Abstract
In this short review, the authors performed a database search and summarize current knowledge of the management of patients with pineal cysts (PCs) and investigate the role of surgical treatment. The scientific literature on the surgical treatment of PCs is sparse and encompasses only case series with little over 200 operated patients combined. All included papers reported favorable results after pineal cyst surgery with improvement of symptoms in most patients. Microsurgical resection of PCs, preferably using the supracerebellar-infratentorial approach, could be considered as a viable treatment option in symptomatic patients. Even patients with non-specific symptoms are reported to improve after surgery. However, evidence offered by this literature review is very limited and therefore our conclusions must be tempered by the restricted set of data. For ethical reasons, a randomized controlled trial is not an acceptable approach, and therefore patient registry could be a useful tool to identify a subset of symptomatic patients that might benefit from pineal cyst resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patients with a pineal cyst (PC) are increasingly referred to neurosurgeons and neurologists because of a widespread availability of magnetic resonance (MR) diagnostics. Patients frequently present with non-specific symptoms and rarely with hydrocephalus or Parinaud’s syndrome. Whereas patients with hydrocephalus and Parinaud’s syndrome are clearly indicated for surgery, most patients with non-specific symptoms are managed conservatively. Occasionally, some neurosurgeons indicate patients with severe non-specific symptoms for surgical treatment, although it is not widely accepted in the neurosurgical community [13]. The first documented report of PC resection in the literature is from Pussep in 1914 [18]. Since then, several hundreds other cases have been reported, typically as case reports or as small retrospective studies. Large, prospective studies are lacking. Indication criteria for surgical treatment are vague in cases of non-specific symptoms and sometimes are based on the gut feeling of the surgeon rather than on objective criteria.
In this short review, we summarize current knowledge of the management of patients with pineal cysts and investigate the role of surgical treatment.
Material and methods
The PUBMED database was searched for publications using the terms “pineal cyst” and “pineal gland cyst” on June 8, 2017. Searches were restricted to the English language and human studies but without limitation on the publication date. The present authors checked and selected all articles according to the inclusion criteria described below. References in each included study were checked to find additional relevant studies. We included studies that fulfilled the following criteria:
-
Case series with more than eight patients that underwent surgery for PC
-
Case series of adult patients
The exclusion criteria were the following:
-
Only cerebrospinal fluid shunting surgery
-
Insufficient clinical data
For each study, we determined the number of operated patients, PC size, surgical approach, length of follow-up, surgical complication, clinical outcome, and recurrence rate.
Results
Our search found 323 papers. After selection and cross-reference checking, ten studies matched our inclusion criteria [3, 6,7,8,9,10,11, 14, 17, 22]. Nine of the ten studies used a retrospective study design and one a prospective design [14]. Papers that were not included were case reports (37 papers), radiological studies (61 papers), surgical series of less than eight patients with PCs (11 papers), pediatric studies (34 papers), and others (170 papers).
The ten included studies comprised 211 patients who underwent surgery for PCs (Table 1). Cyst size ranged from 7 to 35 mm. Surgical procedures that were performed included microsurgical supracerebellar infratentorial and occipital transtentorial PC resection, microsurgical and endoscopic PC fenestration and stereotactic aspiration, and biopsy. Improvement in presenting symptoms occurred in 42.9–100% of the patients. The complication rate varied widely, from as low as 0.0% to as high as 77.8%.
None of the studies included a control group of patients with PCs that would be treated conservatively.
Discussion
Rationale for surgical treatment
In general, surgical treatment is reserved for patients with symptomatic pineal cysts. Surgery might also be considered in atypical cases in which the histological diagnosis is uncertain. “Typical” MR appearance of a PC is a smooth-walled cystic lesion in the pineal region with rim enhancement after gadolinium not thicker than 2 mm [2]. However, cases of multilobular PCs or heterogenous contrast enhancement may arouse suspicion of a tumor. Watchful waiting with serial MR is one of the treatment strategies. Yet, definitive diagnosis of a PC can be based on only a histological tissue examination and therefore surgery might be justified. Several cases have been described in the literature in which pineal tumor was misdiagnosed as PCs [16, 21].
Which patient with PC is symptomatic?
