Abstract
The cavernous sinus (CS) has one of the most complex anatomical networks of the skull base and because of the diversity of its contents is involved in many pathological processes. Nevertheless, anatomical literature concerning the CS is still controversial, so a systematic literature review was performed to find out the microanatomy of the medial wall of the CS and its clinical importance on sellar pathologies. Experimental studies from English-language literature between 1996 and 2010 were identified in MEDLINE, LILACS, and Cochrane databases. After analysis, two tables were prepared exhibiting the major points of each article. Fourteen experimental studies were included in the tables. Four studies concluded that the medial wall of the CS is composed of a loose, fibrous structure, and the remaining ten presumed that the medial wall is formed by a dural layer that constitutes the lateral wall of the sella. The lack of definition standards and of methodological criteria led to variation in the results among different studies. Thus, this hindered results comparison, possibly explaining the different observations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
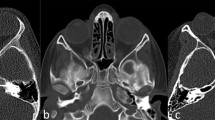
The cavernous sinus (CS) has one of the most complex anatomical networks of the skull base and because of the diversity of its contents is involved in many pathological processes [20]. It is located on either side of the sphenoid sinus, sella, and pituitary gland extending from the superior orbital fissure to the petrous ridge of the temporal bone (Fig. 1). The median size of the cavernous sinus is 2 cm in length and 1 cm in width [7, 16]. Its structure and content are complex and matter of discussion. The term “cavernous sinus” was designated by Winslow (Exposition Anatomique de la Structure du Corps Humain. London: Prevast, 1734) because he believed that it resembled the cavernous body of the penis. This name was discussed and considered inappropriate, later being substituted by the term “lateral sellar compartment” [1, 13].
Its structure is defined by a dural envelope surrounding the cavernous segment of the internal carotid artery [17]. The envelope not only contains the dural carotid cavernous artery and its branches, but a segment of the abducent nerve and a sympathetic nerve complex—considered by its continuity with the internal carotid nerve [1, 26]. It is also the site of a venous confluence that receives the veins that drain the orbit and Sylvian fissure, as well as the anterior and middle cranial fossae. The CS communicates freely with the basilar sinus, superior and inferior petrosal sinuses, and the intercavernous sinuses [17]. The oculomotor nerve, trochlear nerve, and the ophthalmic and maxillary divisions of the trigeminal nerve are, generally, contained within the lateral wall and project into the sinus [7, 16]. In addition to this wall, the CS has a roof or superior wall, posterior wall, and medial wall, the latter being the topic under review.
The anatomy of the medial wall of the CS, although thoroughly described [3–5, 9, 14, 26, 28], still generates controversy. It is classically taught that the pituitary gland is contained within a dural layer that limits the CS medially [7] (Fig. 2). However, some recent reports suggest that there is not a medial dural wall—rather, there exists a loose fibrous tissue that separates the pituitary capsule [4, 9], or merely its own capsule formed by fine collagen fibers that separate the gland [28].
Due to this discussion, a systematic review of English literature concerning the CS was performed to evaluate the microanatomy of the medial wall. This review not only has theoretical interest for the study of anatomy but also has important clinical and surgical implications. Because of its nature, the medial wall has a significant role in the determination of the direction of growth of pituitary adenomas, and in the surgical strategy, because of frequent extension of adenomas and the pituitary gland from the sella into the CS [26].
Methods
We performed MEDLINE, LILACS, and Cochrane database searches for reports on CS medial wall published between 1996 and 2010. Analysis was performed of experimental studies in humans and animals published in the English language. We used the following key words in different permutations: anatomy, cavernous sinus, dural wall, lateral sellar compartment, and medial wall.
The search performed identified 23 studies related to the microanatomy of the medial wall of the CS. Of the literature investigated, nine studies were excluded since they did not fulfill the methodological criteria of inclusion because of the following: descriptive observational studies, i.e., case report (four studies), prospective (cohort) and retrospective (control group) analytical observational studies (one and four studies, respectively).
Of all studies thus collected, we checked the reference lists for additional studies. This method of cross-reference checking was continued until no further publications were found. With the results, two tables were prepared showing the characteristics of each article.
