Abstract
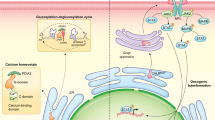
Calreticulin (CRT) is a multifunctional protein found within the endoplasmic reticulum (ER). In addition, CRT participates in the formation and development of tumors and promotes the proliferation and migration of tumor cells. When a malignant tumor occurs in the human body, cancer cells that die from immunogenic cell death (ICD) expose CRT on their surface, and CRT that is transferred to the cell surface represents an "eat me" signal, which promotes dendritic cells to phagocytose the tumor cells, thereby increasing the sensitivity of tumors to anticancer immunotherapy. Expression of CRT in tumor tissues is higher than in normal tissues and is associated with disease progression in many malignant tumors. Thus, the dysfunctional production of CRT can promote tumorigenesis because it disturbs not only the balance of healthy cells but also the body's immune surveillance. CRT may be a diagnostic marker and a therapeutic target for cancer, which is discussed extensively in this review.

Similar content being viewed by others
Abbreviations
- AML:
-
Acute myeloid leukemia
- BC:
-
Breast cancer
- CRT:
-
Calreticulin
- ecto-CRT:
-
Plasma membrane surface calreticulin
- ER:
-
Endoplasmic reticulum
- EMT:
-
Epithelial-mesenchymal transition
- EC:
-
Esophageal cancer
- ICD:
-
Immunogenic cell death
- IFN:
-
Type I interferon
- JAK2:
-
Janus kinase 2
- MPNs:
-
Myeloproliferative neoplasms
- MPL:
-
Myeloproliferative leukemia protein
- NPC:
-
Nasopharyngeal carcinoma
- PCR:
-
Polymerase chain reaction
References
Fucikova J, et al. Calreticulin and cancer. Cell Res. 2021;31(1):5–16.
Li Y, et al. Bioinformatics analysis for the role of CALR in human cancers. PLoS ONE. 2021;16(12): e0261254.
Ostwald TJ, MacLennan DH. Isolation of a high affinity calcium-binding protein from sarcoplasmic reticulum. J Biol Chem. 1974;249(3):974–9.
McCauliffe DP, et al. The 5’-flanking region of the human calreticulin gene shares homology with the human GRP78, GRP94, and protein disulfide isomerase promoters. J Biol Chem. 1992;267(4):2557–62.
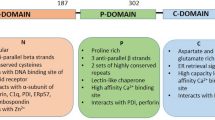
Michalak M, et al. Calreticulin, a multi-process calcium-buffering chaperone of the endoplasmic reticulum. Biochem J. 2009;417(3):651–66.
Opas M, et al. Regulation of expression and intracellular distribution of calreticulin, a major calcium binding protein of nonmuscle cells. J Cell Physiol. 1991;149(1):160–71.
Smith MJ, Koch GL. Multiple zones in the sequence of calreticulin (CRP55, calregulin, HACBP), a major calcium binding ER/SR protein. EMBO J. 1989;8(12):3581–6.
Michalak M, et al. Calreticulin: one protein, one gene, many functions. Biochem J. 1999;344(Pt 2):281–92.
Nakamura K, et al. Functional specialization of calreticulin domains. J Cell Biol. 2001;154(5):961–72.
Burns K, et al. Calreticulin: from Ca2+ binding to control of gene expression. Trends Cell Biol. 1994;4(5):152–4.
Pike SE, et al. Calreticulin and calreticulin fragments are endothelial cell inhibitors that suppress tumor growth. Blood. 1999;94(7):2461–8.
Goicoechea S, et al. The anti-adhesive activity of thrombospondin is mediated by the N-terminal domain of cell surface calreticulin. J Biol Chem. 2002;277(40):37219–28.
Coppolino MG, Dedhar S. Calreticulin. Int J Biochem Cell Biol. 1998;30(5):553–8.
Baksh S, Michalak M. Expression of calreticulin in Escherichia coli and identification of its Ca2+ binding domains. J Biol Chem. 1991;266(32):21458–65.
Michalak M, et al. Calreticulin. Biochem J. 1992;285(Pt 3):681–92.
Breier A, Michalak M. 2,4,6-Trinitrobenzenesulfonic acid modification of the carboxyl-terminal region (C-domain) of calreticulin. Mol Cell Biochem. 1994;130(1):19–28.
