Abstract
Research on the dynamics of vegetation distribution in relation to past climate can provide valuable insights into terrestrial ecosystems’ response to climate change. However, paleoenvironmental data sources are often scarce. The integration of ecological niche modeling and paleoecological data can fill in this knowledge gap. In order to elucidate the potential impacts of past and future climate change on the distribution of multiple vegetation types, we used 433 occurrence points of 100 species to build distribution models of five vegetation types occurring in southern Brazil, based on past, current, and future scenarios. Past models indicated the existence of a steppe domain during the Last Glacial Maximum, with forest expansion during the Mid-Holocene, which is consistent with paleoenvironmental data. The current distribution model identified a large area that was climatically suitable for ecotones in an important protection area threatened by agribusiness. The optimistic projections for 2070 predicted an expansion of mixed ombrophilous and seasonal semi-deciduous forests to a higher altitude and latitude, respectively. The pessimistic projections predicted a catastrophic scenario, with the extinction of the steppe and the savanna and a major increase of areas unsuitable for all vegetation types. The ombrophilous dense forest remained stable in all time scenarios, even in the pessimistic future projection. The results of the present study reinforce the need for the implementation of policies that will reduce greenhouse gas emissions that drive global climate change, which may lead to the extinction not only of species but also of landscapes as we know them today.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climatic fluctuations during the Quaternary, mainly the glacial and interglacial cycles in Pleistocene (Barnola et al. 1987), have driven many changes in the distribution range of organisms globally (Hewitt 2000; Bueno et al. 2017). Paleoenvironmental studies have shown that the vegetation mosaic that covers Southern Brazil and consists of forests, grasslands (steppes), and savannas was the result of climatic fluctuations, which alternated between hot/wet and cold/dry periods (Behling 1997, 1998; Ledru et al. 1998; Behling and Negrelle 2001; Behling et al. 2004). With the exception of the Pampas, which cover the southernmost portion of Brazil, these different vegetation types in Southern Brazil are mainly found in two global biodiversity hotspots: the Atlantic Forest and the Cerrado (Myers et al. 2000). This distribution suggests that this vegetation mosaic features exceptional concentrations of endemic species, and also experiences excessive habitat loss.
In terrestrial ecosystems, deforestation is the major cause of extinctions (Sala et al. 2000; Newbold et al. 2015; WWF 2018); however, climate change may have an equal or even greater impact on biodiversity loss (Selwood et al. 2014), especially in biodiversity hotspots (Malcolm et al. 2006; Jantz et al. 2015). According to the Intergovernmental Panel on Climate Change (IPCC), the global temperature is likely to increase by up to 4.8 °C by the end of the century (IPCC 2014). South America is the continent with the highest extinction risks caused by climate change (Urban 2015) and the Atlantic Forest is a hotspot particularly vulnerable to global warming (Bellard et al. 2014).
Research on the dynamics of vegetation distribution in relation to past climate can provide valuable information on terrestrial ecosystems’ response to future climate change (Blois et al. 2013; Nolan et al. 2018). However, there are still many gaps in terms of knowledge on the identity and distribution of species (Cardoso et al. 2011; Diniz-Filho et al. 2013; Hortal et al. 2014). The existing knowledge on the structure of the Atlantic Forest is based on only 0.01% of the forest that remains (de Lima et al. 2015). A strategy that can fill in these biodiversity knowledge gaps, while also generating information on the effects of the climate on vegetation distribution, is ecological niche modeling (ENM).
ENM is a computational method that can estimate the potential distribution of species by associating occurrence points with environmental variables, characterizing environmental conditions that are appropriate for the species, and identifying where and how these suitable environments are distributed in space, time, and geographic scenarios (Pearson 2007; Peterson and Soberón 2012). The integration of ENM results and paleoecological data provides a detailed geographic framework for addressing many biogeographic questions of great value to ecology and conservation, such as identifying factors that determine the limits of species’ distribution and assessing the impacts of climate change on this distribution (Peterson et al. 2004; Svenning et al. 2011).
Although ENM tools are most commonly used in species distribution analysis, they also provide valuable information on ecosystems and biomes (Sobral-Souza et al. 2015, 2018; Manish et al. 2016; Wang et al. 2017). The State of Paraná (Southern Brazil) is situated in an interesting phytogeographic location, in which tropical and subtropical floral elements are mixed, and multiple disjunct and relictual vegetation types occur, which make Paraná a vast field on which to conduct biogeographic studies (Kaehler et al. 2014). The vegetation mosaic of Paraná is formed by distinct forest intervals with herbaceous and shrub formations, resulting from different environmental conditions (Roderjan et al. 2002; Marques et al. 2011). Furthermore, in Paraná State exist areas where two Brazilian hotspots, the Atlantic Forest and the southernmost portion of the Cerrado, meet (Myers et al. 2000), which is a rare occurrence; these areas are characterized by high endemism (Ritter et al. 2010; Maia and Goldenberg 2014; Pereira and Labiak 2018) and ecotone formations (Bianchi et al. 2012; Moro et al. 2012; Batista and Bastos 2014; Caxambú et al. 2015).
In this study, we used the vegetation types of Paraná as models to elucidate the potential impact of past and future climate change on the distribution and conservation of the multiple vegetation types in a rare contact zone of the two aforementioned Brazilian hotspots. More specifically, our goals were (1) to demonstrate the power of ENM in studying the distribution of different vegetation types in relation to climate, (2) to reconstruct the vegetation distribution during the Last Glacial Maximum (LGM) and Mid-Holocene (MID) and compare these results with paleoenvironmental data, (3) to identify suitable areas for ecotone occurrences, and (4) to estimate the changes in each vegetation type from the LGM to 2070, with the use of both optimistic and pessimistic future projections for the latter.
Material and methods
Occurrence points of vegetation types
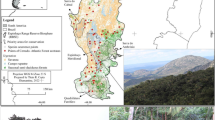
As a guide, we used the distribution of vegetation types in Paraná that we obtained from Maack (1948), who described the original vegetation cover before it was replaced by farmland and pasture land (Da Silva and Passos 2010). Maack identified five major vegetation types (Fig. 1): ombrophilous dense forest (ODF), which covers the eastern portion of the State and is characterized by a high canopy, where trees reach heights of up to 40 m; mixed ombrophilous forest (MOF), which covers plateau portions of the State and is characterized by the presence of Araucarias, the Paraná pine (Araucaria angustifolia); seasonal semi-deciduous forest (SSF), localized in western and northern regions, which also has a high canopy, but the trees partially lose their leaves in the driest months; steppe (STE), which is situated at higher altitudes and is formed by grasslands and shrubs; savanna (SAV), which is located in the southernmost portion of the Cerrado and formed by arboreal and shrubby woody plants.