Results
Fourteen studies were chosen for tabulation. Of these, four (172 samples, 44.7% of all samples included in this review) referred to the medial wall of the CS as a loose fibrous structure [4, 9], as its own pituitary capsule of fine collagen fibers [28], or as two layers composed of the lateral part of the pituitary capsule and the fibrous layer [21]. Ten studies (213 samples, 55.3% of all samples included in this review) identified a dural layer [12] forming the medial wall of the CS and separating the CS from the sella and pituitary capsule [14, 25, 26], or separating the CS directly from the pituitary gland [2, 3, 6, 8, 23, 27] (Fig. 3). The data from study designs that did not demonstrate a dural wall between the CS and the pituitary fossa are presented in Table 1, while those that discuss the presence of a dural layer are presented in Table 2.
As one can observe in both tables, the samples (385 were included in this review) vary in the following way: adult cadavers (155; 40.2%), asymptomatic adults (90; 23.4%), embryos and fetuses (76; 19.7%), skull bases (33; 8.6%), adult primates (11; 2.8%), patients with nonfunctioning pituitary adenoma with encasement of the intracavernous carotid artery (10; 2.6%), and patients with nonfunctioning pituitary adenoma without extension to the CS (10; 2.6%).
With regard to the methodology, in those studies that presented results confirming a fibrous tissue separating the pituitary fossa from the lateral sellar compartment, all of them utilized magnetic resonance imaging (MRI) [4, 9, 28] and/or histological staining methods (the Van Gieson [28], hematoxylin and eosin, and/or Masson’s trichrome [4, 9, 21]). Of these studies, three included histological sections and cadaveric dissection under surgical microscope [4, 9, 21], while in one study, transphenoidal surgery was assessed by the Mann–Whitney U test in cases and controls beyond the histological section [28]. The second group, which identified the medial wall of the CS as comprised of a dural layer, did not utilize MRI in their methodology. However, 61.5% prosecuted microscopic dissection [2, 3, 8, 12, 14, 25–27], and 38.5% used histological section with Masson’s trichrome, hematoxylin and eosin, or hemalaun, eosin, and saffron[6, 8, 14, 23, 27]. In one study, the thickness of the pituitary capsule and the inferior and lateral pituitary fossae was measured, and the collagen structure from these layers was analyzed [14], while in another study, computer measurements of medial wall of the CS thickness were compared in the three different regions (posterior, middle, and anterior) and also on the left vs. the right sides [27].
Discussion
The CS is anatomically complex, as confirmed by Yasuda et al. [25, 26] in his two studies that reported the existence of a dural wall in between the lateral surface of the pituitary gland and the CS. The results of Yasuda et al. [25], as well as those of Marinkovic et al. [12] and Tobenas-Dujardin et al. [23], considered that the medial wall of the CS is composed from meningeal dura in the sellar portion and from periosteum in the sphenoid portion.
This hypothesis has been debated by Kehrli et al. [9], Yokoyama et al. [28], Dietemann et al. [4], and Songtao et al. [21], whose studies suggested the absence of this dural medial wall. According to Kehrli et al. [9], a dural wall limiting the CS medially does not exist, but a perforated, fibrous tissue originating from the mesenchyma. The only dense connective tissue separating the pituitary from the surrounding tissues is the pituitary capsule itself. Songtao et al. [21] conceded that the lateral part of the pituitary capsule and the fibrous layer constituted the medial wall of the CS. However, Destrieux et al. [3] assumed that this pituitary capsule is formed from dura mater, the medial wall of the CS being the lateral part of this dural sac that encases the pituitary gland. Dura mater separates the CS from the pituitary gland not only in humans but in primates as well [6]. Accordingly, the investigations of Yilmazlar et al. [27] showed that the medial wall of the CS is a distinct dural layer that forms a barrier between the medial venous space of the cavernous sinus and the pituitary gland. As stated by Campero et al. [2], the medial wall of the CS has two areas—sellar and sphenoidal, both made up of one dural layer.
It is interesting to note that Yokoyama et al. [28] also observed small histological defects in the pituitary capsule, which consists of a very thin layer of collagen fibers that form the medial wall of the CS. Three of 30 cuts from 10 adult cadavers suggest that these defects are important sites of adenoma extension. However, Yasuda et al. [26] did not encounter such defects in 40 CS exams, explaining that the tendency pituitary tumors have toward extending into the CS is due to the medial wall being formed by a single, thin layer of dura. Kursat et al. [11] measured the thickness of the lower third of the medial wall and suggest that it could be the major determinant of parasellar extension when the diaphragma sellae is thick and the stalk opening is complete. This low anatomical resistance against chronic tumor growth may facilitate expansion of pituitary tumors into the CS [10]. According to Yilmazlar et al. [27], no macro- or microscopic defects were observed in any of the medial wall of the CS in 18 specimens. The thinness of the posterior medial wall of the CS suggests that this is the most likely path for extension of pituitary tumors into the CS.
