Abstract
The ability of xylanolytic enzymes produced by Aspergillus fumigatus RP04 and Aspergillus niveus RP05 to promote the biobleaching of cellulose pulp was investigated. Both fungi grew for 4–5 days in liquid medium at 40°C, under static conditions. Xylanase production was tested using different carbon sources, including some types of xylans. A. fumigatus produced high levels of xylanase on agricultural residues (corncob or wheat bran), whereas A. niveus produced more xylanase on birchwood xylan. The optimum temperature of the xylanases from A. fumigatus and A. niveus was around 60–70°C. The enzymes were stable for 30 min at 60°C, maintaining 95–98% of the initial activity. After 1 h at this temperature, the xylanase from A. niveus still retained 85% of initial activity, while the xylanase from A. fumigatus was only 40% active. The pH optimum of the xylanases was acidic (4.5–5.5). The pH stability for the xylanase from A. fumigatus was higher at pH 6.0–8.0, while the enzyme from A. niveus was more stable at pH 4.5–6.5. Crude enzymatic extracts were used to clarify cellulose pulp and the best result was obtained with the A. niveus preparation, showing kappa efficiency around 39.6% as compared to only 11.7% for that of A. fumigatus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In the last decades, an increasing number of studies aimed to develop environmentally clean and non-toxic methods for industrial processes. For instance, an enzymatic step in the process of cellulose pulp bleaching would contribute to reduce the use of chlorine-containing reagents. According to Valcheva [1], the utilization of enzymes, particularly xylanases, results in an easier bleaching in subsequent stages and a better pulp brightness. Endo-1,4-β-xylanase (1,4-β-D-xylan xylanohydrolase; EC 3.2.1.8) catalyzes the hydrolysis of glycosidic bonds in the xylan backbone, reducing the degree of polymerization of the substrate [2]. This improves the access of bleaching reagents into the cellulose fibers facilitating the elimination of lignin in subsequent alkaline extraction [3].
The industrial application of xylanases takes place mainly in Scandinavia, North America and China [1, 3]. In the last decades, an increased number of studies were devoted to the biobleaching of cellulose pulp [1–9]. In order to induce xylanase synthesis from microbial sources, agricultural residues, such as rye flakes, wheat bran, oat flakes, corn flakes, crushed corncob, rice straw, sugar cane bagasse and others can be used [4, 8]. The use of agricultural residues as alternative carbon sources reduces the production costs and the price of the final product. Among the filamentous fungi employed to produce xylanase, the Aspergillus genus is one of the most explored. For example, production of xylanase and assays of biobleaching of cellulose pulp have been reported for Aspergillus caespitosus [8], A. nidulans and A. awamori [4], A. niger An76 and A. aculeatus [2].
The aim of the present study was to describe xylanases from two fungi isolated from Brazilian soil, which were identified as Aspergillus fumigatus and Aspergillus niveus, and to test the adequacy of these enzymes for cellulose pulp biobleaching. These fungi produced high levels of xylanases [10] with special characteristics, such as high stability and high optimum temperature, in comparison to others reported in the literature [11–13].
Materials and methods
Organisms and culture conditions
The Aspergillus strains were isolated from soil and decomposing leaves from a reforestation area of the Campus of São Paulo University at Ribeirão Preto, SP, Brazil. These strains were identified as A. fumigatus RP04 and A. niveus RP05, and are deposited at the Pernambuco Federal University, PE, Brazil. Stock cultures of both strains were propagated at 35°C on slants of solid oatmeal medium [14]. Both microorganisms were grown in different liquid media, such as SR [15], CP [16], Khanna [17], Czapeck [18], Vogel [19] and M-5 [20], inoculated with 4 × 105 spores/ml (final concentration) in 25 ml of medium. Most cultures were supplemented with 1% birchwood xylan, but other carbon sources were also tested. Cultivation was either static or agitated at 100 rpm; the incubation temperatures and times varied according with the experiment.
Extraction of xylanases and protein determination
Mycelia were harvested by filtration, rinsed with distilled water, blotted on filter paper and stored at −15°C until use. The mycelial mass was homogenized by grinding in a mortar with glass beads at 4°C. After addition of 10 ml of 100 mM McIlvaine buffer (citrate-phosphate buffer) [21] pH 5.0–5.5 for A. fumigatus or 4.0–4.5 for A. niveus, cell disruption was continued and the slurry was centrifuged at 12,100g, 15 min. The supernatant was used to estimate growth as total protein, according to Lowry et al. [22], using bovine serum albumin as standard.
