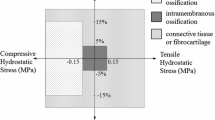
Abstract
Mechanical cues are known to regulate tissue differentiation during skeletal healing. Quantitative characterization of this mechano-regulatory effect has great therapeutic potential. This study tested an existing theory that shear strain and interstitial fluid flow govern skeletal tissue differentiation by applying this theory to a scenario in which a bending motion applied to a healing transverse osteotomy results in cartilage, rather than bone, formation. A 3-D finite element mesh was created from micro-computed tomography images of a bending-stimulated callus and was used to estimate the mechanical conditions present in the callus during the mechanical stimulation. Predictions regarding the patterns of tissues—cartilage, fibrous tissue, and bone—that formed were made based on the distributions of fluid velocity and octahedral shear strain. These predictions were compared to histological sections obtained from a previous study. The mechano-regulation theory correctly predicted formation of large volumes of cartilage within the osteotomy gap and many, though not all patterns of tissue formation observed throughout the callus. The results support the concept that interstitial fluid velocity and tissue shear strain are key mec- hanical stimuli for the differentiation of skeletal tissues.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Angele P, Schumann D, Angele M, Kinner B, Englert C, Hente R, Fuchtmeier B, Nerlich M, Neumann C, Kujat R (2004) Cyclic, mechanical compression enhances chondrogenesis of mesenchymal progenitor cells in tissue engineering scaffolds. Biorheology 41(3–4): 335–346
Bailon-Plaza A, Vander Meulen MC (2003) Beneficial effects of moderate, early loading and adverse effects of delayed or excessive loading on bone healing. J Biomech 36: 1069–1077. doi:10.1016/S0021-9290(03)00117-9
Blenman PR, Carter DR, Beaupre GS (1989) Role of mechanical loading in the progressive ossification of a fracture callus. J Orthop Res 7(3): 398–407. doi:10.1002/jor.1100070312
Byrne DP, Lacroix D, Planell JA, Kelly DJ, Prendergast PJ (2007) Simulation of tissue differentiation in a scaffold as a function of porosity, Young’s modulus and dissolution rate: application of mechanobiological models in tissue engineering. Biomaterials 28(36): 5544–5554. doi:10.1016/j.biomaterials.2007.09.003
Carter DR, Beaupre GS, Giori NJ, Helms JA (1998) Mechanobiology of skeletal regeneration. Clin Orthop Relat Res 355(Suppl): S41–S55. doi:10.1097/00003086-199810001-00006
Carter DR, Blenman PR, Beaupre GS (1988) Correlations between mechanical stress history and tissue differentiation in initial fracture healing. J Orthop Res 6(5): 736–748. doi:10.1002/jor.1100060517
Claes LE, Heigele CA (1999) Magnitudes of local stress and strain along bony surfaces predict the course and type of fracture healing. J Biomech 32(3): 255–266. doi:10.1016/S0021-9290(98)00153-5
Cullinane DM, Fredrick A, Eisenberg SR, Pacicca D, Elman MV, Lee C, Salisbury K, Gerstenfeld LC, Einhorn TA (2002) Induction of a neoarthrosis by precisely controlled motion in an experimental mid-femoral defect. J Orthop Res 20(3): 579–586. doi:10.1016/S0736-0266(01)00131-0
Cullinane DM, Salisbury KT, Alkhiary Y, Eisenberg S, Gerstenfeld L, Einhorn TA (2003) Effects of the local mechanical environment on vertebrate tissue differentiation during repair: does repair recapitulate development?. J Exp Biol 206(Pt 14): 2459–2471. doi:10.1242/jeb.00453
Davisson T, Kunig S, Chen A, Sah R, Ratcliffe A (2002a) Static dynamic compression modulate matrix metabolism in tissue engineered cartilage. J Orthop Res 20: 842–848. doi:10.1016/S0736-0266(01)00160-7
Davisson T, Sah RL, Ratcliffe A (2002b) Perfusion increases cell content and matrix synthesis in chondrocyte three-dimensional cultures. Tissue Eng 8(5): 807–816. doi:10.1089/10763270260424169
Epari DR, Taylor WR, Heller M, Duda GN (2006) Mechanical conditions in the initial phase of bone healing. Clin Biomech (Bristol, Avon) 21: 646–655. doi:10.1016/j.clinbiomech.2006.01.003
Gerstenfeld LC, Alkhiary YM, Krall EA, Nicholls FH, Stapleton SN, Fitch JL, Bauer M, Kayal R, Graves DT, Jepsen KJ et al (2006) Three-dimensional reconstruction of fracture callus morphogenesis. J Histochem Cytochem 54(11): 1215–1228. doi:10.1369/jhc.6A6959.2006
