Abstract
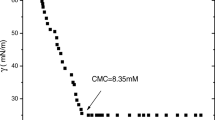
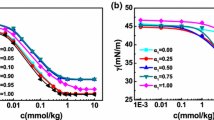
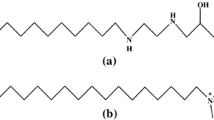
The interactions of non-ionic amphiphilic diblock copolymer poly(oxyethylene/oxybutylene) (E39B18) with anionic surfactant sodium dodecyl sulphate (SDS) and cationic surfactant hexadecyltrimethylammonium bromide (CTAB) were studied by using various techniques such as surface tension, conductivity, steady-state fluorescence and dynamic light scattering. Surface tension measurements were used to determine the critical micelle concentration (CMC) and thereby the free energy of micellization (ΔG mic), free energy of adsorption (ΔG ads), surface excess concentration (Γ) and minimum area per molecule (A). Conductivity measurements were used to determine the critical micelle concentration (CMC), critical aggregation concentration (CAC), polymer saturation point (PSP), degree of ionization (α) and counter ion binding (β). Dynamic light scattering experiments were performed to check the changes in physiochemical properties of the block copolymer micelles taken place due to the interactions of diblock copolymers with ionic surfactants. The ratio of the first and third vibronic peaks (I 1/I 3) indicated the polarity of the pyrene micro environment and was used for the detection of micelle as well as polymer-surfactant interactions. Aggregation number (N), number of binding sites (n) and free energy of binding (ΔG b) for pure surfactants as well as for polymer-surfactant mixed micellar systems were determined by the fluorescence quenching method.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Goddard, E.D. and Ananthapadmanabhan, K.P., “Interaction of surfactants with polymers and proteins”, CRC Press, Boca Raton, FL, 1993, p. 36
Piculell, L., Guillemet, F., Thuresson, K., Shubin, V. and Ericsson, O., Adv. Colloid Interface Sci., 1996, 63: 1
Kwak, J.C.T., “Polymer-Surfactant Systems”, Dekker, New York, 1998
Claesson, P.M., Dedinaite, A. Poptoshev, E., Phys. Chem. Polyelectrolytes, 2001, 101: 3661
Goddard, E.D., J. Colloid Interface Sci., 2002, 256: 228
Tuzar, Z. and Kratochvil, P., Adv. Colloid A Interface Sci., 1976, 6: 201
Dai, S., Tam, K.C. and Li, L., Macromolecules, 2001, 34: 7049
Wang, G. and Olofsson, G., J. Phys. Chem. B, 1998, 102: 9276
Thuresson, K., Nystrom, B., Wang, G. and Lindman, B., Langmuir, 1995, 11: 3730
Dai, S. and Tam, K.C., Langmuir, 2004, 20: 2177
Wang, G. and Olofsson, G., J. Phys. Chem. B, 1998, 102: 927
Shah, S.S., Jamroz, N.U. and Sharif, Q.M., J. Colloids and Surfaces A Physicochemical and Engineering Aspects, 2001, 178: 199
Mata, J., Varade, D. and Bahadu, P., Termochim. Acta, 2005, 428: 147
Wang, C. and Tam, K.C., Langmuir, 2008, 18: 6484
Kang, H., Peng, B., Liang, Y., Han, X. and Liu, H., J. Colloid Interface Sci., 2008, 32: 74
Taboada, P., Castro, E., Barbosa, S. and Mosquera, V., Chem. Phys., 2005, 314: 299
Blokhus, A.M. and Klok, K., J. Colloid Interface Sci., 2000, 230: 448
Xia, G., Hui, Y. and Rong, G., Chinese J. Chem., 2008, 26: 1589
Josephine, C., Brackman, J. and Engberts, B.F.N., Langmuir, 1992, 8: 424
Tachiya, M., Chem. Phys. Lett., 1975, 33: 289
Bravo, C., Leis, J.R. And Pena, E., J. Phys. Chem., 1992, 96: 1957
Tartar, H.V., J. Colloid Sci., 1959, 14: 115
Ekwall, P., Mandell, L. and Solyom, P., J. Colloid Interface Sci., 1971, 35: 519
Lin, C.M., Chen, Y.Z. and Tao, H.K., React. Funct. Polym., 2009, 69: 539
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was financially supported by Pakistan science foundation (PSF) and Higher Education Commission (HEC) Pakistan.
Rights and permissions
About this article
Cite this article
Rehman, N., Khan, A., Bibi, I. et al. Micellar parameters of diblock copolymers and their interactions with ionic surfactants. Chin J Polym Sci 30, 217–226 (2012). https://doi.org/10.1007/s10118-012-1114-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-012-1114-6