Abstract
The availability of reproducible broth microdilution (BMD) methods including inter log2 antibiotic dilutions for measuring Staphylococcus aureus (SA) vancomycin minimum inhibitory concentrations (MICs) within the susceptible range is needed to elucidate the impact of vancomycin MICs on clinical outcomes of invasive SA infections. Here, we report on the development of a very precise BMD method that incorporates the following incremental antibiotic concentrations: 0.50, 0.62, 0.75, 0.87, 1.0, 1.25, 1.40, 1.50, 1.60, 1.75, and 2.0 μg/mL. The intra- and inter-assay coefficients of variation of this method were around 20%. The mean of the differences in MIC values for all isolates obtained across two independent runs performed at one center was 0.04 μg/mL [95% confidence interval (CI), 0.011–0.07 μg/mL] and that for ten isolates measured at two different centers was 0.04 μg/mL (95% CI, 0–13 μg/mL). Vancomycin MIC values differed by less than 0.1 μg/mL between runs for most isolates. Storage of isolates at −20 °C for up to 3 months had no impact on the vancomycin MIC values. The mean vancomycin MIC values obtained by the Etest using a standard inoculum (0.5 McFarland) were significantly higher (p ≤ 0.001) than those measured by BMD and the MIC values measured by the two methods correlated poorly (Rho, 0.319; p = 0.148). Nevertheless, the mean MIC values measured by the Etest using lower inocula (107 or 106 CFU/mL) and those measured by BMD were comparable and correlated significantly (p = 0.004 for 107 CFU/mL and p = 0.029 for 106 CFU/mL).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bloodstream infections caused by Staphylococcus aureus (SA), which are either susceptible (MSSA) or resistant to methicillin (MRSA), carry high morbidity and mortality rates [1]. There has been controversy in recent years as to whether or not bacteremia and endocarditis due to SA strains with elevated vancomycin minimum inhibitory concentration (MIC) values, but within the susceptible range (≥1.5 μg/mL), as measured by the Etest, are associated with poorer clinical outcomes (see [2,3,4] for systematic reviews and meta-analyses). These studies are methodologically flawed owing to the suboptimal intra- and inter-assay precision of the vancomycin Etest method [5], so that miscategorization of isolates as having low (≤1 μg/mL) versus high (≥1.5 μg/mL) susceptibility could well have occurred [5]. In addition, it is well known that vancomycin MIC values measured by the Etest correlate poorly with those determined by broth microdilution (BMD) and agar dilution assays, these currently being the reference methods according to international agencies (i.e., Clinical and Laboratory Standards Institute, CLSI, and the European Committee on Antimicrobial Susceptibility Testing, EUCAST) [6,7,8]. Conventional BMD methods use serial 2-fold doubling dilutions of antimicrobials, whereas the Etest method provides additional MICs that fall between these concentrations. Thus, given the narrow MIC margin for isolate categorization (low vs. highly susceptible), the BMD method, as traditionally performed, is inappropriate for shedding light on the above issue. Here, we report on the development of a robust BMD method that incorporates several “inter log2 dilution” concentrations of vancomycin.
Materials and methods
Bacterial isolates
In total, 50 SA isolates (MSSA, n = 28 and MRSA, n = 22) recovered from blood cultures (BD BACTEC™ Plus Aerobic/F and Plus Anaerobic/F medium bottles incubated in an automated continuous monitoring blood culturing instrument, BACTEC™ FX, Becton Dickinson, BD Diagnostics, Franklin Lakes, USA) at the Microbiology Service of Hospital Clínico Universitario (HCU) were selected for this study. These isolates had been cryopreserved at −70 °C in glycerol-containing brain-heart infusion (BHI) broth for a median of 556 days (524–588 days). The isolates were genotypically characterized by means of a commercially available DNA gene microarray (Staphylococcus aureus Genotyping Kit 2.0; Clondiag, Jena, Germany), as previously described [9]. For the current study, care was taken to include a wide variety of SA clonal types; namely, among MSSA isolates: CC5 (n = 2), CC6 (n = 2), CC9 (n = 1), CC12 (n = 3), CC15 (n = 2), CC22 (n = 1), CC30 (n = 8), CC45 (n = 7), CC97 (n = 1), and CC121 (n = 1), and among MRSA, CC5 (n = 14), CC8 (n = 2), CC22 (n = 3), and CC88 (n = 1). All isolates were susceptible to vancomycin as determined by the Etest and none were heteroresistant as assessed by the Etest macromethod using a 2.0 McFarland inoculum [9].