Special attention should be given to the Peker et al. [14] study that analyzed the collagen structure in the layers of the medial wall. In their findings, they confirmed that the dense fibrous capsule involves the pituitary gland. Externally from the pituitary gland, there exists a dural layer forming the lateral wall of the sella. As well, they declared that the infiltration of the CS by pituitary adenoma occurs because of lateral wall weakness (formed by only one thin layer of dura) of the pituitary fossa and from the degree of biochemical destruction of fibrous collagen within the capsule.
The preoperative knowledge of the integrity of the medial wall of the CS is very important since invasion of parasellar structures occurs in 6–10% of pituitary adenomas [15]. Tumoral invasion increases the risk of surgical morbidity, diminishing the chance of a complete cure. As well, invasion indicates the velocity of proliferation and a biologically more aggressive tumoral behavior [15].
MRI is superior to CT scan in distinguishing the pituitary gland, the sellar region, and its surroundings. However, both techniques fail to portray the medial wall of the CS adequately [24] as it would be too thin to view easily via MRI, thus increasing the difficulty of preoperative evaluation of the adenoma invasion [3]. According to Yasuda et al. [26], the fine nature and single dural layer of the medial wall explains the difficulty that Dietemann et al. [4] described in visualizing its structure with MRI imaging. This study was composed of 50 radiographs of normal pituitary glands, and the medial wall of the CS was discovered in a minority of the cases.
However, the work of Dietemann et al. [4] also does not demonstrate a dural wall in histological cuts in adults and embryos using the Masson’s trichrome stain, but a loose, circumferential, fibrous tissue separating a dense glandular capsule from the nervous and vascular structures of the CS. This explains the parasellar extension and the high incidence of recurrence of intracavernous pituitary tumors—after rupturing the pituitary capsule, due to the absence of a solid barrier between the CS and the pituitary fossa.
The medial wall of the CS can invaginate to the interior of the CS depending on the lateral expansion of the pituitary adenoma. This concept, contrary to the invasion and rupture of the dural wall, explains the broad frequency of reports concerning intracavernous invasion of pituitary adenoma in the literature [18].
Thus, a majority of data from CT and MRI scans that indicated intracavernous invasion is, in reality, the compression or invagination in the medial wall. In fact, anatomic studies [16, 22] describe that such invagination of the medial wall exists in almost a third of normal pituitary glands [19].
Surgical technique based on meningeal anatomy is important for CS surgeries. The presence or absence of tumoral invasion into weak points can influence microsurgical results [8].
Conclusions
Despite the excellent quality of the studies, it remains necessary to investigate further, performing new studies that allow for more fluid comparison between results. The incompatible results occur, more than likely, because of the utilization of different methods and varying definitions within the articles. Thus, continuation of the studies is needed to arrive at a consensus.