Sonnichsen B, et al. Retention and retrieval: both mechanisms cooperate to maintain calreticulin in the endoplasmic reticulum. J Cell Sci. 1994;107(Pt 10):2705–17.
Wijeyesakere SJ, et al. The C-Terminal Acidic Region of Calreticulin Mediates Phosphatidylserine Binding and Apoptotic Cell Phagocytosis. J Immunol. 2016;196(9):3896–909.
John LM, Lechleiter JD, Camacho P. Differential modulation of SERCA2 isoforms by calreticulin. J Cell Biol. 1998;142(4):963–73.
Milner RE, et al. Calreticulin, and not calsequestrin, is the major calcium binding protein of smooth muscle sarcoplasmic reticulum and liver endoplasmic reticulum. J Biol Chem. 1991;266(11):7155–65.
Mesaeli N, et al. Calreticulin is essential for cardiac development. J Cell Biol. 1999;144(5):857–68.
Faustino RS, et al. Calreticulin secures calcium-dependent nuclear pore competency required for cardiogenesis. J Mol Cell Cardiol. 2016;92:63–74.
Johnson S, et al. The ins and outs of calreticulin: from the ER lumen to the extracellular space. Trends Cell Biol. 2001;11(3):122–9.
Raghavan M, et al. Calreticulin in the immune system: ins and outs. Trends Immunol. 2013;34(1):13–21.
Lu YC, Weng WC, Lee H. Functional roles of calreticulin in cancer biology. Biomed Res Int. 2015;2015: 526524.
Panaretakis T, et al. Mechanisms of pre-apoptotic calreticulin exposure in immunogenic cell death. EMBO J. 2009;28(5):578–90.
Scorrano L, et al. BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science. 2003;300(5616):135–9.
Tufi R, et al. Reduction of endoplasmic reticulum Ca2+ levels favors plasma membrane surface exposure of calreticulin. Cell Death Differ. 2008;15(2):274–82.
Obeid M, et al. Calreticulin exposure is required for the immunogenicity of gamma-irradiation and UVC light-induced apoptosis. Cell Death Differ. 2007;14(10):1848–50.
Kepp O, et al. Surface-exposed and soluble calreticulin: conflicting biomarkers for cancer prognosis. Oncoimmunology. 2020;9(1):1792037.
Tesniere A, et al. Molecular characteristics of immunogenic cancer cell death. Cell Death Differ. 2008;15(1):3–12.
Osman R, et al. Calreticulin release at an early stage of death modulates the clearance by macrophages of apoptotic cells. Front Immunol. 2017;8:1034.
Clarke C, Smyth MJ. Calreticulin exposure increases cancer immunogenicity. Nat Biotechnol. 2007;25(2):192–3.
Ohkuro M, et al. Calreticulin and integrin alpha dissociation induces anti-inflammatory programming in animal models of inflammatory bowel disease. Nat Commun. 2018;9(1):1982.
Ito H, Seyama Y, Kubota S. Calreticulin is directly involved in anti-alpha3 integrin antibody-mediated secretion and activation of matrix metalloprotease-2. Biochem Biophys Res Commun. 2001;283(2):297–302.
Iwahashi N, et al. Calreticulin Regulates Syncytialization Through Control of the Synthesis and Transportation of E-Cadherin in BeWo Cells. Endocrinology. 2019;160(2):359–74.
Cheng WF, et al. Tumor-specific immunity and antiangiogenesis generated by a DNA vaccine encoding calreticulin linked to a tumor antigen. J Clin Invest. 2001;108(5):669–78.
Chiang WF, et al. Calreticulin, an endoplasmic reticulum-resident protein, is highly expressed and essential for cell proliferation and migration in oral squamous cell carcinoma. Oral Oncol. 2013;49(6):534–41.
Klampfl T, et al. Somatic mutations of calreticulin in myeloproliferative neoplasms. N Engl J Med. 2013;369(25):2379–90.
Nangalia J, et al. Somatic CALR mutations in myeloproliferative neoplasms with nonmutated JAK2. N Engl J Med. 2013;369(25):2391–405.
Belcic Mikic T, Pajic T, M. Sever, CALR mutations in a cohort of JAK2 V617F negative patients with suspected myeloproliferative neoplasm. Sci Rep. 2019;9(1):19838.
