Abstract
We investigated the influence of light quality on the vulnerability of pepper plants to water deficit. For this purpose plants were cultivated either under compact fluorescence lamps (CFL) or light-emitting diodes (LED) providing similar photon fluence rates (95 µmol m−2 s−1) but distinct light quality. CFL emit a wide-band spectrum with dominant peaks in the green and red spectral region, whereas LEDs offer narrow band spectra with dominant peaks at blue (445 nm) and red (665 nm) regions. After one-week acclimation to light conditions plants were exposed to water deficit by withholding irrigation; this period was followed by a one-week regeneration period and a second water deficit cycle. In general, plants grown under CFL suffered more from water deficit than plants grown under LED modules, as indicated by the impairment of the photosynthetic efficiency of PSII, resulting in less biomass accumulation compared to respective control plants. As affected by water shortage, plants grown under CFL had a stronger decrease in the electron transport rate (ETR) and more pronounced increase in heat dissipation (NPQ). The higher amount of blue light suppressed plant growth and biomass formation, and consequently reduced the water demand of plants grown under LEDs. Moreover, pepper plants exposed to high blue light underwent adjustments at chloroplast level (e.g., higher Chl a/Chl b ratio), increasing the photosynthetic performance under the LED spectrum. Differently than expected, stomatal conductance was comparable for water-deficit and control plants in both light conditions during the stress and recovery phases, indicating only minor adjustments at the stomatal level. Our results highlight the potential of the target-use of light quality to induce structural and functional acclimations improving plant performance under stress situations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Light quality strongly influences biochemical and physiological processes, impacting morphology, growth and development of plants. In the last decades, a significant number of publications provided precise information on the effect of light quality on plant growth, morphogenesis and photosynthetic responses (Abidi et al. 2012; Brown et al. 1995; Hogewoning et al. 2010a, b; Murakami et al. 2014; Muraoka et al. 2002; Schuerger et al. 1997; Terfa et al. 2013). In controlled (climate chamber) and semi-controlled (greenhouse) environments, compact fluorescence lamps (CFL) and high pressure sodium lamps are commonly used to provide photosynthetic active radiation (PAR) to the plants. The spectrum of these artificial lighting systems is often characterized by comparatively wide-band peaks containing small amounts of blue and high amounts of green and red light. Nowadays, light-emitting diodes (LED) providing narrow peaks (some nanometers) with high reproducibility and spectral resolution enable to design optimal species-specific light supply and precise investigation of spectral-dependent plant responses (Massa et al. 2008). Currently, most LED systems used for horticultural production in greenhouses (e.g., interlighting modules) provide mainly red and blue light. Red and blue light, as compared to green light, are the most efficiently absorbed wavelengths by photosynthetic pigments, and provide most energy for photosynthesis (Massa et al. 2008). Nevertheless, under strong white light conditions, green light might drive leaf photosynthesis even more efficiently than red light (Terashima et al. 2009). Differently, blue light in excess might initiate a chloroplast avoidance response as well as a decrease in mesophyll conductance resulting in a decrease of the photosynthetic efficiency (Loreto et al. 2009; Wada 2013).
Specific morphological and physiological acclimations to different light might support plants coping with abiotic stress factors. Recently, we demonstrated that light quality, particularly the amount of blue light, affects the vulnerability of pepper plants to UV stress (Hoffmann et al. 2015). Other authors suggest that light induced acclimations might also affect drought vulnerability. Among others, light quality mediates stomatal aperture and affects transpiration (Chen et al. 2012; Zeiger and Field 1982). As shown in cucumber leaves, both leaf hydraulic and stomatal conductance were lower in plants cultivated in the absence of blue light while the osmotic-induced water deficit caused a stronger decrease of net photosynthesis in plants grown under monochromatic red light (Savvides et al. 2012). In this scope, Savvides and colleagues suggest damages of the photosynthetic apparatus, which only occurred under the monochromatic red light.
Apart from the light mediated stomatal aperture, adjustments at sub-cellular level might determine the vulnerability to drought. In case of slow dehydration, the acclimation avoids the activation of cyclic pathways and preserves the pH gradients even under drought (Bürling et al. 2014). Nevertheless, chloroplasts are a major site of damage when plants are exposed to severe stress (Walters 2005). Particularly the photosystem II (PSII) is known to be sensitive to abiotic stresses and might constrain photosynthesis in such situations (Björkman and Powles 1984; Havaux 1992; Murchie and Lawson 2013). In plants that suffer from drought, maximum photosynthesis is reached at a lower photon fluence rate than in plants under well-watered conditions (Lawlor and Cornic 2002). This can lead to an imbalance between energy harvesting and the capacity of energy utilization, particularly under high light conditions. Lower capacity of energy utilization results in higher dissipation of the absorbed energy and lower conversion into photochemistry (Demmig-Adams and Adams 2003). Specific light-induced adjustments in the composition and function of the photosynthetic apparatus might increase the capacity of energy utilization and thus determine the vulnerability to drought. In general terms, it is assumed that plants grown under high light/blue light conditions have a wider acclimation capacity being less susceptible to the exposition to stress factors (Lichtenthaler 1996). This is due to structural changes of chloroplasts that can also be triggered by blue light and are usually observed in sun-exposed or high-light leaves (Buschmann et al. 1978; Lichtenthaler et al. 2013). Typical characteristics of these so called “sun-type chloroplasts” are enhanced phenylquinone contents and higher Chl a/Chl b ratios that improve the photosynthetic performance (Lichtenthaler 1984). In general, leaves acclimated to high light can afford a higher Chl a/Chl b ratio as there is less need for light harvesting.
