Abstract
The objective of this study is to evaluate the influence of the addition of garden waste (GW) on the performance of food waste (FW) anaerobic digestion. In this regard, a bench scale experiment was performed via biochemical methane potential test. The reactors were composed of flasks with a total volume of 310 mL and a useable volume of 120 mL, containing the waste mixtures in the different studied conditions. All reactors were incubated under 36 °C. A factorial design was used to evaluate the influence of three factors on methane production: inoculum and substrate ratio (I:S), volatile solid (VS) content of the mixture and type of substrate. The results indicated that the condition referred to an I:S ratio of 1:1, higher VS content (VSmix) and only FW as a substrate achieved the higher methane production and differed significantly from the others. This combination resulted in 67.5% of VS removal, 509 NmL of cumulative CH4 and a specific methane yield of 159 NmLCH4 gVS−1. The results obtained in this research indicated that the addition of GW on the anaerobic digestion of FW did not positively influence the methane production.
Graphic abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anaerobic digestion (AD) is an efficient alternative to organic waste treatment. In the past few years, this technology has gained especial attention as it recovers energy in form of biogas, which can be used to energetic purposes [1, 2]. Currently, the technical and scientific community are focused on establishing new process optimization techniques to treat different sources of organic wastes.
Food waste represents a large share of municipal solid waste and it is composed by a high organic matter content. As a result of its biodegradation products, the improper disposal of this type of waste leads to multiple environmental impacts. Besides, the large quantity of organic waste disposed in landfills decreases, as a consequence, their lifespan [1, 3].
Anaerobic digestion of food waste has been applied in a few countries, achieving good results regarding methane production. However, some adversities are still found when treating food waste, especially related to system stability [4]. The anaerobic digestion of residues that are consisted of low cellulose content is limited by methanogenesis, as the great amount of easily degradable organic matter leads to a high production of volatile fatty acids (VFA), which can inhibit the further methane conversion [5].
Co-digestion of food waste with lignocellulosic waste—such as garden (leaves and branches) and agriculture waste (different culture straws, grain residues)—may lead to process optimization. The combination of these residues improves material properties, resulting, for instance, in a better C:N ratio, which enhances the biological treatment performance [5,6,7,8,9].
The biochemical methane potential (BMP) test is a widely used procedure that evaluates the biogas production from the biodegradation of a given type of waste. This procedure supports the study of operational conditions optimization, aiming at a further application of the process in larger scales [10]. The inoculum and substrate ratio (I:S) is an important factor in anaerobic digestion mainly due to the introduction of a balanced microbial population in the anaerobic system [11]. The range of I:S generally used in studies, ranges from 4:1 to 1:2. The decrease in the I:S ratio may cause accumulation of fatty acids and high ammonia concentrations in the reactor, and consequently lower methane production [7, 12]. Furthermore, different residues require different proportions due to their particular characteristics.
In this context, the objective of this research is to evaluate the methane production potential from food waste anaerobic digestion, and also to verify if the co-digestion of food waste with garden waste enhances methane production.
Material and methods
Substrate and inoculum
The inoculum consisted of sludge provided from an anaerobic reactor, which is part of a Wastewater Treatment Plant (WWTP). Food waste (FW) refers to the post-consumption leftovers obtained from the meals served in the university restaurant of the State University of Londrina, Paraná, Brazil. The FW was removed from the food trays by water jet and it was subsequently shredded, sieved (mesh size of 5 mm), homogenized, stored and frozen to −18 °C. The garden waste (GW) consisted of grass (paspalum notatum) clippings collected from the university’s lawn mowing. After collection, GW was left for five days under a covered area to dry naturally and it was posteriorly submitted to milling, a physical pre-treatment, in a knife mill with a mesh size of 1.19 mm.
Experimental system
The bench scale reactors were consisted of borosilicate glass flasks. Each reactor had a total volume of 310 mL, a useable volume of 120 mL and a lid containing two outlets. One outlet was connected to a manometer to measure the pressure within the reactors from the produced biogas. The maximum pressure reading was 3 kgf cm−2 (in a scale of 0.05 kgf cm−2). In the second outlet, a brass needle valve was attached and used to purge the gas to further perform its quantitative analysis.