Edahiro Y, Araki M, Komatsu N. Mechanism underlying the development of myeloproliferative neoplasms through mutant calreticulin. Cancer Sci. 2020;111(8):2682–8.
Marty C, et al. Calreticulin mutants in mice induce an MPL-dependent thrombocytosis with frequent progression to myelofibrosis. Blood. 2016;127(10):1317–24.
Chachoua I, et al. Thrombopoietin receptor activation by myeloproliferative neoplasm associated calreticulin mutants. Blood. 2016;127(10):1325–35.
Pecquet C, et al. Calreticulin mutants as oncogenic rogue chaperones for TpoR and traffic-defective pathogenic TpoR mutants. Blood. 2019;133(25):2669–81.
Elf S, et al. Mutant Calreticulin Requires Both Its Mutant C-terminus and the Thrombopoietin Receptor for Oncogenic Transformation. Cancer Discov. 2016;6(4):368–81.
Shide K, et al. Calreticulin mutant mice develop essential thrombocythemia that is ameliorated by the JAK inhibitor ruxolitinib. Leukemia. 2017;31(5):1136–44.
Lim KH, et al. Expression of CALR mutants causes mpl-dependent thrombocytosis in zebrafish. Blood Cancer J. 2016;6(10): e481.
Shide K, et al. Calreticulin haploinsufficiency augments stem cell activity and is required for onset of myeloproliferative neoplasms in mice. Blood. 2020;136(1):106–18.
Kaur A, Raghavan M. A calreticulin tail: C-terminal mutants of calreticulin allow cancer cells to evade phagocytosis. Mol Cell. 2020;77(4):683–5.
Di Buduo CA, et al. Defective interaction of mutant calreticulin and SOCE in megakaryocytes from patients with myeloproliferative neoplasms. Blood. 2020;135(2):133–44.
Liu P, et al. Secreted calreticulin mutants subvert anticancer immunosurveillance. Oncoimmunology. 2020;9(1):1708126.
Belcic Mikic T, et al. The contemporary approach to CALR-positive myeloproliferative neoplasms. Int J Mol Sci. 2021;22(7):3371.
Pronier E, et al. Targeting the CALR interactome in myeloproliferative neoplasms. JCI Insight. 2018;3(22):122703.
Han L, et al. Calreticulin-mutant proteins induce megakaryocytic signaling to transform hematopoietic cells and undergo accelerated degradation and Golgi-mediated secretion. J Hematol Oncol. 2016;9(1):45.
Nieborowska-Skorska M, et al. Ruxolitinib-induced defects in DNA repair cause sensitivity to PARP inhibitors in myeloproliferative neoplasms. Blood. 2017;130(26):2848–59.
Hui W, et al. Alterations of signaling pathways in essential thrombocythemia with calreticulin mutation. Cancer Manag Res. 2021;13:6231–8.
Chao MP, et al. Calreticulin is the dominant prophagocytic signal on multiple human cancers and is counterbalanced by CD47. Sci Transl Med. 2010;2(63):63ra94.
Obeid M, et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. 2007;13(1):54–61.
Fucikova J, et al. Calreticulin exposure by malignant blasts correlates with robust anticancer immunity and improved clinical outcome in AML patients. Blood. 2016;128(26):3113–24.
Ivashkiv LB, Donlin LT. Regulation of type I interferon responses. Nat Rev Immunol. 2014;14(1):36–49.
Yang PM, et al. Sequential interferon beta-cisplatin treatment enhances the surface exposure of calreticulin in cancer cells via an interferon regulatory factor 1-dependent manner. Biomolecules. 2020;10(4):643.
Vaes RDW, et al. Biomarkers of radiotherapy-induced immunogenic cell death. Cells. 2021;10(4):930.
Truxova I, et al. Calreticulin exposure on malignant blasts correlates with improved natural killer cell-mediated cytotoxicity in acute myeloid leukemia patients. Haematologica. 2020;105(7):1868–78.
Galluzzi L, Kroemer G. Calreticulin and type I interferon: An unsuspected connection. Oncoimmunology. 2017;6(3): e1288334.
Chen X, et al. Calreticulin promotes immunity and type I interferon-dependent survival in mice with acute myeloid leukemia. Oncoimmunology. 2017;6(4): e1278332.
