Abstract
Due to the growing world population and changing eating habits, there is an increasing demand in sustainable alternative protein sources, whereas the available land for the production of plant and animal protein decreases owing to desertification and urbanization. Furthermore, the rapidly decreasing resources of fossil fuels necessitate more sustainable production cycles combined with well-conceived land use. This includes the establishment of novel utilization pathways for hitherto not or insufficiently used biomass. In this context, insects offer prospective alternatives, since they represent highly efficient and, due to evolutionary processes, highly optimized bioreactors that have the ability to effectively and autonomously convert biomass into biochemical compounds such as proteins, fat, and chitin by combined mechanical, chemical, and microbiological degradation. Furthermore, insects are a vastly underutilized bioresource and need to be exploited for the bioconversion and valorization also of hitherto not usable organic residues to food, feed, chemicals, enzymes, and other bioactive compounds. Mentionable is here also the production of attractants, repellants, defensive, and other chemicals such as antimicrobial peptides that open up new opportunities for therapeutical and biotechnological applications, for example regarding plant pest management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With approximately just under one million described species, the class of insects is the class with the most number of species within the kingdom Animalia (Grimaldi and Engel 2005), and it is estimated that there are several million more to date undiscovered or undescribed species. Consequently, there is a huge potential of serviceable, beneficial insect species that are hitherto unexploited.
In comparison with conventional livestock, insects can be produced more sustainably, since they have higher feed conversion efficiencies, lower water and land requirements, and lower greenhouse gas emissions (Oonincx et al. 2010). Additionally, insects play an important role in food security because they deliver ecological services such as plant pollination, bio-waste degradation, for example of dead plant material, and natural pest control, and because in some cases they also serve as food and feed.
Of the almost one million insect species described, approximately 2037 are consumed as food worldwide (Jongema 2015). Accordingly, there is still immense potential in insect biodiversity and the use of to date unexploited insect species as for example food and feed. Edible insects are generally rich in protein and fat and can provide valuable minerals, vitamins and energy. Therefore, they have a huge potential to contribute to food and feed as well as protein and nutrient security (Rumpold and Schlüter 2013a).
In animal feed, insect protein has the potential to completely or partly replace soy bean and fish meal in conventional livestock production and aquaculture (Veldkamp et al. 2012; Barroso et al. 2014; Makkar et al. 2014; Stamer et al. 2014). This can prevent further erosion of forests for the cultivation of soy and overfishing for the production of fishmeal and thus contribute to forest as well as aquatic biodiversity.
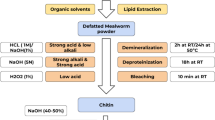
Additionally, insects have the potential to valorize organic by-products, such as food waste or lignocellulosic waste from the bioethanol production. Moreover, in addition to proteins, fats, minerals, and vitamins (Rumpold and Schlüter 2013a), they can, for example, provide chitin for medical (Lee et al. 2008; Muzzarelli 2010) and other applications, valuable enzyme systems for bioconversion processes also derived from their residential gut microbiota (Engel and Moran 2013; Koenig et al. 2013), antimicrobial peptides as alternative antibiotics and therapeutic (Chernysh et al. 2002; Hull et al. 2012; Poeppel et al. 2014), or tools against phytopathogens in plant protection strategies (Vilcinskas and Gross 2005).
Overall, huge research efforts are required in order to fully apprehend the potential and challenges of the bioresource insects.
Edible insects as food and feed
Insects as food
While the consumption of insects by humans appears like a far-fetched notion in the so-called Western world, entomophagy is traditionally practiced by more than two billion people worldwide (van Huis et al. 2013). Of the insect species consumed as food that have been described in the literature (Jongema 2015), 31 % are beetles (Coleoptera), 18 % are caterpillars (Lepidoptera), 14 % are bees, wasps, and ants (Hymenoptera), and 13 % are grasshoppers, locusts, and crickets (Orthoptera) (van Huis et al. 2013). Since up to today most insects consumed are collected in the wild (van Huis et al. 2013), an increase in insect consumption based on gathering will lead to overexploitation and endangering of species in flora and fauna. There are already reports of decreasing populations of species in Mexico due to overcollection by humans (Ramos-Elorduy 2006) and in Japan presumably due to the use of pesticides (Payne 2015). Thus it is recommended to establish safe and sustainable mass-rearing processes on an industrial scale for an efficient and ecological use of edible insects as food and feed (Rumpold and Schlüter 2013b). Before cultivation and application of foreign insect species, their potential hazard on the indigenous ecosystem has to be considered to prevent a negative impact on the ecosystem and the endangerment of indigenous species (Kenis et al. 2009).
With regard to conventional livestock, insects possess several advantageous properties. In addition to the environmental aspects concerning resource efficiency mentioned before, insects have shorter life cycles, can be multivoltine, i.e., undergo more than one life cycle per year, have more offspring, and have a high feed conversion efficiency, i.e., need less feed for the production of 1 kg of biomass than for example needed for the production of beef and pork (Oonincx and de Boer 2012).
Edible insects are rich in protein and fat and provide minerals and vitamins (Rumpold and Schlüter 2013a; Feedipedia.org 2015; Nowak et al. 2016). Species of the order Orthoptera (grasshoppers, locusts, crickets) with protein contents of more than 77 % on a dry matter basis have been reported (Ramos-Elorduy et al. 1998) and fat contents as high as 77 % (based on dry matter) have been found for the Lepidopteran (moths and butterflies) larvae of Phasus triangularis (Ramos-Elorduy et al. 1997, 1998). In general, they meet amino acid requirements of humans and are high in unsaturated fatty acids (Rumpold and Schlüter 2013a). A comparison of 85 mineral compositions and 80 vitamin contents of edible insects as derived from literature with recommended daily intakes for adults showed that, in general, consumed insects are rich copper, iron, magnesium, manganese, phosphorous, selenium, and zinc as well as riboflavin, pantothenic acid, biotin, and in some cases folic acid (Rumpold and Schlüter 2013a).
