Abstract
Chitosan-poly(propylene imine) dendrimer (CS-PPI) hybrid was successfully prepared and grafted on wool substrate. The impact of key operational grafting factors, i.e., pH, temperature, and CS-PPI concentration on grafting yield, was investigated through dyeing with two commercial reactive dyes, C.I. Reactive Orange 122 (RO122) and C.I. Reactive Red 195 (RR195). The optimal grafting values obtained were pH 6, temperature 70 °C, and 20 % owf CS-PPI concentration. The performance of CS-PPI-grafted wool was investigated in terms of dyeing ability, color fastness, and shrinkage of wool fabrics. Grafted wool actually showed lowered optimal dyeing temperature of 40 °C as compared to those of pristine wool (80 °C). The color depth (K/S) comparison of CS-PPI grafted wool dyed with no salt usage in dye bath with those of pristine wool conventionally dyed with 30 % owf salt revealed that both grafted and pristine samples had similar color depth suggesting elimination of salt from reactive dye bath so-called “salt free dyeing” for grafted wool. No remarkable change in color fastness of dyed grafted wool fabrics against wash, rub, light, and perspiration was observed and all were in good and acceptable level. In addition, the shrink-proofing property of CS-PPI grafted wool fabrics was also enhanced. The results of this study clearly indicated that CS-PPI hybrid is a high potential novel bio-compatible compound which can be successfully used in finishing of wool in both eco-friendliness and energy saving standpoint in dyeing process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wool is a well known important renewable natural fiber in both textile and hand-woven carpet industry. This is owing to its peculiar distinctive natural properties such as excellent elasticity and resiliency, heat insulation, moisture absorption, good dyeing ability with both chemical and natural dyes etc. In spite of introduction of numerous other textile fibers into the industry, there is no alternative fiber including all the natural properties of wool as a Stone Age fiber (Julia` et al. 2000; Mori and Inagaki 2006).
Dyeing process is carried out at different stages of manufacture to add color and sophistication to textiles and raise product value. A wide variety of dyes, techniques, and apparatus are available for wool textiles dyeing and among them acid, metal complex, and reactive dyes are widely used (Zhang and Zhang 2014; Sadeghi-Kiakhani et al. 2014). Reactive dyes have become very popular due to their brilliancy, variety of hues, high wet fastness, convenient usage, and high applicability. They have also some dyeing problems, such as low dye-ability (low dye build-up), requirements of large amount of auxiliary agents, and high volume of discharged wastewater, which must be improved (Holme 2003; Mall et al. 2002; Molino et al. 2005; Pascual and Julia 2001).
Felting is an adverse character of wool products which takes place as an outcome of the directionally reliant to frictional coefficient of the wool fibers. To diminish felting, this directional dependency must be decreased (Pascual and Julia 2001). To reduce above drawbacks, cationization of wool through chemical modifications with compounds containing cationic groups was performed (Holme 2003; Arivithamani et al. 2014). As it is well accepted that surface property of fibers plays an essential role in the functional and appealing properties of the fabrics, many surface modifications by chemical treatments are capable to change textile properties (Mall et al. 2002; Molino et al. 2005; Mansour 2010).
Chitosan is deacetylated derivative of chitin obtained from crustaceans-like crab and shrimp shell wastes. It has many remarkable useful chemical and physical properties such as biodegradability, non-toxicity, antimicrobial activity, antioxidant, etc. (Enescu 2008; Jocic et al. 2005; Yang et al. 2010). In the textile industry, chitosan has been widely applied to provide antimicrobial properties, increase dyeing ability, and prepare beneficial fibers (Zhang 2008; Froehing 2001). In addition, Chitosan can be used to increase the cationic property on wool fabric thanks to amino groups present in its chemical structure. It was reported that chitosan pretreatment successfully reduces the difference of dyeing performance between damaged and undamaged wool fibers through the increasing of dyeing rate and dyeing ability (Jocic et al. 2005).
Dendrimers as biologically active macromolecules having branched structure, many reactive end groups, highly ordered, and compacted shape. This unique structure creates the best places for the host molecules between the branches (Blencowe et al. 2009; Calabrett et al. 2007; Sashiwa et al. 2000). Recently, dendrimers have been applied for the elimination of dyes from colored effluents, extraction of dyes, increasing the dyeing ability of textiles, and development the antimicrobial finishing of textiles (Sashiwa et al. 2002; Sadeghi-Kiakhani et al. 2013a, b).