References
Bleys RL, Janssen LM, Groen GJ (2001) The lateral sellar nerve plexus and its connections in humans. J Neurosurg 95(1):102–110
Campero A, Campero AA, Martins C, Yasuda A, Rhoton AL Jr (2010) Surgical anatomy of the dural walls of the cavernous sinus. J Clin Neurosci 17(6):746–750. doi:10.1016/j.jocn.2009.10.015
Destrieux C, Kakou MK, Velut S, Lefrancq T, Jan M (1998) Microanatomy of the hypophyseal fossa boundaries. J Neurosurg 88(4):743–752
Dietemann JL, Kehrli P, Maillot C, Diniz R, Reis M Jr, Neugroschl C, Vinclair L (1998) Is there a dural wall between the cavernous sinus and the pituitary fossa? Anatomical and MRI findings. Neuroradiology 40(10):627–630
Dolenc VV (1989) Anatomy and surgery of the cavernous sinus. Springer, New York
Domingues RJ, Muniz JA, Tamega OJ (1999) Morphology of the walls of the cavernous sinus of Cebus apella (tufted capuchin monkey). Arq Neuropsiquiatr 57(3B):735–739
Gray H (1995) Angiologia. In: Williams PL, Warwick R, Dyson M, Bannister LH (eds) Gray anatomia, vol 1, 37th edn. Guanabara Koogan, Rio de Janeiro, pp 754–755
Kawase T, van Loveren H, Keller JT, Tew JM (1996) Meningeal architecture of the cavernous sinus: clinical and surgical implications. Neurosurgery 39(3):527–534, discussion 534–526
Kehrli P, Ali M, Reis M Jr, Maillot C, Dietemann JL, Dujovny M, Ausman JI (1998) Anatomy and embryology of the lateral sellar compartment (cavernous sinus) medial wall. Neurol Res 20(7):585–592
Knappe UJ, Konerding MA, Schoenmayr R (2009) Medial wall of the cavernous sinus: microanatomical diaphanoscopic and episcopic investigation. Acta Neurochir (Wien) 151(8):961–967, discussion 967
Kursat E, Yilmazlar S, Aker S, Aksoy K, Oygucu H (2008) Comparison of lateral and superior walls of the pituitary fossa with clinical emphasis on pituitary adenoma extension: cadaveric-anatomic study. Neurosurg Rev 31(1):91–98, discussion 98–99
Marinkovic S, Gibo H, Vucevic R, Petrovic P (2001) Anatomy of the cavernous sinus region. J Clin Neurosci 8(Suppl 1):78–81
Parkinson D (1998) Lateral sellar compartment O.T. (cavernous sinus): history, anatomy, terminology. Anat Rec 251(4):486–490
Peker S, Kurtkaya-Yapicier O, Kilic T, Pamir MN (2005) Microsurgical anatomy of the lateral walls of the pituitary fossa. Acta Neurochir (Wien) 147(6):641–648, discussion 649
Pinker K, Ba-Ssalamah A, Wolfsberger S, Mlynarik V, Knosp E, Trattnig S (2005) The value of high-field MRI (3 T) in the assessment of sellar lesions. Eur J Radiol 54(3):327–334
Rhoton AL, Renn WH, Harris FS (1978) Microsurgical anatomy of the sellar region and cavernous sinus. In: Rand RW (ed) Microneurosurgery, 2nd edn. Mosby, Saint Louis, pp 71–92
Rhoton AL Jr (2002) The cavernous sinus, the cavernous venous plexus, and the carotid collar. Neurosurgery 51(4 Suppl):S375–410
Roux FX, Kalamarides M, Devaux B, Leriche B, Nataf F, Brami F, Meder JF, Destrieux C, Santini JJ (1996) Intracavernous invagination of pituitary macro-adenomas. Ann Endocrinol (Paris) 57(5):403–410
Roux FX, Obreja C, Moussa R, Devaux B, Nataf F, Turak B, Page P, Meder JF (1998) Intracavernous extension of hypophyseal macroadenomas: infiltration or invagination? Neurochirurgie 44(5):344–351
Sen C, Chen CS, Post KD (1997) Microsurgical anatomy of the skull base and approaches to the cavernous sinus. Thieme, New York
Songtao Q, Yuntao L, Jun P, Chuanping H, Xiaofeng S (2009) Membranous layers of the pituitary gland: histological anatomic study and related clinical issues. Neurosurgery 64(3 Suppl):1–9, discussion 9–10
Taptas JN (1982) The so-called cavernous sinus: a review of the controversy and its implications for neurosurgeons. Neurosurgery 11(5):712–717
Tobenas-Dujardin AC, Duparc F, Laquerriere A, Muller JM, Freger P (2003) Embryology of the walls of the lateral sellar compartment: apropos of a continuous series of 39 embryos and fetuses representing the first 6 months of intra-uterine life. Surg Radiol Anat 25(3–4):252–258
Vieira JO Jr, Cukiert A, Liberman B (2004) Magnetic resonance imaging of cavernous sinus invasion by pituitary adenoma diagnostic criteria and surgical findings. Arq Neuropsiquiatr 62(2B):437–443
Yasuda A, Campero A, Martins C, Rhoton AL Jr, de Oliveira E, Ribas GC (2005) Microsurgical anatomy and approaches to the cavernous sinus. Neurosurgery 56(1 Suppl):4–27, discussion 24–27
Yasuda A, Campero A, Martins C, Rhoton AL Jr, Ribas GC (2004) The medial wall of the cavernous sinus: microsurgical anatomy. Neurosurgery 55(1):179–189, discussion 189–190
Yilmazlar S, Kocaeli H, Aydiner F, Korfali E (2005) Medial portion of the cavernous sinus: quantitative analysis of the medial wall. Clin Anat 18(6):416–422
Yokoyama S, Hirano H, Moroki K, Goto M, Imamura S, Kuratsu JI (2001) Are nonfunctioning pituitary adenomas extending into the cavernous sinus aggressive and/or invasive? Neurosurgery 49(4):857–862, discussion 862–853
Acknowledgment
The authors wish to thank Cassius Vinicius Reis, MD (Belo Horizonte, MG) for providing the figures.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Takeshi Mikami, Sapporo, Japan
The authors reported on variations in the morphology of the cavernous sinus medial wall, which has a complex anatomy and adjoins important neurovascular structures. The morphological details are interesting, and an anatomical review of the region would benefit neurosurgeons seeking to develop effective surgical strategies for dealing with pituitary lesions, procedures that reduce the risk of surgical complications when such lesions are excised. In particular, the recent introduction of endoscopic endonasal transsphenoidal techniques for surgery of the pituitary gland has spurred development of numerous modifications and approaches aimed at providing wider surgical views, including exposure of the cavernous sinus so that tumors located within the cavernous sinus can be excised. Therefore, detailed knowledge of cavernous sinus medial wall anatomy is increasingly important.