Biochemical methane potential test
The experiment was conducted using a two-level factorial design for three factors (23 design) to evaluate the factors: substrate (FW and FW + GW); inoculum and substrate ratio—I:S (1:1 and 2:1) and the volatile solid content—VS (VSmix. and VSmix./2). The I:S factors selected for this experiment were those that resulted in the best methane production performance when previously tested in an experiment conducted with different ratios. In the conditions with the addition of GW, 20% of the OLR of the FW was replaced by GW. In regard to VS content, the values were related to the VS obtained from the mixture of inoculum and substrate (VSmix.), and half of this value after dilution with water (VSmix./2). Additional control experiments using just inoculum as substrate was also performed. In this case, the total volume of CH4 produced has to be below 20% of the produced volume in both treatments [10].
All treatments were performed in triplicate. The combinations of the studied factors are presented in Table 1. After the preparation of inoculum and substrate mixtures in the different conditions, sodium carbonate buffer was added in all samples until the buffer concentration in each mixture reached 2000 mg L−1. The flasks were hermetically closed, incubated in an oven under mesophilic temperature (36 ± 1 °C) and stirred manually on a daily basis. Initially, the experiment duration was determined as the period until the daily biogas production reached less than 1% of the cumulative production for each condition [10, 13]. However, during the experiment, some of the reactors presented low production periods, followed by its progressive increase. As a result, the monitoring period was extended, totalizing 70 days.
The volume of produced biogas and the concentration of CH4, CO2 and H2S were monitored throughout the experiment. The purges were carried out when the pressure exerted by the gas in the manometer reached between 0.75 and 1.10 kgf cm−2 by coupling a hosepipe between the biogas outlet valve and the inlet of the portable gas analyzer (Dräger® X-am 7000). The physicochemical analyses of the initial and final samples from the BMP test were also performed for pH, alkalinity, TS, and VS parameters. These analyses, related to the monitoring of the reactors and also to the waste characterization were based in the procedures established on the Standard Methods for the Examination of Water and Wastewater [14].
Biogas measurements
The pressure exerted in the manometers was daily recorded and posteriorly converted into daily volume of produced biogas (between current and previous day), based on the ideal gas law and Clapeyron equation, according to Eq. 1 [15, 16]:
where Vol. between T and (T + 1) is the volume of gas produced in a time interval (mL), T is time (days), PF is flask pressure (mbar), VUF is useful volume of flask filled with gas (L), C is molar volume of an ideal gas (22.41 L mol−1), R is the ideal gas constant (83.14 L mbar mol−1 K−1), TF is the flask temperature (K).
Water vapor is a biogas component and overestimates its volume in a range of 2% to 8%, in normal temperature and pressure (NTP) conditions [17]. The pressure is a function of the temperature, so for a given temperature of 36 °C, the pressure resulted in 59.3 mbar.
The volume of produced biogas data was also adjusted to the NTP, using the local atmospheric pressure, according to Eq. 2:
where VolNTP is the volume of cumulative biogas in the period adjusted to NTP (NmL), Volcum is the volume of cumulative biogas in the period (mL), TNTP is the normal temperature (237.15 K), TF is the flask temperature in the incubation environment (K), Patm is the local atmospheric pressure in the day of the measurement (mbar), Pw is the water vapor pressure (59.3 mbar) and Po is the normal atmospheric pressure (1.013 mbar).
Statistical analysis
The statistical analysis was carried out in the Software R [18]. The goodness of fit of the models’ equation was expressed by the adjusted coefficient of determination \(R_{{{\text{adjusted}}}}^{2}\) and its statistical significance was evaluated with the F test. The assumptions homogeneity of variances, normality and independence of errors were evaluated by the tests [19,20,21] respectively, to validate the model. The averages were compared utilizing the Tukey test. The significance level of the analyses was 5%.
