Abstract
Photodynamic antimicrobial chemotherapy (PACT) is an antimicrobial approach that uses photosensitizers (PS) in combination with light sources at specific wavelengths aiming the production of reactive oxygen species. The long illumination time necessary to active PS is a challenge in PACT. Thus, this study investigated the antimicrobial effect of a novel single source of light-emitting diode (LED) light that covers the entire spectrum of visible light beyond interchangeable probes at high power intensity. Blue and red LED probes were used into different exposure times to active different concentrations of curcumin (C) and toluidine blue (T) on planktonic suspensions of Streptococcus mutans UA 159 (S. mutans). S. mutans were standardized and submitted to (1) PACT treatment at three concentrations of C and T exposure at three radiant exposures of a blue LED (BL) (C+BL+) and a red LED (RL) (T+RL+), (2) C (C+BL−) or T alone (T+RL−), (3) both LED lights (C−BL+ and T−RL+), and (4) neither PS nor LED illumination (control group: C−BL− and T−RL−). Aliquots of the suspensions were diluted and cultured on blood agar plates. The number of colony-forming units was calculated after 48 h. The groups submitted to PACT presented a lethal photokilling rate to all PS concentrations at tested dosimetries. The comparison to control group when PS and LED lights used alone demonstrated no decrease in the number of viable bacterial counts. The novel LED device in combination with curcumin and toluidine blue promoted an effective photoinactivation of S. mutans suspensions at ultrashort light illumination times.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
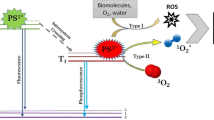
Photodynamic antimicrobial chemotherapy (PACT) is a therapy modality that employs the combination of visible light and photosensitizers (PS) or dyes [1]. The PS binds the membrane of target cell, thus, exposed to visible light at specific wavelength (specific for each PS) that leads the production of different reactive oxygen species (ROS) such as singlet oxygen generating a sequence of biological events as apoptosis of the cells or death of the microorganisms [2, 3].
As known, dental caries is an infectious disease caused by the unbalance of the microorganisms present in the oral cavity [4]. Streptococcus mutans has been implicated as a cariogenic bacteria because of its relatively high numbers in plaque prior to the appearance of carious lesions, its capacity for rapid degradation of carbohydrates with the formation of abundant acid, and its ability to tolerate low pH environments [5, 6].
PACT is emerging as a suitable process to reduce bacterial contamination in oral cavity, increasing the therapeutical success [7]. Due to multifactorial role of this disease, PACT presents advantages in relation to the most common treatments, including mechanical debridement, which includes selective targeting, noninvasive nature, easy repeatability, and rapidness besides offering good result at low costs [5, 8].
PACT has been applied by the combination of several PS with different light sources [7, 9, 10]. An ideal PS should have a low toxicity to host cells when is not activated by the light. It should present a pure composition, stable shelf life, ideally water solubility or be soluble in a harmless aqueous solvent mixture, and produce a high ROS rate in a short period of illumination time [11–13]. None of the available PS fulfills all of these characteristics; however, some investigations have indicated that toluidine blue (TBO) exhibits phototoxicity against a broad range of bacteria at low densities of energy, it is a small phenothiazine dye molecule, and it does not cause in vitro toxicity or DNA damage to keratinocytes [14–16]. On the other hand, the use of this dye might cause tooth and restoration staining, which makes its applicability a clinical challenge [7].
Curcumin is a natural yellow pigment extracted from the rhizomes of Curcuma longa and presents a wide range of pharmacological effects, including anti-inflammatory, anti-carcinogenic, and anti-infection activities [17, 18]. Several studies have attested its photokilling property in PACT when activated with proper light sources [19, 20]. Still, affordable costs, easy handling, and great effectiveness represent advantages on the use of this PS [21, 22]. In addition, no burning sensation, oral soreness, or ulcers were found when curcumin was utilized in an in vivo approach [23]. However, its solubility in water is highly limited being indicated the use of oils and synthetic solvents to allow its dissolution. Overall, both PS when exposed to proper light wavelengths are able to membrane diffusion by lipid membrane peroxidation generating strand breaks in the organism's nucleic acid, with resultant genetic mutation and photodamage [19, 24].
