Abstract
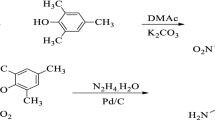
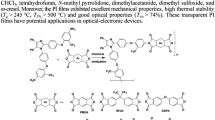
A novel non-coplanar aromatic diamine monomer, 3,3′-ditertbutyl-4,4′-diaminodiphenyl-4″-naphthylmethane (TAPN) was synthesized by a condensation reaction of 2-tertbutylaniline and 1-naphthaldehyde under catalyst hydrochloric acid. The structure of the monomer was confirmed by FTIR, NMR, elementary analysis and mass spectrometry. A series of aromatic polyimides (PIs) were synthesized via conventional one-step polycondensation from TAPN and various commercial aromatic dianhydrides. All of the PIs exhibit excellent solubility in common organic solvents, even in low boiling point solvents such as chloroform (CHCl3), tetrahydrofuran (THF) and acetone. The PIs present outstanding thermal stability with the glass transition temperature (T g) ranged from 299 °C to 350 °C, and the temperature at 10% weight loss ranged from 490 °C to 504 °C, and high optical transparency with the cutoff wavelengths of 306–356 nm. Moreover, the flexible and tough PI films have prominent mechanical properties with tensile strengths in the range of 77.6–90.5 MPa, tensile modulus in the range of 1.8–2.4 GPa and elongation at break in the range of 6.3%–9.5%, as well as lower dielectric constant (2.89–3.12 at 1 MHz) and lower moisture absorption (0.35%–0.66%).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ding, M.X., Prog. Polym. Sci., 2007, 32: 623
Liaw, D.J., Wang, K.Li., Huang, Y.C., Lee, K.R., Lai, J.Y. and Ha, C.S., Prog. Polym. Sci., 2012, 37: 907
Ghosh, A., Sen, S.K., Banerjee, S. and Voit, B., RSC Adv., 2012, 2: 5900
Hasegawa, M. and Horie, K., Prog. Polym. Sci., 2001, 26: 259
Huang, X.H., Huang, W., Zhou, Y.F. and Yan, D.Y., Chinese J. Polym. Sci., 2011, 29(4): 506
Zhao, J.J., Gong, C.L., Zhang, S.J., Shao, Y. and Li, Y.F., Chin. Chem. Lett., 2010, 21: 277
Huang, X.H., Huang, W., Liu, J.Y., Meng, L.L. and Yan, D.Y., Polym. Int., 2012, 61: 1503
Zhang, S.J., Bu, Q.Q., Li, Y.F., Gong, C.L., Xu, X.Y. and Li, H., Mater. Chem. Phys., 2011, 128: 392
Huang, W., Yan, D.Y., Lu, Q.H. and Tao, P., J. Polym. Sci., Part A: Polym. Chem., 2002, 40(2): 229
Huang, W., Yan, D.Y. and Lu, Q.H., Macromol. Rapid Commun., 2001, 22(18): 1481
Yi, L., Li, C.Y., Huang, W. and Yan, D.Y., J. Polym. Res., 2014, 21: 572
Zhang, S.J., Li, Y.F., Ma, T., Zhao, J.J., Xu, X.Y., Yang, F.C. and Xiang, X.Y., Polym. Chem., 2010, 1: 485
Ghaemy, M. and Khajeh, S., Chinese J. Polym. Sci., 2011, 29(4): 465
Liaw, D.J., Chang, F.C., Leung, M.K., Chou, M.Y. and Muellen, K., Macromolecules, 2005, 39: 4024
Liaw, D.J., Wang, K.L., Chang, F.C., Lee, K.R. and Lai, J.Y., J. Polym. Sci., Part A: Polym. Chem., 2007, 45: 2367
Taghavi, M., Ghaemy, M., Hassanzadeh, M. and Amini Nasab, S.M., Chinese J. Polym. Sci., 2013, 31(4): 679
Thiruvasagama, P., Des. Monomers Polym., 2014, 17(2): 166
Wang, C.Y., Zhao, X.Y. and Li, G., Chin. J. Chem., 2012, 30: 2466
Kudo, K., Yoshizawa, T., Hamada, T., Li, J., Sakamoto, S. and Shiraishi, S., Macromol. Rapid Commun., 2006, 27: 430
Matsumoto, T., Kawabata, S. and Takahashi, R., High Perform. Polym., 2006, 18: 719
Liu, J.G., He, M.H., Zhou, H.W., Qian, Z.G., Wang, F.S. and Yang, S.Y., J. Polym. Sci., Part A: Polym. Chem., 2002, 40: 110
Wang, C.S. and Yang, R.W., J. Appl. Polym. Sci., 1997, 66: 609
Gao, Y.F., Zhou, Y.M., He, M., Wang, H.Y., Cui, Y.P. and Zhang, T., Des. Monomers Polym., 2014, 17(6): 590
Sen, S.K., and Banerjee, S., RSC Adv., 2012, 2: 6274
Fang, C., Zhou, Y.M., He, M., Bu, X.H., Yin, K., Weng, J.J. and Zhang, Z.W., J. Appl. Polym. Sci., 2013, 128(1): 80
Wang, J.Y., Liu, C., Su, G.X. and Jian, X.G., High Perform. Polym., 2012, 24: 356
Ree, M., Macromol. Res., 2006, 14: 1
Guo, Y.Z., Shen, D.X., Ni, H.J., Liu, J.G. and Yang, S.Y., Prog. Org. Coat., 2013, 76: 768
Ishii, J., Yokotsuka, H., Saito, T. and Hasegawa, M., J. Photopolym. Sci. Technol., 2011, 24: 287
Pakhuruddin, M.Z., Ibrahim, K. and Aziz, A.A., Optoelectron. Adv. Mat., 2013, 7: 377
Zhao, X.J., Liu, J.G., Rui, J.M., Fan, L. and Yang, S.Y., J. Appl. Polym. Sci., 2007, 103: 1442
Qiu, Z.M., Wang, J.H., Zhang, Q.Y., Zhang, S.B., Ding, M.X. and Gao, L.X., Polymer, 2006, 47: 8444
Langsam, M. and Burgoyne, W.F., J. Polym. Sci., Part A: Polym. Chem., 1993, 31: 909
Dine-Hart, R.A. and Wright, W.W., Makromol. Chem., 1971, 143: 189
Kohl, P.A., Annu. Rev. Chem. Biomol. Eng., 2011, 2: 379
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by the National Natural Science Foundation of China (Nos. 51163003 and 21264005), the fund of Guangxi Natural Science Foundation (Nos. 2014GXNSFAA118040 and 2013GXNSFDA019008) and Guangxi Funds for Specially-appointed Expert.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Liu, Cj., Mei, M., Pei, Xl. et al. Aromatic polyimides with tertbutyl-substituted and pendent naphthalene units: synthesis and soluble, transparent properties. Chin J Polym Sci 33, 1074–1085 (2015). https://doi.org/10.1007/s10118-015-1658-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-015-1658-3