Abstract
It is well known that surgery is the mainstay treatment for duodenal adenocarcinoma. However, the optimal extent of surgery is still under debate. We aimed to systematically review and perform a meta-analysis of limited resection (LR) and pancreatoduodenectomy for patients with duodenal adenocarcinoma. A systematic electronic database search of the literature was performed using PubMed and the Cochrane Library. All studies comparing LR and pancreatoduodenectomy for patients with duodenal adenocarcinoma were selected. Long-term overall survival was considered as the primary outcome, and perioperative morbidity and mortality as the secondary outcomes. Fifteen studies with a total of 3166 patients were analyzed; 995 and 1498 patients were treated with limited resection and pancreatoduodenectomy, respectively. Eight and 7 studies scored a low and intermediate risk of publication bias, respectively. The LR group had a more favorable result than the pancreatoduodenectomy group in overall morbidity (odd ratio [OR]: 0.33, 95% confidence interval [CI] 0.17–0.65) and postoperative pancreatic fistula (OR: 0.13, 95% CI 0.04–0.43). Mortality (OR: 0.96, 95% CI 0.70–1.33) and overall survival (OR: 0.61, 95% CI 0.33–1.13) were not significantly different between the two groups, although comparison of the two groups stratified by prognostic factors, such as T categories, was not possible due to a lack of detailed data. LR showed long-term outcomes equivalent to those of pancreatoduodenectomy, while the perioperative morbidity rates were lower. LR could be an option for selected duodenal adenocarcinoma patients with appropriate location or depth of invasion, although further studies are required.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Duodenal adenocarcinoma is a rare tumor that accounts for less than 0.5% of all cases of gastrointestinal cancer. Nevertheless, the duodenum is the most common location for adenocarcinoma of the small bowel, followed by the jejunum and the ileum [1,2,3,4,5]. Duodenal adenocarcinoma has a reported 5-year survival rate for resected tumors of 25–75% [6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23], which is significantly better than that of other periampullary cancers. Surgery is the mainstay treatment for duodenal adenocarcinoma and several surgical strategies, including endoscopic resection, partial (wedge) resection, segmental (limited) resection, and pancreaticoduodenectomy (PD), are used to treat duodenal adenocarcinoma depending on the location or the depth of tumor invasion. Although PD enables a more extensive removal of the regional lymph nodes, it is often associated with a higher incidence of postoperative morbidity and mortality when compared with segmental resection [24]. Partial (wedge) resection and segmental resection are usually indicated for select tumors with no risk of lymph node metastasis but are not suitable for endoscopic resection [25, 26]. Even though the incidence of such tumors has increased because of the advances in endoscopic and radiologic diagnostic modalities, the optimal extent of surgery for duodenal adenocarcinoma remains an issue for debate.
The aim of this study was to perform a systematic review and meta-analysis of studies that compared limited resection (LR) with PD for patients with primary duodenal cancer, with long-term overall survival as the primary outcome and perioperative morbidity and mortality as the secondary outcomes.
Materials and methods
The PRISMA [27] checklist for systematic reviews and meta-analyses were followed to conduct this study.
Information resources
An electronic database search was performed to identify published studies from inception to January 2020 using PubMed and the Cochrane Library.
Search strategy
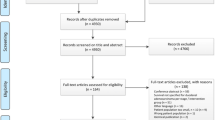
Both free text and MESH terms were used for searching relevant studies. The keywords used were ‘duodenal neoplasm’, ‘duodenal cancer’, and ‘duodenal adenocarcinoma’ combined with ‘pancreatoduodenectomy’, ‘pancreaticoduodenectomy’, ‘duodenal neoplasms/surgery’, ‘duodenopancreatectomy’, ‘partial resection’, ‘segmental resection’, ‘limited resection’, and ‘simple resection’ using the Boolean operator ‘AND’ (Fig. 1).
Inclusion criteria
To be included in the analysis, the studies had to include:
-
1.