One of the disadvantages of PACT is the time for PS activation by the light. Teixeira et al. [25] achieved significant decreases in the viability of in vitro biofilms of S. mutans, but only after 15 min of irradiation using a red light-emitting diode (LED) (55 J cm−2) in the presence of TBO at 100 μg ml−1. A time of almost 30 min was necessary to photoinactivate Candida species using curcumin activated by an LED with maximum emission at 455 nm [21]. Therefore, the novel sources of light that could enhance the photodynamic efficacy by producing high ROS proportions at short period of time for the translation of PACT from the laboratory to a clinical application are highly needed.
The present study aimed to investigate the antimicrobial effect of a novel source of noncoherent light in the activation of TBO and curcumin by two different interchangeable probes that cover the red and blue spectrum of light on planktonic suspensions of S. mutans. This is the first step for the improvement of a potential use of PACT for the treatment of oral infections.
Materials and methods
Light source
A single source of a noncoherent light that produces the full spectrum of visible light was used (LumaCare LC-122 A, LumaCare Medical Group, Newport Beach, CA, USA). This light offers interchangeable flat probes (beam diameter = 12 mm; spot area = 113.1 mm2) at specific frequencies that are connected with a simple interlocking connection. To this study, two different fiber optic probes were used to cover the blue and red light spectrum. The blue light (wavelength range, 400–440 nm) was used to activate curcumin and presented an output range with a central wavelength peak at 420 ± 20 nm at power density (Pd) of 95.5 mW cm−2 (set power (P) = 105 mW). The radiant exposures tested to this specific wavelength were 24, 48, and 72 J.cm−2. To achieve these values, light irradiation time was calculated according to a previous study [26]. Thus, the suspensions contained in round-shape acrylic containers (bacterial inoculum and photosensitizer at equal volumes−100 μL−final volume = 200 μL; diameter = 8 mm; area = 50 mm2) were irradiated for 252, 505, and 757 s with the samples receiving the total dose of 12, 24, and 36 J, respectively. The red light (wavelength range, 570–690 nm) provided an absorption spectra with a central wavelength at 635 ± 10 nm with a Pd of 1,460 mW cm−2 (set power (P) = 1,650 mW) and 18, 35, and 53 J cm−2 of radiant exposures were tested to irradiate TBO. The exposure times corresponding to each tested radiant exposures were 12, 24, and 36 s with the samples receiving the total dose of 9, 17.5, and 26.5 J, respectively. A work distance to both PS was fixed at 5 mm that corresponds to the distance between the light source and cell line surface—safe distance to avoid heating sample as stated by manufacturer.
Photosensitizers
To this present investigation, curcumin (C) (Sigma-Aldrich, St. Louis, MI, USA) was dissolved in dimethyl sulfoxide (DMSO) to obtain a stock solution of 50 mM. On the day of the experiment, this solution was diluted in deionized water to obtain final concentrations at 2.5, 1.25, and 0.75 mM (keeping the final concentration of DMSO at 10 % for all of them). For the toluidine blue (T) (Sigma-Aldrich, St. Louis, MI, USA), a 1,000-μM stock solution was prepared in deionized water and subsequently dissolved at concentrations of 100, 50, and 25 μM. On the day of the experiment, both PS stock solutions were diluted and kept in the dark until its use.
Preparation of the microorganisms
A standard suspension of S. mutans (ATCC 700610 UA 159) was inoculated in buffered tryptone yeast extract containing 1 % (wt/vol) glucose and incubated for 18–24 h at 37 °C under microaerophilic condition (5 % of CO2). This bacterial culture was then centrifuged at 4,000 rpm for 5 min and the supernatant was discarded. Then, the cell pellet was re-suspended in sterile phosphate buffered saline, and the number of cells was adjusted by means of a spectrophotometer (wavelength at 540 nm using a 1.5 optical density unit equals ~ 2 × 109 cells mL−1).