Apart from the processes described above at the stomata and chloroplast level, light induced morphological acclimations can affect the vulnerability to drought. In many plant species, a large aerial biomass (commonly with high contents of water) and a big transpiring surface is related to high water loss to the environment. Under high light conditions, plants are usually shorter and exhibit a smaller leaf area to reduce transpiration and to improve their drought resistance (Lichtenthaler 1996). In a similar sense, light quality might strongly affect plant morphology. In this context the ratios blue:green and red:far red are of central importance; blue light has been reported to promote a compact growth habit, whereas green and far red light enhance stem elongation (Sellaro et al. 2010; Zhang et al. 2011).
In general, there is a considerable lack of information concerning the interaction between light quality and the physiological and morphological responses of plants to water deficit. A more precise knowledge of such interactions would open new prospects for the target-oriented use of light to prepare or ‘prime’ plants for future stresses. Practical examples include the acclimatization of vegetable plantlets prior to their planting in the field, and potentially, the preparation of ornamental plants to better support the often adverse conditions at selling-points and at the customers home. Facing this situation and the promising perspectives, our main objective was to assess the effects of light quality and water deficit, as well as the impact of their interaction, on photosynthetic performance and plant growth.
The above described effects of light on plant physiology and morphology led us to hypothesize that specific light-mediated acclimations at plant and leaf levels reduce the vulnerability of pepper plants to water deficit. Due to the wide transpiring leaf surface and high stomatal conductance, pepper has high water requirements, and is therefore a good model plant for drought stress experiments (Alvino et al. 1994; Doorenbos and Kassam 1986). The sensitivity of pepper to water deficit is demonstrated in a large number of publications dealing with physiological responses as well as plant growth and biomass production (Delfine et al. 2001; De Pascale et al. 2003; González-Dugo et al. 2007; Ferrara et al. 2011; Kulkarni and Phalke 2009; Sezen et al. 2006; Smittle et al. 1994; Sziderics et al. 2010). In addition, findings demonstrate the sensitivity of pepper plants to light quality, including light- induced alterations in plant growth, dry matter partitioning and anatomical features of leaves and stems (Brown et al. 1995; Schuerger et al. 1997). In the present study, pepper plants were cultivated either under a wide-band light spectrum provided by CFL, or under LEDs offering narrow band spectra with dominant peaks at 445 and 665 nm. Chlorophyll fluorescence was used as a reliable technique to determine modifications in PSII photochemistry (Chaerle and Van der Straeten 2000, 2001; Flexas et al. 1999; Lichtenthaler and Miehé 1997; Maxwell and Johnson 2000; Sarijeva et al. 2007). Stomatal conductance was assessed with a portable gas exchange equipment. Plant biomass as well as the leaf contents of chlorophyll a + b (Chl a + b) and proline served as indicative parameters to evaluate the impact of light quality and water deficit.
Materials and methods
Plant material, growth conditions and experimental setup
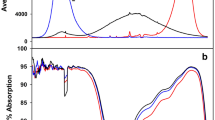
The experiment was conducted in a custom-built climate chamber. Pepper (Capsicum annuum L., 'Ziegenhorn Bello', Austrosaat AG, Graz, Austria) seeds were sown in trays filled with a mixture of peat (60 %), sand (20 %) and perlite (20 %) and allocated under white compact fluorescence lamps (CFL) with main peaks at 435 nm, 545 nm and 612 nm, respectively (Fig. 1) (MASTER PL-L 4P, Philips, Amsterdam, Netherlands). The photosynthetic photon fluence rate of CFL was set to 95 µmol m−2 s−1 whereby 14 % of the light energy was provided by blue, 40 % by green and 46 % by red light. Three weeks after seed sowing, plantlets were transplanted into pots (11 cm, ES round 8°, Goettinger, Lamprecht-Verpackungen GmbH, Göttingen, Germany), each filled with 250 g of the above described peat-sand-perlite mixture, and cultivated under the same environmental conditions for three more weeks. Next, half of the plants were kept under CFL while the remaining plants were allocated under LED modules (a prototype optimized for our research purposes; Ushio Lighting Inc., Tokyo, Japan). The LED-modules are characterized by a 2:1 combination of red and blue LEDs with single peaks respectively at 665 and 445 nm (Fig. 1). The photosynthetic photon fluence rate of the LED modules was also set to 95 ± 5 µmol m−2 s−1 whereby 36 % of the energy was provided by blue light and 64 % by red light. The light spectra were recorded at plant height with a high-resolution spectroradiometer (FieldSpec®3, ASD Inc., Boulder, USA). Plants were cultivated under a photoperiod of 12 h, with day/night temperature of 21/20 °C and relative humidity of 82 %.