Results and discussion
Characterization of the inoculum and substrates
Table 2 presents the data obtained from the characterization of the inoculum and the substrates (FW and GW). The VS/TS content of FW achieved 93.9%, demonstrating its biodegradation potential due to the high presence of organic matter. In this study, FW had a pH of 4.37 and did not contained alkalinity from carbonates and bicarbonates. Other studies also mentioned these properties in FW [22, 23]. The pH of the inoculum close to neutrality (6.8) and its high alkalinity (2.3 g L−1) contributed to the FW treatment, since the latter presented acidic characteristics.
The values of carbohydrate (38%), lipid (12%) and protein (49%) content in FW are related to the waste composition. A high percentage of proteins enhances the biogas production. However, it also results in ammoniacal nitrogen and H2S formation [24]. Also important is the low C:N ratio of FW (6.1) as a consequence of the high nitrogen content (7.8%). The ideal C:N ratio for anaerobic digestion is situated around 20 to 30, so the low ratio can be prejudicial to the process performance. Thus, the addition of GW as a substrate, which presented a C:N ratio of 30, beneficiates the treatment by balancing these two nutrients.
The characterization of the GW also resulted in a high organic matter content, indicated by a VS/TS of 88.4%. Nevertheless, the organic fraction of GW is distinguished from the FW due to the presence of lignin, cellulose, and hemicellulose in elevated concentrations. The content of cellulose (35%), hemicellulose (16%) and lignin (29%) are similar to another study, with the residue of grass digestion and characterized by containing 30.3% of cellulose, 25.7% of hemicellulose and 22.9% of lignin [5].
Operational parameters
Table 3 shows the average values of pH and alkalinity at the beginning and at the end of the BMP experiment. Alkalinity and pH are two related parameters. Thus, the initial alkalinity is recommended to be within an adequate range to ensure process stability, since the formation of organic acids during the process consumes the alkalinity into bicarbonate, decreasing, consequently, the pH. Once the initial pH in this study resulted in values between 6.0 and 6.7, it was chosen to add the buffer sodium carbonate to ensure that the pH would stay within the adequate range throughout the process. As a result, the initial values of alkalinity remained between 1655 and 4492 mg L−1. These concentrations belong to the indicated range observed in literature (from 1000 to 5000 mg L−1) [24].
After the buffer addition, the initial pH values extrapolated the maximum indicated pH, reaching from 8.6 to 9.6. Still, at the end of the experiment, they decreased and prevailed close to neutrality, between 7.2 and 7.8. So, the elevated pH at the beginning of the experiments might have inhibited the acidification during the anaerobic process.
Performance parameters
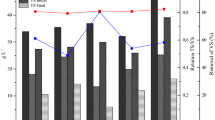
The average values of total solids (TS), volatile solids (VS) and VS removal efficiency, related to the initial and final samples of the BMP experiment are shown in Table 4. The TS of the samples were related to previously defined conditions, thus, the TS of A2, A4, B2 and B4 resulted in approximately half of the A1, A3, B1 and B3 values, respectively. The TS comprises both VS and fixed solids (FS), therefore, for the treatment efficiency evaluation, it is important to also consider the VS content, which represents the organic matter fraction of the sample.
In general, A-treatments (only with FW as substrate) resulted in higher VS removal efficiencies than B-treatments. A2 demonstrated the highest performance, with a removal of 67.5%. The low removal efficiencies of the treatments in which GW was present might be related to the difficulty in biodegradation of some specific GW material, like lignin [25].
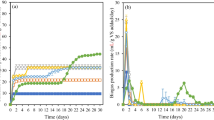
Figure 1 displays the results of the daily biogas production rate, the cumulative biogas production, and the methane content of each condition during the experiment. The daily biogas production rate curves (Fig. 1a) demonstrated an elevated biogas production in the first days of the experiment. This fact is related to the high availability of both organic matter and microorganisms within the system, which induced the first steps of anaerobic digestion, converted the compounds to be utilized in methanogenesis, and consequently produced biogas. After the conversion of the easily biodegradable compounds, the biogas production decreased as the remaining compounds were more difficult to biodegrade. The maximum production rates reached 251 and 160 NmL day−1 in treatments A1 and B1, respectively, both in the first day of experiment.