Taking into consideration the pressing global need for alternative protein and food sources, insects offer thus a high-quality nutrient-rich alternative. However, the European law regarding the use of insect protein and other novel protein sources in food and feed products is indistinct (van der Spiegel et al. 2013). Legal aspects such as the Novel Food legislation [Regulation (EC) No 258/97] and moreover consumer acceptance are up to now major challenges for the successful marketing of edible insects as food in Europe. Other challenges include microbial food safety, and the allergenic, toxic, and zoonotic potential of insects as also identified in a risk profile by the European Food Safety Authority, where 12 insect species with the biggest potential for the use as food and feed in the European Union have been listed (EFSA 2015). Despite their risk potential, food insects have a huge potential to contribute to global food as well as protein security.
The potential of insects as feed
Due to a rising world population accompanied by a change in eating habits and increasing meat consumption, food and feed security are global challenges of the twenty-first century. In particular in the European feed sector, the demand in protein is met by vast imports of particularly soybean but also of fish meal. Around 14.3 million tonnes soybeans and 28.3 million tonnes soybean cake have been imported into the European Union (EU) in 2011. Main exporting countries include Argentina, Brazil, and the USA (FAO 2014). In addition, the EU (including the candidate countries) is one of the main consumers of fish meal at 1.2 million tons per year. In 2002, 33 % of fish meal was used in aquaculture, 32 % as pig feed, and 29 % for poultry production in the EU (Oliveira-Goumas 2004). An increased cultivation of soy leads to deforestation and endangers the rainforest biodiversity (Fearnside 2001), and the intensive use of fish meal endangers the aquatic biodiversity, for example, due to overfishing (Naylor et al. 2000, 2009). In this context, the use of sustainable insect-based feed could thus contribute to protein security and biodiversity.
However, there are still legal hurdles prohibiting the use of insects as feed for conventional livestock and aquaculture in Europe that are mostly due to the so-called feed ban [Commission Regulation (EC) No 999/2001], an EU regulation on the use of processed animal protein (PAP) in feed for farmed animals, installed for the prevention of transmissible spongiform encephalopathies (TSE). That is rather unexpected since insects are a natural feed source of among others birds and fish. However, there is evidence that insects such as the meat-eating fly Sarcophaga carnaria can function as vectors transmitting TSE from one animal to another providing the dietary exposure of the fly larvae to infected animal brain and its subsequent consumption by another animal (Post et al. 1999). Concluding, it has to be assured that feed utilized for farmed insects intended for the use as feed and food is free of infected animal protein. This is ensured by the feed ban that also applies for farmed food and feed insects.
The general applicability of insects as feed has been shown in numerous investigations and has been summarized in several reviews (Veldkamp et al. 2012; Barroso et al. 2014; Makkar et al. 2014; Rumpold and Schlüter 2014). Studies on the application of insects as poultry feed suggest that insects could replace up to 15 % of soy bean meal in a poultry diet and that fish meal could be completely replaced by insect meal in poultry feed. Insects investigated as poultry feed included caterpillars (Lepidoptera) (Ijaiya and Eko 2009), fly larvae (Diptera) (Hwangbo et al. 2009), beetle larvae (Coleoptera) (Ramos-Elorduy et al. 2002), and grasshoppers and crickets (Orthoptera) (Wang et al. 2005; Anand et al. 2008), which demonstrates that no insect order excels and insects in general have potential as poultry feedstuff.
In fish feeding trials, the potential but also the limitations of insects as fish meal replacement were shown (Barroso et al. 2014; Makkar et al. 2014). Mostly dipterans, especially the black soldier fly Hermetia illucens, were applied as fish feed (Bondari and Sheppard 1987; St-Hilaire et al. 2007b; Ogunji et al. 2008; Sealey et al. 2011; Kroeckel et al. 2012; Olaniyi and Salau 2013), since their amino acid profile resembled the amino acid profiles of fish meal, and Hermetia was hence believed to most likely succeed as fish meal replacement. However, it was discovered that its potential as fish meal replacement was furthermore limited by its fatty acid composition that was of minor value compared to fish meal. This limitation could be reduced by supplementing the fly larvae with omega-3 fatty acids via fish offal (St-Hilaire et al. 2007a). More research and rearing activities are required in order to efficiently exploit insects as fish feed and as sustainable fish meal replacement in aquaculture. In addition, further insect species should be taken into consideration.
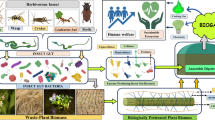
Insect bioconversion and organic residue valorization
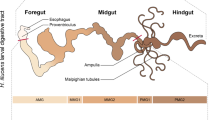
Insects host a large variety and complex community of microorganisms in their guts and other body regions. The microorganisms resident in insects are mostly beneficial and contribute to nutrition by upgrading nutrient-poor diets or by supporting the digestion of recalcitrant food components. Furthermore, they contribute to the protection from predators, parasites, and pathogens, to inter- and intraspecific communication, and to modulation of immune responses. In addition, they affect the host’s efficiency as disease vectors and govern mating and reproductive systems (Engel and Moran 2013; Douglas 2015). Many are still uncharacterized, and considering the diversity of insects and their differing feeding habits, it is expected that there is a huge potential of novel microbes to be discovered (Dharne et al. 2006). Furthermore, there is an enormous potential for the biodegradation of organic residues via insects and their symbiotic microorganisms. This includes the valorization of organic waste such as manure or food waste and the digestion of otherwise not readily degraded organic residues such as lignin and lignocellulosic plant material into valuable insect biomass.