Enzymatic assays
Xylanase activity was determined in the crude filtrate by measuring the reducing groups released from birchwood xylan [23]. The reaction mixture consisted of 200 μl of 1% (w/v) xylan in McIlvaine buffer at the ideal pH for each enzyme, and 200 μl of enzymatic extract appropriately diluted. The reaction mixture was incubated at 70°C for the extract of A. fumigatus and 65°C for that of A. niveus. One unit was defined as the amount of enzyme that releases 1 μmol of reducing sugar per minute determined according to Miller [23]. Total activity (total U) was defined as units/ml multiplied by the total sample volume. Identical conditions of assay were employed for cellulase, amylase and pectinase determination, using as substrate 1% (w/v) carboxymethyl-cellulose, avicel or filter paper for cellulase, polygalacturonic acid for polygalacturonase and starch for amylase activities. The assay temperature was 60°C for these other enzymes. One unit was defined as the amount of enzyme that releases 1 μmol of reducing sugar per minute, using monogalacturonic acid as standard for polygalacturonase, or glucose for cellulase and amylase.
Effects of temperature and pH
The effect of temperature on xylanase activity was analyzed using crude filtrate from A. fumigatus and A. niveus cultures. The assays were performed in McIlvaine buffer at the ideal pH for each enzyme, incubated at 25–80°C, with intervals of 5°C. The effect of pH was assayed using the same buffer in the pH range 2.5–8.0, at the ideal temperature for each enzyme. Thermal inactivation was analyzed with enzymes incubated at 70°C for 1 h and the assays were performed at the optima of temperature and pH for each enzyme. At different time intervals, aliquots were withdrawn and residual activities were measured as described above. The pH stability was analyzed incubating the enzymes at 25°C with McIlvaine buffer in different pH values (range 2.5–8.0) for 1 h, and after that the assays were carried out at the optima of temperature and pH for each enzyme.
Biobleaching
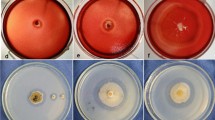
The amount of enzyme used for this treatment was 10 U/g of dried cellulose pulp extracted from Eucalyptus grandis. All calculations and procedures were carried out according to TAPPI, Atlanta GA [24] methodology. The volume of enzyme or distilled water added corresponded to 10% (dry weight) of the pulp mixture. The samples were incubated in polyethylene bags at the adequate temperature for 1 h, after that the cellulose pulp was filtered on a Büchner funnel, rinsed with 200 ml of distilled water and used for determination of kappa, viscosity and brightness parameters.
Reproducibility of results
All results are the mean of at least three independent experiments.
Results and discussion
Optimization of xylanase production
In order to find the best culture conditions, both Aspergilli were grown on six different liquid media. The best enzymatic yields were obtained in Vogel or Czapeck media for A. fumigatus RP04 and A. niveus RP05, respectively, (Table 1).
The time-course of growth and xylanase production for A. fumigatus and A. niveus was followed in Vogel and Czapeck media, respectively, supplemented with 1% birchwood xylan, for 196 h. A. fumigatus grew along all the experimental time (Fig. 1a), reaching the maximum of xylanase production at 96 h (Fig. 1c). By the end of the experiment, a decrease in xylanase activity was observed, probably due to inhibition of the enzyme by end products. For A. niveus, the peak of growth was observed at 72 h (Fig. 1a), but the maximal enzyme production occurred at 120 h, suggesting partial autolysis of the mycelium at the end of cultivation. Comparing xylanase production (total units) of both fungi (Fig. 1a, c) it can be seen that A. fumigatus produced 2.1-fold more xylanase than A. niveus, but in contrast, due to lesser growth, the specific activity (U/mg protein) of A. niveus xylanase was 25.5-fold higher than that of A. fumigatus. The time required for production of xylanase for A. fumigatus was 96 h and it is in agreement with that reported for A. awamori and A. oryzae [4]. A. tamarii required 120 h, similar to A. niveus RP05 [4].