To our knowledge, no reports are available in the open literatures concerning the reactive dyeing of wool altered with CS-PPI hybrid. Therefore, in this study, the wool was altered with CS-PPI, dyed using two commercial reactive dyes by conventional exhaustion method, and the impacts of the operational parameters such as pH, CS-PPI concentration, and temperature of grafting process on the dyeing ability of wool were explored. Moreover, fastness properties of dyed wool against light, wash, rub and perspiration were evaluated and assessed. Finally, the effect of CS-PPI treatment on the shrink-proof property of the wool fabric is evaluated.
Experimental
Materials and apparatus
Commercial scoured wool fabrics with plain weave (warp density of 60 inch−1 and weft density of 53 inch−1) were used in this study. Chitosan was provided by Kitotak Co. (degree of deacytilation (DD): 85 %, MW: 1,000 kDa, Iran). Two commercially reactive dyes, namely, C.I. Reactive Orange 122 (RO122) and C.I. Reactive Red 195 (RR195) provided by Dystar Co. were used as received without further purification for wool dyeing trials. All chemicals used were analytical grade.
The wool dyeing was performed in acidic media of pH 5 (glacial acetic acid, Merck) using laboratory HT dyeing machine. UV–visible absorption spectra were measured using a Cecil 9200 double beam spectrophotometer. The reflectance characteristics of the dyed samples were measured through Gretag Macbeth spectrophotometer Color-Eye 7000 A, color eye reflection spectrophotometer (D65 illumination, 10° observer).
Preparation of CS-PPI composite
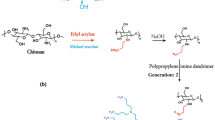
Chitosan (1, Scheme 1) was dissolved in the acetic acid and water/methanol mixture. Then, ethyl acrylate was introduced into the solution. After stirring at 50 °C for 10 days upon completion the reaction, the solution was quenched and precipitated in acetone saturated with NaHCO3. After that, the precipitate was collected by filtration, and then the filtrates were dispersed in H2O saturated with NaHCO3. The resulting mixture was dialyzed against H2O, and lyophilized to obtain N-carboxyethyl chitosan ethyl ester (2, Scheme 1). For the preparation of N-carboxyethyl chitosan (3, Scheme 1), the prepared compound (2, Scheme 1) was added to NaOH solution; the mixture was stirred for 2 h, dialyzed, and lyophilized as above mentioned. Finally, the precipitated powders were obtained in quantitative yield of 95 %.
Compound (3, Scheme 1) was dispersed in methanol; PPI (G = 2) was added to the prepared suspension, and the mixture was stirred at ambient temperature. After three days, the solvent was evaporated; the obtained powder was dispersed in NaOH solution at room temperature for 2 h, dialyzed, and lyophilized to achieve CS-PPI. The summarized preparation steps of CS-PPI are shown in Scheme 1, and were explained in detail in our previous work (Sadeghi-Kiakhani et al. 2013a, b).
Preparation of wool fabrics
Wool fabric was scoured in 3 % NaOH at 50–60 °C for 60 min. In order to increase functional groups in wool surface, the wool was oxidized using 1.5 % (owf) KMnO4 solution at boil for 2 min. A fine powder of CS-PPI was dissolved in citric acid solution (pH 4–5), and the grafting of wool fabric with CS-PPI was carried out according to the pad-dry-cure method as follows:
The scoured wool was immersed in different concentrations of CS-PPI solutions for 24 h at various temperatures. The fabrics were then dried for 5 min at 70 °C, cured in an oven at 120 °C for 5 min. In this study, the effects of some important operational parameters on the grafting yield were explored. These parameters were CS-PPI concentrations (5, 10, 15, 20, and 25 % owf), grafting temperatures (30, 50, 70, and 90 °C), and grafting pH (2, 4, 6, and 8). Then, the dyeing was accomplished with reactive dyes, and the fastness properties of the treated wool fabrics were assessed.
Dyeing method
Pristine and grafted wool fabrics were dyed with two reactive dyes in the bath containing 2 % (owf) dye, 1 % (owf) Albegal B (Ciba Co.), acetic acid (pH 5) at T = 60 °C, the procedure recommended by the dye manufacturer; the procedure was slightly modified for dyeing trials at 40 °C. Dyeing was performed in a laboratory dyeing machine at a liquor ratio of 1:20. The samples were wetted for 5 min in the dye bath at 30 °C before the addition of dye. Dyeing time range of 10–120 min was explored at both 40 and 60 °C dyeing temperatures. Finally, fabrics were rinsed with hot and then cold water, and dried at the ambient temperature.