Goodship AE, Kenwright J (1985) The influence of induced micromovement upon the healing of experimental tibial fractures. J Bone Joint Surg Br 67(4): 650–655
Hente R, Fuchtmeier B, Schlegel U, Ernstberger A, Perren SM (2004) The influence of cyclic compression and distraction on the healing of experimental tibial fractures. J Orthop Res 22: 709–715. doi:10.1016/j.orthres.2003.11.007
Hung CT, Mauck RL, Wang CC, Lima EG, Ateshian GA (2004) A paradigm for functional tissue engineering of articular cartilage via applied physiologic deformational loading. Ann Biomed Eng 32(1): 35–49. doi:10.1023/B:ABME.0000007789.99565.42
Isaksson H, Comas O, van Donkelaar CC, Mediavilla J, Wilson W, Huiskes R, Ito K (2007) Bone regeneration during distraction osteogenesis: mechano-regulation by shear strain and fluid velocity. J Biomech 40(9): 2002–2011. doi:10.1016/j.jbiomech.2006.09.028
Isaksson H, van Donkelaar CC, Huiskes R, Ito K (2006a) Corroboration of mechanoregulatory algorithms for tissue differentiation during fracture healing: Comparison with in vivo results. J Orthop Res 24(5): 898–907. doi:10.1002/jor.20118
Isaksson H, Wilson W, van Donkelaar CC, Huiskes R, Ito K (2006b) Comparison of biophysical stimuli for mechano-regulation of tissue differentiation during fracture healing. J Biomech 39(8): 1507–1516. doi:10.1016/j.jbiomech.2005.01.037
Kelly DJ, Prendergast PJ (2005) Mechano-regulation of stem cell differentiation and tissue regeneration in osteochondral defects. J Biomech 38(7): 1413–1422. doi:10.1016/j.jbiomech.2004.06.026
Kelly DJ, Prendergast PJ (2006) Prediction of the optimal mechanical properties for a scaffold used in osteochondral defect repair. Tissue Eng 12(9): 2509–2519. doi:10.1089/ten.2006.12.2509
Knippenberg M, Helder MN, Doulabi BZ, Semeins CM, Wuisman PIJM, Klein-Nulend J (2005) Adipose tissue-derived mesenchymal stem cells acquire bone cell-like responsiveness to fluid shear stress on osteogenic stimulation. Tissue Eng 11(11/12): 1780–1788. doi:10.1089/ten.2005.11.1780
Lacroix D, Chateau A, Ginebra MP, Planell JA (2006) Micro-finite element models of bone tissue-engineering scaffolds. Biomaterials 27(30): 5326–5334. doi:10.1016/j.biomaterials.2006.06.009
Lacroix D, Prendergast PJ (2000) A homogenization procedure to prevent numerical instabilities in poroelastic tissue differentiation models. In: Eighth annual symposium: computational methods in orthopaedic biomechanics
Lacroix D, Prendergast PJ (2002) A mechano-regulation model for tissue differentiation during fracture healing: analysis of gap size and loading. J Biomech 35(9): 1163–1171. doi:10.1016/S0021-9290(02)00086-6
Mauck RL, Byers BA, Yuan X, Tuan RS (2007) Regulation of cartilaginous ECM gene transcription by chondrocytes and MSCs in 3D culture in response to dynamic loading. Biomech Model Mechanobiol 6: 113–125. doi:10.1007/s10237-006-0042-1
McKibbin B (1978) The biology of fracture healing in long bones. J Bone Joint Surg Br 60-B(2): 150–162
Perren M, Cordey J (1980) The concept of interfragmentary strain. In: Uhthoff HK(eds) Current concepts of internal fixation of fractures. Springer, Berlin, pp 63–77
Prendergast PJ, Huiskes R, Soballe K (1997) ESB Research Award 1996. Biophysical stimuli on cells during tissue differentiation at implant interfaces. J Biomech 30(6): 539–548. doi:10.1016/S0021-9290(96)00140-6
Riddle RC, Taylor AF, Genetos DC, Donahue HJ (2006) MAP kinase and calcium signaling mediate fluid flow-induced human mesenchymal stem cell proliferation. Am J Physiol Cell Physiol 290: C776–C784. doi:10.1152/ajpcell.00082.2005
Sah RL, Kim YJ, Doong JY, Grodzinsky AJ, Plaas AH, Sandy JD (1989) Biosynthetic response of cartilage explants to dynamic compression. J Orthop Res 7(5): 619–636. doi:10.1002/jor.1100070502
Salisbury KT, Einhorn TA, Gerstenfeld LC, Morgan EF (2005) Mechanobiological regulation of molecular expression and tissue differentiation during bone healing. Vail, Colorado. Compendex, pp 1530–1531
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hayward, L.N.M., Morgan, E.F. Assessment of a mechano-regulation theory of skeletal tissue differentiation in an in vivo model of mechanically induced cartilage formation. Biomech Model Mechanobiol 8, 447–455 (2009). https://doi.org/10.1007/s10237-009-0148-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-009-0148-3