Time-course and culture temperature from Aspergillus fumigatus and Aspergillus niveus for xylanase production. Total protein (a, b) and total activity (c, d) during different cultivation periods at 40°C (a, c) and on different temperatures during 96 h for A. fumigatus or 120 h for A. niveus (b, d). Microorganisms were cultivated in Czapeck (A. niveus) or Vogel (A. fumigatus) media supplemented with 1% birchwood xylan. Symbols: Filled circle with dash, Aspergillus fumigatus; Empty circle with dash, Aspergillus niveus
An interesting effect of temperature was observed (Fig. 1 b, d). Despite of the maximal xylanase levels being attained with incubation at 40°C (Fig. 1d), the optimum temperature for growth was distinct, being 40°C for A. fumigatus and 30°C for A. niveus (Fig. 1b). This result suggested a thermotolerant character for A. fumigatus, as previously reported for A. phoenicis [2, 6] and A. caespitosus [8]. For other Aspergillus species, such as A. awamori, A. niger, A. nidulans, A. oryzae and A. tamari, the best xylanase production occurs when the fungi are grown at lower temperatures [4].
Still aiming to improve xylanase production, cultures from both fungi were carried out under the optimized conditions, with agitation (100 rpm), or under static conditions. For both strains, the best growth was obtained with agitated cultures (Fig. 2a), but in contrast, static conditions (Fig. 2b) improved xylanase production and the enzyme levels were 2.3-fold higher for A. fumigatus and 1.46-fold for A. niveus, as compared to agitated cultures.
Influence of aeration in the cultivation of Aspergillus fumigatus and Aspergillus niveus for xylanase production. Total protein (a) and total activity (b) under agitation (100 rpm) or on static conditions. Microorganisms were cultivated in Czapeck (A. niveus) or Vogel (A. fumigatus) media supplemented with 1% birchwood xylan. White column Aspergillus fumigatus, gray column Aspergillus niveus
Aspergillus fumigatus produced approximately 3–4-fold more xylanase than A. niveus (Fig. 1c). Comparing with the literature, A. fumigatus produced 16–55% more xylanase than other fungi, such as A. caespitosus [8], A. nidulans and A. awamori [2, 4], while A. niveus produced 25 and 39% more xylanase than A. awamori and A. nidulans, respectively [2, 4], and 64% more xylanase in comparison with other strains of A. niveus used for prebleaching tests [13]. It is important to emphasize that the best enzymatic production under static conditions may lower the production cost.
Effect of alternative carbon sources on xylanase production
Aspergillus fumigatus RP04 and A. niveus RP05 were cultured in the optimized media, supplemented with different carbon sources (Table 2). A. fumigatus produced 5.0–6.0% more xylanase on powdered corncob, wheat bran and crushed corncob as compared to birchwood xylan. Rice straw and rye flakes were as favorable to production of xylanase as was birchwood xylan, and could be good alternative carbon sources. Xylanase from A. niveus was produced preferentially on wheat bran and powdered corncob, but birchwood xylan, xylose, E. grandis xylan and oat spelt xylan also were good substrates for the production of this enzyme. The possibility of using agricultural residues to produce enzymes may lower the production costs resulting in a cheaper product [25]. But it is known that agro-industrial residues are complex and when they are used as carbon source may induce the fungi to produce other enzymes in the crude extract. Because of that, it was decided to use xylan as the only carbon source to continue this study. In order to assure that cellulase activities were absent, we assayed the activity of the crude extract with three different substrates (carboxymethyl-cellulose, avicel or filter paper) as described in Sect. “Materials and methods”, and no activity was detected. Other enzyme activities, such as amylase and pectinase were found in diminished levels, suggesting constitutive activities for both fungi.
Characterization of xylanase activity in the crude extract
It is important for practical applications that industrial enzymes display some adequate properties, such as resistance to temperature and pH. The temperature optimum for A. fumigatus and A. niveus was 70 and 60–65°C, respectively (Fig. 3a). The temperature optimum observed for A. fumigatus was the same as that of the thermophilic fungus Thermomyces lanuginosus [26], but it was different of those reported for most Aspergilli [2, 6, 8], including other strains of A. fumigatus [12]. A. niveus exhibited an optimum temperature similar to those of A. fischeri, A. kawachii, A. oryzae, A. sojae and A. sydowii [2, 5].