In the era of widespread availability of MR imaging, patients with non-specific symptoms or unrelated symptoms often undergo brain scanning; in some of these patients PC is discovered. In most of these patients, PCs are merely an incidental finding and they should be assured of the benign nature of the PC. However, in some patients, PC might play a role in the pathogenesis of clinical symptoms and defining this subgroup of patients is therefore of paramount importance.
A classical textbook presentation of pineal lesions should be hydrocephalus and Parinaud’s syndrome, which is explained by direct pressure of the pineal mass on the mesencephalon. Nevertheless, PCs rarely grow large enough to compress surrounding structures and cause these symptoms. Hajnsek et al. did not diagnose hydrocephalus or Parinaud’s syndrome in a series of 56 patients [9] and Majovsky et al. reported only one patient in series of 21 patients [14]. On the other hand, Berhouma et al. reported obstructive hydrocephalus in 18 (75%) of 24 operated patients [3]. These results are most likely subject to selection bias and probably caused by different indication criteria employed in different departments.
Nonspecific symptoms seen in patients with PCs may include headache, vertigo, nausea, fatigue, diplopia, tremor, seizures, and paresthesia. Seifert et al. studied the association between PC and headache. These authors compared 51 patients with PC and 51 matched controls without PC [20]. Patients with PC had headaches twice as often as the controls, suggesting the existence of causality. The exact mechanism remains unknown, although several hypotheses have been proposed (e.g., a melatonin disturbance or compression of the vein of Galen) [4, 5].
Is surgery effective?
The present research has shown clinical improvement ranging from as low as 42.9% to as high as 100%. However, more recent studies have shown a clinical improvement of over 90%. Unfortunately, five of the ten studies in our review do not specifically address clinical outcome. Measuring the outcome is very heterogenic: some authors simply state, “symptoms were resolved” [9,10,11] and others created their own ad hoc scales [6]. In our series, we employed a standardized validated questionnaire designed to measure outcome after surgery for Chiari malformation (Chicago Chiari Outcome Scale) [1]. Today, there is a need to develop a specific outcome measuring tool in patients with PCs.
Improvement in symptoms of increased intracerebral pressure due to hydrocephalus or oculomotor palsy caused by the direct compression of mesencephalon is expected when the mass effect of PC is removed. Nevertheless, most patients in the reviewed series presented with non-specific symptoms and yet high improvement was achieved. Even an unusual, rare presentation of PC (such as seizures, monoparesthesia, face numbness, tremor, ataxia, hemiparesis, or syncope) could resolve after cyst resection. The exact mechanism of relief of non-specific symptoms is not easily explained by PC resection; instead, some might argue for the suspicion of a placebo effect [12]. However, we regard a placebo effect as a less plausible explanation because of a relatively high symptom improvement rate and because of the long-lasting effect of the surgery.
What type of surgery?
If surgical treatment of PCs were considered, modern neurosurgery offers microsurgery, endoscopy, and stereotaxy in its armamentarium. Two main microsurgical approaches are a supracerebellar infratentorial and an occipital transtentorial approach to the pineal region. A stereotactic approach enables puncture and an aspiration of the PC and may serve as a biopsy as well. An endoscopic route towards the PC is tranventricular and allows the treating of a hydrocephalus by performing a third ventriculostomy.
Microsurgery
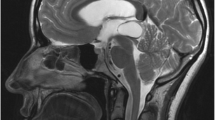
In most cases, the goal of surgery is PC resection (Fig.1), which is achieved microsurgically either by a supracerebellar infratentorial (SCIT) or an occipital transtentorial approach (OTT). The SCIT approach is a straightforward extracerebral technique via the natural corridor between the superior aspect of the cerebellum and the tentorium (Fig. 2). Preferably, the unilateral SCIT approach is used to protect a dominant transverse sinus and spare some bridging veins between the cerebellum and the tentorium that are in the surgical corridor. A radical pineal cyst resection is achieved in almost all cases. Possible complications include diplopia and venous infarction of the cerebellum. In the largest surgical series, all patients underwent surgery using the SCIT approach in a sitting position. In this patient group, the complication rate was 0% [9].