One week after the plants were assigned to the respective lighting systems (CFL or LED) the water supply treatments were initiated. After the substrate was saturated with a modified Hoagland nutrient solution (pH 6.2) drought stress was induced by withholding water for 7 days [53–59 days after sowing (das)], followed by a recovery period (60–66 das) and a second cycle of water deficit (67–73 das). Water loss (evaporation + transpiration) was determined gravimetrically throughout the experiment. Control plants were irrigated daily throughout the experiment with the nutrient solution. The experimental treatments were identified as follows:
-
CFL_c = Compact fluorescence lamps, control
-
CFL_wd = Compact fluorescence lamps, water deficit
-
LED_c = Light-emitting diodes, control
-
LED_wd = Light-emitting diodes, water deficit
Growth parameters
Stem length was measured from the cotyledon to the youngest branching point. Measurements were done at least twice a week throughout the experiment. At the end of each experimental phase (59, 66, 73 das) fresh mass (FM) of leaves and stems was determined separately using a precision scale (BP210S, Sartorius, Göttingen, Germany) while leaf area was measured with a leaf area meter (LI-3100, LI-COR, Lincoln, Nebraska, USA). Stems and leaves were then frozen at −25 °C, lyophilized (Gamma 1–16 LSC; Christ, Osterode am Harz, Germany) and weighed again in order to quantify their respective dry mass (DM).
Stomatal conductance
Stomatal conductance (Gs) was recorded weekly with a portable infrared gas analyzer (CIRAS-1, PP Systems, Amesbury, USA) equipped with a standard 2.5 cm2 leaf cuvette (PLC B, PP Systems, Amesbury, USA). Measurements were carried out under the CFL adopting standardized settings: CO2 concentration 350 ± 5 ppm, photon fluence rate 100 ± 5 µmol m−2 s−1 PAR, a boundary layer resistance (Rb) of 0.27 m2 s mol−1 and a leaf chamber air flow rate of 200 ml min−1.
Chlorophyll fluorescence
Chlorophyll fluorescence was recorded weekly using an imaging pulse-amplitude-modulated fluorometer (Imaging PAM, Heinz-Walz GmbH, Effeltrich, Germany). Fluorescence images (640 × 480 pixels) were taken by a black and white CCD camera on fully expanded leaves at the third leaf level. To standardize measuring conditions and to make sure that all PSII reaction centers are open when the maximal photochemical quenching is determined, plants were dark-adapted for 30 min prior to the evaluations (see review of Maxwell and Johnson 2000). After recording the ground fluorescence (F0), a light saturation pulse was given to determine the maximum fluorescence yield (Fm). The yield of variable chlorophyll fluorescence (Fv) was calculated as Fm – F0 and the maximal PSII quantum yield was calculated as Fv/Fm. After the first saturation pulse actinic light was switched on and saturation pulses were applied at 20 s intervals in order to determine the maximum chlorophyll fluorescence in the light adapted state (Fm′). The electron transport rate (ETR) was calculated according to Björkman and Demmig (1987) and Krall and Edwards (1992) as: (Fm′–F)/Fm′) × PAR × 0.5 (fraction of excitation energy distributed to PSII) × 0.84 (leaf absorbance coefficient). Hereby, F describes the (nearly) steady state fluorescence signal of an illuminated leaf, assessed immediately prior to the application of a saturation pulse. Non-photochemical quenching (NPQ) which indicates the heat dissipation of excitation energy in the antenna system was calculated as: (Fm − Fm′)/Fm′. For each image captured by the CCD camera four areas of interest were selected and averaged.
Biochemical indicators
Proline concentration
Proline concentration in the samples was determined colorimetrically according to the method described by Bates et al. (1973) and Dolatabadian et al. (2008), slightly modified by us. Briefly, 3 ml sulfosalicylic acid (3 % w/v) was added to 0.1 g dried and ground leaf material, and the mixture was homogenized and centrifuged at 4,000 rpm for 15 min (Varifuge 3.0R, Heraeus Sepatech GmbH, Hanau, Germany). Next, 0.2 ml of the supernatant were added to 1.8 ml sulfosalicylic acid, 2 ml glacial acetic acid and 2 ml ninhydrine acid and incubated in a hot water bath (100 °C) for 1 h. After cooling to 20 °C, 4 ml toluene was added and mixed. The absorbance of the supernatant was measured at 520 nm with a UV-spectrophotometer (Lambda 35 UV/VIS Spectrophotometer, PerkinElmer, Waltham, USA). Proline concentrations were calculated from a standard curve.
Chlorophyll concentration
Chlorophyll concentration was determined colorimetrically after methanolic extraction, according to the method as described elsewhere (Holden 1976; Strobl and Türk 1990). For the extraction 5 ml methanol was added to 0.05 g dry and ground leaf material, mixed and centrifuged at 4,000 rpm for 15 min (Varifuge 3.0R, Heraeus Sepatech GmbH, Hanau, Germany). The supernatant was then decanted into 50 ml flasks. The pellet was extracted three more times until the extract was colourless. The collected supernatants were filled up with methanol to a defined volume (50 ml). Thereafter the absorbencies (A) were measured with a UV/VIS spectrophotometer (Lambda 35, Perkin-Elmer, Waltham, USA) at 650 nm and 665 nm. The following equations were used to calculate the concentrations of chlorophyll a (Chl a), chlorophyll b (Chl b) and total chlorophyll (Chl a + b):
Statistics
Statistical analyses were performed by one-way analysis of variance (Anova, P ≤ 0.05) using SPSS statistic software (PASW statistics version 20.0, SPSS Inc., Chicago, USA). As PostHoc analysis, Duncan´s multiple range test (P ≤ 0.05) was used to determine differences among the four treatments. The impact of light quality and water supply as well as the interaction between the two factors was determined by a two-factor analysis of variance (P ≤ 0.05).