Effect of temperature (a) and pH (b) in the enzyme reaction; thermostability (c) and pH stability (d) of the extracellular xylanases produced by A. fumigatus and A. niveus. The microorganisms were grown in Czapeck (A. niveus) or Vogel (A. fumigatus) supplemented with 1% birchwood xylan and the reactions were carried out as: a Xylanases activity were determined using McIlvine buffer, pH 5.0, at temperatures range from 25 to 80°C; b xylanases pH were determined at 65°C for A. niveus or 70°C for A. fumigatus using McIlvine buffer on different pHs (2.5–8.0); c thermal stabilities were carried out incubating both enzymes at 70°C and after the residual activities were determined at 65°C, pH 4.5–5.0 for A. niveus or at 70°C pH 5.0–5.5 for A. fumigatus; d pH stability were determined incubating both enzymes at different pH (2.5–8.0) at 25°C, during 1 h and after the residual activities were determined at 65°C, pH 4.5–5.0 for A. niveus or at 70°C pH 5.0–5.5 for A. fumigatus. Symbols: Filled circle with dash, Aspergillus fumigatus; Empty circle with dash Aspergillus niveus
For A. fumigatus the pH optimum was 5.0–5.5 and for A. niveus, 4.5–5.0 (Fig. 3b). In comparison with other fungi, these values of pH optimum are close to those of A. niger, A. fischeri, A. sojae and A. nidulans [2, 5], and to one of the xylanases (xyl II) produced by A. caespitosus [8]. Others fungal xylanases are alkalophilic, as those from Thermomyces lanuginosus [26], A. nidulans KK-99, A. terreus [2], and one of the xylanases (xyl I) from A. caespitosus [8].
Even though A. fumigatus produced approximately 3–4-fold more xylanase than A. niveus, the thermostability of both enzymes was similar at 60°C for 30 min. After this period, the residual activity of A. fumigatus xylanase diminished considerably and, after 120 min the xylanase from A. fumigatus retained only 10% of activity, although the xylanase from A. niveus still maintained 30% of the initial activity (Fig. 3c). Although the thermostability of the xylanase from A. niveus was higher, the temperature optimum of the xylanase from A. fumigatus was 70°C, while the xylanase from A. niveus corresponded to 60–65°C (Fig. 3a), suggesting that the substrate xylan may protect the enzyme.
The stability of the xylanases at different pH was also tested (Fig. 3d) and the results were different for both fungi. A. fumigatus presented a xylanase more stable in pH from 6.0 to 8.0, while the xylanase from A. niveus was more stable from 4.5 to 6.0. A. oryzae [2], A. fischeri [2, 4] and Termomyces lanuginosus [26] also presented a considerable stability on ranges of pH from 5.0 up to 8.0.
Assays of cellulose biobleaching using xylanase
The xylanases produced by A. fumigatus and A. niveus were tested on cellulose pulp biobleaching. The amount of enzyme was 10 U/g of dry pulp, for 1 h, at the temperature optimum of each enzyme. To compare the efficiency of the crude extracts from both fungi in biobleaching, the kappa number was determined. Kappa number is an indication of the lignin content or bleach ability of wood pulp. It estimates the amount of chemicals required to obtain a pulp with a given degree of brightness. Thus, it is expected to lower the kappa number after the enzyme treatment. The best result was obtained with the crude extract from A. niveus (Table 3), that decreased 4.6 points the kappa number in comparison with the control. When the cellulose pulp was treated with xylanases from A. fumigatus, a decrease of 0.9 points was observed, corresponding to a kappa efficiency of 39.6% for A. niveus and 11.7% for A. fumigatus. Using xylanase from A. niveus, the brightness improved 3.4 points, against 2 points using xylanase from A. fumigatus. The viscosity decreased 9.2% when xylanase from A. fumigatus was used, but the xylanase from A. niveus promoted no decrease of viscosity, confirming that this crude filtrate was free of cellulase. The presence of cellulase in the xylanolytic extract may affect the cellulose pulp properties, and is a constant preoccupation of the users [4, 7, 26]. Comparing these results to those obtained with other microorganisms such as Termomyces lanuginosus [26] and A. caespitosus (xyl I and II) [8], we observed that the kappa number was reduced only 1.0, 0.2 (xyl I) or 1.5 (xyl II) and 1.1 points, respectively, comparing to their control after at least 1 h of treatment using a quantity of xylanase equivalent to 10 U/g of dry pulp. A reduction of 3.3 points in kappa number (similar to our A. niveus strain) was observed when xylanase from Arthrobacter sp. MTCC 5214 was used; but to reach this performance in the treatment, it was necessary to double the amount of enzyme and the time of treatment (20 U/g of dry pulp/2 h) and, unfortunately, using Arthrobacter sp. MTCC 5214 xylanase a decrease of 7.5% in the cellulose viscosity was observed [27].