Another common microsurgical approach is OTT, which is also extracerebral, but in contrast to SCIT, the pineal region is accessed supratentorially along the falcotentorial angle (Fig. 2). A slight retraction to the occipital lobe and incision of the tentorial apex are necessary to create a surgical corridor towards the PC. The most common complication after OTT is hemianopia, which occurs transiently in 16.1–79% of the patients and permanently in 0–4.1% [3, 19, 23]. Berhouma et al. used OTT in 20 patients with PC and achieved radical resection in 70% with a complication rate of 20% (four patients with transient hemianopia) [3]. Because of the relatively high prevalence of postoperative hemianopia with the OTT approach, we exclusively use SCIT for PC resection. It is surprising that recent series on pineal cysts resection report rather a high complication rate [3, 10].
Microsurgery shows the highest rate of clinical improvement, with more than 90% showing improvement [6, 9, 10, 14]. Eide et al. reported on 21 operated patients: 15 had their PC resected and six had their PC microscopically fenestrated. The authors found significantly better results in the resection group [6]. In our series of patients with PC resection, we spared some pineal tissue (less than half of the PC) to prevent total melatonin secretion loss [14]. Despite this sparing strategy, we still achieved improvement in 95.2% of our patients. These results suggest superiority of total or near-total PC resection over fenestration.
The matter of physiological changes after radical PC resection is not yet fully resolved. In our previous study, we examined melatonin and cortisol secretion profiles in four patients before and after PC resection. In all cases, patients experienced a loss of endogenous pineal melatonin production, which equated with pinealectomy. Surprisingly, cortisol secretion substantially increased in patients after surgery. Presently, we do not have an explanation for that finding [15].
Stereotaxy
Kreth et al. reported the largest case series using stereotactic aspiration of PCs (n = 14 patients). The authors encountered no complications, but clinical improvement was achieved in only 42.9% of the patients. No recurrence was noted in this series, although Mena et al. reported the regrowth of a PC in their series with only one patient treated with stereotactic aspiration [17].
Endoscopy
An advent of neuroendoscopy has resulted in the introduction of new techniques for pineal region surgery that allowed the surgeon to address both PC and coexisting hydrocephalus (Fig. 2). However, the complete removal of a PC is usually not possible. Tirakotai et al. performed an endoscopic fenestration and biopsy in nine patients. No complications occurred in this series but one PC recurred. Regrettably, the authors do not state the clinical outcome of the patients.
Study limitations
Our paper has two main limitations that need to be addressed. First, all ten studies in this review are case studies, which represent only a level of evidence 4 according to the Oxford Centre for Evidence-based Medicine. Second, a potential publication bias might have manifested itself in the present review. Publication bias occurs when studies with positive findings are more likely to be published than studies with negative findings, which can distort the perception of a topic by the medical community. Because of such bias, reports showing infectivity of pineal cyst resection might have never reached the scientific literature.
Conclusion
The scientific literature on the surgical treatment of PCs is sparse and encompasses only case series with little over 200 operated patients combined. All included papers reported favorable results after pineal cyst surgery with improvement of symptoms in most patients. Microsurgical resection of PCs, preferably using the supracerebellar-infratentorial approach, could be considered as a viable treatment option in symptomatic patients. Even patients with non-specific symptoms are reported to improve after surgery. However, evidence offered by this literature review is very limited, and therefore our conclusions must be tempered by the restricted set of data. For ethical reasons, a randomized controlled trial is not an acceptable approach, and therefore patient registry could be a useful tool to identify a subset of symptomatic patients that might benefit from pineal cyst resection.