A comparison between LR and PD
-
2.
Primary adenocarcinoma of the duodenum at diagnosis (clearly documented)
-
3.
Adverse early postoperative events including 30-day postoperative mortality, overall postoperative complications, such as procedure related complications (anastomosis leakage, pancreatic fistula, biliary fistula, fluid collection, and abscess) and general complications (wound infection, ileus, acute renal failure, urinary tract infections, delirium, and respiratory, and cardiovascular complications)
-
4.
Long-term outcomes including overall survival
Exclusion criteria
Studies were excluded from the analysis if: (a) the outcomes of interest were not clearly reported; (b) it was impossible to extract or calculate the necessary data from the published results; and (c) the sample size was ≤ 8.
Data extraction
Five reviewers (P.B., R.H., S.N, S.K, and H.E) independently extracted the following data variables from each study: first author, country, year of publication, study population characteristics including age and gender, study design, inclusion and exclusion criteria, length of follow-up, and number of subjects undergoing LR and PD. There was 100% agreement on the collected data among the five reviewers.
Statistical analysis
Meta-analysis was conducted by the Review Manager (RevMan) Meta-analysis software, v. 5.1.6. The 95% confidence intervals (CIs) were calculated as estimates of precision for odds ratio (OR). The statistical tests were two-sided, and P < 0.05 was considered statistically significant. The dichotomous variables were analyzed based on ORs with 95% CI. For the outcomes being considered, the reference categories were selected such that an OR < 1 favored the LR approach. The studies were weighted in the meta-analysis by the inverse variances of their effect estimates, that is, the validities of the included studies. Heterogeneity was considered not statistically significant when the Cochrane Q test P value was > 0.1. The I2 statistic, a transformation of the Q test, was used to assess the consistency of the effect sizes. In case of heterogeneity, meta-analysis was performed using the random-effects model. In addition, an I2 value of less than 25% was defined as low heterogeneity, a value between 25 and 50% was defined as moderate heterogeneity, and a value of > 50% was defined as high heterogeneity [28]. In cases of I2 values less than 30%, fixed effects models were used throughout. Analysis of long-term survival was performed by obtaining the numbers at risk and combined with either the quoted survival rates or the values read from enlarged plots of the Kaplan–Meier curves to produce the estimates. Where numbers at risk were not quoted, constant censoring over the period of follow-up was assumed in the estimation. A meta-analysis of long-term survival from the included studies using hazard ratios could not be performed because only one study (Cloyd et al.) [19] reported the hazard ratio between the LR and PD groups. The other included studies may have had an insufficient number of patients to perform a multivariate analysis. Funnel plots with an Egger test were used to screen for publication bias.
Assessment of study quality
The methodological quality of all the included studies was assessed with the validated Newcastle–Ottawa scale [29]. Studies scoring > 6 were considered to be high quality and the maximum score was 9.
Definition
LR was defined as one of following surgical procedures: transduodenal excision [30], partial/full thickness resection (wedge resection) or pancreas-sparing segmental duodenectomy (PSD) [31,32,33,34,35]. PD (Whipple’s procedure) [36] is the standard procedure to treat periampullary tumors and other benign disorders that require removal of the distal bile duct, the duodenum (or some parts of the stomach), and the head of the pancreas. Morbidity was classified using the Clavien–Demartines–Dindo system [37]. Postoperative pancreatic fistula (POPF), post pancreatectomy hemorrhage (PPH), and delayed gastric emptying time (DGE) were defined according to the International Study Group (ISGPF) criteria [38,39,40]. Postoperative mortality was defined as death within 30 days of surgery from any cause.
Results
Selection of trials and patient’s characteristics
Fourteen studies were retrospective cohort studies and one was a prospective cohort study. No randomized controlled trial was found in our search. The Newcastle–Ottawa scale (NOS) was used for quality assessment.