The duration of the biodegradation depends on the type of substrate used and on the operational conditions. Generally, the biogas production stabilizes after 30 monitoring days, as obtained by other researchers [5, 7]. In this study, the production lasted longer, and this fact might be related to the elevated initial pH.
The cumulative biogas production curves (Fig. 1b) showed that in A1 the production stabilized from the 5th to the 25th day and then increased again. This fact may have occurred due to the presence of some compounds of greater difficulty in degradation, which may have been degraded at different times. B1 resulted in the higher cumulative production until the 60th monitoring day. However, at this point, a considerable increase in A1 occurred, reaching at the end the highest cumulative biogas production. Both treatments obtained the greater values related to cumulative biogas production, with 1082 NmL in A1 and 931 NmL in B1.
Each condition resulted in different biogas production levels, which might be related to factors like the elevated initial pH and substrate composition. After the beginning of the biodegradation, probably from hydrolysis and acidogenesis steps, pH dropped and reached a value closer to ideal for the occurrence of further methanogenesis. This may be possibly related to the fact that this experiment produced biogas for a longer period. Furthermore, the components that were harder to degrade might have been degraded in several stages, leading to different production levels.
Regarding methane concentration in the biogas (Fig. 1c), its maximum value did not occur simultaneously with the maximum biogas production period. In the beginning of the experiment the biogas generated is mainly a result of the first steps of anaerobic digestion and it is composed of a low methane content. The higher concentrations of methane were obtained from 30th day of experiment onwards, varying according to the conditions. A1 and A3 presented the higher methane content of the experiment, reaching 71 and 68%, respectively.
In Table 5 it is presented for each studied condition the average concentrations of CH4, CO2 and H2S, the cumulative biogas volume and the specific methane yield (SMY). The average methane concentration values were situated within the expected range for organic waste anaerobic digestion [24] treatment A1 resulted in the highest content of CH4, as well as of CO2 and H2S. That can be related to the greater organic matter decomposition for this condition and also to the generation of the three gases in the different steps of anaerobic digestion.
The specific methane yield is represented by the total volume of produced methane as a function of the added VS mass. The highest values were obtained in treatment A1 and A2, both containing only FW and with an I:S ratio of 1:1. The treatments that GW was added to the substrate resulted in a lower SMY, which can be related not only to the higher initial VS content, but also because the substrate was harder to degrade since the structure of the lignocellulosic biomass is responsible for creating a natural obstacle against the activity of microorganisms. It is noteworthy that the chemical and morphological characteristics of this type of substrate are one of the main difficulties in converting lignocellulosic materials into biofuels or other products of interest [25].
Comparing these results with the ones achieved [6], both presented similarities regarding SMY. The authors obtained a SMY improvement when food waste was added to garden waste anaerobic digestion. In their case, a replacement of 20% of the garden waste by food waste resulted in a higher methane production, with a specific methane yield of 120 LCH4 kgVS−1 added to the reactor. Previous authors [5] achieved a specific production of 279 LCH4 kgVS−1 when grass was co-digested with food waste (proportion of 2.5:1.5 in gVS).
As a consequence of its high calorific value, methane is the gas with the greatest interest to energy recovery purposes. Therefore, the statistical analysis was performed based on the results of its cumulative production. Methane production was evaluated by the factorial design 23, in which the factors substrate, I:S ratio and VS content were varied. The assumptions for the analysis of variance (ANOVA) were satisfied, as the proposed model attended to the assumptions of normality (Shapiro–Wilk, p value > 0.05), homogeneity of variance (Breusch-Pagan, p value > 0.05) of the residues and independence of errors (Durbin-Watson, p value > 0.05). The ANOVA demonstrated that substrate, I:S, VS and their interactions—except the interaction between substrate and I:S—presented a significative effect in methane production (Table 6).
Considering the significance of the three-way interaction, the factor substrate is dependent on the levels of the I:S and VS content factors. So, Turkey test was performed to assess the effect of each factor when the other two factor levels of this study were fixed, as presented in Tables 7, 8, 9. Table 7 shows the effects of the variance on I:S when substrate and VS content factors were fixed.