Aspergillus niveus provided a better result than A. fumigatus, but the performance of the xylanase obtained from the second strain was also interesting, and should receive more attention. Even when A. fumigatus is commonly described as a pathogenic fungus [28], it is still used to produce enzymes such as xylanase [11], applied in industry [12]. Other possibility is to carry on studies about this interesting xylanase intending to insert and express the gene in non-pathogenic yeasts, as it was done by Liu et al. [29].
Conclusions
In this work, A. niveus and A. fumigatus were described as good xylanase producers, but A. niveus xylanase provided the most promising characteristics to be industrially applied in the cellulose pulp biobleaching, and should receive more attention. This fungus should be studied more in order to better understand A. niveus xylanase synthesis system, which may enlarge the number of enzymes available in the market to be used in the paper industries.
References
Valcheva E, Valchev I, Yotova L (2001) Kinetics of enzyme action Cartazyme NS-10 prior to bleaching of kraft pulp. Biochem Eng J 7:223–226. doi:10.1016/S1369-703X(00)00127-3
Polizeli MLTM, Rizzatti ACS, Monti R, Terenzi HF, Jorge JA, Amorim DS (2005) Xylanases from fungi: properties and industrial applications. Appl Microbiol Biotechnol 67:577–591. doi:10.1007/s00253-005-1904-7
Zhao J, Li X, Qu Y, Gao P (2002) Xylanase pretreatment leads to enhanced soda pulping of wheat straw. Enzyme Microb Technol 30:734–740. doi:10.1016/S0141-0229(02)00050-9
Techapun C, Poosaran N, Watanabe M, Sasaki K (2003) Thermostable and alkaline-tolerant microbial cellulase-free xylanases produced from agricultural wastes and properties required for use in pulp bleaching bioprocess: a review. Process Biochem 38:1327–1340. doi:10.1016/S0032-9592(02)00331-X
Beg QK, Kapoor M, Mahajan L, Hoondal GS (2001) Microbial xylanases and their industrial applications: a review. Appl Microbiol Biotechnol 56:326–338. doi:10.1007/s002530100704
Rizzatti ACS, Sandrim VC, Jorge JA, Terenzi HF, Polizeli MLTM (2004) Influence of temperature on the properties of xylanolytic enzymes of the thermotolerant fungus Aspergillus phoenicis. J Ind Microbiol Biotechnol 31:88–93. doi:10.1007/s10295-004-0120-2
Salles BC, Medeiros RG, Bao SN, Silva FG Jr, Filho EXF (2005) Effect of celulase-free xylanases from Acrophialophora nainiana and Humicola grisea var. thermoidea on eucalyptus kraft pulp. Process Biochem 40:343–349. doi:10.1016/j.procbio.2004.01.008
Sandrim VC, Rizzatti ACS, Terenzi HF, Jorge JA, Milagres AMF, Polizeli MLTM (2005) Purification and biochemical characterization of two xylanases produced by Aspergillus caespitosus and their potential for kraft pulp bleaching. Process Biochem 40:1823–1828. doi:10.1016/j.procbio.2004.06.061
Lu W, Li D, Wu Y (2003) Influence of water activity and temperature on xylanase biosynthesis in pilot-scale solid-state fermentation by Aspergillus sulphureus. Enzyme Microb Technol 32:305–311. doi:10.1016/S0141-0229(02)00292-2
Guimarães LHS, Peixoto-Nogueira SC, Michelin M, Rizzatti ACS, Sandrim VC, Zanoelo FF et al (2006) Screening of filamentous fungi for production of enzymes of biotechnological interest. Braz J Microbiol 37:474–480
Thiagarajan S, Jeya M, Gunasekaran P (2005) Improvement of xylanase production in solid-state fermentation by alkali-tolerant Aspergillus fumigatus MKU1 using a fractional factorial design. Indian J Exp Biol 43(10):887–891
Savitha S, Sadhasivam S, Swaminathan K (2007) Application of Aspergillus fumigatus xylanase for quality improvement of waste paper pulp. Bull Environ Contam Toxicol 78:217–221. doi:10.1007/s00128-007-9132-8