This analysis comprised 15 studies with a total of 3166 patients, of which 995 patients (31.4%) were treated by LR and 1498 patients were treated by PD (Tables 1 and 2). The patient classifications for each surgical procedure according to tumor depth and location are summarized in Supplementary Table 3. Patients who were selected for LR had a higher percentage of tumors in the third and fourth portion of the duodenum (available data from five studies) and a slightly higher percentage of T1-stage tumors (available data from three studies). The indications for LR and classification of LR procedures are summarized in Supplementary Table 4. Most of the indications for LR were tumors located in the third or fourth portion of the duodenum and a T1a/T1b-stage tumor.
Perioperative outcomes
To evaluate the safety of the procedure of the LR group and the PD group, the ORs of postoperative mortality and morbidity were calculated using the data extracted from the 13 included studies, including 951 patients in the LR group and 1340 patients in the PD group.
Morbidity
The meta-analysis showed more a favorable result in the LR group than in the PD group in terms of overall morbidity (OR: 0.33, 95% CI 0.17–0.65) and occurrence of postoperative pancreatic fistula (OR: 0.13, 95% CI 0.04–0.43) (Fig. 2a, b).
a Results of the meta-analysis on overall morbidity after limited resection versus after pancreaticoduodenectomy. b Results of the meta-analysis on POPF after limited resection versus after pancreaticoduodenectomy. LR limited resection, PD pancreaticoduodenectomy, CI confidence interval, POPF postoperative pancreatic fistula
Mortality
The meta-analysis showed no significant difference between the LR group and the PD group in terms of mortality (OR: 0.96, 95% CI 0.70–1.33) (Fig. 3). It should be noted that the patients in the study by Cloyd et al. [19] had a markedly higher weight than other studies; therefore, we performed a sensitivity analysis after excluding the data from the Cloyd study, which showed that the LR group had a more favorable outcome compared to the PD group in terms of mortality (OR: 0.39, 95% CI: 0.16–0.94) (Supplementary Fig. 5).
Long-term outcomes
Overall survival was calculated using the data extracted from Kaplan–Meier curves from eight studies including 855 patients in the LR group and 1101 patients in the PD group.
The meta-analysis showed no significant difference in overall survival between the LR group and the PD group (OR: 0.61, 95% CI 0.33–1.13; Fig. 4).
Publication bias and quality of the included studies
The results of the test for publication bias are presented in Supplemental Figs. 1–4. A publication bias in term of mortality may exist in this present study (P = 0.05035). The results of the quality assessment test of the included studies are shown in Fig. 5. Eight studies had a low risk of bias and seven studies had an intermediate risk of bias.
Discussion
Negative margin resection with regional lymphadenectomy is the only therapeutic option to cure duodenal adenocarcinoma and should be attempted to achieve long-term survival [12, 41]. Patients’ survival greatly depends on regional lymph node metastases. Patients with regional lymph node metastasis had a significantly shorter survival time than without [3, 13, 14, 16, 20, 41,42,43,44,45,46] and lymph node metastasis was also an independent prognostic factor in many studies [13, 14, 20, 41,42,43,44,45, 47]. On the contrary, T stage, tumor size, and location were not associated with patient outcomes [12, 13, 41, 48,49,50]. Several controversies exist regarding the optimum extent of surgery for regional lymphadenectomy. Several authors [12, 17, 51,52,53,54] recommend PD over LR to treat duodenal adenocarcinoma because it has a greater possibility of removing all lymph node metastases. However, patients treated through PD have higher morbidity and mortality rates than those treated through LR, despite the recent advances in surgical techniques and postoperative care. LR was recommended to treat tumors that are located within the distal part of the duodenum and reportedly resulted in lower morbidity and mortality with satisfactory long-term overall survival [6,7,8, 13,14,15, 55]. However, this approach may result in incomplete regional lymphadenectomy which could affect patient survival [56]. On the other hand, there is another opinion that the number of resected lymph nodes does not differ between LR and PD [14, 47].