Distribution of vegetation types in the State of Paraná (Southern Brazil) according to Maack (1948), and occurrence points (dots) used to build distributions models in Maxent
We selected a set of species to represent each vegetation type, prioritizing endemic species and species with concentrated distributions in regions around Paraná, including the States of São Paulo, Mato Grosso do Sul, Santa Catarina, and Rio Grande do Sul (Online Resource 1.1). The species set was selected based on information from Brazilian Flora 2020 (floradobrasil.jbrj.gov.br/), a project that includes descriptions, identification keys, and illustrations of all plants, algae, and fungi species known in Brazil.
Occurrence records were obtained from the GBIF and SpeciesLink databases. We removed duplicate records, or those from which data were missing or were unreliable. We filtered the records so that no pair of points was closer than 10 km, a treatment suggested by Boria et al. (2014) in order to reduce the effects of sampling bias and spatial autocorrelation on model performance. In sum, we used 433 occurrence records of 100 species (Fig. 1, Online Resource 1.2), which consisted of 101 STE (39 species), 63 SAV (15 species), 89 SSF (10 species), 120 MOF (12 species), and 60 ODF (24 species) records.
Environmental variables and climate change scenarios
We obtained environmental variables for the current climatic conditions as well as for past and future climate projections from the WorldClim database at a spatial resolution of 30 arcsec, except for the LGM layer, for which data were available only at 2.5 arcmin. In order to reduce the dimensionality and collinearity of the environmental layers, we used Pearson’s correlation coefficient to remove variables with high cross-correlation values (> ± 0.6; Online Resource 1.3 and Online Resource 2), thereby reducing the set of 19 variables to four variables, which were temperature annual range (Bio07), mean temperature of warmest quarter (Bio10), precipitation of wettest quarter (Bio16), and precipitation of driest quarter (Bio17). This set of variables condensed the climate conditions in Paraná, including seasonal variations, and may have had an impact on vegetation distribution.
The same bioclimatic variables were obtained for past climatic conditions during the LGM (22 ka) and MID (6 ka) and for future projections, for which we formulated an optimistic (RCP 2.6) and a pessimistic (RCP 8.5) scenario for 2070. We used the bioclimatic variables derived from two global climate models (GCMs), CCSM4 and MIROC-ESM, because they are available in WorldClim for both past and future scenarios. The use of a range of GCMs and future emission scenarios is more appropriate for ecological studies because climatic predictions can vary greatly among GCMs, and considering both optimistic and pessimistic scenarios may be important in conservation management and political negotiations (Harris et al. 2013, 2014; Porfirio et al. 2014; Varela et al. 2015). Online Resource 2 provides a more detailed description of each bioclimatic variable projected in each time scenario by these GCMs.
Ecological niche models
We used Maxent 3.4.1 to identify environmentally suitable sites for the persistence of each vegetation type in past, current, and future climate scenarios. We chose Maxent over other available modeling methods because of its superior performance and suitability for presence-only data (Phillips et al. 2006) and for vegetation types (Wang et al. 2017); additionally, Maxent has been cited in more than 1000 published papers since 2006 (Merow et al. 2013); it is free to use and became open source in 2017 (Phillips et al. 2017).
Seventy percent of the occurrence data of each vegetation type were randomly allocated to model calibration and the remaining 30% to model evaluation, with 100 bootstrap replications. We used the following validation metrics: area under the receiver operating characteristic curve function (AUC) (Fielding and Bell 1997), calculated by Maxent; true skill statistics (TSS) (Allouche et al. 2006), corresponding to the maximum training sensitivity plus specificity threshold; and the Boyce Index (Boyce et al. 2002), calculated using the “ROCR” and “ecospat” R packages. We created response curves showing how the variation of each variable affected the probability of the presence of each vegetation type using the DAT outputs (*_only.dat) generated by Maxent.
Vegetation shifts and paleoenvironmental data
The vegetation type with the highest suitability for a given grid cell was assigned as the most likely vegetation type of that grid cell, based on the methodology proposed by Wang et al. (2017). Grid cells below the threshold corresponding to the maximum sensitivity plus specificity (Liu et al. 2013) for all vegetation types were classified as undefined. We estimated the percentage of persistence and changing areas of each vegetation type relatively to their distributions between each time scenario. We also compiled paleoenvironmental records published by other authors and compared these with models built for the LGM and MID scenarios. In sum, we used seven LGM and 12 MID records (Online Resource 1.4).
Climatically suitable areas for ecotones
We converted suitability models from continuous to binary values (presence or absence) using as a threshold the tau value corresponding to the Cloglog output in Maxent (tau = 0.632). Tau is the minimum value given to a grid cell considered a typical location of a specie (Phillips et al. 2017). We defined as climatically suitable areas for ecotones the grid cells in which the suitability values of 2 or more vegetation types reached values above the threshold. The climatically suitable areas for ecotones identified in this study were close to the ecotone concept defined by the Brazilian Vegetation Technical Manual (IBGE 2012); in this, ecotones are described as areas of tension between two or more vegetation types, which is similar to one of the first definitions given by Clements (1905). Thus, we considered as climatically suitable areas for ecotones not only simple transition areas but also regions where there was a high probability of different vegetation types coexisting.
More details about the analysis are provided in the Online Resource 3, which includes a fully reproducible, dynamic document that combines code, rendered results (figures and tables), and explanations.
Results
The statistical validation measures suggested that the models of all vegetation types performed well (Table 1). Vegetation types with narrow distribution (STE, SAV, and ODF) performed better than those with broader distribution (SSF and MOF).
According to the response curves (Fig. 2), the areas with the lowest temperature variation were more suitable for ODF and the areas with the highest temperature variation were more suitable for SSF. The coolest areas were more suitable for STE, while the warmest areas were more suitable for ODF and SSF. SAV and MOF have highly suitability in areas with intermediate temperatures. The driest areas were more suitable for SSF, while the wettest areas were more suitable for ODF. STE, SAV, and MOF have highly suitability in areas with intermediate precipitation.