The absorbance of the dye solution was measured at λmax of each dye prior and after dyeing tests. Then, the percentage of dye bath exhaustion (E %) was calculated using Eq. 1, where A 0 and A 1 were the absorbance of the dye solution prior and after the dyeing process, respectively. The amount of dye fixed (F) was calculated using Eq. 2, where A 2 was the absorbance of the dye bath after soaping process.
The impacts of pH, CS-PPI concentration and temperature of grafting process on the dyeing ability of treated wool were explored using K/S data calculated from the Kubelka–Munk (Eq. 3), where K is the absorbance coefficient, S is the scattering coefficient, R is the reflectance value at λmax (wavelength of maximum absorption). K/S data were directly correlated with the dye concentration present on the dyed substrate.
Color difference (\( \Delta E \)) of dyed wool fabrics was obtained using Eq. 4:
where L* is lightness; a* and b* are the chromaticity coordinates.
Fastness properties
Wash fastness was measured by the standard ISO 105 C06 C2S:1994 (E) method. The washing was conducted for 30 min at 60 °C, rinsed with cold water, air dried, and analyzed. Light fastness test ISO 105 B02:1988 (E) was evaluated with the xenon arc lamp using blue reference samples. The rub fastness test was completed according to ISO105-X12:1993 (E) using a crockmeter. For the wet rub test, the testing squares were thoroughly immersed in distilled water; the rest of the procedure was the same as in the dry test. The staining on the white test cloth was assessed according to the gray scale. The perspiration fastness was assessed in acidic and alkali media by the standard ISO 105 - E04:1994 (E) method. The samples were prepared by stitching a piece of dyed wool fabric between two pieces of adjacent fabrics, all of same length, and then immersed in the acid and alkaline solutions for 30 min. The staining on the adjacent fabrics was evaluated according to the gray scale.
Shrink-proofing property
The treated fabrics were measured for their shrink-proofing property according to the AATCC TM 187-2001 method.
The measurements of colorimetric and shrink-proofing properties, etc. were carried out three times. The reproducibility of the experimental data, calculated as relative standard deviation, was acceptable ≤4 %.
Results and discussion
Dyeing ability and related parameters
Some researchers have reported that chitosan and its derivatives are very effective in the alteration of wool and enhancement of the dyeing rate and build-up of various acid, reactive, and natural dyes (Enescu 2008; Giri Dev et al. 2009). However, the dyeing properties of CS-PPI grafted wool using reactive dyes have not been investigated so far. Hence, it has been considered in the current research.
Effect of CS-PPI concentration on the dyeing ability of wool
The amount of exhausted dye as a function of CS-PPI concentration is shown in Fig. 1a. It is well observed that the exhaustion values of altered wool are markedly higher, especially with higher concentrations of CS-PPI, as compared to untreated wool. The dye exhaustion steadily increased with the increase in CS-PPI concentration and reached the maximum at around 20 % (owf) CS-PPI. This phenomenon could be the direct consequence of an increase in the population of free amino groups on the wool surface upon CS-PPI grafting, and/or formation of hydrogen bonds with the active radical group of the reactive dyes giving rise to sequentially increased dyeing property of the grafted wool. Moreover, another explanation could be the interaction of hydroxyl, carboxyl, and amide groups in wool polymer chain being prone to form hydrogen bond with amino groups of CS-PPI, and consequently, the improvement in the dye exhaustion (Sadeghi-Kiakhani et al. 2013b; Zargarkazemi et al. 2014). The results demonstrated that the appropriate CS-PPI concentration for grafting process was 20 % (owf).
Effect of grafting pH on the dyeing ability of wool
Grafting pH has an important role on the grafting yield. According to the results of previous section, the dye build up on wool was increased with an increase in the CS-PPI concentration owing to the increase in number of dye receptive sites. Hence, the amount of CS-PPI grafted on wool as a function of grafting solution pH (2–8) was evaluated through dye taken up and is shown in Fig. 1b. The results showed nearly similar build up values for treated wool at pH above 6 as compared with untreated wool. This means no grafting occurred at pH higher than 6. This phenomenon could be ascribed to the decrease of the solubility of CS-PPI at pH > 6 which results in decrease or loss the grating reaction between wool and CS-PPI in the grafting step (Zargarkazemi et al. 2014).