Photodynamic therapy application
S. mutans suspensions were incubated with different concentrations of tested PS at a final volume of 200 μL distributed in container with 8 mm diameter. Curcumin solution was kept in the dark with S. mutans suspension for 60 s (pre-irradiation time) [26] whereas TBO was kept for 5 min [27]. After the incubation time, the solution (PS/bacterial suspension) was irradiated to achieve the tested radiant exposures to both PSs. PACT groups were treated with both PS and light (C+BL+ and T+RL+, where C, curcumin; T, TBO; BL, blue LED; and RL, red LED). Figures 1 and 2 show the diagram of PACT application to curcumin and TBO, respectively. To determine whether PS alone induced any toxic effects on bacterial viability, bacteria suspension were exposed to PS under identical conditions to those described above, but not exposed to LED (treated only with C or T: group C+BL− and T+RL−). Exposing cells to irradiation determined the isolated effect of blue and red LED with no previous exposure to PS (treated only with blue LED and red LED: group C−BL+ and T−RL+). The control situations consisted of S. mutans suspensions not exposed to PS or LED lights (control groups: C−BL− and T−RL−). Aliquots of suspensions after treatments were used to perform tenfold serial dilutions, and the diluted samples were plated onto 5 % defibrinated sheep blood agar (Sigma Chemicals, Co.) and then incubated at 37 °C, 5 % CO2 for 48 h to investigate the number of viable microorganisms. After incubation, the total number of colony forming units (CFU) was determined and number of CFUs per millimeter of suspension (CFU mL−1) was obtained and transformed into logarithm (log 10).
Statistical analysis
In order to verify the differences between the experimental situations to both tested PS, the variable reduction in viable bacterial colony counts promoted by each treatment was analyzed by two-way ANOVA and Tukey's test. The p value was considered as <0.05 to statistical significance. The BioEstat 5.0 software for Windows (Sociedade Civil Mamiraua, Manaus, AM, Brazil) was used for data analysis.
Results
The effect of PACT using curcumin and TBO achieved a high photokilling rate (~99.9 %) to all tested PS concentrations in combination with the studied light fluencies (p < 0.05). These outcomes for curcumin and TBO are summarized on Tables 1 and 2, respectively. In spite of these results, the groups submitted to PS alone (C+BL− and T+RL−) and lights (C−BL+ and T−RL+) presented no reduction in CFU mL−1 when compared among the control groups (p > 0.05).
Discussion
The results of this first study demonstrate that the novel LED light source effectively activated TBO and curcumin–PACT killing S. mutans cells in suspension in a very short period of time. The data is showing that we were able to reduce the time from several minutes to seconds of exposure. In addition, no considerable thermal damage was verified when the lights were applied to the S. mutans, demonstrating that even though the temperature was no measured on this present experiment, the results demonstrated no bacterial reduction to groups treated only with blue LED and red LED with no statistical differences in comparison with control groups (Tables 1 and 2). The use of this technology as a technique to inactivate microorganisms has a considerable impact on the health field. PACT could be used as a complementary therapy for bacterial infection control and previous prophylaxis on dental appointments among other examples.
Some in vivo approaches have attested the positive role of PACT in controlling some of the most prevalent bacteria-induced diseases of humans—caries and inflammatory periodontal disease [23, 28]. Araújo et al. [23] achieved a high decrease in the number of S. mutans present in the saliva of volunteers by a mouthrinse of curcumin exposed to a blue LED device for 5 min. To corroborate with this findings, Braun et al. [28] assessed the adjunctive effect of PACT in patients with chronic periodontitis. They found that phenothiazine chloride activated with a laser device added to conventional anti-infective approaches improved nonsurgical periodontal therapy. These studies have proved that PACT is a promising tool in the dental practice, although the time needed for the treatment may be an issue for the clinical application of PACT in the dental field. The LumaCare™ device used in the present study is a source of light that produces the entire spectrum of visible light by changing different probes at specific wavelengths. Multiple protocols using different PS can be activated by one light source eliminating the high cost of lasers or the need of multiple LED arrays. In this way, one device would be enough to activate PS with different properties, including curcumin and TBO. At same time, this light source presents a high potency (1,460 mW cm−2) in the red wavelength, and for this reason, the illumination times were extremely short (less than 1 min) in comparison to current studies regarding TBO as a photosensitizer [5, 25, 27]. On the other hand, the blue LED (95.5 mW cm−2) has a lower penetration depth into the tissue compared to red light due to its low potency associated with scattering and absorption by biomolecules results in longer illumination times (~4 min). This outcome related to curcumin–blue LED are in agreement with previous published studies [21–23, 26].