References
Aliaga L, Hekman KE, Yassari R, Straus D, Luther G, Chen J, Sampat A, Frim D (2012) A novel scoring system for assessing Chiari malformation type I treatment outcomes. Neurosurgery 70(3):656–665
Barboriak DP, Lee L, Provenzale JM (2001) Serial MR imaging of pineal cysts: implications for natural history and follow-up. AJR Am J Roentgenol 176(3):737–743
Berhouma M, Ni H, Delabar V, Tahhan N, Memou Salem S, Mottolese C, Vallee B (2015) Update on the management of pineal cysts: case series and a review of the literature. Neurochirurgie 61(2–3):201–207
Eide PK, Pripp AH, Ringstad GA (2016) Magnetic resonance imaging biomarkers indicate a central venous hypertension syndrome in patients with symptomatic pineal cysts. J Neurol Sci 363:207–216
Eide PK, Ringstad G (2016) Increased pulsatile intracranial pressure in patients with symptomatic pineal cysts and magnetic resonance imaging biomarkers indicative of central venous hypertension. J Neurol Sci 367:247–255
Eide PK, Ringstad G (2017) Results of surgery in symptomatic non-hydrocephalic pineal cysts: role of magnetic resonance imaging biomarkers indicative of central venous hypertension. Acta Neurochir 159(2):349–361
Fain JS, Tomlinson FH, Scheithauer BW, Parisi JE, Fletcher GP, Kelly PJ, Miller GM (1994) Symptomatic glial cysts of the pineal gland. J Neurosurg 80(3):454–460
Fetell MR, Bruce JN, Burke AM, Cross DT, Torres RA, Powers JM, Stein BM (1991) Non-neoplastic pineal cysts. Neurology 41(7):1034–1040
Hajnsek S, Paladino J, Gadze ZP, Nanković S, Mrak G, Lupret V (2013) Clinical and neurophysiological changes in patients with pineal region expansions. Coll Antropol 37(1):35–40
Kalani M, Wilson D, Koechlin N, Abuhusain H, Dlouhy B, Gunawardena M, Nozue-Okada K, Teo C (2015) Pineal cyst resection in the absence of ventriculomegaly or Parinaud’s syndrome: clincial outcomes and implications for patient selection. J Neurosurg 123(2):352–356
Kreth FW, Schätz CR, Pagenstecher A, Faist M, Volk B, Ostertag CB (1996) Stereotactic management of lesions of the pineal region. Neurosurgery 39(2):280–289 91
Kulkarni AV (2015) Editorial: pineal cyst resection. J Neurosurg 123(2):350–351
Májovský M, Netuka D, Beneš V (2016) Clinical management of pineal cysts: a worldwide online survey. Acta Neurochir 158(4):663–669
Májovský M, Netuka D, Beneš V (2017) Conservative and surgical treatment of patients with pineal cysts: a prospective case series of 110 patients. World Neurosurg. doi:10.1016/j.wneu.2017.05.155
Májovský M, Řezáčová L, Sumová A, Pospíšilová L, Netuka D, Bradáč O, Beneš V (2017) Melatonin and cortisol secretion profile in patients with pineal cyst before and after pineal cyst resection. J Clin Neurosci 39:155–163
Márquez J, Rivero M (2011) Pineal cysts. J Neurosurg Pediatr 8(4):422–422
Mena H, Armonda RA, Ribas JL, Ondra SL, Rushing EJ (1997) Nonneoplastic pineal cysts: a clinicopathologic study of twenty-one cases. Ann Diagn Pathol 1(1):11–18
Pussep L (1914) Die operative Entfernung einer Zyste der Glandula pinealis. Neurol Zentralbl 33:560–563
Qi S, Fan J, Zhang X, Zhang H, Qiu B, Fang L (2014) Radical resection of nongerminomatous pineal region tumors via the occipital transtentorial approach based on arachnoidal consideration: experience on a series of 143 patients. Acta Neurochir 156(12):2253–2262
Seifert CL, Woeller A, Valet M, Zimmer C, Berthele A, Tölle T, Sprenger T (2008) Headaches and pineal cyst: a case-control study. Headache 48(3):448–452
Starke RM, Cappuzzo JM, Erickson NJ, Sherman JH (2016) Pineal cysts and other pineal region malignancies: determining factors predictive of hydrocephalus and malignancy. J Neurosurg 1–6
Tirakotai W, Schulte DM, Bauer BL, Bertalanffy H, Hellwig D (2004) Neuroendoscopic surgery of intracranial cysts in adults. Childs Nerv Syst 20(11–12):842–851
Yoshimoto K, Araki Y, Amano T, Matsumoto K, Nakamizo A, Sasaki T et al (2013) Clinical features and pathophysiological mechanism of the hemianoptic complication after the occipital transtentorial approach. Clin Neurol Neurosurg 115(8):1250–1256
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by grant no. MO 1012NK funded by the Ministry of Defense of the Czech Republic, grant no. 15-32791A funded by the Ministry of Health of the Czech Republic and grant no. Q25/LF1/2 funded by Charles University. The sponsors had no role in the design or conduct of this research.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Májovský, M., Netuka, D. & Beneš, V. Is surgery for pineal cysts safe and effective? Short review. Neurosurg Rev 41, 119–124 (2018). https://doi.org/10.1007/s10143-017-0876-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-017-0876-2