Measurement of vancomycin MICs by a BMD method
Frozen stocks were thawed and the isolates were subcultured overnight on trypticase soy agar plates with 5% sheep’s blood (BD). Vancomycin MICs were measured by using BMD panels prepared in-house following CLSI recommendations [10, 11]. Vancomycin powder (100 mg) with a potency of ≥900 μg of vancomycin per mg was obtained from Sigma Aldrich Chemicals (St. Louis, MO, USA). Vancomycin was redissolved in sterile water and used to prepare antimicrobial solutions at concentrations of 0.50, 0.62, 0.75, 0.87, 1.0, 1.25, 1.40, 1.50, 1.60, 1.75, and 2.0 μg/mL in cation-adjusted Mueller–Hinton broths (Sigma Aldrich). A volume of 100 μL of each vancomycin solution was dispensed into U-bottomed 96-well sterile plates (Greiner CELLSTAR, Bio-One from Sigma Aldrich). Isolates were resuspended in sterile demineralized water to achieve a 0.5 McFarland standard as measured by the Vitek DensiCheck nephelometer (bioMérieux diagnostics, Marcy-l’Étoile, France) and then diluted in Mueller–Hinton broth (Sigma Aldrich) to achieve a 105 CFU/mL suspension. A volume of 100 μL of the appropriate antibiotic dilution was dispensed per well. Then, a volume of 5 μL of each isolate suspension was transferred to each well. The BMD results were read visually after 24 h of incubation at 35 °C in ambient air. We found no inter-observer variability in MIC readings (all authors examined BMD plates while optimizing the assay). ATCC 29213 was used for quality control. MICs were recorded as the lowest concentrations that inhibited bacterial growth.
Measurement of vancomycin MICs by broth Etest
The Etest was performed using Etest strips from bioMérieux (Marcy-l’Étoile, France) on Muller–Hinton agar (BD), as previously reported [5]. Three different bacterial inocula were used in these experiments: 1 × 108 CFU/mL (0.5 McFarland), 1 × 107 CFU/mL, and 1 × 106 CFU/mL. The plates were read visually by EMM and RF after 24 h of incubation at 35 °C in ambient air. SA ATCC 29213 was used for quality control.
Testing schedule and data analysis
For the assessment of the intra-assay (within-run) and inter-assay precision of the BMD method, all 50 SA isolates were tested in quadruplicate in two different runs (run 1 and run 2). The time elapsed between the two runs was 6 h. The same SA subcultures were used for runs 1 and 2. Out of the 50 SA isolates, 34 were retested in quadruplicate using frozen panels (cryopreserved at −20 °C during 1 month). To evaluate the inter-center reproducibility of the assay, ten isolates (five MSSA and five MRSA) were tested (in quadruplicate) in parallel (same subculture, same day) at the Microbiology Unit of Hospital San Francisco de Borja (HSFB) and at the Microbiology Service of HCU. To assess the potential effect of bacterial cryopreservation on vancomycin MIC values measured by the BMD method, bacterial isolates were frozen at −20 °C, recovered from storage at 1 and 3 months, and subcultured once prior to repeat BMD testing (in quadruplicate). The intra-assay (within-run) coefficient of variation (CV) was calculated as follows: the CV for each isolate was calculated by dividing the standard deviation (SD) of the set of measurements (four for each isolate) by the mean (of four measurements) and multiplying by 100. Finally, the average of the individual CVs was denoted as the intra-assay CV. The inter-assay and inter-center coefficients of variation were calculated using the pooled standard deviation divided by the overall mean of all duplicated samples (run 1 and run 2, or run at HCU and run at HSFB) and then multiplying by 100. A number of isolates were tested by the Etest (three different inocula) in singlets in two different runs in order to determine the inter-assay CV of the method. The degree of correlation between vancomycin MIC values obtained by the BMD method and the Etest were analyzed by the Spearman’s rank test. The Wilcoxon signed-rank test was used for the analyses of differences between paired samples (non-parametric distribution). p-Values <0.05 were deemed to be statistically significant. Statistical analyses were performed with the aid of the software SPSS version 23.