Results
Growth parameters
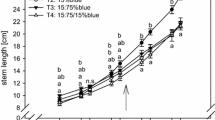
Light quality significantly affected stem length from 63 das until the end of the experiment. Thus, differences in stem length between the control plants (CFL_c, LED_c) were evident during the regeneration period (59–67 das), and became progressively bigger in the course of the experiment (Fig. 2), with highest values for CFL_c plants. This trend was confirmed by the FM and DM of the stems (Table S1). These parameters were also affected by light quality, with pronounced stem growth in plants cultivated under CFL. Water availability significantly affected stem length after 57 das. Drought stress limited plant elongation (Fig. 2), stem FM and stem DM (Table S1) in plants of both light treatments; nevertheless the effect was more pronounced in plants grown under CFL, as also indicated by the statistical interaction of light quality and water availability at the end of the experiment.
While leaf area was not significantly influenced by light quality (Table 1), drought stress limited expansion of leaf area similarly in plants of both light treatments. However, light quality had a significant impact on leaf DM, whereas the drought-induced limitation of leaf FM and DM formation was more pronounced under CFL. In addition, the percent dry matter, indicated by (DM × 100)/FM (Table 1) and the ratio leaf DM/stem DM (Table S1), were also influenced by light quality (P ≤ 0.02).
Biochemical indicators
Water deficit induced the synthesis and accumulation of proline in plants grown under CFL and LEDs as shown at the end of the water deficit periods. Thereby, drought- exposed plants grown under CFL had significantly higher proline concentration than those grown under LEDs (Fig. 3), although the statistics didn´t show a significant impact of light quality. During recovery, proline content decreased to that found in controls.
Proline concentration in pepper leaves as influenced by light quality (CFL, LED) and water supply. Evaluations were done at the end of the first drought period (59 das), at the end of the regeneration period (66 das) and after the second drought cycle (73 das). Mean ± SE (n ≥ 8) followed by the same letters do not differ significantly according to the Duncan test (P ≤ 0.05)
Light quality (CFL, LED) did not affect the total chlorophyll contents. In contrast, a significant impact of water supply was determined after rewatering (66 das), showing significantly lower contents of Chl a + b in plants of the water deficit treatments (Table 2). Light quality significantly changed the Chl a/Chl b ratio, that was higher in plants cultivated under the LED light.
Stomatal conductance (Gs) and maximal photochemical efficiency (Fv/Fm)
Five days after assignment to the lighting systems (CFL or LED) plants grown under CFL had significantly lower Gs than plants cultivated under the LEDs (Fig. 4). At this point Gs were significantly affected by the different light treatments (P ≤ 0.01). Exposure to water deficit decreased Gs in plants of both light treatments at 57 das; from this time until the end of the experiment the previously determined difference between plants of the two control treatments (CFL_c, LED_c) could not be observed anymore. During rewatering, plants previously exposed to water deficit (CFL_wd, LED_wd) reached the same stomatal conductance as the respective control groups. In the second drought cycle Gs decreased stronger in plants grown under CFL. While during the first drought cycle only water supply affected Gs, during the second drought cycle light quality (p ≤ 0.05) and water supply (P ≤ 0.01) affected the stomatal conductance. An interaction of both factors was not observed.
Stomatal conductance (left) and maximum photochemical efficiency of PSII (Fv/Fm) (right) of control and water deficit plants grown under CFL or LEDs. Gray regions indicate the periods of water deficit. Mean ± SE (n ≥ 8) followed by the same letters do not differ significantly according to the Duncan test (P ≤ 0.05)
The maximum photochemical efficiency, Fv/Fm, was not affected by light quality whereas water deficit significantly lowered Fv/Fm in plants of both light treatments (Fig. 4). At the end of the first drought period plants grown under LEDs were more affected by water deficit. In contrast, plants grown under fluorescence lamps showed a stronger decrease in Fv/Fm during the second drought cycle.
Electron transport rate (ETR) and non-photochemical quenching (NPQ)
After 1 week of acclimation (52 das) a significant impact of light treatments on ETR induction was evident (Fig. 5). At the start and end of the time-resolved measurements, ETR values were similar; however, the light treatments induced distinct curve patterns. In particular, plants grown under LEDs had significantly lower ETR from 100 to 180 s after kinetic induction. Five days after the first drought stress induction (57 das) ETR in the timeframe of 200–320 s was strongly impaired in plants that were cultivated under the CFL. At this point, ETR was significantly affected by light quality and water availability, although an interaction was not observed. Rewatering of plants (63 das) raised the ETR in plants that had been exposed to the stress, irrespective of light treatments. At the end of the second drought stress period (72 das) ETR was significantly lower in water deficit plants whereby plants grown under CFL were more affected. Here, ETR was affected by light quality and water availability and also an interaction of both factors was observed shortly after the first illumination.