Sun J, et al. Diallyl disulfide down-regulates calreticulin and promotes C/EBPalpha expression in differentiation of human leukaemia cells. J Cell Mol Med. 2019;23(1):194–204.
Wemeau M, et al. Calreticulin exposure on malignant blasts predicts a cellular anticancer immune response in patients with acute myeloid leukemia. Cell Death Dis. 2010;1: e104.
Zhang Y, et al. A siRNA-assisted assembly strategy to simultaneously suppress “self” and upregulate “eat-me” signals for nanoenabled chemo-immunotherapy. ACS Nano. 2021;15(10):16030–42.
Zhao J, et al. Diallyl disulfide suppresses growth of HL-60 cell through increasing histone acetylation and p21WAF1 expression in vivo and in vitro. Acta Pharmacol Sin. 2006;27(11):1459–66.
Yi L, et al. Involvement of calreticulin in cell proliferation, invasion and differentiation in diallyl disulfide-treated HL-60 cells. Oncol Lett. 2016;12(3):1861–7.
Tesniere A, et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene. 2010;29(4):482–91.
Huang Y, et al. Comparison of the effects of photon, proton and carbon-ion radiation on the ecto-calreticulin exposure in various tumor cell lines. Ann Transl Med. 2019;7(20):542.
Zheng Y, et al. Calreticulin increases growth and progression of natural killer/T-cell lymphoma. Aging (Albany NY). 2020;12(23):23822–35.
Han A, et al. Calreticulin is a critical cell survival factor in malignant neoplasms. PLoS Biol. 2019;17(9): e3000402.
Pekarikova A, et al. Calreticulin is a B cell molecular target in some gastrointestinal malignancies. Clin Exp Immunol. 2010;160(2):215–22.
Liu YS, et al. Calreticulin nuclear translocalization alleviates CaM/CaMKII/CREB signaling pathway to enhance chemosensitivity in HDAC inhibitor-resistant hepatocellular carcinoma cells. Aging (Albany NY). 2022;14(12):5097–115.
Cheng CY, Su CC. Tanshinone IIA inhibits Hep-J5 cells by increasing calreticulin, caspase 12 and GADD153 protein expression. Int J Mol Med. 2010;26(3):379–85.
Feng R, et al. Calreticulin down-regulation inhibits the cell growth, invasion and cell cycle progression of human hepatocellular carcinoma cells. Diagn Pathol. 2015;10:149.
Bezu L, et al. eIF2alpha phosphorylation: A hallmark of immunogenic cell death. Oncoimmunology. 2018;7(6): e1431089.
Ye J, et al. Calreticulin: a potential diagnostic and therapeutic biomarker in gallbladder cancer. Aging (Albany NY). 2021;13(4):5607–20.
Toquet C, et al. Altered Calreticulin expression in human colon cancer: maintenance of Calreticulin expression is associated with mucinous differentiation. Oncol Rep. 2007;17(5):1101–7.
Vougas K, et al. Two-dimensional electrophoresis and immunohistochemical study of calreticulin in colorectal adenocarcinoma and mirror biopsies. J BUON. 2008;13(1):101–7.
Chih HW, et al. Bullatacin, a potent antitumor annonaceous acetogenin, inhibits proliferation of human hepatocarcinoma cell line 2.2.1.5 by apoptosis induction. Life Sci. 2001;69(11):1321–31.
Fan F, et al. Bullatacin triggers immunogenic cell death of colon cancer cells by activating endoplasmic reticulum chaperones. J Inflamm (Lond). 2021;18(1):23.
Colangelo T, et al. The miR-27a-calreticulin axis affects drug-induced immunogenic cell death in human colorectal cancer cells. Cell Death Dis. 2016;7: e2108.
Kopecka J, et al. Nitric oxide and P-glycoprotein modulate the phagocytosis of colon cancer cells. J Cell Mol Med. 2011;15(7):1492–504.
Du XL, et al. Calreticulin promotes cell motility and enhances resistance to anoikis through STAT3-CTTN-Akt pathway in esophageal squamous cell carcinoma. Oncogene. 2009;28(42):3714–22.
Shi F, et al. Calreticulin promotes migration and invasion of esophageal cancer cells by upregulating neuropilin-1 expression via STAT5A. Clin Cancer Res. 2014;20(23):6153–62.