In weak acidic media (pH 4–6), the higher grafting yield was achieved. This could be because of the enhanced solubility of CS-PPI and facile reaction of free reactive sites of CS-PPI with wool macromolecular chain. However, the strong acidic media (pH 2) may damage the chemical structure of wool at high temperature for a long time (Sadeghi-Kiakhani et al. 2013b; Ranjbar-Mohammadi et al. 2010). Thus, the exhaustion values of the strong acidic pH grafted wool were not noticeable as compared to the data of weak acidic media.
Effect of grafting temperature on the dyeing ability of wool
A considerable amount of energy could be saved by optimizing the temperature process as an important operational parameter in the grafting process (Zargarkazemi et al. 2014). Exhaustion values of treated wool with CS-PPI at different grafting temperatures are demonstrated in Fig. 1c. As it is seen, exhaustion values of the treated wool increased with rising temperature up to 70 °C. This confirms the role of temperature in the accelerating and enhancement of the interactions between wool and CS-PPI. Therefore, the suitable temperature for grafting was found to be 70 °C.
Thus, the optimum values of three important grafting reaction parameters for wool pretreatment in the range studied can be summarized as follows: pH 4–6, CS-PPI 20 % (owf); Temperature 70 °C for 24 h.
Effect of dyeing time on the dye exhaustion
As shown in Fig. 2, the CS-PPI-grafted wool exhibited higher dyeing rate and higher dye exhaustion as compared to pristine wool. Moreover, the higher the concentration of CS-PPI, the faster the dyeing rate was observed. This can be explained by the surface alteration of the wool, and accordingly, increasing the number of positive dye receptive sites on the wool surface, and the faster adsorption of dyes from dye bath on to the wool.
According to the graphs in Fig. 2, the modified wool reached equilibrium at around 60 min using both dyes while the untreated wool still required higher amount of time for saturation in the studied time range. In the analysis of a dyeing system including particular dye and fiber, a decrease in dyeing saturation time as well as the enhancement in the amount of dye exhaustion is the great importance since noticeable amount of time, energy, and chemicals could be saved in both environmental and economical points of view. This goal was attained in this study by a decrease in the required amount of dye for a desired depth of color on the substrate, and a decrease in the dyeing time and energy.
Effect of dyeing temperature on dyeing ability of wool
The dye uptake of wool samples at 40 and 60 °C as a function of CS-PPI concentration is shown in Fig. 3. For comparison purpose, the raw wool exhaustion at 80 °C is shown in Fig. 3, as well. Pristine wool substrates dyed at 80 °C exhibited similar close values to CS-PPI-treated wool dyed at 40 °C. Moreover, pretreatment with CS-PPI resulted in considerable enhancement in dye bath exhaustion at lower temperatures of dyeing, where with CS-PPI higher concentrations the dye uptake at 40 °C was even higher than untreated wool dyed at 80 °C. An increase in dyeing temperature to 60 °C caused to gradual improvement in dye exhaustion, but the enhancement was not noticeable.
Dyeing data trend showed that optimum dyeing temperature using CS-PPI-grafted wool with reactive dyes was considerably decreased and shifted toward lower temperatures. This is worthwhile in terms of saving of energy in wool reactive dyeing as well as ecological dyeing with enhanced dye exhaustion with subsequent lower colorant left in discharged wastewater.
Effect of salt concentration on dye exhaustion
The use of inorganic salts like sodium sulfate in wool dyeing for increasing the affinity of acid or reactive dyes is well known. In dye bath, it retards dye migration toward substrate and thus the better levelness in dyeing could be obtained. Figure 4 shows the impact of salt concentration (0–40 % owf) on the dyeing ability of pristine wool samples. The CS-PPI-treated wool samples dyed with no salt addition, for comparison purpose, are shown in Fig. 4 as well. K/S values were enhanced by salt amount up to nearly 30 % (owf). Therefore, 30 % (owf) salt concentration was considered as an optimum concentration and used in the untreated wool dyeing. Interestingly, it was found that K/S values of CS-PPI-treated wool samples were similar to the values of wool fabric dyed with salt 30 % (owf). This finding emphasized that the treated wool could be successfully dyed with same color depth with no salt consumption as compared to those of untreated wool. In other words, in wool dyeing with reactive dyes, the salt was eliminated from the dye bath with no adverse impact on dye uptake, and thus “salt free dyeing” characteristic was imparted. This finding is worthwhile in terms of an environmentally friendly dyeing process with less hazards of discharged wastewater.
Fastness properties
Textiles are exposed to repeated wash, light, rub, and perspiration during their usage. Hence, durability of the finish applied on the textile material at above conditions is of great importance (Giri Dev et al. 2009). Fastness data have been measured and the results are reported in Table 1.