Electron transport rate (ETR) of control and water deficit plants grown under CFL or LEDs. Measurements were taken 1 week after light adaptation (52 das), during the first drought period (57 das), during the regeneration period (63 das) and during the second drought cycle (72 das). Mean ± SE (n = 8) followed by the same letters do not differ significantly according to the Duncan test (P ≤ 0.05)
Similarly, the non-photochemical quenching, NPQ, revealed a significant impact of light quality before induction of water deficit (Fig. 6, 52 das) and during the first drought cycle. As shown in the kinetic curves, NPQ reached its maximum earlier in plants grown under CFL as compared to plants grown under LEDs. However, 57 das drought-exposed plants had a stronger increase of NPQ as compared to the well-watered plants (160–320 s), particularly for those grown under CFL. An interaction of light quality and water availability could not be observed at 57 das. During rewatering (63 das) NPQ of drought-stressed plants approached the level of the respective control plants. However, the higher values at 80–100 s in CFL_wd plants indicate that a complete recovery was not reached and also the statistics showed a significant impact of light quality and water availability. At the end of the second drought stress period, NPQ increased similarly in water deficit plants of both light treatments. Although the statistics no longer indicate an effect of light quality on thermal energy dissipation, it is conspicuous that NPQ was higher in plants grown under the CFL.
Non photochemical quenching (NPQ) of control and water deficit plants grown under CFL or LEDs. Measurements were taken 1 week after light adaptation (52 das), during the first drought period (57 das), during the regeneration period (63 das) and during the second drought cycle (72 das). Mean ± SE (n = 8) followed by the same letters do not differ significantly according to the Duncan test (P ≤ 0.05)
Discussion
In this paper we aimed to exploit the impact of light quality, provided either by compact fluorescent lamps or light emitting diodes, on the response of pepper plants to water deficit. In this context, we hypothesized that specific light-mediated acclimations at plant and leaf levels decrease the vulnerability of pepper plants to drought.
Contribution of growth parameters
Water loss of plants to the environment strongly depends on their biomass and the transpiring surface area, the later influencing the response of plants to a temporary water deficit. As we show here, the overall plant growth (stem length, FM, DM) was constrained in plants cultivated under the LED illumination, whereas the leaf/stem DM ratio increased, both typical adaptations to a blue enriched light spectrum (Brown et al.1995; Schuerger et al. 1997). Contrary to our expectations, light quality had no impact on total leaf area, which otherwise might have had a strong impact on transpiration. However, similar results were found by Xiaoying et al. (2012) who investigated the impact of different LED irradiations on growth characteristics of cherry tomato plants. Although we assume that the described modifications in plant growth influenced the susceptibility to drought, their real contribution cannot be estimated properly. By considering the parameters of total leaf area (Table 1), plant height (Fig. 2) and total plant FM and DM (Fig. S1), we note that there was no significant difference between control plants (CFL_c and LED_c) and water deficit plants (CFL_wd and LED_wd). Significant differences became apparent only during the regeneration and the second drought cycle.
Contribution of physiological acclimations
Light quality mediates stomatal aperture and thus affects transpiration rate. In particular, stomatal movements are strongly regulated by blue/UV-A light absorbing phototropins and cryptochromes (Chen et al. 2012). During the period of light acclimation (Fig. 4), a higher amount of blue light in the LED spectrum significantly triggered stomatal opening and the statistics also confirmed the impact of light quality during the second drought cycle. Nevertheless, in the course of the experiment Gs of plants grown under LEDs approached the values measured in plants grown under CFL. Thus, besides the photosynthetic relevance of stomatal conductance, as it regulates transpiration and affects gas exchange, other processes must have significantly influenced the photosynthetic performance. Working with osmotic stress, Savvides et al. (2012) report a stronger decrease of net photosynthesis in plants grown under monochromatic red light than in a blue light containing light spectrum, although stomatal conductance was not affected.