Wang XM, et al. PTP1B contributes to calreticulin-induced metastatic phenotypes in esophageal squamous cell carcinoma. Mol Cancer Res. 2013;11(9):986–94.
Liu SH, et al. Honokiol confers immunogenicity by dictating calreticulin exposure, activating ER stress and inhibiting epithelial-to-mesenchymal transition. Mol Oncol. 2015;9(4):834–49.
Kong Q, Zhang Z, Liang Z. Upregulating miR-637 aggravates endoplasmic reticulum stress-induced apoptosis in gastric cancer cells by suppressing Calreticulin. Anim Cells Syst (Seoul). 2020;24(5):267–74.
Lee PC, et al. Calreticulin regulates vascular endothelial growth factor-A mRNA stability in gastric cancer cells. PLoS ONE. 2019;14(11): e0225107.
Chen CN, et al. Identification of calreticulin as a prognosis marker and angiogenic regulator in human gastric cancer. Ann Surg Oncol. 2009;16(2):524–33.
Wang L, et al. Calreticulin enhances gastric cancer metastasis by dimethylating H3K9 in the E-cadherin promoter region mediating by G9a. Oncogenesis. 2022;11(1):29.
Hong SH, et al. An autoantibody-mediated immune response to calreticulin isoforms in pancreatic cancer. Cancer Res. 2004;64(15):5504–10.
Matsukuma S, et al. Calreticulin is highly expressed in pancreatic cancer stem-like cells. Cancer Sci. 2016;107(11):1599–609.
Sheng W, et al. Calreticulin promotes EGF-induced EMT in pancreatic cancer cells via Integrin/EGFR-ERK/MAPK signaling pathway. Cell Death Dis. 2017;8(10): e3147.
Sheng W, et al. Calreticulin promotes EMT in pancreatic cancer via mediating Ca(2+) dependent acute and chronic endoplasmic reticulum stress. J Exp Clin Cancer Res. 2020;39(1):209.
Sheng W, et al. Overexpression of calreticulin contributes to the development and progression of pancreatic cancer. J Cell Physiol. 2014;229(7):887–97.
Huang X, et al. Calreticulin couples with immune checkpoints in pancreatic cancer. Clin Transl Med. 2020;10(1):36–44.
Yoneda A, Minomi K, Tamura Y. Heat shock protein 47 confers chemoresistance on pancreatic cancer cells by interacting with calreticulin and IRE1alpha. Cancer Sci. 2021;112(7):2803–20.
Zamanian M, et al. Calreticulin mediates an invasive breast cancer phenotype through the transcriptional dysregulation of p53 and MAPK pathways. Cancer Cell Int. 2016;16:56.
Cruz-Ramos E, Sandoval-Hernandez A, Tecalco-Cruz AC. Differential expression and molecular interactions of chromosome region maintenance 1 and calreticulin exportins in breast cancer cells. J Steroid Biochem Mol Biol. 2019;185:7–16.
Eric-Nikolic A, et al. Overexpression of calreticulin in malignant and benign breast tumors: relationship with humoral immunity. Oncology. 2012;82(1):48–55.
Calvillo-Rodriguez KM, et al. PKHB1, a thrombospondin-1 peptide mimic, induces anti-tumor effect through immunogenic cell death induction in breast cancer cells. Oncoimmunology. 2022;11(1):2054305.
Eric A, et al. Effects of humoral immunity and calreticulin overexpression on postoperative course in breast cancer. Pathol Oncol Res. 2009;15(1):89–90.
He D, et al. Ecotropic virus integration-1 and calreticulin as novel prognostic markers in triple-negative breast cancer: A retrospective cohort study. Oncol Lett. 2019;18(2):1847–55.
Lwin ZM, et al. Clinicopathological significance of calreticulin in breast invasive ductal carcinoma. Mod Pathol. 2010;23(12):1559–66.
Wu L, et al. EVI1 acts as an oncogene and positively regulates calreticulin in breast cancer. Mol Med Rep. 2019;19(3):1645–53.
Liu X, et al. HIF-1-regulated expression of calreticulin promotes breast tumorigenesis and progression through Wnt/beta-catenin pathway activation. Proc Natl Acad Sci U S A. 2021;118(44):e2109144118.
Liu R, et al. Calreticulin as a potential diagnostic biomarker for lung cancer. Cancer Immunol Immunother. 2012;61(6):855–64.