The current vegetation distribution predicted by Maxent (Fig. 3) is similar to the phytogeographical map created by Maack (1948; Fig. 1). This distribution made possible the identification of a large area that was climatically suitable for ecotones and consisted of STE, SAV, and MOF in the central eastern region of Paraná, which coincides with the Devonian Scarp, an important protection area in Paraná State.
Distribution models of Paraná vegetation types during the Last Glacial Maximum (LGM) (22 ka), Mid-Holocene (MID) (6 ka), and current period as well as in 2070 (RCP 2.6 and RCP 8.5); these scenarios were derived from CCSM4 and MIROC-ESM. Stripped areas indicate climatically suitable areas for ecotones and grid cells indicate two or more vegetation types with suitability values above the tau value (0.632). In the LGM and MID models, points indicate paleoenvironmental records (Online Resource 1.4)
In general, the projections showed the same patterns in the GCMs derived from CCSM4 (Fig. 4) and MIROC-ESM (Fig. 5). Both models predicted a domain consisting of STE and MOF during the LGM, with SAV and ODF stretches in the north and on the coast, respectively. During the MID, both models indicated an expansion of certain forest types, mainly MOF over STE and SSF over MOF. The paleoenvironmental profiles with which a vegetation type was associated were located within or in very close proximity to the modeled distribution range of the vegetation type in both past scenarios (Fig. 3, Online Resource 1.4). Between the MID and the current scenario, both models showed a further expansion of MOF over STE and a southward expansion of SSF.
Percentage of persistence and change of the dominant vegetation types among the time scenarios produced by CCSM4. The values above the bars indicate the area (%) covered by each vegetation type before a change occurred. (a) Changes between the Last Glacial Maximum and Mid-Holocene, (b) changes between the Mid-Holocene and the current period, (c) changes between the current period and 2070 (RCP 2.6), and (d) changes between the current period and 2070 (RCP 8.5). The Y-axis takes into account only one vegetation type per grid cell (the dominant vegetation type with the highest suitability), and does not take into account the presence of more than one vegetation type in areas classified as ecotones
Percentage of persistence and change of the dominant vegetation types among the time scenarios produced by MIROC-ESM. The values above the bars indicate the area (%) covered by each vegetation type before a change occurred. (a) Changes between the Last Glacial Maximum and Mid-Holocene, (b) changes between the Mid-Holocene and the current period, (c) changes between the current period and 2070 (RCP 2.6), and (d) changes between the current period and 2070 (RCP 8.5). The Y-axis takes into account only one vegetation type per grid cell (the dominant vegetation type with the highest suitability), and does not take into account the presence of more than one vegetation type in areas classified as ecotones
The optimistic projections for 2070 (RCP 2.6) predicted a major expansion of MOF over STE in high-elevation areas and a southward expansion of SSF over MOF and SAV. The suitable ecotone areas were reduced by 66% and 82% in the optimistic scenarios derived from CCSM4 and MIROC-ESM, respectively. The pessimistic projection for 2070 (RCP 8.5) predicted a catastrophic scenario, in which STE and SAV are expected to be virtually extinct, MOF is restricted to small patches, SSF expands to the southernmost portion of Paraná, and large areas become unsuitable for all vegetation types. The only exception is ODF, which remained stable even in the pessimistic projections. In neither of the pessimistic scenarios there were suitable ecotone areas.
Discussion
The characterization of vegetation responses to climate is important for assessing the potential impact of climate change on terrestrial ecosystems (Seddon et al. 2016). In this study, we utilized for the first time data generated by ENM to describe the responses of different vegetation types to bioclimatic variables. The response curves of vegetation types to bioclimatic variables were consistent with the general descriptions of Roderjan et al. (2002) and of others who conducted studies in Paraná and reported the presence of ODF in regions with high temperature and rainfall (Marques et al. 2011), MOF in wet and cold regions (Fritzsons et al. 2018), SAV in areas that were colder compared with its core region further north (Bustamante et al. 2012; Oliveira et al. 2014), STE in the coldest regions usually associated with the highest altitudes (Iganci et al. 2011), and SSF in regions with high temperatures and with a defined dry and warm period (Higuchi et al. 2013).
In addition to the response curves, the good predictive ability of the models was reaffirmed by the congruence between the paleoenvironmental data and the projections to past periods. In general, paleoenvironmental studies in Paraná report a STE domain during the LGM, with a few records of MOF, and a forest expansion over STE and SAV caused by temperature or precipitation increases. Continental (Behling 1998; Behling and Negrelle 2001; Cruz et al. 2006) and marine (Gu et al. 2017) records from adjacent regions in São Paulo and Santa Catarina also indicate the existence of a grassland domain in the highlands and a forest and grassland mosaic in the lowlands during the LGM, with a marked expansion of forests over grasslands during the MID. Although there is no direct evidence of the presence of ODF during the LGM in Paraná, there is evidence of an expansion of this vegetation type in São Paulo owing to high precipitation levels in tropical and subtropical latitudes between 24 ka and 21 ka ago (Ledru et al. 2005). The integration of the ENM results with paleoenvironmental data offers the potential for an enhanced contribution of paleobiology to ecology and conservation biology, thereby offering a quantitative ecological perspective that may inform conservation management decisions by estimating future climate change impacts (Svenning et al. 2011).
The current vegetation distribution predicted by Maxent (Fig. 3) also evidenced the predictive power of the models, as it was similar to the phytogeographical map created by Maack (1984; Fig. 1), which is considered the most important Paraná vegetation record because it was described before original vegetation was replaced by farmland and pasture land (Da Silva and Passos 2010). In this study, ENM results were used for the first time to identify climatically suitable areas for ecotones, which are places of great importance in terms of conservation and the study of evolutionary and ecological processes (Smith et al. 1997; Kark et al. 2007; Kark 2017). Also worth mentioning is the conservation unit called Devonian Scarp, a topographic step with peaks rising up to 1290 m and slopes up to 450 m (Maack 2002), which, according to the current predicted distribution, had a high probability of coexisting with STE, SAV, and MOF. Floristic surveys in Devonian Scarp confirm the existence of STE with SAV and MOF patches (Takeda et al. 1996; Carmo et al. 2010; Da Silva and Passos 2010; Moro and Carmo 2010; Ritter et al. 2010; Carmo and Assis 2012).