Waste management systems using omnivorous insects
In numerous publications the potential of the omnivorous black soldier fly (H. illucens) for the bioconversion of manure is emphasized (El Boushy 1991; Sheppard et al. 1994; Diener et al. 2009; Zhou et al. 2013; Lalander et al. 2015). Sheppard et al. (1994) described a so-called manure management system for laying hens using larvae reducing and converting 50 % of the poultry manure and resulting in a potential feed with protein contents of 42 % and fat contents of 35 %. However, within the EU, this system is not applicable for the food or feed production, since a production animal can only be reared on certificated feed which does not include manure. On the other hand, if the insects are intended for non-food applications, rearing on manure is applicable.
For example, due to its high fat content, the potential and feasibility of rearing black soldier fly larvae on manure for the biodiesel production was shown (Li et al. 2011a, b). This is in accordance with findings by Yang and Liu (2014) and Yang et al. (2014b) that concluded that grease derived from swine manure-grown housefly larvae (Yang et al. 2014b) and blowfly (Chrysomya megacephala) larvae (Yang and Liu 2014) could be applied for biodiesel production. It was argued that biodiesel based on fat from fly larvae reared on manure had advantages over crop oil-based biodiesel since its production does not compete with food resources or land use but used waste nutrients for insect growth (Li et al. 2011a). Applications for insect residues such as the protein fraction need to be found for economic use of insect fat for the biodiesel production, however.
Regarding the use of insects for the bioconversion of other organic residues than manure, there are few scientific publications. One example is an investigation of the rearing of the black soldier fly H. illucens on coffee pulp, a residue from the coffee production. For a year-round availability, the coffee pulp had been ensilaged under anaerobic conditions beforehand (Larde 1990). Diener et al. successfully reared black soldier fly larvae on municipal waste in Costa Rica (2011). Furthermore, Žáková und Borkovcová (2013) investigated the weight reduction of 14 different organic wastes such as gardening waste, waste from plant material, food waste, and catering waste by H. illucens on a laboratory scale and observed the highest reduction in waste from plant material. Rearing of H. illucens on compost leachates was also feasible and resulted, in addition to recycling of nitrogen, carbon, and phosphorous and conversion thereof into biomass by insects, in reduced waste disposal costs and reduced harmful environmental effects of the compost leachates (Popa and Green 2012). Hem et al. (2008) showed the potential of H. illucens for the degradation of fermented palm kernel meal, a waste product of the palm oil production, and as feed for aquaculture such as the tilapia culture. Due to its low usability of containing proteins (18 %) and fat (12–20 %) for monogastric animals, palm kernel meal has limited use as feed for example in aquaculture (Heuze et al. 2015) for example in aquaculture directly. Hence, the rearing of insects on fermented palm kernel meal and the resulting conversion into insect biomass signifies a valorization of the palm kernel meal.
Furthermore, the potential of mealworm larvae (Tenebrio molitor) for the degradation of organic residues and as feed for poultry was ascertained. The investigated insect diets with a protein content of 20 % consisted of mealworm beetle excreta, yeast, and different dried organic wastes, for example, from the fruit and vegetable production. Subsequent feeding trials of broiler chicks showed a successful replacement of up to one-third of the soy bean meal present in the diet with dried and ground mealworm larvae (Ramos-Elorduy et al. 2002). Due to its high fat content, the potential of T. molitor for the biodiesel production was also investigated. The larvae were reared on decayed vegetables for 9 weeks, their fat was extracted, and the obtained grease was used for biodiesel production. It was concluded that biodiesel production from fat of T. molitor larvae is feasible (Zheng et al. 2013).
In addition, it was observed, that waxworms (larvae of the Indian mealmoth Plodia interpunctella) were capable of chewing and eating polyethylene (PE) films. PE, a not readily biodegradable synthetic organic compound, could be biodegraded via symbiotic PE-degrading microorganisms in the guts of the waxworm larvae (Yang et al. 2014a). This is promising evidence for the biodegradation of PE by insects and their symbiotic bacteria. However, an incubation time of 60 days with the respective, from the mealmoth larvae isolated PE-degrading microorganisms Enterobacter asburiae YT1 and Bacillus sp. YP1 resulted in a degradation of 6–10 % of a PE film (Yang et al. 2014a), which does not appear to be economically efficient. In comparison, the efficiency of biodegradation of PE films by the waxmoth and by a combination of both isolated bacteria would be interesting.
All those examples emphasize the general potential of insects for the bioconversion of organic residues. However, there is still research required on the suitability of rearing respective insect species on respective organic waste and by-products, if a pretreatment like a drying or fermentation step of the organic residue is necessary, and how the conversion rate to insect biomass can be optimized. In addition, an added value of the bioconversion of organic residues via insects in comparison to direct application of those organic residues for example in feed needs to be verified.
Degradation of lignin by insects
In contrast to the industrial fermentation of sugar and starch containing plant material to ethanol, the production of so-called second-generation biofuels from agricultural residues remains inefficient and expensive (Brune 2014). This is due to the natural resistance of the plant cell wall against enzymatic or microbial degradation (biomass recalcitrance) (Himmel et al. 2007). For technical processing, chemical and physical pretreatment steps are performed for the separation of lignin. This increases the ethanol yield but results in non-degraded lignin as a by-product. For an efficient biofuel production from lignin-containing biomass, cost as well as energy-efficient, environmentally friendly processes for the complete degradation of lignified biomass and more sustainable methods for lignin degradation are urgently needed.
A biodegradation of lignin can be achieved using wood decay fungus such as white rot, bacteria, soil bacteria, ruminants and wood-feeding (xylophagous) insects. As aforementioned, insects are divers and represent an enormous bioresource, but they are yet mostly unexploited in technological processes. Generally, insects possess several for technical reasons interesting properties such as resource efficiency, low space requirements, and depending on the species and rearing conditions, short life cycles. Due to their feeding habits, insects have a high potential for the organic waste degradation and valorization. Of high interest in this regard are, in addition to omnivorous or saprophagous species, especially lignivorous insects that are able to degrade lignocellulose.