The degree to which plants are affected by environmental stresses strongly depends on their acclimation and recovery potentials, which are both influenced by the growth conditions (Ziska et al. 1992). Due to specific changes in the composition (e.g., increased Chl a/Chl b ratio) and structure of chloroplasts, it is assumed that plants grown under high light or blue light have a larger acclimation potential, enhancing their tolerance against various stresses (Lichtenthaler 1996). The increase in the Chl a/Chl b ratio is characteristic for sun-type chloroplasts that possess more electron transport chains on chlorophyll basis (Buschmann et al. 1978; Lichtenthaler 1984). As expected, the higher amount of blue light in the LED spectrum enhanced the Chl a/Chl b ratio, improving the photosynthetic performance during drought. Moreover, the relative amounts of PSII and PSI might have been changed, affecting the photosynthetic performance (Chow et al. 1990). In general terms, optimization of electron transport in the thylakoid membrane might be reached by adjustment of PSII/PSI stoichiometry to the prevailing light conditions through changes in the relative amounts of photosystems, as well as through accumulation of Chl a and/or Chl b (Anderson et al. 1995; Dietzel et al. 2008). In our study, Fv/Fm and the steady state values of ETR and NPQ were not affected by the different light treatments. Similarly, measurements of CO2 exchange confirmed these trends (data not shown). Although light curves would have provided precise information on the full photosynthetic potential (Ralph and Gademann 2005), our main objective was to analyze the photosynthetic performance of our experimental plants under similar light intensities as those used for cultivation. The kinetic curves of ETR and NPQ do reveal pronounced differences between the experimental groups. The faster induction of NPQ observed in CFL-grown plants (Fig. 6) may indicate a higher demand for energy dissipation shortly after first illumination. These plants were more affected by water deficits with more decreased ETR (Fig. 5) and increased NPQ (Fig. 6) than those grown under LEDs. This suggests a large imbalance between energy harvesting and the capacity for photosynthetic energy utilization in drought-stressed plants grown under CFL. A faster increase of ETR demonstrates that the photosynthetic induction is faster, indicating that the rate to which electrons are transported away from PSII, a process associated with stomatal opening and light-induced activation of enzymes, reaches the steady state more quickly (Maxwell and Johnson 2000). Analogous to that, a faster decrease of NPQ indicates that light use becomes more efficient as less energy is converted into heat and is therefore potentially available for the photochemical process. In this context, long-term dark relaxation curves might provide additional information on the different factors (high energy quenching, state transition and photoinhibition) that contributed to NPQ and should be considered for further examination (Quick and Stitt 1989; Walters and Horton 1991). In the course of the experiment, ETR and NPQ proved to be more sensitive to water deficit than Fv/Fm. Amongst others, Chaves (1991), Epron and Dreyer (1991) and Havaux (1992) highlighted that even a strong desiccation had only a marginal effect on Fv/Fm, demonstrating a remarkable resistance of the photosynthetic apparatus to dehydration. In contrast to that, Flexas et al. (1999) confirmed that ETR depends significantly on soil–water availability. Discrepancies among the cited studies might be related to the different plant species, the nature of dehydration and the measuring conditions. Finally, we conclude that differences in light spectral composition, particularly the amount of blue light, caused structural and functional acclimations at plant, leaf and cellular levels. While the morphological acclimations contributed to different water requirements, the photosynthetic acclimations, which were affected by the composition of chloroplasts, reduced the susceptibility to short-term water deficit. In contrast, stomatal conductance only had minor effects. Additional, species-specific research on the impact of light compositions on plant growth, stomatal behavior and photosynthesis might contribute for higher efficiency of plant production under greenhouse conditions and plant factories.
Abbreviations
- A:
-
Absorbance
- c:
-
Control
- das:
-
Days after sowing
- D:
-
Diameter
- CFL:
-
Compact fluorescence lamps
- Chl:
-
Chlorophyll
- DM:
-
Dry mass
- ETR:
-
Electron transport rate
- F:
-
Fluorescence yield
- Fm :
-
Maximum chlorophyll fluorescence of a dark adapted leaf
- Fm´:
-
Maximum chlorophyll fluorescence in the light adapted state
- F0 :
-
Ground fluorescence of a dark adapted leaf
- Fv :
-
Variable chlorophyll a fluorescence level from a dark adapted leaf (Fv = Fm − F0)
- FM:
-
Fresh mass
- Gs:
-
Stomatal conductance
- LED:
-
Light-emitting diode
- NPQ:
-
Non-photochemical quenching
- PAR:
-
Photosynthetic active radiation
- PSI:
-
Photosystem I
- PSII:
-
Photosystem II
- ROS:
-
Reactive oxygen species
- wd:
-
Water deficit
- Vol:
-
Volume
References
Abidi F, Girault T, Douillet O, Guillemain G, Sintes G, Laffaire M, Ben Ahmed H, Smiti S, Huché-Thélier L, Leduc N (2012) Blue light effects on rose photosynthesis and photomorphogenesis. Plant Biol 15:67–74