The condition VSmix./2 with FW + GW was the only that presented no significant difference, when comparing 1:1 and 2:1 ratio. In the other studied conditions, there was statistical difference between the proportions. The higher cumulative methane volumes were obtained in the inoculum and substrate proportions of 1:1.
In Table 8 it is shown the effects of VS content variation when substrate and I:S ratio factors are fixed. In all levels of substrate and I:S, the higher methane production volumes were obtained when VS content were also increased (VSmix.). Also, VSmix. was statistically different from VSmix./2 in the 5% of significance level.
Lastly, the results on Table 9 are referred to the effects on the substrate factor when I:S and VS content factors are fixed. There was significant difference between treatments with FW and FW + GW only when VS content was higher (VSmix.), for both 1:1 and 2:1 ratio. In the condition of VSmix./2 there was no significant difference between the two substrates.
The combination 1:1 and VSmix. resulted in the greatest methane production and it differed significantly from the others. Also, within this combination, there was significant difference between only FW as a substrate and FW + GW. The condition with an I:S of 1:1, VSmix. and only FW as a substrate obtained the higher cumulative methane production (509 NmL), which varied when substrate changed to FW + GW, resulting in 437 NmL of produced methane. In this way, the BMP experiment for the studied conditions revealed that the addition of GW in the substrate had no positive influence on methane production, if compared to the higher production conditions.
Conclusions
When evaluating the studied conditions of the biochemical methane potential test, it was observed by the factorial design that the inoculum and substrate ratio (I:S) of 1:1, the VS content of the mixture (VSmix.) and the food waste as the only substrate (FW) resulted in higher cumulative methane production. Treatment A1 obtained the greatest methane production and had statistically differed from all other treatments, achieving a cumulative CH4 production of 509 NmL and a specific methane yield of 159 NmL gVS−1. Co-digestion of food waste with garden waste (FW + GW) presented a significant reduction in methane production, when applying a high VS content condition. This reduction might be related to the greater difficulty in degrading lignocellulosic compounds. As a suggestion for future studies, it would be interesting to study different types of pre-treatment of GW aiming to facilitate its biodegradation and consequently methane production.
Availability of data and material
Not available.
References
Yong Z, Dong Y, Zhang X, Tan T (2015) Anaerobic co-digestion of food waste and straw for biogas production. Renew Energ 78:527–530
Ren Y, Yu M, Wu C, Wang Q, Gao M, Huang Q, Liu Y (2018) A comprehensive review on food waste anaerobic digestion: research updates and tendencies. Bioresour Technol 247:1069–1076
Woon KS, Lo IMC (2016) A proposed framework of food waste collection and recycling for renewable biogas fuel production in Honk Kong. Waste Manage 47:3–10
Ye M, Liu J, Ma C, Li YY, Zou P, Qian G, Xu Z (2018) Improving the stability and efficiency of anaerobic digestion of food waste using additives: a critical review. J Clean Prod 192:316–326
David A, Govil T, Tripathi AK, Mcgeary J, Farrar K, Sani RK (2018) Thermophilic anaerobic digestion: enhanced and sustainable methane production from co-digestion of food and lignocellulosic wastes. Energies 11:1–13
Brown D, Li Y (2013) Solid state anaerobic co-digestion of yard waste and food waste for biogas production. Bioresour Technol 127:275–280
Panigrahi S, Sharma HB, Dubey BK (2020) Anaerobic co-digestion of food waste with pretreated yard waste: a comparative study of methane production, kinetic modeling and energy balance. J Clean Prod 243:1–9
Perin JKH, Borth PLB, Torrecilhas AR, Cunha LS, Kuroda EK, Fernandes F (2020) Optimization of methane production parameters during anaerobic co-digestion of food waste and garden waste. J Clean Prod 272:1–7
Uma S, Thalla AK, Devatha CP (2020) Co-digestion of food waste and switchgrass for biogas potential: effects of process parameters. Waste Biomass Valoriz 11:827–839