Well-known wood-degrading insects include species from the orders Isoptera (termites), Coleoptera (beetles), and Blattodea (cockroaches). Termites belong to the most important lignocellulose-digesting insects. In a first step, they mechanically grind the wood particles into micrometer-size particles by their mandibles and mix them with cellulolytic enzymes excreted from their salivary glands. Subsequently, the lignocellulose is digested symbiotically by the termites’ own enzymes and enzymes from the symbiotic microbiota of the termite gut, a lignocellulose-degrading symbiotic ecosystem (Koenig et al. 2013), also referred to as a “minute intestinal bioreactor” (Brune 2014). Additionally, a lignin degradation (side-chain oxidation, ring hydroxylation, and demethylation) was verified (Geib et al. 2008). The digestion of the feed takes from several hours (Geib et al. 2008) to 24 h (Koenig et al. 2013). This leads to the conclusion that termites not only degrade wood considerably more efficient than fungi by themselves, but that termites are also superior to biogas plants if retention times of energy plants are considered (Koenig et al. 2013).
Due to their high efficiency, termites represent a promising alternative for the industrial conversion of lignocellulose and lignin for the production of biofuels. In addition to the industrial fermentation of lignified plant fibers to biofuels, their enzymatic systems open up opportunities for biotechnological applications and the production of novel enzymes (Brune 2014). However, disadvantageous for a technical application is the rather difficult breeding of termites being social insects in comparison to solitarily living insect species.
Xylophagous beetles include for example the Asian long-horned beetle (Anoplophora glabripennis) that just like termites has the ability to degrade lignin. Lignin-degrading reactions such as side-chain oxidation or elimination, ring hydroxylation, and demethylation, analogous to lignin degradation by fungus or termites, have been observed. Similarly to termites, there are indications for the development of a symbiosis between the beetle and his gut microbiota that enables the digestion of living wood tissue, not attacked by fungus (Geib et al. 2008).
A xylophagous species of the order Blattodea (cockroaches) is Cryptocercus punctulatus, whose intestinal microflora is already well researched (Berlanga et al. 2009). However, there is not much mentioned in scientific publications about the degradation pathways of lignin or lignin-containing biomass. A molecular phylogenetic comparison resulted in a close relation between cockroaches and termites. Cockroaches are considered as paraphyletic. This is reflected by the bacterial communities in their guts (Schauer et al. 2012) and leads to the assumption that xylophagous species of the order Blattodea have the ability to digest lignin and lignified biomass.
It can be concluded that xylophagous insects represent a potential to date unexploited biological system, an organic bioreactor, for the industrial degradation of lignin and lignified biomass. It needs to be investigated which insect species is generally appropriate for what kind of biomass biodegradation and to what extent a utilization of industrial rearing systems of certain species is advantageous and feasible. In addition, utilizing of wood and other pests, it needs to be ascertained at all costs that an escape into the environment is prevented. Depending on the selected species, the utilization of the degradation products after conversion of the lignin into biomass needs to be evaluated.
Insect biotechnology: production of bioactive ingredients
Semiochemicals
Insects produce a high variety of multifunctional natural products functioning, for example, as attractants or repellants such as semiochemicals (pheromones, allomones, kairomones). Those bioactive agents are either produced by the insect itself or produced by symbiotic microorganisms, or precursors are sequestered from plants and other animals (Blum 1996). These insect-derived natural bioactive products offer a variety of valuable applications.
One example is cantharidin, a monoterpene produced by beetles of the families Meloidae and Oedemeridae as chemical defense as well as attractant (Frenzel and Dettner 1994). Its anticancer and antitumor activities have been shown but due to its extreme toxicity for mammals its application is limited. Therefore, derivatives and analogues with higher bioactivity, higher selectivity, and lower toxicity were developed as potential chemotherapeutic anticancer agents (Puerto Galvis et al. 2013). Antitumor activity was also shown for pederin, a vesicant toxic amide from a rove beetle of the genus Paederus used as a chemical defense against predators. Pederin, which is suggested to be synthesized by a bacterial symbiont of female beetles, blocks protein biosynthesis (Kellner and Dettner 1995; Piel 2002). It was furthermore shown that a cloning of genes responsible for the biosynthesis of invertebrate products such as pederin from bacterial symbionts without the need for culturing was possible enabling microbial production of these products (Piel 2002).
Antimicrobial peptides
Additionally, insects represent a rich resource for antimicrobial peptides (Vilcinskas and Gross 2005) produced by the insects themselves or their residential microorganisms for the defense against microbes, including bacteria, fungi, viruses, and protozoa (Zasloff 2002). For example, an isolation of bacilli from different compartments of the guts of several insect species and subsequent screening for biologically active metabolites resulted in peptides with antibacterial or antifungal activity (Gebhardt et al. 2002). Furthermore, antiviral and antitumor capacities were observed. For example, alloferons, slightly cationic peptides isolated from the blood of an experimentally infected blow fly Calliphora vicina showed enhanced antitumor and antiviral resistance in mice. Evidence was found for antiviral activity against influenza viruses, herpes simplex and hepatitis B and C infections (Chernysh et al. 2002).
Although a number of antimicrobial peptides with promising bioactivities have been isolated from insects, their commercial use is impeded by factors like loss of bioactivity under physiological conditions, low peptide stability, potential toxicity, high costs, difficulties in mass production and legal approval. This can be overcome by the creation of improved synthetic analogues and derivatives (Hull et al. 2012). With the pressing need for novel antibiotics and since resistance of pathogens against antimicrobial peptides is less probable than observed for conventional antibiotics, antimicrobial peptides open up new opportunities for the creation of future antibiotics and other chemotherapeutic agents (Zasloff 2002; Hull et al. 2012).