Alvino A, Centritto M, De Lorenzi F (1994) Photosynthesis response of sunlit and shade pepper (Capsicum annuum) leaves at different positions in the canopy under two water regimes. Aust J Plant Physiol 21:377–391
Anderson JM, Chow WS, Park YI (1995) The grand design of photosynthesis: acclimation of the photosynthetic apparatus to environmental cues. Photosynth Res 46:129–139
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207
Björkman O, Demmig B (1987) Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. Planta 170:489–504
Björkman O, Powles SB (1984) Inhibition of photosynthetic reactions under water stress: interaction with light level. Planta 161:490–504
Brown CS, Schuerger AC, Sager JC (1995) Growth and photomorphogenesis of pepper plants under red light-emitting diodes with supplemental blue or far-red lighting. J Am Soc Hortic Sci 120:808–813
Bürling K, Ducruet J-M, Cornic G, Hunsche M, Cerovic ZG (2014) Assessment of photosystem II thermoluminescence as a tool to investigate the effects of dehydration and rehydration on the cyclic/chlororespiratory electron pathways in wheat and barley leaves. Plant Sci 223:116–123
Buschmann C, Meier D, Kleudgen HK, Lichtenthaler HK (1978) Regulation of chloroplast development by red and blue light. Photochem Photobiol 27:195–198
Chaerle L, Van Der Straeten D (2000) Imaging techniques and the early detection of plant stress. Trends Plant Sci 5(11):495–501
Chaerle L, Van Der Straeten D (2001) Seeing is believing: imaging techniques to monitor plant health. Biochem Biophys Acta 1519:153–166
Chaves MM (1991) Effects of water deficits on carbon assimilation. J Exp Bot 42:1–16
Chen C, Xiao Y-G, Li X, Ni M (2012) Light-regulated stomatal aperture in Arabidopsis. Mol Plant 5:566–572
Chow WS, Melis A, Anderson JM (1990) Adjustments of photosystem stoichiometry in chloroplast improve the quantum efficiency of photosynthesis. Proc Natl Acad Sci USA 87:7502–7506
De Pascale S, Ruggiero C, Barbieri G (2003) Physiological responses of pepper to salinity and drought. J Am Soc Hortic Sci 128:48–54
Delfine S, Loreto F, Alvino A (2001) Drought-stress effects on physiology, growth and biomass production of rainfed and irrigated bell pepper plants in the mediterranean region. J Am Soc Hortic Sci 126:297–304
Demmig-Adams B, Adams WW III (2003) Photoprotection against excess light via zeaxanthin-dependent energy dissipation. In: Larcher W (ed) Physiological plant ecology. Springer, Heidelberg, pp 359–360
Dietzel L, Bräutigam K, Pfannschmidt T (2008) Photosynthetic acclimation: state transitions and adjustment of photosystem stoichiometry—functional relationships between short-term and long-term light quality acclimation in plants. FEBS J 275:1080–1088
Dolatabadian A, Sanavy SAMM, Chashmi NA (2008) The effects of foliar application of ascorbic acid (vitamin C) on antioxidant enzymes activities, lipid peroxidation and proline accumulation of Canola (Brassica napus L.) under conditions of salt stress. J Agron Crop Sci 194:206–213
Doorenbos J, Kassam AH (1986) Yield response to water, irrigation and drainage. Paper 33. FAO, Rome, Italy
Epron D, Dreyer E (1991) Effects of severe dehydration on leaf photosynthesis in Quercus petraea (Matt.) Liebl.: photosystem II efficiency, photochemical and nonphotochemical fluorescence quenching and electrolyte leakage. Tree Physiol 10:273–284
Ferrara A, Lovelli S, Di Tommaso T, Perniola M (2011) Flowering, growth and fruit setting in greenhouse bell pepper under water stress. J Agron 10:12–19
Flexas J, Escalona JM, Medrano H (1999) Water stress induces different levels of photosynthesis and electron transport rate regulation in grapevines. Plant Cell Environ 22:39–48
González-Dugo V, Orgaz F, Fereres E (2007) Responses of pepper to deficit irrigation for paprika production. Sci Hortic 114:77–82
Havaux M (1992) Stress tolerance of photosystem II in vivo. Antagonistic effects of water, heat and photoinhibition stresses. Plant Physiol 100:424–432
Hoffmann A, Noga G, Hunsche M (2015) High blue light improves acclimation and photosynthetic recovery of pepper plants exposed to UV stress. Environ Exp Bot 109:254–263
Hogewoning SW, Douwstra P, Trouwborst G, van Ieperen W, Harbinson J (2010a) An artificial solar spectrum substantially alters plant development compared with usual climate room irradiance spectra. J Exp Bot 61:1267–1276
Hogewoning SW, Trouwborst G, Maljaars H, Poorter H, van Ieperen W, Harbinson J (2010b) Blue light dose-responses of leaf photosynthesis, morphology, and chemical composition of Cucumis sativus grown under different combinations of red and blue light. J Exp Bot 61:3107–3117
Holden M (1976) Chlorophylls. In: Goodwin TW (ed) Chemistry and biochemistry of plant pigments, 2nd edn. Academic Press, London, pp 1–37
Krall JP, Edwards GE (1992) Relationship between photosystem II activity and CO 2 fixation in leaves. Physiol Plant 86:180–187
Kulkarni M, Phalke S (2009) Evaluating variability of root size system and its constitutive traits in hot pepper (Capsicum annum L.) under water stress. Sci Hortic 120:159–166
Lawlor DW, Cornic G (2002) Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ 25:275–294
Lichtenthaler HK (1984) Differences in morphology and chemical composition of leaves grown at different light intensities and qualities. In: Baker NR, Davies WJ, Ong KC (eds) Control of leaf growth. Cambridge University Press, Cambridge, pp 201–222
Lichtenthaler HK (1996) Vegetation stress: an introduction to the stress concept in plants. J Plant Physiol 148:4–14
