385–394
Huang J, Nara K, Lian C, Zong K, Peng K, Xue S, Shen G (2012) Ectomycorrhizal fungal communities associated with Masson pine (Pinus massoniana Lamb.) in Pb–Zn mine sites of central south China. Mycorrhiza 22:589–602
Huang J, Nara K, Zong K, Wang J, Xue S, Peng K, Shen G, Lian C (2014) Ectomycorrhizal fungal communities associated with Masson pine (Pinus massoniana) and white oak (Quercus fabri) in a manganese mining region in Hunan Province, China. Fungal Ecol 9:1–10
Jentschke G, Godbold DL (2000) Metal toxicity and ectomycorrhizas. Physiol Plant 109:107–116
Krznaric E, Verbruggen N, Wevers Jan HL, Carleer R, Vangronsveld J, Colpaert Jan V (2009) Cd-tolerant Suillus luteus: a fungal insurance for pines exposed to Cd. Environ Pollut 157:1581–1588
Li MS (2006) Ecological restoration of mineland with particular reference to the metalliferous mine wasteland in China: a review of research and practice. Sci Total Environ 357:38–53
Lindsay WL, Norvell WA (1978) Development of a DTPA soil test for zinc, iron, manganese, and copper. Soil Sci Soc Am J 42:421–428
Nara K (2006) Ectomycorrhizal networks and seedling establishment during early primary succession. New Phytol 169:169–178
Olsen SR, Sommers LE (1982) Methods of soil analysis. Part 2. Chemical and microbiological properties
Prasad MNV, Freitas H (2000) Removal of toxic metals from solution by leaf, stem and root phytomass of Quercus ilex L. (holly oak). Environ Pollut 110:277–283
Pulford I, Watson C (2003) Phytoremediation of heavy metal-contaminated land by trees: a review. Environ Int 29:529–540
Roy S, Khasa DP, Greer CW (2007) Combining alders, frankiae, and mycorrhizae for the revegetation and remediation of contaminated ecosystems. Botany 85:237–251
Singh OV, Labana S, Pandey G, Budhiraja R, Jain RK (2003) Phytoremediation: an overview of metallic ion decontamination from soil. Appl Microbiol Biotechnol 61:405–412
Smith SE, Read DJ (2010) Mycorrhizal symbiosis. Academic Press, London
Van Tichelen KK, Vanstraelen T (1999) Nutrient uptake by intact mycorrhizal Pinus sylvestris seedlings: a diagnostic tool to detect copper toxicity. Tree Physiol 19:189–196
Zhou Y, Yue S, Zhou T (2010) Migration of heavy metals in Yangshanchong tailings impoundment in Tongling, Anhui Province. Res Environ Sci 23:497–503
Acknowledgments
This study received grants-in-aid from the Japan Society for the Promotion of Sciences (20380087).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zong, K., Huang, J., Nara, K. et al. Inoculation of ectomycorrhizal fungi contributes to the survival of tree seedlings in a copper mine tailing. J For Res 20, 493–500 (2015). https://doi.org/10.1007/s10310-015-0506-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10310-015-0506-1