The results obtained indicated that TBO without light activation slightly reduced the CFU per milliliter numbers in the dark (to all tested concentrations), which is normally found in exogenous photosensitive drugs. However, the CFU per milliliter reduction was not statistically significance when compared to the other controls. Study performed by Paulino et al. [1] demonstrated TBO toxicity when used in concentrations higher than 5 μM whereas Chan and Lai [29] demonstrated a decrease in CFUs for the Streptoccocus sanguis with concentrations of 2 × 104 M of methylene blue in the dark.
According to Araújo et al. [22], curcumin without light activation did not lead to a significant reduction in the colony-forming units of S. mutans and Lactobacillus acidophilus. A percentage of log reduction ranging from 3–5 log 10 was found when a solution of curcumin was used on planktonic suspension of S. mutans [26]. Nevertheless, when compared to the present study, those previous investigations used much higher concentrations of curcumin (~1.6 times higher).
In our study, neither PS nor light when used alone had a bactericidal effect. In fact, our results highlighted the need for dye–light conjugation to ensure the effectiveness of PACT. The application of the novel light device in the presence of the PS achieved a lethal photosensitization (~99.9 %) of S. mutans suspensions in low concentrations of PS at only seconds of light exposure. This outcome is in agreement with current photodynamic studies [30, 31]. This is probably due to the fact that both dyes are activated by a light source made specifically for PACT treatment resulting in an effective combination of PS and LED light.
The effectiveness of PACT is mostly related to ROS production and its interaction with the target cell organelles. Some studies suggested that a high rate of ROS is achieved after shorter illumination times of curcumin [21, 32]. In the present study, we found ~4 min illumination time of curcumin to be an optimal protocol to reach a considerable photoinactivation (5.97 log of bacterial reduction when compared to the control). We can hypothesize that due to the great water solubility of TBO in the hydrophobic region of the cell membrane allied to its low molecular weight (305.83 g mol−1) was able to cause membrane damage due to lipid peroxidation, which is the probable action mechanism of TBO-mediated PACT [9].
Photodynamic chemotherapy success is not only based on the direct production of free radicals or ROS. Rolim et al. [27] proved that erythrosine (163.5 μM) exposed to a broad blue LED produced a low rate of ROS (among another PS tested) and was still able to produce an antimicrobial effect, demonstrating that other factors may be involved on PACT's effectivity [33]. Further studies need to be taken into consideration to elucidate PACT's mechanism of action, including concentration of dyes, type of microorganisms and their organization (suspension or biofilm), type of light source, and other parameters should be taken into consideration to elucidate the actual PACT mechanism.
This study is the first step to the development of an optimum PACT treatment for oral infections, considering there is no previous study regarding LumaCare™ using the blue and red LED in this field. As part of the next steps, we are now applying the methods used in this study on microorganisms organized as biofilms.
The results of this study attested the efficacy of a novel LED device on PACT field on oral bacteria most involved with dental caries. The biggest advantage of this novel LED device is the decrease in the treatment time, making it more viable for clinical applications. Yet, the concomitant use of curcumin and TBO reinforces the antimicrobial activity of these PS as prior substances to photodynamic applications. Further studies must be conducted due to the broad range of variables present in this therapy, aiming its clinical applicability.