Angayarknni J, Palaniswamy M, Pradeep BV, Swaminathan K (2006) Biochemical substitution of fungal xylanases for prebleaching of hardwood kraft pulp. Afr J Biotechnol 5(10):921–929
Emerson R (1941) An experimental study of the life cycles and taxonomy of Allomyces. Lloydia 4:77–144
Rizzatti ACS, Jorge JA, Terenzi HF, Rechia CGV, Polizeli MLTM (2001) Purification and properties of a thermostable extracellular β-d-xylosidase produced by a thermotolerant Aspergillus phoenicis. J Ind Microbiol Biotechnol 26:156–160. doi:10.1038/sj.jim.7000107
Peixoto SC, Jorge JA, Terenzi HF, Polizeli MLTM (2003) Rhizopus microsporus var. rhizopodiformis: a thermotolerant fungus with potential for production of thermostable amylases. Int Microbiol 6:269–273. doi:10.1007/s10123-003-0140-1
Khanna P, Sundari SS, Kumar NJ (1995) Production, isolation and partial purification of xylanase from Aspergillus sp. World J Microbiol Biotechnol 11:242–243. doi:10.1007/BF00704661
Wiseman A (1975) Handbook of enzyme biotechnology. Wiley & Sons, New York, p 148
Vogel HF (1964) Distribution of lysine pathways among fungi: evolutionary implications. Am Nat 98:435–446. doi:10.1086/282338
Peralta RM, Terenzi HF, Jorge JA (1990) α-d-glycosidase activities of Humicola grisea: biochemical and kinetic characterization of a multifunctional enzyme. Biochim Biophys Acta 1033:243–249
McIlvaine TC (1921) A buffer solution for colorimetric comparison. J Biol Chem 49:183–186
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:267–275
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–429. doi:10.1021/ac60147a030
Test methods TAPPI technical association of the pulp and paper industry. TAPPI Press, Atlanta, 1996
Aachary AA, Prapulla SG (2008) Corncob-induced endo-1, 4-beta-d-xylanase of Aspergillus oryzae MTCC 5154: production and characterization of xylobiose from glucuronoxylan. J Agric Food Chem 56(11):3981–3988. doi:10.1021/jf073430i
Li XT, Jiang ZQ, Li LT, Yang SQ, Feng WY, Fan JY et al (2005) Characterization of cellulase-free, neutral xylanase from Thermomyces lanuginosus CBS 288.54 and its biobleaching effect on wheat straw pulp. Bioresour Technol 96:1370–1379. doi:10.1016/j.biortech.2004.11.006
Khandeparkar R, Bhosle NB (2007) Application of thermoalkalophilic xylanase from Arthrobacter sp. MTCC5214 in biobleaching of kraft pulp. Bioresour Technol 98(4):897–903. doi:10.1016/j.biortech.2006.02.037
Taubitz A, Bauer B, Heesemann J, Ebel F (2007) Role of respiration in the germination process of the pathogenic mold Aspergillus fumigatus. Curr Microbiol 54(5):354–360. doi:10.1007/s00284-006-0413-y
Liu MQ, Weng XY, Sun JY (2006) Expression of recombinant Aspergillus niger xylanase A in Pichia pastoris and its action on xylan. Protein Expr Purif 48(2):292–299
Acknowledgments
This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). J. A. J., H.·F. T. and M. L. T. M. P. are Research Fellows of CNPq. S.·C. P.·N. was a recipient of a CNPq fellowship and this study is part of her Doctoral Thesis. M. M. was a recipient of a FAPESP fellowship. The authors thank Ricardo F. Alarcon and Mauricio de Oliveira for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Carvalho Peixoto-Nogueira, S., Michelin, M., Betini, J.H.A. et al. Production of xylanase by Aspergilli using alternative carbon sources: application of the crude extract on cellulose pulp biobleaching. J Ind Microbiol Biotechnol 36, 149–155 (2009). https://doi.org/10.1007/s10295-008-0482-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-008-0482-y