In addition, antimicrobial peptides are a novel approach for the engineering of disease resistant crops (Vilcinskas and Gross 2005). For example, a novel antimicrobial peptide from the Common Green Bottle Fly Lucilia sericata has been discovered. This peptide, named lucimycin, has antifungal activity against fungal plant pathogens, among others against an oomycete of the genus Phytophtora (Poeppel et al. 2014). Phytophtora infestans is the most important potato plant pest called late blight and responsible for the great Irish famine in the nineteenth century and still causes losses of over 3 billion US dollars each year worldwide (Bengyella and Pranab 2010). For the reduction in potato harvest losses and a higher resistance to the late blight, it is aimed to develop transgenic potato plants expressing lucimycin (Poeppel et al. 2014). This shows the potential of insect biotechnology and the genetic exploitation of defense mechanisms of insects.
Conclusion: challenges and opportunities exploiting insects
Overall, it can be summarized that insects are a highly diverse and vastly underutilized bioresource with many potential applications. This includes the bioconversion and valorization of organic residues and the production of food, feed, chemicals, enzymes, and bioactive compounds. Selected examples are given in Table 1.
Due to their high nutritional value, insects have a long tradition as a food source in many parts of the world. They offer a great opportunity as a sustainable animal protein source and represent a prospective resource-efficient alternative to meat in the human diet. Furthermore, food insects have the potential to contribute to global food as well as protein security. Challenges for food insects in the so-called Western world include particularly legal barriers and consumer acceptance. In regions where entomophagy is traditionally practiced, the development of safe and sustainable mass-rearing systems for the production of food insects is required, since an increasing insect consumption based on gathering will lead to overexploitation and endangering of species in flora and fauna. In addition, before cultivation and application of foreign insect species, their potential hazard on the indigenous ecosystem has to be considered to prevent a negative impact on the ecosystem and the endangerment of indigenous species.
The use of sustainable insect-based feed has also the potential to contribute to protein security and biodiversity. For example, a replacement of soy and fish meal in feed can contribute to forest and aquatic diversity. Although the general applicability of insects as feed has been shown and insects represent a natural feed source especially for poultry and fish, EU regulations to date prohibit the use of processed insects in feed. A revision of the respective regulations is expected.
Generally, the potential of insects for the bioconversion of organic residues such as manure, food waste, urban waste, even PE and lignocellulosic residues into insect biomass was shown. For organic waste degradation and especially for manure degradation, application of flies and moreover of the omnivorous black soldier fly (H. illucens) prevailed. It can be concluded that such insects have a huge potential for organic waste reduction and valorization and concomitant bioconversion to valuable insect biomass to be applied for example as feed or for the biodiesel production. Here, holistic and sustainable approaches are required.
In addition, the degradation of lignocellulose for the biofuel production by wood-feeding insects has a high potential and represents a sustainable alternative for the biofuel production. Moreover, insects can be applied as models for the industrial lignin digestion and be exploited for the production of enzymes. However, it has to be ensured that the lignivorous insects cannot escape into the environment to preserve trees, forests and facilities constructed from wood.
Studying communication strategies and defense mechanisms against predators and microbes of insects, it was discovered that they sequester or produce a multitude of remarkable chemical substances such as toxins and antimicrobial peptides that can be exploited for pest control or the creation of novel therapeutics. Antimicrobial peptides open up new opportunities for the creation of future antibiotics and antitumor and anticancer agents. Furthermore, they can be exploited for the genetic engineering of disease resistant crops. As aforementioned, challenges for the commercial use of antimicrobial peptides include loss of bioactivity under physiological conditions, low peptide stability, potential toxicity, high costs, difficulties in mass production and legal approval. The development of more efficient, non-toxic, and more stable synthetic analogues and derivatives offers possible solutions to these problems.
Overall, it can be concluded that there is still a large amount of research required regarding identification and application of known and to date unknown insect species appropriate for respective bioconversion processes and other applications in order to extensively exploit the potential of insects as a bioresource.
References
Anand H, Ganguly A, Haldar P (2008) Potential value of Acridids as high protein supplement for poultry feed. Int J Poult Sci 7:722–725