Recently, a bill (PL 527/2016) was proposed to reduce the Devonian Scarp area by 68%, on the premise that its current borders are overly restricting land use by farmers (Paraná 2016). The bill was shelved in 2018, but it can be raised for discussion any time. The results of the present study reinforce the importance of the Devonian Scarp as a conservation unit. This area offers protection to a vegetation mosaic formed by the oldest vegetation in Paraná (STE), which is the result of the encounter of two Brazilian hotspots (the Atlantic Forest and the southern border of the Cerrado SAV), and by the vegetation type that is the symbol of Paraná State (MOF), where the Paraná pine and mate tea (Ilex paraguariensis) are found, which are species of economic and medicinal importance that are represented in the Paraná State flag (Martins-Ramos et al. 2010).
Both GCM scenarios indicate that the ODF has remained fairly stable throughout the past 22 ka and should remain stable in the future, even in the pessimistic projection. Climatically stable areas are related to centers of high diversity and endemism (Harrison and Noss 2017), play an important role in understanding the evolutionary history of biodiversity, and can contribute to protecting it against climate change (Keppel et al. 2012, 2015). Other studies have identified the coast of Paraná as a climatically stable area (Carnaval and Moritz 2008; Ferro et al. 2014; Carvalho and Del Lama 2015; Costa et al. 2018) and a center of endemism (Murray-Smith et al. 2009) and high diversity (Zwiener et al. 2017a), which highlights the importance of conserving this forest.
The models suggest that climate change is a threat to biodiversity. In the optimistic projections for 2070 (RCP 2.6), the models predicted a major expansion of MOF over STE in high-elevation areas, and a southward expansion of SSF over MOF and SAV. Indeed, species distributions have recently shifted to higher altitudes and higher latitudes (Chen et al. 2011) and they are likely to affect ecosystem functioning and human well-being (Pecl et al. 2017). The MOF in Paraná harbors species of economic and medicinal importance (Martins-Ramos et al. 2010). STE areas are often used as forage resources by livestock, play an important role in greenhouse gas mitigation (O’Mara 2012; Andrade et al. 2015), and are more reliable carbon sinks than forests (Dass et al. 2018). A notable reduction of suitable areas was observed for both vegetation types, even in the optimistic scenario, which coincides with the UNFCCC Paris Agreement that aims to limit global warming well below 2 °C and as close to 1.5 °C as possible (UNFCCC 2015).
In the pessimistic projection for 2070 (RCP 8.5), the models produced an even more worrying scenario, which predicted the extinction of STE, SAV, and the ecotones. The SSF is unlikely to expand to the southernmost portion of Paraná. The extent to which populations will migrate and adapt will depend upon several factors, such as the effective population size and dispersal ability (Aitken et al. 2008). The SSF is currently restricted to small fragments, mostly in agricultural areas (Dettke et al. 2008), and it may stop the movement of species (Heller and Zavaleta 2009). The immense increase of areas identified as unsuitable for all vegetation types in the pessimistic projections is also worthy of concern. Under a pessimistic scenario, the synergistic effects of future land use changes and global warming are projected to reduce the natural vegetation cover by up to 58% in hotspots (Jantz et al. 2015), with an accompanying disruption of ecosystem services and impact on biodiversity (Nolan et al. 2018). The unsuitable areas identified in this study may be covered by either widespread native species (Zwiener et al. 2017b), species from other vegetation types not included in this study (e.g., from seasonal deciduous forests), or invasive species (Walther et al. 2009; Bradley et al. 2010; Gallardo et al. 2017; Lamsal et al. 2018). Therefore, climate change may enhance biotic homogenization by facilitating the invasions of widespread native species and alien species.
In addition to validation metrics, other indirect inferences such as response curves and paleoenvironmental data reinforce the predictive power of the models. However, the model results should be interpreted with caution. Vegetation distribution also depends upon complex ecological relationships involving predictors not included in model calibration and predictions, such as the soil, microclimate, dispersal capacity, biotic interactions, time-lag effects, and human activities (Pearson 2006; Wisz et al. 2013; Wu et al. 2015; Wang et al. 2017). For example, combining bioclimatic and soil properties may prevent an overestimation of the current vegetation cover and allow us to better predict the transitions between biomes (Arruda et al. 2017). However, incorporating soil variables in past and future projections can be problematic owing to a lack of information on past pedoenvironments. Keeping soil variables constant in the past or future projections may result in unlikely scenarios because some pedogenic processes occur over short time periods (Targulian and Krasilnikov 2007) and soil properties change along with climate and vegetation by means of complex feedbacks (Stärz et al. 2016). There are significant gaps in our understanding of the migratory capacity of plants in a rapid climate change scenario (Corlett and Westcott 2013) and the inclusion of these variables in ENM is an important challenge to improve the robustness of models in projecting the distribution of vegetation under future conditions.
Conclusions
In this study, we estimated the current, past, and future distribution of multiple vegetation types in a contact zone of two Brazilian hotspots, by using a niche modeling approach. The Atlantic Forest and the Cerrado are highly threatened by fragmentation and land use changes, and here we showed that climate change may further intensify biodiversity loss. Future projections reinforce the need for policies to reduce greenhouse gas emissions that drive global climate change, which may lead to the extinction not just of species but also of landscapes as we know them today.