Lichtenthaler HK, Miehé JA (1997) Fluorescence imaging as a diagnostic tool for plant stress. Trends Plant Sci 2:316–320
Lichtenthaler HK, Babani F, Navrátil M, Buschmann C (2013) Chlorophyll fluorescence kinetics, photosynthetic activity, and pigment composition of blue-shade and half-shade leaves as compared to sun and shade leaves of different trees. Photosynth Res 117:355–366
Loreto F, Tsonev T, Centritto M (2009) The impact of blue light on leaf mesophyll conductance. J Exp Bot 60:2283–2290
Massa GD, Kim HH, Wheeler RM, Mitchell CA (2008) Plant productivity in response to LED lighting. HortScience 43:1951–1956
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
Murakami K, Matsuda R, Fujiwara K (2014) Light-induced systemic regulation of photosynthesis in primary and trifoliate leaves of Phaseolus vulgaris: effects of photosynthetic photon flux density (PPFD) versus spectrum. Plant Biology 16:16–21
Muraoka H, Tang Y, Koizumi H, Washitani I (2002) Effects of light and soil water availability on leaf photosynthesis and growth of Arisaema heterophyllum, a riparian forest understorey plant. J Plant Res 115:419–427
Murchie EH, Lawson T (2013) Chlorophyll fluorescence analysis: a guide to good practice and understanding some new applications. J Exp Bot 64:3983–3998
Quick WP, Stitt M (1989) An examination of factors contributing to non-photochemical quenching of chlorophyll fluorescence in barley leaves. Biochim Biophys Acta 977:287–296
Ralph PJ, Gademann R (2005) Rapid light curves: a powerful tool to assess photosynthetic activity. Aquat Bot 82:222–237
Sarijeva G, Knapp M, Lichtenthaler HK (2007) Differences in photosynthetic activity, chlorophyll and carotenoid levels, and in chlorophyll fluorescence parameters in green sun and shade leaves of Ginko and Fagus. J Plant Physiol 164:950–955
Savvides A, Fanourakis D, van Ieperen W (2012) Co-ordination of hydraulic and stomatal conductances across light qualities in cucumber leaves. J Exp Bot 63:1135–1143
Schuerger AC, Brown CS, Stryjewski EC (1997) Anatomical features of pepper plants (Capsicum annuum L.) grown under red light-emitting diodes supplemented with blue or far-red light. Ann Bot 79:273–282
Sellaro R, Crepy M, Trupkin SA, Karayekov E, Buchovsky AS, Rossi C, Casal JJ (2010) Cryptochrome as a sensor of the blue/green ratio of natural radiation in Arabidopsis. Plant Physiol 154:401–409
Sezen SM, Yazar A, Eker S (2006) Effect of drip irrigation regimes on yield and quality of field grown bell pepper. Agr Water Manage 81:115–131
Smittle DA, Lamar Dickens W, Stansell JR (1994) Irrigation regimes affect yield and water use by bell pepper. J Am Soc Hortic Sci 119:936–939
Strobl A, Türk R (1990) Untersuchungen zum Chlorophyllgehalt einiger subalpiner Flechtenarten. Phyton Ann Rei Bot A 30:247–264
Sziderics AH, Oufir M, Trognitz F, Kopecky D, Matusikova I, Hausman JF, Wilhelm E (2010) Organ-specific defence strategies of pepper (Capsicum annuum L.) during early phase of water deficit. Plant Cell Rep 29:295–305
Terashima I, Fujita T, Inoue T, Chow WS, Oguchi R (2009) Green light drives leaf photosynthesis more efficiently than red light in strong white light: revisiting the enigmatic question of why leaves are green. Plant Cell Physiol 50:684–697
Terfa MT, Solhaug KA, Gislerod HR, Olsen JE, Torre S (2013) A high proportion of blue light increases the photosynthesis capacity and leaf formation rate of Rosa x hybrida but does not affect time to flower opening. Physiol Plantarum 148:146–159
Wada M (2013) Chloroplast movement. Plant Sci 210:177–182
Walters RG (2005) Towards an understanding of photosynthetic acclimation. J Exp Bot 56:435–447
Walters RG, Horton P (1991) Resolution of components of noon-photochemical chlorophyll fluorescence quenching in barley leaves. Photosynth Res 27:121–133
Xiaoying L, Shirong G, Taotao C, Zhigang X, Tezuka T (2012) Regulation of the growth and photosynthesis of cherry tomato seedlings by different light irradiations of light emitting diodes (LED). Afr J Biotechnol 11:6169–6177
Zeiger E, Field C (1982) Photocontrol of the functional coupling between photosynthesis and stomatal conductance in the intact leaf. Plant Physiol 70:370–375
Zhang T, Maruhnich SA, Folta KM (2011) Green light induces shade avoidance symptoms. Plant Physiol 157:1528–1536
Ziska LH, Teramura AH, Sullivan JH (1992) Physiological sensitivity of plants along an elevational gradient to UV-B radiation. Am J Bot 79:863–871
Acknowledgments
The authors thank Mr. Toshihiko Oishi, Ushio Europe B. V. (The Netherlands), and the group of technical engineers from Ushio Lighting Inc. (Japan) for developing and making the LED panels available for this study. We are grateful to Prof. Dr. Uwe Rascher, Institute of Bio- and Geoscience (IBG-2), Jülich Research Center, for loaning the spectroradiometer. Many thanks to Libeth Schwager, INRES Horticultural Science, for her support in the laboratory and to Elif Köllhofer for her assistance during the experimental phase. Finally, we acknowledge the Theodor-Brinkmann-Graduate School (Faculty of Agriculture, University of Bonn) for providing a scholarship to the first author. We also appreciate the critical-constructive comments of the anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hoffmann, A.M., Noga, G. & Hunsche, M. Acclimations to light quality on plant and leaf level affect the vulnerability of pepper (Capsicum annuum L.) to water deficit. J Plant Res 128, 295–306 (2015). https://doi.org/10.1007/s10265-014-0698-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-014-0698-z