References
Paulino TP, Ribeiro KF, Thedei G Jr, Tedesco AC, Ciancaglini P (2005) Use of hand held photopolymerizer to photoinactivate Streptococcus mutans. Arch Oral Biol 50:353–359
Dougherty TJ, Gomer CJ, Henderson BW (1998) Photodynamic therapy. J Natl Cancer Inst 90:889–905
Lulic M, Leiggener Gorog I, Salvi GE, Ramseier CA, Matheos N, Lang NP (2009) One year outcomes of repeated adjunctive photodynamic therapy during periodontal maintenance: a proof-of-principle randomized controlled clinical trial. J Clin Periodotol 36:661–666
Selwitz RH, Ismail AI, Pitts NB (2007) Dental caries. Lancet 369:51–59
Zanin IC, Goncalves RB, Junior AB et al (2005) Susceptibility of Streptococcus mutans biofilms to photodynamic therapy: an in vitro study. J Antimicrob Chemother 56:324–330
Svensater G, Welin J, Wilkins JC (2001) Protein expression by planktonic and biofilm cells of Streptococcus mutans. FEMS Microbiol Lett 205:139–146
Giusti JS, Santos-Pinto L, Pizzolito AC, Helmerson K, Carvalho-Filho E, Kurachi C, Bagnato VS (2008) Antimicrobial photodynamic action on dentin using a light-emitting diode light source. Photomed Laser Surg 26:281–287
Wood S, Nattress B, Kirkham J, Shore R, Brookes S, Griffiths J, Robinson C (1999) An in vitro study of the use of photodynamic therapy for the treatment of natural oral plaque biofilms formed in vivo. J Photochem Photobiol B Biol 50:1–7
Wood S, Metcalf D, Devine D, Robinson C (2006) Erythrosine is photosensitizer for the photodynamic therapy of oral plaque biofilms. J Antimicrob Chemoter 57:680–684
Vilela SF, Junqueira JC, Barbosa JO, Majewski M, Munin E, Jorge AO (2012) Photodynamic inactivation of Staphylococcus aureus and Escherichia coli biofilms by malachite green and phenothiazine dyes: an in vitro study. Arch Oral Biol 57:704–710
Meisel P, Kocker T (2005) Photodynamic therapy for periodontal diseases: state of the art. J Photochem Photobiol B 79:159–170
Junqueira JC, Ribeiro MA, Rossoni RS, Barbosa JO, Querido, Jorge AOC (2010) Antimicrobial photodynamic therapy: photodynamic antimicrobial effects of malachite green on Staphylococcus, Enterobacteriaceae, and Candida. Photomed Laser Surg 28:S67–S72
Castano AP, Demidova TN, Hamblin MR (2004) Mechanisms in photodynamic therapy: part one—photosensitizers, photochemistry and cellular localization. Photodiagnosis Photodyn Ther 1:279–293
Wainwright M (1998) Photodynamic antimicrobial chemotherapy (PACT). J Antimicrob Chemoter 42:13–28
Usacheva MN, Teichert MC, Biel MA (2001) Comparison of the methylene blue and toluidine blue O bacterial efficacy against gram-positive and gram-negative microorganisms. Laser Surg Med 29:165–173
Souza RC, Junqueira JC, Rossoni RD, Pereira CA, Munin E, Jorge AO (2010) Comparison of the photodynamic fungicidal efficacy of methylene blue, toluidine blue, malachite green and low power laser irradiation alone against Candida albicans. Lasers Med Sci 25:385–389
Gupta KK, Bharne SS, Rathinasamy K, Naik NR, Panda D (2006) Dietary antioxidant curcumin inhibits microtubule assembly through tubulin binding. FEBS J 273:5320–5332
Ruby AJ, Kuttan G, Babu KD, Rajasekharan KN, Kuttan R (1995) Anti-tumour and antioxidant activity of natural curcuminoids. Cancer Lett 94:79–83
Bruzell EM, Morisbak E, Tønnesen HH (2005) Studies on curcumin and curcuminoids XXIX. Photoinduced cytotoxicity of curcumin in selected aqueous preparations. Photochem Photobiol Sci 20:523–530