Barroso FG, de Haro C, Sanchez-Muros M-J, Venegas E, Martinez-Sanchez A, Perez-Banon C (2014) The potential of various insect species for use as food for fish. Aquaculture 422:193–201. doi:10.1016/j.aquaculture.2013.12.024
Bengyella L, Pranab R (2010) Engineered pathogenesis related and antimicrobial proteins weaponry against Phytopthora infestans in potato plant: a review. Biotechnol Mol Biol Rev 5:61–66
Berlanga M, Paster BJ, Guerrero R (2009) The taxophysiological paradox: changes in the intestinal microbiota of the xylophagous cockroach Cryptocercus punctulatus depending on the physiological state of the host. Int Microbiol 12:227–236. doi:10.2436/20.1501.01.102
Blum MS (1996) Semiochemical parsimony in the Arthropoda. Annu Rev Entomol 41:353–374. doi:10.1146/annurev.en.41.010196.002033
Bondari K, Sheppard DC (1987) Soldier fly Hermetia illucens L. larvae as feed for channel catfish, Ictalurus punctatus (Rafinesque), and blue tilapia Oreochromis aureus (Steindacher). Aquac Fish Manag 18:209–220. doi:10.1111/j.1365-2109.1987.tb00141.x
Brune A (2014) Symbiotic digestion of lignocellulose in termite guts. Nat Rev Microbiol 12:168–180. doi:10.1038/nrmicro3182
Chernysh S, Kim SI, Bekker G, Pleskach VA, Filatova NA, Anikin VB, Platonov VG, Bulet P (2002) Antiviral and antitumor peptides from insects. Proc Natl Acad Sci USA 99:12628–12632. doi:10.1073/pnas.192301899
Dharne M, Patole M, Shouche YS (2006) Microbiology of the insect gut: tales from mosquitoes and bees. J Biosci 31:293–295. doi:10.1007/bf02704100
Diener S, Zurbruegg C, Tockner K (2009) Conversion of organic material by black soldier fly larvae: establishing optimal feeding rates. Waste Manag Res 27:603–610. doi:10.1177/0734242x09103838
Diener S, Solano NMS, Gutierrez FR, Zurbruegg C, Tockner K (2011) Biological treatment of municipal organic waste using black soldier fly larvae. Waste Biomass Valoriz 2:357–363. doi:10.1007/s12649-011-9079-1
Douglas AE (2015) Multiorganismal insects: diversity and function of resident microorganisms. Annu Rev Entomol 60:17–34. doi:10.1146/annurev-ento-010814-020822
EFSA (2015) Risk profile related to production and consumption of insects as food and feed. EFSA J 13:4257. doi:10.2903/j.efsa.2015.4257
El Boushy AR (1991) House-fly pupae as poultry manure converters for animal feed: a review. Bioresour Technol 38:45–49. doi:10.1016/0960-8524(91)90220-e
Engel P, Moran NA (2013) The gut microbiota of insects: diversity in structure and function. Fems Microbiol Rev 37:699–735. doi:10.1111/1574-6976.12025
FAOstat (2014) http://faostat3.fao.org/home/E. Accessed 18 Dec 2014
Fearnside PM (2001) Soybean cultivation as a threat to the environment in Brazil. Environ Conserv 28:23–38
Feedipedia.org (2015) www.feedipedia.org. Accessed 4 Nov 2015
Frenzel M, Dettner K (1994) Quantification of cantharidin in canthariphilous ceratopogonidae (Diptera), anthomyiidae (Diptera) and cantharidin-producing oedemeridae (Coleoptera). J Chem Ecol 20:1795–1812. doi:10.1007/bf02066223
Gebhardt K, Schimana J, Muller J, Fiedler HP, Kallenborn HG, Holzenkampfer M, Krastel P, Zeeck A, Vater J, Hotzel A, Schmid DG, Rheinheimer J, Dettner K (2002) Screening for biologically active metabolites with endosymbiotic bacilli isolated from arthropods. Fems Microbiol Lett 217:199–205. doi:10.1016/s0378-1097(02)01065-0
Geib SM, Filley TR, Hatcher PG, Hoover K, Carlson JE, Jimenez-Gasco MdM, Nakagawa-Izumi A, Sleighter RL, Tien M (2008) Lignin degradation in wood-feeding insects. Proc Natl Acad Sci USA 105:12932–12937. doi:10.1073/pnas.0805257105
Grimaldi DA, Engel MS (2005) The evolution of insects. Cambridge University Press, Cambridge
Hem S, Toure S, Sagbla C, Legendre M (2008) Bioconversion of palm kernel meal for aquaculture: experiences from the forest region (Republic of Guinea). Afr J Biotechnol 7:1192–1198
Heuze V, Tran G, Sauvant D, Noblet J, Renaudeau D, Bastianelli D, Lebas F (2015) Feedipedia: palm kernel meal. http://www.feedipedia.org/node/43. Accessed 4 Nov 2015
Himmel ME, Ding S-Y, Johnson DK, Adney WS, Nimlos MR, Brady JW, Foust TD (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315:804–807. doi:10.1126/science.1137016
Hull R, Katete R, Ntwasa M (2012) Therapeutic potential of antimicrobial peptides from insects. Biotechnol Mol Biol Rev 7:31–47
Hwangbo J, Hong EC, Jang A, Kang HK, Oh JS, Kim BW, Park BS (2009) Utilization of house fly-maggots, a feed supplement in the production of broiler chickens. J Environ Biol 30:609–614
Ijaiya AT, Eko EO (2009) Effect of replacing dietary fish meal with silkworm (Anaphe infracta) caterpillar meal on growth, digestibility and economics of production of starter broiler chickens. Pak J Nutr 8:845–849
Jongema Y (2015) List of edible insects of the world (June 1, 2015). Laboratory of entomology, Wageningen University. http://www.ent.wur.nl/UK/Edible+insects/Worldwide+species+list/. Accessed 1 Oct 2015
Kellner RLL, Dettner K (1995) Allocation of pederin during lifetime of Paederus rove beetles (Coleoptera: Staphylinidae): evidence for polymorphism of hemolymph toxin. J Chem Ecol 21:1719–1733. doi:10.1007/bf02033672
Kenis M, Auger-Rozenberg MA, Roques A, Timms L, Pere C, Cock M, Settele J, Augustin S, Lopez-Vaamonde C (2009) Ecological effects of invasive alien insects. Biol Invasions 11:21–45. doi:10.1007/s10530-008-9318-y