Holliger C, Alves M, Andrade D, Angelidaki I, Astals S, Baier U, Bougrier C et al (2016) Towards a standardization of biomethane potential tests. Water Sci Technol 74:2515–2522
Haider M, Zeshan R, Yousaf S, Malik RN, Visvanathan C (2015) Effect of mixing ratio of food waste and rice husk co-digestion and substrate to inoculum ratio on biogas production. Bioresour Technol 190:451–457
Latifi P, Karrabi M, Danesh S (2019) Anaerobic co-digestion of poultry slaughterhouse wastes with sewage sludge in batch-mode bioreactors (effect of inoculum-substrate ratio and total solids). Renew Sust Energ Rev 107:288–296
Edwiges T, Frare L, Mayer B, Lins L, Triolo JM, Flotats X, Costa MSSM (2018) Influence of chemical composition on biochemical methane potential of fruit and vegetable waste. Waste Manage 71:618–625
APHA, AWWA, WEF (2017) Standard methods for examination of water and wastewater, 23 edn. American Public Health Association, Washington, 1546 p
El-Mashad HM, Zhang R (2010) Biogas production from co-digestion of dairy manure and food waste. Bioresour Technol 101:4021–4028
Labatut RA, Angenent LT, Scott NR (2011) Biochemical methane potential and biodegradability of complex organic substrates. Bioresour Technol 102:2255–2264
Strömberg S, Nistor M, Liu J (2014) Towards eliminating systematic errors caused by the experimental conditions in biochemical methane potential (BMP) tests. Waste Manage 34:1939–1948
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Breusch TS, Pagan AR (1979) A simple test for heteroscedasticity and random coefficient variation. Econometrica 47:1287–1294
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrica 52:591–611
Durbin J, Watson GS (1951) Testing for serial correlation in least squares regression. II. Biometrika 38:159–177
Kuczman O, Gueri MVD, De Souza SNM, Schirmer WN, Alves HJ, Secco D, Buratto WG, Ribeiro CB, Hernandes FB (2018) Food waste anaerobic digestion of a popular restaurant in Southern Brazil. J Clean Prod 196:382–389
Strazzera G, Battista F, Garcia NH, Frison N, Bolzonella D (2018) Volatile fatty acids production from food wastes for biorefinery platforms: a review. J Environ Manage 226:78–288
Deublein D, Steinhauser A (2008) Biogas from waste and renewable resources: an introduction. Wiley-Vch Verlag Gmbh & Co. KgaA, Weinheim
Wu M, Yan ZY, Zhang XM, Xu FX, Sun RC (2016) Integration of mild acid hydrolysis in γ-valerolactone/water system for enhancement of enzymatic saccharification from cotton stalk. Bioresour Technol 200:23–28
Acknowledgements
This research was financed by institutions Fundação Araucária (Paraná/Brazil), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES (Brazil), Companhia Paranaense de Energia—COPEL (Paraná/Brazil) and Agência Nacional de Energia Elétrica -ANEEL/technical and scientific cooperation contract nº.4600013405 (Brazil).
Funding
This research was financed by institutions Fundação Araucária (Paraná/Brazil), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES (Brazil), Companhia Paranaense de Energia—COPEL (Paraná/Brazil) and Agência Nacional de Energia Elétrica—ANEEL/technical and scientific cooperation contract nº.4600013405 (Brazil).
Author information
Authors and Affiliations
Contributions
Priscila Liane Biesdorf Borth: Methodology, Validation, Investigation, Data curation, Writing—original draft, Writing – review & editing, Visualization. Jessica Klarosk Helenas Perin: Methodology, Validation, Investigation, Data curation, Writing—original draft. Arthur Ribeiro Torrecilhas: Methodology, Validation, Investigation, Data curation. Nicole Caldas Pan: Software, Formal analysis. Emília Kiyomi Kuroda: Conceptualization, Methodology, Resources, Supervision, Project administration, Funding acquisition. Fernando Fernandes: Conceptualization, Methodology, Resources, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Consent to participate
All authors agree with its submission.
Consent for publication
All authors agree with this work publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Borth, P.L.B., Perin, J.K.H., Torrecilhas, A.R. et al. Biochemical methane potential of food and garden waste co-digestion with variation in solid content and inoculum:substrate ratio. J Mater Cycles Waste Manag 23, 1974–1983 (2021). https://doi.org/10.1007/s10163-021-01270-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-021-01270-z