Data show that wash fastness rating for staining of adjacent fabrics in the case of dyed fabrics is generally good and those for change in color is also acceptable. It should be mentioned that no change in color was observed at higher CS-PPI concentrations; wash fastness was very good with rating of 4–5, which was independent of CS-PPI concentrations; very good fastness to rubbing in both dry and wet states was obtained with dry rubbing fastness as high as 4–5, while wet rubbing fastness was in the range of 3–4; Perspiration fastness properties (in both acidic and alkaline media) of dyed wool samples in terms of ratings for staining of adjacent fabrics and change in color were very good. In addition, the lower ratings for change in color at both acidic and alkaline conditions for CS-PPI-grafted woolen samples as compared to dyed pristine wool represent the more sensitivity of the grafted samples to pH which may be owing to the degradation of the dyes at acidic and basic conditions.
In general, it could be concluded that the fastness properties of dyed fabrics are almost independent from CS-PPI concentration where no adverse effect on general fastness properties was observed.
Colorimetric properties
CIE L*a*b* values for dyed wool fabrics are presented in Table 2. The colorimetric data confirmed that the treated fabrics had a little difference as compared with untreated fabrics. In general, dyed CS-PPI-grafted wool in optimal grafting condition was light and bright. The variations in colorimetric data (L *, a * and b * values) can be attributed to the new mutual chemical interaction between functional groups of dye-CS-PPI-wool system.
From the leveling standpoint of dyed wool fabric, the results in Table 3 demonstrate that the leveling of the wool fabrics dyed at 60 °C is all within the acceptable range of 1.5. It is also illustrated that dyeing at lower temperature of 40 °C led to leveling which is also within the acceptable range.
Shrink-proofing property
According Table 4 data, the shrinkage of the untreated fabrics in both the warp and the weft direction is larger as compared to CS-PPI-treated fabrics. This behavior may be due to the fact that in untreated fabrics, the yarn is stretched stiff during weaving, whereas in treated fabrics fiber scales were entirely covered with reactive polymers or an even layer of protective thin membrane formed over the surface. In turn, the latter would create the stiffness of the yarn, the structure of the fabric, and weaving density more obstructive to the movement of singular fibers. Under these situations, the fibers in the treated fabric would become stronger to external forces and not slip over each other, and consequently, the fabric would become more shrink-proof (Yang et al. 2010). Therefore, when the osmosis of CS-PPI in the fabric is better, the surface of the fiber covered by CS-PPI becomes larger, and treated fabric with CS-PPI has better shrink-proofing properties (Yang et al. 2010). It was also noticed that shrinkage in the warp direction was always greater than that of the weft direction. This difference is because the warp yarn is stretched more firmly than the weft yarn during weaving, which is also the significant factor why wool fabrics are disposed to shrink when heated. It was found that the shrink-proofing of treated fabrics is upgraded with increase in the concentration of CS-PPI.
Conclusions
From the results of the present study, following conclusions can be drawn:
Chitosan-poly(propylene imine) (CS-PPI) hybrid was introduced as a potential non-toxic bio-polymer for multifunctional finishing of wool; with grafting CS-PPI onto the wool, the dyeing ability of wool was noticeably enhanced with no adverse impact on other characteristics of dyed fabrics; similar color depth was obtained for dyed grafted wool at 40 °C and that of dyed untreated wool at 80 °C, and thus the optimal dyeing temperature with reactive dyes was markedly decreased. It was found that salt could be eliminated from reactive dye bath when wool grafted with CS-PPI. Moreover, CS-PPI is an effective shrinkage reduction finish for wool which considerably enhanced shrink-proof characteristic of wool fabrics. Color fastness of the treated fabrics was acceptable against washing, light, and perspiration. Overall, CS-PPI could be considered as a novel efficient eco-friendly finishing compound for multifunctional treatment of wool fabrics.