This systematic review and meta-analysis compared the two surgical strategies used for the management of patients with duodenal adenocarcinoma. We aimed to find the most appropriate treatment to decrease postoperative morbidity and mortality (as morbidity may cause a lower survival rate [57]), and increase long-term survival. The results showed that the LR group had lesser overall postoperative morbidity and POPF, no significant difference in overall mortality, and comparable long-term overall survival compared to the PD group. However, the results of this meta-analysis may be affected by selection bias, as patients who underwent LR had a higher number of tumors in distal portions of the duodenum and superficial (T1) lesion than the PD group. Therefore, it is a matter of decision-making by the surgeon to choose which patients should undergo LR or PD. Further, when considering the risk of nodal metastasis based on the depth of tumor invasion and the pattern of lymph node metastases, Salera et al. [45] showed that no patients with T1 tumors had lymph node metastasis and the prevalence of pN1 disease was significantly associated with pT (depth of tumor) category. Kato et al. [18] also reported the absence of lymph node metastasis when the tumor was limited to the mucosa or submucosa. Sakamoto et al. [58] and Kato et al. [18] determined the distribution of lymph node metastasis based on tumor location or tumor depth of invasion in the duodenum which included anterior and posterior pancreatoduodenal, infrapyloric, common hepatic, pericholedochal and superior mesenteric arterial, and para-aotic. These findings indicate that tumors located deeper than the mucosa have a greater risk of metastasizing to the regional lymph nodes. Therefore, PD is still the standard treatment in primary duodenal adenocarcinoma and is recommended for all tumors that have invaded to the submucosa to ensure complete regional lymphadenectomy. Additionally, LR can be recommended in select patient with superficial lesions located in suitable locations that allow LR to be safely performed (e.g., locations other than second portion of the duodenum).
The major limitation of this review is that most of the included studies were retrospective with a small sample size. Indeed, no randomized controlled trial was included. Therefore, this study suffered from selection biases and other confounding factors, such as the difference in tumor biology and aggressiveness in each patient, which affected the accuracy of the comparison between the two surgical approaches. Also, due to the lack of detailed data from the included studies in this meta-analysis, a comparison between the two groups stratified by tumor depth (T categories) and tumor location was not possible. According to the Funnel plot for the comparison of mortality for all studies of LR versus PD (Supplementary Fig. 3), although the p value was marginally not significant, this was likely to have a publication bias (test for funnel plot asymmetry: P = 0.05035).
Therefore, the recommendations based on our study’s results may not be fully conclusive, but nonetheless, may still provide a reference for a surgeon’s decision-making to treat primary duodenal carcinoma.
Conclusion
Negative margin surgical resection and regional lymphadenectomy are the only appropriate therapeutic approaches for duodenal adenocarcinomas. Although the standard treatment for duodenal cancer is PD, the long-term outcomes are comparable between LR and PD, with the perioperative morbidity rates being lower for LR, provided negative margins can be ensured and regional lymphadenectomy can be performed. Therefore, LR with dissection of the regional lymph nodes may be a suitable option for some selected patients depending on the tumor location and depth of invasion, although further studies are required.