Koenig H, Li L, Froehlich J (2013) The cellulolytic system of the termite gut. Appl Microbiol Biotechnol 97:7943–7962. doi:10.1007/s00253-013-5119-z
Kroeckel S, Harjes AGE, Roth I, Katz H, Wuertz S, Susenbeth A, Schulz C (2012) When a turbot catches a fly: evaluation of a pre-pupae meal of the black soldier fly (Hermetia illucens) as fish meal substitute: growth performance and chitin degradation in juvenile turbot (Psetta maxima). Aquaculture 364:345–352. doi:10.1016/j.aquaculture.2012.08.041
Lalander CH, Fidjeland J, Diener S, Eriksson S, Vinneras B (2015) High waste-to-biomass conversion and efficient Salmonella spp. reduction using black soldier fly for waste recycling. Agron Sustain Dev 35:261–271. doi:10.1007/s13593-014-0235-4
Larde G (1990) Recycling of coffee pulp by Hermetia illucens (Diptera, Stratiomyidae) larvae. Biol Wastes 33:307–310. doi:10.1016/0269-7483(90)90134-e
Lee CG, Da Silva CA, Lee J-Y, Hartl D, Elias JA (2008) Chitin regulation of immune responses: an old molecule with new roles. Curr Opin Immunol 20:684–689. doi:10.1016/j.coi.2008.10.002
Li Q, Zheng L, Cai H, Garza E, Yu Z, Zhou S (2011a) From organic waste to biodiesel: black soldier fly, Hermetia illucens, makes it feasible. Fuel 90:1545–1548. doi:10.1016/j.fuel.2010.11.016
Li Q, Zheng L, Qiu N, Cai H, Tomberlin JK, Yu Z (2011b) Bioconversion of dairy manure by black soldier fly (Diptera: Stratiomyidae) for biodiesel and sugar production. Waste Manag 31:1316–1320. doi:10.1016/j.wasman.2011.01.005
Makkar HPS, Tran G, Henze V, Ankers P (2014) State-of-the-art on use of insects as animal feed. Anim Feed Sci Technol 197:1–33. doi:10.1016/j.anifeedsci.2014.07.008
Muzzarelli RAA (2010) Chitins and chitosans as immunoadjuvants and non-allergenic drug carriers. Mar Drugs 8:292–312. doi:10.3390/md8020292
Naylor RL, Goldburg RJ, Primavera JH, Kautsky N, Beveridge MCM, Clay J, Folke C, Lubchenco J, Mooney H, Troell M (2000) Effect of aquaculture on world fish supplies. Nature 405:1017–1024. doi:10.1038/35016500
Naylor RL, Hardy RW, Bureau DP, Chiu A, Elliott M, Farrell AP, Forster I, Gatlin DM, Goldburg RJ, Hua K, Nichols PD (2009) Feeding aquaculture in an era of finite resources. Proc Natl Acad Sci USA 106:15103–15110. doi:10.1073/pnas.0905235106
Nowak V, Persijn D, Rittenschober D, Charrondiere UR (2016) Review of food composition data for edible insects. Food Chem 193:39–46. doi:10.1016/j.foodchem.2014.10.114
Ogunji JO, Kloas W, Wirth M, Neumann N, Pietsch C (2008) Effect of housefly maggot meal (magmeal) diets on the performance, concentration of plasma glucose, cortisol and blood characteristics of Oreochromis niloticus fingerlings. J Anim Physiol Anim Nutr 92:511–518. doi:10.1111/j.1439-0396.2007.00745.x
Olaniyi CO, Salau BR (2013) Utilization of maggot meal in the nutrition of African cat fish. Afr J Agric Res 8:4604–4607
Oliveira-Goumas B (2004) European parliament directorate general for research working paper: the fish meal and fish oil industry its role in the common fisheries policy (FISH 113 EN) vol FISH 113 EN. European Parliament Luxemburg
Oonincx DGAB, de Boer IJM (2012) Environmental impact of the production of mealworms as a protein source for humans: a life cycle assessment. PLoS One. doi:10.1371/journal.pone.0051145
Oonincx DGAB, van Itterbeeck J, Heetkamp MJW, van den Brand H, van Loon JJA, van Huis A (2010) An exploration on greenhouse gas and ammonia production by insect species suitable for animal or human consumption. PLoS One. doi:10.1371/journal.pone.0014445
Payne CLR (2015) Wild harvesting declines as pesticides and imports rise: the collection and consumption of insects in contemporary rural Japan. J Insects Food Feed 1:57–65. doi:10.3920/JIFF2014.0004
Piel J (2002) A polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles. Proc Natl Acad Sci USA 99:14002–14007. doi:10.1073/pnas.222481399
Poeppel A-K, Koch A, Kogel K-H, Vogel H, Kollewe C, Wiesner J, Vilcinskas A (2014) Lucimycin, an antifungal peptide from the therapeutic maggot of the common green bottle fly Lucilia sericata. Biol Chem 395:649–656. doi:10.1515/hsz-2013-0263
Popa R, Green TR (2012) Using black soldier fly larvae for processing organic leachates. J Econ Entomol 105:374–378. doi:10.1603/ec11192
Post K, Riesner D, Walldorf V, Mehlhorn H (1999) Fly larvae and pupae as vectors for scrapie. Lancet 354:1969–1970. doi:10.1016/s0140-6736(99)00469-9
Puerto Galvis CE, Vargas Mendez LY, Kouznetsov VV (2013) Cantharidin-based small molecules as potential therapeutic agents. Chem Biol Drug Des 82:477–499. doi:10.1111/cbdd.12180
Ramos-Elorduy J (2006) Threatened edible insects in Hidalgo, Mexico and some measures to preserve them. J Ethnobiol Ethnomed 2:51. doi:10.1186/1746-4269-2-51
Ramos-Elorduy J, Moreno JM, Prado E, Perez M, Otero J, Larron De Guevara O (1997) Nutritional value of edible insects from the state of Oaxaca, Mexico. J Food Compos Anal 10:142–157. doi:10.1006/jfca.1997.0530
Ramos-Elorduy J, Pino-M JM, Correa SC (1998) Insectos comestibles del Estado de México y determinación de su valor nutritivo. Anales Inst Biol Univ Nac Autón México Ser Zool 69(1):65–104
Ramos-Elorduy J, Gonzalez EA, Hernandez AR, Pino JM (2002) Use of Tenebrio molitor (Coleoptera: Tenebrionidae) to recycle organic wastes and as feed for broiler chickens. J Econ Entomol 95:214–220. doi:10.1603/0022-0493-95.1.214