References
Aitken SN, Yeaman S, Holliday JA, Wang T, Curtis-McLane S (2008) Adaptation, migration or extirpation: climate change outcomes for tree populations. Evol Appl 1:95–111. https://doi.org/10.1111/j.1752-4571.2007.00013.x
Allouche O, Tsoar A, Kadmon R (2006) Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J Appl Ecol 43:1223–1232. https://doi.org/10.1111/j.1365-2664.2006.01214.x
Andrade BO, Koch C, Boldrini II, Vélez-Martin E, Hasenack H, et al (2015) Grassland degradation and restoration: a conceptual framework of stages and thresholds illustrated by southern Brazilian grasslands. Nat e Conserv 13:95–104. https://doi.org/10.1016/j.ncon.2015.08.002
Arruda DM, Fernandes-Filho EI, Solar RRC, Schaefer CEGR (2017) Combining climatic and soil properties better predicts covers of Brazilian biomes. Sci Nat 104. https://doi.org/10.1007/s00114-017-1456-6
Barnola JM, Raynaud D, Korotkevich YS, Lorius C (1987) Vostok ice core provides 160,000-year record of atmospheric CO2. Nature 329:408–414. https://doi.org/10.1038/329408a0
Batista VG, Bastos RP (2014) Anurans from a Cerrado-Atlantic Forest ecotone in Campos Gerais region, southern Brazil. Check List 10:574–582. https://doi.org/10.15560/10.3.574
Behling H (1997) Late Quaternary vegetation, climate and fire history of the Araucaria forest and campos region from Serra Campos Gerais, Paraná State (South Brazil). Rev Palaeobot Palynol 97:109–121. https://doi.org/10.1590/S1519-69842012000400005
Behling H (1998) Late Quaternary vegetational and climatic changes in Brazil. Rev Palaeobot Palynol 99:143–156. https://doi.org/10.1016/S0034-6667(97)00044-4
Behling H, Negrelle RRB (2001) Tropical rain forest and climate dynamics of the Atlantic lowland, Southern Brazil, during the late Quaternary. Quat Res 56:383–389. https://doi.org/10.1006/qres.2001.2264
Behling H, Pillar VDP, Orlóci L, Bauermann SG (2004) Late Quaternary Araucaria forest, grassland (Campos), fire and climate dynamics, studied by high-resolution pollen, charcoal and multivariate analysis of the Cambará do Sul core in southern Brazil. Palaeogeogr Palaeoclimatol Palaeoecol 203:277–297. https://doi.org/10.1016/S0031-0182(03)00687-4
Bellard C, Leclerc C, Leroy B, Bakkenes M, Veloz S, Thuiller W, Courchamp F (2014) Vulnerability of biodiversity hotspots to global change. Glob Ecol Biogeogr 23:1376–1386. https://doi.org/10.1111/geb.12228
Bianchi JS, Bento CM, Kersten R de A (2012) Epífitas vasculares de uma área de ecótono entre as Florestas Ombrófilas Densa e Mista, no Parque Estadual do Marumbi, PR. Estud Biol 34:37–44. https://doi.org/10.7213/estud.biol.6121
Blois JL, Williams JW, Fitzpatrick MC, Ferrier S, Veloz SD et al (2013) Modeling the climatic drivers of spatial patterns in vegetation composition since the Last Glacial Maximum. Ecography (Cop) 36:460–473. https://doi.org/10.1111/j.1600-0587.2012.07852.x
Boria RA, Olson LE, Goodman SM, Anderson RP (2014) Spatial filtering to reduce sampling bias can improve the performance of ecological niche models. Ecol Model 275:73–77. https://doi.org/10.1016/j.ecolmodel.2013.12.012
Boyce MS, Vernier PR, Nielsen SE, Schmiegelow FKA (2002) Evaluating resource selection functions. Ecol Model 157:281–300. https://doi.org/10.1016/S0304-3800(02)00200-4
Bradley BA, Wilcove DS, Oppenheimer M (2010) Climate change increases risk of plant invasion in the Eastern United States. Biol Invasions 12:1855–1872. https://doi.org/10.1007/s10530-009-9597-y
Bueno ML, Pennington RT, Dexter KG, Kamino LHY, Pontara V, Neves DM, Ratter JA, de Oliveira‐Filho AT (2017) Effects of Quaternary climatic fluctuations on the distribution of Neotropical savanna tree species. Ecography (Cop) 40:403–414. https://doi.org/10.1111/ecog.01860
Bustamante M, Nardoto G, Pinto A, Resende J, Takahashi F et al (2012) Potential impacts of climate change on biogeochemical functioning of Cerrado ecosystems. Braz J Biol 72:655–671. https://doi.org/10.1590/S1519-69842012000400005
Cardoso P, Erwin TL, Borges PAV, New TR (2011) The seven impediments in invertebrate conservation and how to overcome them. Biol Conserv 144:2647–2655. https://doi.org/10.1016/j.biocon.2011.07.024
Carmo MRB do, Assis MA de (2012) Caracterização florística e estrutural das florestas naturalmente fragmentadas no Parque Estadual do Guartelá, município de Tibagi, Estado do Paraná. Acta Bot Bras 26:133–145. https://doi.org/10.1590/S0102-33062012000100015
Carmo MRB do, Moro RS, Nogueira MKFS (2010) A vegetação florestal nos Campos Gerais. In: Patrimônio Natural dos Campos Gerais do Paraná. UEPG, Ponta Grossa
Carnaval AC, Moritz C (2008) Historical climate modelling predicts patterns of current biodiversity in the Brazilian Atlantic forest. J Biogeogr 35:1187–1201. https://doi.org/10.1111/j.1365-2699.2007.01870.x
Carvalho AF, Del Lama MA (2015) Predicting priority areas for conservation from historical climate modelling: stingless bees from Atlantic Forest hotspot as a case study. J Insect Conserv 19:581–587. https://doi.org/10.1007/s10841-015-9780-7
Caxambú MG, Geraldino HCL, Solvalagem ACM (2015) Ferns and lycophytes in two areas of ecotone between seasonal semideciduous forest and mixed ombrophilous forest in Campo Mourão, Paraná, Brazil. Open J For 05:195–209. https://doi.org/10.4236/ojf.2015.52018
Chen IC, Hill JK, Ohlemüller R, Roy DB, Thomas CD (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333:1024–1026. https://doi.org/10.1126/science.1206432
Clements FE (1905) Research methods in ecology. The University publishing company, Lincoln
Corlett RT, Westcott DA (2013) Will plant movements keep up with climate change? Trends Ecol Evol 28:482–488. https://doi.org/10.1016/j.tree.2013.04.003