References
Aparicio T, Zaanan A, Mary F et al (2016) Small Bowel Adenocarcinoma. Gastroenterol Clin North Am 45(3):447–457
Buchbjerg T, Fristrup C, Mortensen MB (2015) The incidence and prognosis of true duodenal carcinomas. Surg Oncol 24(2):110–116
Meijer LL, Alberga AJ, de Bakker JK et al (2018) Outcomes and treatment options for duodenal adenocarcinoma: a systematic review and meta-analysis. Ann Surg Oncol 25(9):2681–2692
Alwmark A, Andersson A, Lasson A (1980) Primary carcinoma of the duodenum. Ann Surg 191(1):13–18
Hu JX, Miao XY, Zhong DW et al (2006) Surgical treatment of primary duodenal adenocarcinoma. Hepatogastroenterology 53(72):858–862
Joesting DR, Beart RW Jr, van Heerden JA et al (1981) Improving survival in adenocarcinoma of the duodenum. Am J Surg 141(2):228–231
Rose DM, Hochwald SN, Klimstra DS et al (1996) Primary duodenal adenocarcinoma: a ten-year experience with 79 patients. J Am Coll Surg 183(2):89–96
Barnes G Jr, Romero L, Hess KR et al (1994) Primary adenocarcinoma of the duodenum: management and survival in 67 patients. Ann Surg Oncol 1(1):73–78
Zhang S, Cui Y, Zhong B et al (2011) Clinicopathological characteristics and survival analysis of primary duodenal cancers: a 14-year experience in a tertiary centre in South China. Int J Colorectal Dis 26(2):219–226
Chung WC, Paik CN, Jung SH et al (2011) Prognostic factors associated with survival in patients with primary duodenal adenocarcinoma. Korean J Intern Med 26(1):34–40
Santoro E, Sacchi M, Scutari F et al (1997) Primary adenocarcinoma of the duodenum: treatment and survival in 89 patients. Hepatogastroenterology 44(16):1157–1163
Sohn TA, Lillemoe KD, Cameron JL et al (1998) Adenocarcinoma of the duodenum: factors influencing long-term survival. J Gastrointest Surg 2(1):79–87
Bakaeen FG, Murr MM, Sarr MG et al (2000) What prognostic factors are important in duodenal adenocarcinoma? Arch Surg. 135(6):635–641 ((discussion 41-2))
Kaklamanos IG, Bathe OF, Franceschi D et al (2000) Extent of resection in the management of duodenal adenocarcinoma. Am J Surg 179(1):37–41
Tocchi A, Mazzoni G, Puma F et al (2003) Adenocarcinoma of the third and fourth portions of the duodenum: results of surgical treatment. Arch Surg 138(1):80–85
Cecchini S, Correa-Gallego C, Desphande V et al (2012) Superior prognostic importance of perineural invasion vs. lymph node involvement after curative resection of duodenal adenocarcinoma. J Gastrointest Surg. 16(1):113–120 ((discussion 20))
Onkendi EO, Boostrom SY, Sarr MG et al (2012) 15-year experience with surgical treatment of duodenal carcinoma: a comparison of periampullary and extra-ampullary duodenal carcinomas. J Gastrointest Surg 16(4):682–691
Kato Y, Takahashi S, Kinoshita T et al (2014) Surgical procedure depending on the depth of tumor invasion in duodenal cancer. Jpn J Clin Oncol 44(3):224–231
Cloyd JM, Norton JA, Visser BC et al (2015) Does the extent of resection impact survival for duodenal adenocarcinoma? Analysis of 1,611 cases. Ann Surg Oncol 22(2):573–580
Jiang QL, Huang XH, Chen YT et al (2016) Prognostic factors and clinical characteristics of patients with primary duodenal adenocarcinoma: a single-center experience from China. Biomed Res Int 2016:6491049
Lee CHA, Shingler G, Mowbray NG et al (2018) Surgical outcomes for duodenal adenoma and adenocarcinoma: a multicentre study in Australia and the United Kingdom. ANZ J Surg 88(3):E157–E161
Rangelova E, Blomberg J, Ansorge C et al (2015) Pancreas-preserving duodenectomy is a safe alternative to high-risk pancreatoduodenectomy for premalignant duodenal lesions. J Gastrointest Surg 19(3):492–497
Kohga A, Yamamoto Y, Sano S et al (2017) Surgical strategy for T1 duodenal or ampullary carcinoma according to the depth of tumor invasion. Anticancer Res 37(9):5277–5283
DeOliveira ML, Winter JM, Schafer M et al (2006) Assessment of complications after pancreatic surgery: a novel grading system applied to 633 patients undergoing pancreaticoduodenectomy. Ann Surg. 244(6):931–937 ((discussion 7-9))
Perez A, Saltzman JR, Carr-Locke DL et al (2003) Benign nonampullary duodenal neoplasms. J Gastrointest Surg 7(4):536–541
Yan JQ, Peng CH, Yang WP et al (2010) Surgical management of benign duodenal tumours. ANZ J Surg 80(7–8):526–530
Moher D, Liberati A, Tetzlaff J et al (2010) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8(5):336–341
Higgins JP, Altman DG, Gotzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Wells G, Shea B, O'Connell D, Peterson j, Welch V, Losos M, et al (2000) The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis2000.