Rumpold BA, Schlüter OK (2013a) Nutritional composition and safety aspects of edible insects. Mol Nutr Food Res 57:802–823. doi:10.1002/mnfr.201200735
Rumpold BA, Schlüter OK (2013b) Potential and challenges of insects as an innovative source for food and feed production. Innov Food Sci Emerg Technol 17:1–11. doi:10.1016/j.ifset.2012.11.005
Rumpold BA, Schlüter OK (2014) Nutrient composition of insects and their potential application in food and feed in Europe. Food Chain 4:129–139. doi:10.3362/2046-1887.2014.013
Schauer C, Thompson CL, Brune A (2012) The bacterial community in the gut of the cockroach Shelfordella lateralis reflects the close evolutionary relatedness of cockroaches and termites. Appl Environ Microbiol 78:2758–2767. doi:10.1128/aem.07788-11
Sealey WM, Gaylord TG, Barrows FT, Tomberlin JK, McGuire MA, Ross C, St-Hilaire S (2011) Sensory analysis of rainbow trout, Oncorhynchus mykiss, fed enriched black soldier fly prepupae, Hermetia illucens. J World Aquac Soc 42:34–45. doi:10.1111/j.1749-7345.2010.00441.x
Sheppard CD, Newton LG, Thompson SA, Savage S (1994) A value added manure management system using the black soldier fly. Bioresour Technol 50:275–279. doi:10.1016/0960-8524(94)90102-3
Stamer A, Wesselss S, Neidigk R, Hoerstgen-Schwark G (2014) Black soldier fly (Hermetia illucens) larvae-meal as an example for a new feed ingredient’s class in aquaculture diets. Paper presented at the 4th ISOFAR scientific conference “Building Organci Bridges”, at the organic world conference, Istanbul, Turkey, 13–15 Oct
St-Hilaire S, Cranfill K, McGuire MA, Mosley EE, Tomberlin JK, Newton L, Sealey W, Sheppard C, Irving S (2007a) Fish offal recycling by the black soldier fly produces a foodstuff high in omega-3 fatty acids. J World Aquac Soc 38:309–313. doi:10.1111/j.1749-7345.2007.00101.x
St-Hilaire S, Sheppard C, Tomberlin JK, Irving S, Newton L, McGuire MA, Mosley EE, Hardy RW, Sealey W (2007b) Fly prepupae as a feedstuff for rainbow trout, Oncorhynchus mykiss. J World Aquac Soc 38:59–67. doi:10.1111/j.1749-7345.2006.00073.x
van der Spiegel M, Noordam MY, van der Fels-Klerx HJ (2013) Safety of novel protein sources (Insects, Microalgae, Seaweed, Duckweed, and Rapeseed) and legislative aspects for their application in food and feed production. Compr Rev Food Sci Food Saf 12:662–678. doi:10.1111/1541-4337.12032
van Huis A, van Itterbeeck J, Klunder HC, Mertens E, Halloran A, Muir G, Vantomme P (2013) Edible insects: future prospects for food and feed security vol 171. FAO Forestry paper. FAO, Rome
Veldkamp T, van Duinkerken G, Van Huis A, Lakemond CMM, Ottevanger E, Bosch G, Van Boekel MAJS (2012) Insects as a sustainable feed ingredient in pig and poultry diets - a feasibility study. Wageningen UR Livestock Research, Lelystad. https://www.wageningenur.nl/upload_mm/2/8/0/f26765b9-98b2-49a7-ae43-5251c5b694f6_234247%5B1%5D
Vilcinskas A, Gross J (2005) Drugs from bugs: the use of insects as a valuable source of transgenes with potential in modern plant protection strategies. J Pest Sci 78:187–191. doi:10.1007/s10340-005-0114-5
Wang D, Zhai SW, Zhang CX, Bai YY, An SH, Xu YN (2005) Evaluation on nutritional value of field crickets as a poultry feedstuff Asian-Australas. J Anim Sci 18:667–670
Yang S, Liu Z (2014) Pilot-scale biodegradation of swine manure via Chrysomya megacephala (Fabricius) for biodiesel production. Appl Energy 113:385–391. doi:10.1016/j.apenergy.2013.07.056
Yang J, Yang Y, Wu W-M, Zhao J, Jiang L (2014a) Evidence of polyethylene biodegradation by bacterial strains from the guts of plastic-eating waxworms. Environ Sci Technol 48:13776–13784. doi:10.1021/es504038a
Yang S, Li Q, Gao Y, Zheng LY, Liu ZD (2014b) Biodiesel production from swine manure via housefly larvae (Musca domestica L.). Renew Energy 66:222–227. doi:10.1016/j.renene.2013.11.076
Žáková M, Borkovcová M (2013) Hermetia illucens application in management of selected types of organic waste. In: The 2nd electronic international interdisciplinary conference, vol 1, 2–6 Sept 2013. EDIS—Publishing Institution of the University of Zilina, pp 367–370. http://www.eiic.cz/archive/?vid=1&aid=2&kid=20201-33, http://www.eiic.cz/archive/?vid=1&aid=3&kid=20201-33&q=f1
Zasloff M (2002) Antimicrobial peptides of multicellular organisms. Nature 415:389–395. doi:10.1038/415389a
Zheng L, Hou Y, Li W, Yang S, Li Q, Yu Z (2013) Exploring the potential of grease from yellow mealworm beetle (Tenebrio molitor) as a novel biodiesel feedstock. Appl Energy 101:618–621. doi:10.1016/j.apenergy.2012.06.067
Zhou F, Tomberlin JK, Zheng L, Yu Z, Zhang J (2013) Developmental and waste reduction plasticity of three black soldier fly strains (Diptera: Stratiomyidae) raised on different livestock manures. J Med Entomol 50:1224–1230. doi:10.1603/me13021
Acknowledgments
This study was supported by the seed money program of the Leibniz Network on Biodiversity (LVB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Rumpold, B.A., Klocke, M. & Schlüter, O. Insect biodiversity: underutilized bioresource for sustainable applications in life sciences. Reg Environ Change 17, 1445–1454 (2017). https://doi.org/10.1007/s10113-016-0967-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10113-016-0967-6