Costa GC, Hampe A, Ledru MP, Martinez PA, Mazzochini GG et al (2018) Biome stability in South America over the last 30 kyr: inferences from long-term vegetation dynamics and habitat modelling. Glob Ecol Biogeogr 27:285–297. https://doi.org/10.1111/geb.12694
Cruz FW, Burns SJ, Karmann I, Sharp WD, Vuille M, Ferrarid JA (2006) A stalagmite record of changes in atmospheric circulation and soil processes in the Brazilian subtropics during the Late Pleistocene. Quat Sci Rev 25:2749–2761. https://doi.org/10.1016/j.quascirev.2006.02.019
Da Silva PAH, Passos E (2010) A paisagem de Vila Velha e seu significado para a teoria dos refúgios e a evolução do Domínio Morfoclimático dos Planaltos das Araucárias. RA’E GA - O Espac Geogr em Anal:155–164. https://doi.org/10.5380/raega.v19i0.15983
Dass P, Houlton BZ, Wang Y, Warlind D (2018) Grasslands may be more reliable carbon sinks than forests in California. Environ Res Lett 13:074027. https://doi.org/10.1088/1748-9326/aacb39
de Lima RAF, Mori DP, Pitta G, Melito MO, Bello C, Magnago LF, Zwiener VP, Saraiva DD, Marques MCM, de Oliveira AA, Prado PI (2015) How much do we know about the endangered Atlantic Forest? Reviewing nearly 70 years of information on tree community surveys. Biodivers Conserv 24:2135–2148. https://doi.org/10.1007/s10531-015-0953-1
Dettke GA, Orfrini AC, Milaneze-Gutierre MA (2008) Composição florística e distribuição de epífitas vasculares em um remanescente alterado de Floresta Estacional Semidecidual no Paraná, Brasil. Rodriguésia 59:859–872. https://doi.org/10.1590/2175-7860200859414
Diniz-Filho JAF, Loyola RD, Raia P, Mooers AO, Bini LM (2013) Darwinian shortfalls in biodiversity conservation. Trends Ecol Evol 28:689–695. https://doi.org/10.1016/j.tree.2013.09.003
Ferro VG, Lemes P, Melo AS, Loyola R (2014) The reduced effectiveness of protected areas under climate change threatens atlantic forest tiger moths. PLoS One 9. https://doi.org/10.1371/journal.pone.0107792
Fielding AH, Bell JF (1997) A review of methods for the assessment of prediction errors in conservation presence / absence models. Environ Conserv 24:38–49. https://doi.org/10.1017/S0376892997000088
Fritzsons E, Wrege MS, Mantovani LE (2018) Climatic aspects related to the distribution of Brazilian pine in the State of Santa Catarina. Floresta 48:503. https://doi.org/10.5380/rf.v48i4.53272
Gallardo B, Aldridge DC, González-Moreno P, Pergl J, Pizarro M et al (2017) Protected areas offer refuge from invasive species spreading under climate change. Glob Chang Biol 23:5331–5343. https://doi.org/10.1111/gcb.13798
Gu F, Zonneveld KAF, Chiessi CM, Arz HW, Pätzold J et al (2017) Long-term vegetation, climate and ocean dynamics inferred from a 73,500 years old marine sediment core (GeoB2107-3) off southern Brazil. Quat Sci Rev 172:55–71. https://doi.org/10.1016/j.quascirev.2017.06.028
Harris RMB, Grose MR, Lee G, Bindoff NL, Porfirio LL et al (2014) Climate projections for ecologists. Wiley Interdiscip Rev Clim Chang 5:621–637. https://doi.org/10.1002/wcc.291
Harris RMB, Porfirio LL, Hugh S, Lee G, Bindoff NL et al (2013) To be or not to be? Variable selection can change the projected fate of a threatened species under future climate. Ecol Manag Restor 14:230–234. https://doi.org/10.1111/emr.12055
Harrison S, Noss R (2017) Endemism hotspots are linked to stable climatic refugia. Ann Bot 119:207–214. https://doi.org/10.1093/aob/mcw248
Heller NE, Zavaleta ES (2009) Biodiversity management in the face of climate change: a review of 22 years of recommendations. Biol Conserv 142:14–32. https://doi.org/10.1016/j.biocon.2008.10.006
Hewitt G (2000) The genetic legacy of the Quaternary ice ages. Nature 405:907–913. https://doi.org/10.1038/35016000
Higuchi P, da Silva AC, Budke JC, Mantovani A, da Bortoluzzi RL C et al (2013) Influência do clima e de rotas migratórias de espécies arbóreas sobre o padrão fitogeográfico de florestas na região sul do Brasil. Cienc Florest 23:539–553. https://doi.org/10.5902/1980509812338
Hortal J, de Bello F, Diniz-Filho JAF, Lewinsohn TM, Lobo JM et al (2014) Seven shortfalls that beset large-scale knowledge of biodiversity. Annu Rev Ecol Evol Syst 46:523–549. https://doi.org/10.1146/annurev-ecolsys-112414-054400
IBGE (2012) Manual Técnico da Vegetação Brasileira. Instituto Brasileiro de Geografia e Estatística - IBGE, Rio de Janeiro
Iganci JR, Heiden G, Miotto STS, Pennington RT (2011) Campos de Cima da Serra: the Brazilian Subtropical Highland Grasslands show an unexpected level of plant endemism. Bot J Linn Soc 167:378–393. https://doi.org/10.1111/j.1095-8339.2011.01182.x
IPCC, 2014 Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, Pachauri RK and Meyer LA (eds.)]. IPCC, Geneva, Switzerland
Jantz SM, Barker B, Brooks TM, Chini LP, Huang Q, Moore RM, Noel J, Hurtt GC (2015) Future habitat loss and extinctions driven by land-use change in biodiversity hotspots under four scenarios of climate-change mitigation. Conserv Biol 29:1122–1131. https://doi.org/10.1111/cobi.12549
Kaehler M, Goldenberg R, Evangelista PHL, Ribas ODS, Vieira AOS, et al (2014) Plantas vasculares do Paraná. Departamento de Botânica, Curitiba
Kark S (2017) Effects of Ecotones on Biodiversity. In: Reference Module in Life Sciences. Elsevier, Oxford, pp 142–148. https://doi.org/10.1016/B978-0-12-809633-8.02290-1
Kark S, Allnutt TF, Levin N, Manne LL, Williams PH (2007) The role of transitional areas as avian biodiversity centres. Glob Ecol Biogeogr 16:187–196. https://doi.org/10.1111/j.1466-8238.2006.00274.x