Zhang RC, Xu XW, Wu D et al (2013) Laparoscopic transduodenal local resection of periampullary neuroendocrine tumor: a case report. World J Gastroenterol 19(39):6693–6698
Poves I, Burdio F, Alonso S et al (2011) Laparoscopic pancreas-sparing subtotal duodenectomy. JOP 12(1):62–65
Cho A, Yamamoto H, Kainuma O et al (2012) Totally laparoscopic pancreas-sparing duodenectomy. Surg Today 42(10):1032–1035
Stauffer JA, Raimondo M, Woodward TA et al (2013) Laparoscopic partial sleeve duodenectomy (PSD) for nonampullary duodenal neoplasms: avoiding a whipple by separating the duodenum from the pancreatic head. Pancreas 42(3):461–466
Konishi M, Kinoshita T, Nakagohri T et al (2007) Pancreas-sparing duodenectomy for duodenal neoplasms including malignancies. Hepatogastroenterology 54(75):753–757
Chung RS, Church JM, vanStolk R (1995) Pancreas-sparing duodenectomy: indications, surgical technique, and results. Surgery 117(3):254–259
Huttner FJ, Fitzmaurice C, Schwarzer G et al (2016) Pylorus-preserving pancreaticoduodenectomy (pp Whipple) versus pancreaticoduodenectomy (classic Whipple) for surgical treatment of periampullary and pancreatic carcinoma. Cochrane Database Syst Rev. 2:CD006053
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Bassi C, Dervenis C, Butturini G et al (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 138(1):8–13
Wente MN, Bassi C, Dervenis C et al (2007) Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 142(5):761–768
Wente MN, Veit JA, Bassi C et al (2007) Postpancreatectomy hemorrhage (PPH): an International Study Group of Pancreatic Surgery (ISGPS) definition. Surgery 142(1):20–25
Poultsides GA, Huang LC, Cameron JL et al (2012) Duodenal adenocarcinoma: clinicopathologic analysis and implications for treatment. Ann Surg Oncol 19(6):1928–1935
Kim MJ, Choi SB, Han HJ et al (2014) Clinicopathological analysis and survival outcome of duodenal adenocarcinoma. Kaohsiung J Med Sci 30(5):254–259
Lee HG, You DD, Paik KY et al (2008) Prognostic factors for primary duodenal adenocarcinoma. World J Surg 32(10):2246–2252
Liang TJ, Wang BW, Liu SI et al (2012) Number of involved lymph nodes is important in the prediction of prognosis for primary duodenal adenocarcinoma. J Chin Med Assoc 75(11):573–580
Sarela AI, Brennan MF, Karpeh MS et al (2004) Adenocarcinoma of the duodenum: importance of accurate lymph node staging and similarity in outcome to gastric cancer. Ann Surg Oncol 11(4):380–386
Hurtuk MG, Devata S, Brown KM et al (2007) Should all patients with duodenal adenocarcinoma be considered for aggressive surgical resection? Am J Surg. 193(3):319–324 ((discussion 24-5))
Solaini L, Jamieson NB, Metcalfe M et al (2015) Outcome after surgical resection for duodenal adenocarcinoma in the UK. Br J Surg 102(6):676–681
Rotman N, Pezet D, Fagniez PL et al (1994) Adenocarcinoma of the duodenum: factors influencing survival. French Association for Surgical Research. Br J Surg. 81(1):83–85
Struck A, Howard T, Chiorean EG et al (2009) Non-ampullary duodenal adenocarcinoma: factors important for relapse and survival. J Surg Oncol 100(2):144–148