Gao F, et al. Calreticulin (CALR)-induced activation of NF-kB signaling pathway boosts lung cancer cell proliferation. Bioengineered. 2022;13(3):6856–65.
Fucikova J, et al. Calreticulin expression in human non-small cell lung cancers correlates with increased accumulation of antitumor immune cells and favorable prognosis. Cancer Res. 2016;76(7):1746–56.
Wu Y, et al. Calreticulin regulates TGF-beta1-induced epithelial mesenchymal transition through modulating Smad signaling and calcium signaling. Int J Biochem Cell Biol. 2017;90:103–13.
Fucikova J, et al. High hydrostatic pressure induces immunogenic cell death in human tumor cells. Int J Cancer. 2014;135(5):1165–77.
Inoue H, et al. Increased plasma levels of damage-associated molecular patterns during systemic anticancer therapy in patients with advanced lung cancer. Transl Lung Cancer Res. 2021;10(6):2475–86.
Furukawa R, et al. Cytotoxic chemotherapeutic agents and the EGFR-TKI osimertinib induce calreticulin exposure in non-small cell lung cancer. Lung Cancer. 2021;155:144–50.
Ye D, et al. LMP1 Up-regulates Calreticulin to Induce Epithelial-mesenchymal Transition via TGF-beta/Smad3/NRP1 Pathway in Nasopharyngeal Carcinoma Cells. J Cancer. 2020;11(5):1257–69.
Han Y, et al. High expression of calreticulin indicates poor prognosis and modulates cell migration and invasion via activating Stat3 in nasopharyngeal carcinoma. J Cancer. 2019;10(22):5460–8.
Alur M, et al. Suppressive roles of calreticulin in prostate cancer growth and metastasis. Am J Pathol. 2009;175(2):882–90.
Zhu N, et al. Calreticulin: an intracellular Ca++-binding protein abundantly expressed and regulated by androgen in prostatic epithelial cells. Endocrinology. 1998;139(10):4337–44.
Zhu N, Wang Z. Calreticulin expression is associated with androgen regulation of the sensitivity to calcium ionophore-induced apoptosis in LNCaP prostate cancer cells. Cancer Res. 1999;59(8):1896–902.
Nguyen MM, et al. Cytoplasmic localization of the androgen receptor is independent of calreticulin. Mol Cell Endocrinol. 2009;302(1):65–72.
Lin YC, et al. Calreticulin regulates beta1-integrin mRNA stability in PC-3 prostate cancer cells. Biomedicines. 2022;10(3):646.
Lin YC, et al. LPA1/3 signaling mediates tumor lymphangiogenesis through promoting CRT expression in prostate cancer. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863(10):1305–15.
Galazis N, et al. Proteomic biomarkers for ovarian cancer risk in women with polycystic ovary syndrome: a systematic review and biomarker database integration. Fertil Steril. 2012;98(6):1590–601.
Abdullah TM, et al. Endoplasmic reticulum stress-induced release and binding of calreticulin from human ovarian cancer cells. Cancer Immunol Immunother. 2022;71(7):1655–69.
Stoll G, et al. Calreticulin expression: Interaction with the immune infiltrate and impact on survival in patients with ovarian and non-small cell lung cancer. Oncoimmunology. 2016;5(7): e1177692.
Acknowledgements
This study was funded by the National key research and development project (NO. 2021YFE0192100), Natural Science Foundation of Hunan Province (NO.2019JJ40258, NO. 2020JJ4083, NO.2021JJ30694), Science and Technology Innovation Project of Hunan Province (NO. 2020SK51703), Innovation and Entrepreneurship Training Program for College Students in Hunan Province (NO.S202110555296), Key Projects of Hunan Provincial Education Department (NO.21A0285), Natural Science Foundation of Hunan Provincial and Municipal Co-funding (NO.2022JJ50029) , Key projects of Shaoyang Science and Technology Bureau (NO.2021GZ031)
Author information
Authors and Affiliations
Contributions
MZ, JX and XB wrote the manuscript; XZ and JL collected literatures; FL and ZZ supervised the work and drew the figures.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, M., Xiao, J., Liu, J. et al. Calreticulin as a marker and therapeutic target for cancer. Clin Exp Med 23, 1393–1404 (2023). https://doi.org/10.1007/s10238-022-00937-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-022-00937-7