Our clinical experience led us to believe that the medial wall of the cavernous sinus was a loose fibrous tissue, so recognition of the dural layer by many anatomical researchers was surprising. As the authors pointed out, however, the definition of these structures may be unclear, and, regrettably, their paper does not reach useful conclusions on this subject. In any case, the medial wall of the cavernous sinus is a weak structure, and it is certain that careful manipulation of it is indispensable during clinical procedures.
Benoit JM Pirotte, Brussels, Belgium
M. Goncalves and coworkers provide here a very useful and detailed critical literature review on the controversial nature of the cavernous sinus medial wall. In this paper, the authors state accurately the anatomical and clinical relevance of that issue. Actually, there is a large variability of methods (anatomical/microscopical dissection, histological/MRI analysis) and description of the thickness and nature of the CS medial wall. The presented analysis appears convincing, and the number of 385 collected samples in the review is significant. The authors did a real effort to collect such data, and they should be encouraged in further investigating in that field. Indeed, there is an urgent need of a consensus on that matter. This paper emphasizes that original systematic anatomical and/or MRI studies are lacking and should be conducted.
Takeshi Kawase, Tokyo, Japan
Because of technological advancement such as endoscopy, the origin of the the cavernous sinus has been the focus of studies again. This paper presents an interesting summary of data from 14 current papers concerning the topic.
It might be a mutual understanding that the parasellar compartment is located in the interdural space between the periosteum of the sphenoid sinus and the meningeal dura of the lateral wall. In those papers, there was a consensus that the lower medial wall between the cavernous sinus and sphenoid sinus could be the periosteal origin. However, their opinions varied on the upper medial wall which separates the cavernous sinus and the pituitary body. One of their common finding of the upper medial wall was “a very thin and loose connective tissue.” However, their opinions on the histological origin were different.
A way to solve the histological question is by considering the three following hypotheses: If the pituitary body could be placed in the epidural space, the medial wall might be the periosteal dura. If it could be in the subdural space, the membrane might be the meningeal dura. However, both the periosteal and meningeal dura are commonly not so loose but more tight, showing different histological findings. It must be considered on the third hypothesis that the pituitary body could be in the interdural space, like the parasellar compartment. We have to recall our memory from current surgery of the cavernous sinus that the “deep layer (inner layer)” of the cavernous sinus was thin and loose, having an easy surgical cleavage plane from the meningeal dura of the lateral wall. By our histological and clinical study, the “deep layer” was found around the cranial nerves, protecting those from injury. I found the nature of the medial wall, with similarity to the “deep layer”, in the following points:
1. A loose, semitransparent, and thin nature.
2. Protection of the pituitary body.
3. Presence of easy surgical cleavage plane from the suprasellar dura (diaphragma sellae).
(Data from Fig. 2F [8])
Rights and permissions
About this article
Cite this article
Gonçalves, M.B., de Oliveira, J.G., Williams, H.A. et al. Cavernous sinus medial wall: dural or fibrous layer? Systematic review of the literature. Neurosurg Rev 35, 147–154 (2012). https://doi.org/10.1007/s10143-011-0360-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-011-0360-3