Ryder NM, Ko CY, Hines OJ et al (2000) Primary duodenal adenocarcinoma: a 40-year experience. Arch Surg. 135(9):1070–1074 ((discussion 4-5))
Moss WM, McCart PM, Juler G et al (1974) Primary adenocarcinoma of the duodenum. Arch Surg 108(6):805–807
Jones BA, Langer B, Taylor BR et al (1985) Periampullary tumors: which ones should be resected? Am J Surg 149(1):46–52
Ouriel K, Adams JT (1984) Adenocarcinoma of the small intestine. Am J Surg 147(1):66–71
Cortese AF, Cornell GN (1972) Carcinoma of the duodenum. Cancer 29(4):1010–1015
Lowell JA, Rossi RL, Munson JL et al (1992) Primary adenocarcinoma of third and fourth portions of duodenum. Favorable prognosis after resection. Arch Surg 127(5):557–560
Lai EC, Doty JE, Irving C et al (1988) Primary adenocarcinoma of the duodenum: analysis of survival. World J Surg 12(5):695–699
Malleo G, Tonsi A, Marchegiani G et al (2013) Postoperative morbidity is an additional prognostic factor after potentially curative pancreaticoduodenectomy for primary duodenal adenocarcinoma. Langenb Arch Surg 398(2):287–294
Sakamoto T, Saiura A, Ono Y et al (2017) Optimal lymphadenectomy for duodenal adenocarcinoma: does the number alone matter? Ann Surg Oncol 24(11):3368–3377
Acknowledgements
We would like to thank the Japan Duodenal Cancer Guideline Committee and many doctors related to this research opportunity. Japan Duodenal Cancer Guideline Committee: S. N. Souya Nunobe, S. K. Shingo Kanaji, H. E. Hidetoshi Eguchi, K. O. Ken-ichi Okada, T. F. Tsutomu Fujii, Y. N. Yuichi Nagakawa, K. K. Kengo Kanetaka, H. Y. Hiroharu Yamashita, S. Y. Suguru Yamada, S. K.Shinji Kuroda, T. A. Toru Aoyama, T. A. Takahiro Akahori, K. N. Kenji Nakagawa, R. H. Ryota Higuchi, H. Y. Hiroki Yamaue, M. S. Masayuki Sho, Y. K. Yasuhiro Kodera.
Funding
This work was supported by the Health and Labor Sciences Research Grant (Grant number H29-GANTAISAKU-IPPAN-013) and JSPS KAKENHI (Grant number 18K08632). The funders had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Consortia
Contributions
Study conception and design of study: RH. Duodenal Guidelines Committee, Surgery Group: SN, SK, HE, KO, TF, YN, KK, HY, SY, SK, TA, TA, KN, RH, HY, MS and YK. Organizing committee meetings and providing opportunities for making article: KO, MS and YK. Acquisition of data: SN, SK, HE, PB and RH. Analysis and interpretation of data: PB. and RH. Drafting of manuscript: PB. and RH. Critical revision of manuscript: SN, SK, HE, MY, KO, MS and YK.
Corresponding author
Ethics declarations
Conflict of interest statement
The authors have no conflicts of interest to declare.
Ethical approval
The authors have no ethical conflicts to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The members of the Japan Duodenal Cancer Guideline Committee are mentioned in “Acknowledgements” section
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Burasakarn, P., Higuchi, R., Nunobe, S. et al. Limited resection vs. pancreaticoduodenectomy for primary duodenal adenocarcinoma: a systematic review and meta-analysis. Int J Clin Oncol 26, 450–460 (2021). https://doi.org/10.1007/s10147-020-01840-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-020-01840-5