Keppel G, Mokany K, Wardell-Johnson GW, Phillips BL, Welbergen JA et al (2015) The capacity of refugia for conservation planning under climate change. Front Ecol Environ 13:106–112. https://doi.org/10.1890/140055
Keppel G, Van Niel KP, Wardell-Johnson GW, Yates CJ, Byrne M et al (2012) Refugia: identifying and understanding safe havens for biodiversity under climate change. Glob Ecol Biogeogr 21:393–404. https://doi.org/10.1111/j.1466-8238.2011.00686.x
Lamsal P, Kumar L, Aryal A, Atreya K (2018) Invasive alien plant species dynamics in the Himalayan region under climate change. Ambio 47. https://doi.org/10.1007/s13280-018-1017-z
Ledru MP, Rousseau DD, Cruz FW, Riccomini C, Karmann I et al (2005) Paleoclimate changes during the last 100,000 yr from a record in the Brazilian Atlantic rainforest region and interhemispheric comparison. Quat Res 64:444–450. https://doi.org/10.1016/j.yqres.2005.08.006
Ledru MP, Salgado-Labouriau ML, Lorscheitter ML (1998) Vegetation dynamics in southern and central Brazil during the last 10,000 yr BP. Rev Palaeobot Palynol 99:131–142. https://doi.org/10.1016/S0034-6667(97)00049-3
Liu C, White M, Newell G (2013) Selecting thresholds for the prediction of species occurrence with presence-only data. J Biogeogr 40:778–789. https://doi.org/10.1111/jbi.12058
Maack R (1948) Notas preliminares sobre clima, solos e vegetação do estado do Paraná. Arq Biol e Biotecnol 2:102–200
Maack R (2002) Geografia Física do Estado do Paraná, 3rd edn. Imprensa Oficial, Curitiba
Maia FR da, Goldenberg R (2014) Melastomataceae from the “Parque Estadual do Guartelá”, Tibagi, Paraná, Brazil: species list and field guide. Check List 10:1316. https://doi.org/10.15560/10.6.1316
Malcolm JR, Liu C, Neilson RP, Hansen L, Hannah L (2006) Global warming and extinctions of endemic species from biodiversity hotspots. Conserv Biol 20:538–548. https://doi.org/10.1111/j.1523-1739.2006.00364.x
Manish K, Telwala Y, Nautiyal DC, Pandit MK (2016) Modelling the impacts of future climate change on plant communities in the Himalaya: a case study from Eastern Himalaya, India. Model Earth Syst Environ 2:92. https://doi.org/10.1007/s40808-016-0163-1
Marques MCM, Swaine MD, Liebsch D (2011) Diversity distribution and floristic differentiation of the coastal lowland vegetation: implications for the conservation of the Brazilian Atlantic Forest. Biodivers Conserv 20:153–168. https://doi.org/10.1007/s10531-010-9952-4
Martins-Ramos D, Bortoluzzi RLC, Mantovani A (2010) Plantas medicinais de um remascente de Floresta Ombrófila Mista Altomontana, Urupema, Santa Catarina, Brasil. Rev Bras Plantas Med 12:380–397. https://doi.org/10.1590/S1516-05722010000300016
Merow C, Smith MJ, Silander JA (2013) A practical guide to MaxEnt for modeling species’ distributions: what it does, and why inputs and settings matter. Ecography (Cop) 36:1058–1069. https://doi.org/10.1111/j.1600-0587.2013.07872.x
Moro RS, Carmo MRB do (2010) A vegetação campestre nos Campos Gerais. In: Patrimônio Natural dos Campos Gerais do Paraná. UEPG, Ponta Grossa
Moro RS, Gomes IA, Pereira TK (2012) Selecting ecotonal landscape units on Meridional Plateau, Southern Brazil. Bosque (Valdivia) 33:23–24. https://doi.org/10.4067/S0717-92002012000300012
Murray-Smith C, Brummitt NA, Oliveira-Filho AT, Bachman S, Moat J, Lughadha EMN, Lucas EJ (2009) Plant diversity hotspots in the Atlantic coastal forests of Brazil. Conserv Biol 23:151–163. https://doi.org/10.1111/j.1523-1739.2008.01075.x
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Newbold T, Hudson LN, Hill SLL, Contu S, Lysenko I et al (2015) Global effects of land use on local terrestrial biodiversity. Nature 520:45–50. https://doi.org/10.1038/nature14324
Nolan C, Overpeck JT, Allen JRM, Anderson PM, Betancourt JL et al (2018) Past and future global transformation of terrestrial ecosystems under climate change. Science 361:920–923. https://doi.org/10.1126/science.aan5360
O’Mara FP (2012) The role of grasslands in food security and climate change. Ann Bot 110:1263–1270. https://doi.org/10.1093/aob/mcs209
Oliveira PTS, Nearing MA, Moran MS, Goodrich DC, Wendland E et al (2014) Trends in water balance components across the Brazilian Cerrado. Water Resour Res 50:7100–7114. https://doi.org/10.1002/2013WR015202
Paraná (2016) Assembleia Legislativa. Projeto de Lei No 527/2016. Altera os limites da APA da Escarpa Devoniana, na forma que especifica a presente Lei. http://portal.alep.pr.gov.br/modules/mod_legislativo_arquivo/mod_legislativo_arquivo.php?leiCod=66840&tipo=I. Accessed 3 Aug 2017
Pearson RG (2006) Climate change and the migration capacity of species. Trends Ecol Evol 21:111–113. https://doi.org/10.1016/j.tree.2005.11.022
Pearson RG (2007) Species’ distribution modeling for conservation educators and practitioners. Am Museum Nat Hist 1:1–50
Pecl GT, Araújo MB, Bell JD, Blanchard J, Bonebrake TC et al (2017) Biodiversity redistribution under climate change: impacts on ecosystems and human well-being. Science 355:eaai9214. https://doi.org/10.1126/science.aai9214
Pereira JBS, Labiak PH (2018) Checklist of ferns and lycophytes from the highlands of Pico Paraná State Park, Paraná, Brazil. Rodriguésia 69:301–307. https://doi.org/10.1590/2175-7860201869203
Peterson AT, Martinez-Meyer E, González-Salazar C (2004) Reconstructing the pleistocene geography of the Aphelocoma jays (Corvidae). Divers Distrib 10:237–246. https://doi.org/10.1111/j.1366-9516.2004.00097.x
Peterson AT, Soberón J (2012) Species distribution modeling and ecological niche modeling: getting the concepts right. Nat a Conserv 10:102–107. https://doi.org/10.4322/natcon.2012.019