References
Arivithamani N, Mary SA, Kumar MS, Giri Dev VR (2014) Keratin hydrolysate as an exhausting agent in textile reactive dyeing process. Clean Technol Environ Policy 16:1207–1215
Blencowe A, Tan JF, Goh TK, Qiao GG (2009) Core crosslinked star polymers via controlled radical polymerization. Polymer 50:5–32
Calabrett MK, Kumar A, McDermott AM, Cai C (2007) Antibacterial activities of poly(amidoamine) dendrimers terminated with amino and poly(ethylene glycol) groups. Biomacromolecules 8:1807–1811
Enescu D (2008) Use of chitosan in surface modification of textile materials. Roumanian Biotechnol Lett 13:4037–4048
Froehing PE (2001) Dendrimers and dyes-A review. Dyes Pigment 48:187–195
Giri Dev VR, Venugopal J, Sudha S, Deepik G, Ramakrishna S (2009) Dyeing and antimicrobial characteristics of chitosan treated wool fabrics with henna dye. Carbohydr Polym 75:646–650
Holme I (2003) Challenge and change in wool dyeing and finishing. Rev Prog Color 33:85–92
Jocic D, Vı´lchez S, Topalovic T, Navarro A, Jovancic P, Rosa Julia M, Erra P (2005) Chitosan/acid dye interactions in wool dyeing system. Carbohydr Polym 60:51–59
Julia` MR, Pascual E, Erra P (2000) Influence of the molecular mass of chitosan on shrinkresistance and dyeing properties of chitosantreated wool. J Soc Dyers Colorists 116:62–67
Mall JK, Sims P, Carr CM (2002) Surface chemical analysis of lipase enzyme treatments on wool and mohair. J Text Inst 93:43–51
Mansour HF (2010) Environment and energy efficient dyeing of woollen fabric with sticta coronate. Clean Technol Environ Policy 12:571–578
Molino R, Espinos JP, Yubero F, Erra P, Gonzalez-Elipe AR (2005) XPS analysis if down stream plasma treated wool: influence of the nature of the gas on the surface modification of wool. Appl Surf Sci 252:1417–1429
Mori M, Inagaki N (2006) Relationship between anti-felting properties and physicochemical properties of wool treated with low-temperature plasma. Res J Text Appar 10:33–45
Pascual E, Julia MR (2001) The role of chitosan in wool finishing. J Biotechnol 89:289–296
Ranjbar-Mohammadi M, Arami M, Bahrami H, Mazaheri F, Mahmoodi NM (2010) Grafting of chitosan as a biopolymer onto wool fabric using anhydride bridge and its antibacterial property. Colloids Surf, B 76:397–403
Sadeghi-Kiakhani M, Arami M, Gharanjig K (2013a) Dye removal from colored textile wastewater using chitosan-PPI dendrimer composite as a biopolymer: optimization, kinetic and isotherm studies. J Appl Polym Sci 127:2607–2619
Sadeghi-Kiakhani M, Arami M, Gharanjig K (2013b) Application of a biopolymer chitosan-poly (propylene) imines dendrimer hybrid as an antimicrobial agent on the wool. Iranian Polym J 22:931–940
Sadeghi-Kiakhani M, Gharanjig K, Arami M (2014) Study on dyeing and fastness properties of wool-polyester blend fabrics using novel mono azonaphthalimide dyes. J Text Inst 105:52–58
Sashiwa H, Makimura Y, Shigemasa Y, Roy R (2000) Chemical modification of chitosan: preparation of chitosan-sialic acid branched polysaccharide composites. Chem Commun 11:909–910
Sashiwa H, Shigemasa Y, Roy R (2002) Chemical modification of chitosan 11: chitosandendrimer composite as a tree like molecule. Carbohydr Polym 49:195–205
Yang HC, Wang WH, Huang KS, Hon MH (2010) Preparation and application of nanochitosan to finishing treatment with anti-microbial and anti-shrinking properties. Carbohydr Polym 79:176–179
Zargarkazemi A, Sadeghi-Kiakhani M, Arami M, Bahrami SH (2014) Modification of wool fabric using prepared chitosan-cyanuric chloride hybrid. J Text Inst. doi:10.1080/00405000.2014.906097
Zhang RP (2008) Influence of wool anti-felting performance with chitosan/protease combination finishing. Wool Text J 4:1–4
Zhang Y, Zhang W (2014) Clean dyeing of cotton fiber using a novel nicotinic acid quaternary triazine cationic reactive dye: salt-free, alkali-free, and non-toxic by-product. Clean Technol Environ Policy. doi:10.1007/s10098-014-0821-9
Acknowledgments
Institute for Color Science and Technology, Tehran, and Tabriz Islamic Art University, Tabriz, are gratefully acknowledged for all the supports throughout this study.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sadeghi-Kiakhani, M., Safapour, S. Eco-friendly dyeing of treated wool fabrics with reactive dyes using chitosanpoly(propylene imine)dendreimer hybrid. Clean Techn Environ Policy 17, 1019–1027 (2015). https://doi.org/10.1007/s10098-014-0855-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-014-0855-z