Dujic J, Kippenberger S, Ramirez-Bosca A, Diaz-Alperi J, Bereiter-Hahn J, Kaufmann R, Bernd A, Hofmann M (2009) Curcumin in combination with visible light inhibits tumor growth in a xenograft tumor model. Int J Cancer 124:1422–1428
Dovigo LN, Pavarina AC, Ribeiro AP, Brunetti IL, Costa CA, Jacomassi DP, Bagnato VS, Kurachi C (2011) Investigation of the photodynamic effects of curcumin against Candida albicans. Photochem Photobiol 87:895–903
Araújo NC, Fontana CR, Bagnato VS, Gerbi ME (2012) Photodynamic effects of curcumin against cariogenic pathogens. Photomed Laser Surg 30:393–399
Araújo NC, Fontana CR, Gerbi MEM, Bagnato VS (2012) Overall-mouth disinfection by photodynamic therapy using curcumin. Photomed Laser Surg 30:96–101
Usacheva MN, Teichert MC, Biel MA (2001) Comparison of the methylene blue and toluidine blue photobactericidal efficacy against gram-positive and gram-negative microorganisms. Lasers Surg Med 29:165–173
Teixeira AH, Pereira ES, Rodrigues LK, Saxena D, Duarte S, Zanin IC (2012) Effect of photodynamic antimicrobial chemotherapy on in vitro and in situ biofilms. Caries Res 46:549–554
Paschoal MA, Tonon CC, Spolidório DMP, Bagnato VS, Giusti JSM, Santos-Pinto L (2013) Photodynamic potential of curcumin and blue LED against Streptococcus mutans in a planktonic culture. Photodiagnosis Photodyn Ther 10:313–319
Rolim JPML, Melo MAS, Guedes SF, Albulquer Filho FB, Souza JR, Nogueira NAP, Zanin ICJ, Rodrigues LKA (2012) The antimicrobial activity of photodynamic therapy against Streptococcus mutans using different photosensitizers. J Photochem Photobiol B106:40–46
Braun A, Dehn C, Krause F, Jepsen S (2008) Short-term clinical effects of adjunctive antimicrobial photodynamic therapy in periodontal treatment: a randomized clinical trial. J Clin Periodontol 35:877–884
Chan T, Lai CH (2003) Bactericidal effects of different laser wavelengths on periodontopathic germs in photodynamic therapy. Lasers Med Sci 18:51–55
Mattiello FD, Coelho AA, Martins OP, Mattiello RD, Ferrão Júnior JP (2011) In vitro effect of photodynamic therapy on Aggregatibacter actinomycetemcomitans and Streptococcus sanguinis. Braz Dent J 22:398–403
Bevilacqua IM, Nicolau RA, Khouri S, Bruqnera A Jr, Teodoro GR, Zângaro RA, Pacheco MT (2007) The impact of photodynamic therapy on the viability of Streptococcus mutans in a planktonic culture. Photomed Laser Surg 25:513–518
Dovigo LN, Pavarina AC, Carmello JC, Machado AL, Brunetti IL, Bagnato VS (2011) Susceptibility of clinical isolates of Candida to photodynamic effects of curcumin. Lasers Surg Med 43:927–934
Fontana CR, Abernethy AD, Som S, Ruggiero K, Doucette S, Marcantonio RC, Boussios CI, Kent R, Goodson JM, Tanner AC, Soukos NS (2009) The antibacterial effect of photodynamic therapy in dental plaque-derived biofilms. J Periodontal Res 44:751–755
Acknowledgments
This research was supported by Brazilian Government Agency—Coordenação de Apoio de Pessoal de Nivel Superior—CAPES Foundation grant BEX #8485/11-9 (scholarship to Marco Aurelio Paschoal).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paschoal, M.A., Lin, M., Santos-Pinto, L. et al. Photodynamic antimicrobial chemotherapy on Streptococcus mutans using curcumin and toluidine blue activated by a novel LED device. Lasers Med Sci 30, 885–890 (2015). https://doi.org/10.1007/s10103-013-1492-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-013-1492-1