Phillips SJ, Anderson RP, Dudík M, Schapire RE, Blair ME (2017) Opening the black box: an open-source release of Maxent. Ecography (Cop) 40:887–893. https://doi.org/10.1111/ecog.03049
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Porfirio LL, Harris RMB, Lefroy EC, Hugh S, Gould SF, Lee G, Bindoff NL, Mackey B (2014) Improving the use of species distribution models in conservation planning and management under climate change. PLoS One 9:e113749. https://doi.org/10.1371/journal.pone.0113749
Ritter LMO, Ribeiro MC, Moro RS (2010) Composição florística e fitofisionomia de remanescentes disjuntos de Cerrado nos Campos Gerais, PR, Brasil - limite austral do bioma. Biota Neotrop 10:379–414. https://doi.org/10.1590/S1676-06032010000300034
Roderjan CV, Galvão F, Kuniyoshi YS, Hatschbach GG (2002) As unidades fitogeográficas do estado do paraná, brasil. Ciência Ambient 24:75–92
Sala OE, Armesto JJ, Berlow E, Bloomfield J, Dirzo R et al (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774. https://doi.org/10.1126/science.287.5459.1770
Seddon AWR, Macias-Fauria M, Long PR, Benz D, Willis KJ (2016) Sensitivity of global terrestrial ecosystems to climate variability. Nature 531:229–232. https://doi.org/10.1038/nature16986
Selwood KE, McGeoch MA, Mac Nally R (2014) The effects of climate change and land-use change on demographic rates and population viability. Biol Rev 90:837–853. https://doi.org/10.1111/brv.12136
Smith TB, Wayne RK, Girman DJ, Bruford MW (1997) A role for ecotones in generating rainforest biodiversity. Science 276:1855–1857. https://doi.org/10.1126/science.276.5320.1855
Sobral-Souza T, Lima-Ribeiro MS, Solferini VN (2015) Biogeography of Neotropical rainforests: past connections between Amazon and Atlantic Forest detected by ecological niche modeling. Evol Ecol 29:643–655. https://doi.org/10.1007/s10682-015-9780-9
Sobral-Souza T, Vancine MH, Ribeiro MC, Lima-Ribeiro MS (2018) Efficiency of protected areas in Amazon and Atlantic Forest conservation: a spatio-temporal view. Acta Oecol 87:1–7. https://doi.org/10.1016/j.actao.2018.01.001
Stärz M, Lohmann G, Knorr G (2016) The effect of a dynamic soil scheme on the climate of the mid-Holocene and the Last Glacial Maximum. Clim Past 12:151–170. https://doi.org/10.5194/cp-12-151-2016
Svenning JC, Fløjgaard C, Marske KA, Nógues-Bravo D, Normand S (2011) Applications of species distribution modeling to paleobiology. Quat Sci Rev 30:2930–2947. https://doi.org/10.1016/j.quascirev.2011.06.012
Takeda IJM, Moro R, Kaczmarech R (1996) Análise florística de um encrave de cerrado no Parque do Guartelá, Tibagi, PR. Publ UEPG, CiBiolSaúde 2:21–31
Targulian VO, Krasilnikov PV (2007) Soil system and pedogenic processes: self-organization, time scales, and environmental significance. Catena 71:373–381. https://doi.org/10.1016/j.catena.2007.03.007
UNFCCC (2015) Adoption of the Paris Agreement. Report No. FCCC/CP/2015/L.9/ Rev.1. https://unfccc.int/resource/docs/2015/cop21/eng/l09r01.pdf. Accessed 24 June 2020
Urban MC (2015) Accelerating extinction risk from climate change. Science 348:571–573. https://doi.org/10.1126/science.aaa4984
Varela S, Lima-Ribeiro MS, Terribile LC (2015) A short guide to the climatic variables of the last glacial maximum for biogeographers. PLoS One 10:1–15. https://doi.org/10.1371/journal.pone.0129037
Walther GR, Roques A, Hulme PE, Sykes MT, Pyšek P et al (2009) Alien species in a warmer world: risks and opportunities. Trends Ecol Evol 24:686–693. https://doi.org/10.1016/j.tree.2009.06.008
Wang S, Xu X, Shrestha N, Zimmermann NE, Tang Z, Wang Z (2017) Response of spatial vegetation distribution in China to climate changes since the Last Glacial Maximum (LGM). PLoS One 12:1–18. https://doi.org/10.1371/journal.pone.0175742
Wisz MS, Pottier J, Kissling WD, Pellissier L, Lenoir J, Damgaard CF, Dormann CF, Forchhammer MC, Grytnes JA, Guisan A, Heikkinen RK, Høye TT, Kühn I, Luoto M, Maiorano L, Nilsson MC, Normand S, Öckinger E, Schmidt NM, Termansen M, Timmermann A, Wardle DA, Aastrup P, Svenning JC (2013) The role of biotic interactions in shaping distributions and realised assemblages of species: implications for species distribution modelling. Biol Rev 88:15–30. https://doi.org/10.1111/j.1469-185X.2012.00235.x
Wu D, Zhao X, Liang S, Zhou T, Huang K, Tang B, Zhao W (2015) Time-lag effects of global vegetation responses to climate change. Glob Chang Biol 21:3520–3531. https://doi.org/10.1111/gcb.12945
WWF (2018) Living Planet Report - 2018: Aiming Higher. WWF, Gland
Zwiener VP, Padial AA, Marques MCM, Faleiro FV, Loyola R, Peterson AT (2017a) Planning for conservation and restoration under climate and land use change in the Brazilian Atlantic Forest. Divers Distrib 23:955–966. https://doi.org/10.1111/ddi.12588
Zwiener VP, Padial AA, Vitule JRS, Grady CJ, Lira-Noriega A (2017b) Climate change as a driver of biotic homogenization of woody plants in the Atlantic Forest. Glob Ecol Biogeogr 27:298–309. https://doi.org/10.1111/geb.12695
Acknowledgments
We are thankful to the thousands of researchers who contributed directly or indirectly to the Brazilian Flora 2020 and to biodiversity databases such as GBIF and SpeciesLink.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES) - Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Wolfgang Cramer
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Trindade, W.C.F., Santos, M.H. & Artoni, R.F. Climate change shifts the distribution of vegetation types in South Brazilian hotspots. Reg Environ Change 20, 90 (2020). https://doi.org/10.1007/s10113-020-01686-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10113-020-01686-7