Results
Precision assessment of the BMD assay
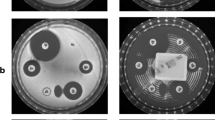
The original vancomycin MIC values of the isolates selected for this study, as measured by the Etest, are shown in Fig. 1a. Likewise, the vancomycin MIC values distribution of isolates measured in the first and second runs are depicted in Fig. 1b, c, respectively. The modal MIC value was 0.75 μg/mL in both runs, and the mean MIC values obtained in the first and second runs were 0.70 μg/mL [95% confidence interval (CI), 0.65–0.74 μg/mL] and 0.74 μg/mL (95% CI, 0.70–0.79 μg/mL), respectively. The intra-assay CVs were 22.4% and 20.9% in runs 1 and 2, respectively, the standard deviation being 0.15 μg/mL in both runs. The mean of the differences of MIC values for all isolates obtained across the two runs was 0.04 μg/mL (95% CI, 0.011–0.07 μg/mL) and the standard deviation of the differences was 0.119 μg/mL. In fact, the vancomycin MIC values differed less than 0.1 μg/mL between runs for most isolates (n = 36) and no more than 0.5 μg/mL for all isolates. The inter-assay CV was 21.7%. The mean of the differences of MIC values for ten isolates obtained across a single run performed in parallel at two different centers (HCU and HSFB) was also 0.04 μg/mL (95% CI, 0–0.08 μg/mL) and the standard deviation of the differences was 0.12 μg/mL.
Distribution of vancomycin minimum inhibitory concentration (MIC) values among isolates used in the study (n = 50). a Original MIC values measured by the Etest. Vancomycin MIC values measured by broth microdilution (BMD) in the first (b) and second (c) runs. The MICs of each isolate in b and c are the mean values of four replicates
Comparable intra- and inter-assay CVs were observed when frozen vancomycin plates were used for vancomycin MIC measurements (intra-assay CV of 21.9% and inter-assay CV of 22.1%). In addition, the MIC values measured when using frozen plates (mean, 0.74 μg/mL; 95% CI, 0.68–0.79 μg/mL) were similar to those measured in the two runs.
Effect of cryopreservation on vancomycin MIC values
A decrease in vancomycin MIC values measured by BMD methods has been reported to occur following SA cryopreservation for a variable period of time [8, 12]. We evaluated whether this was the case in our experimental conditions. A total of 15 SA isolates (MSSA, n = 9 and MRSA, n = 6) were cryopreserved for 1 or 3 months, then thawed and reassayed in quadruplicate. As shown in Fig. 2, cryopreservation had no impact on vancomycin MIC values [median of the mean vancomycin MIC values for each isolate: 0.62 μg/mL at baseline previous to cryopreservation, 1 month, and 3 months (p = 0.569 and p = 0.924 for the differences between baseline and 1 month and baseline and 3 months values, respectively].
Correlation between vancomycin MIC values measured by BMD and the Etest
A total of 17 SA isolates (MSSA, n = 8 and MRSA, n = 9) were tested by the Etest method at the time of the second BMD run using three different inocula. The data are shown in Fig. 3. The mean vancomycin MIC values obtained by the Etest using a standard inocula (0.5 McFarland) (mean, 1.13 μg/mL; 95% CI, 0.98–1.29 μg/mL) were significantly higher (b ≤ 0.001) than those measured by BMD (mean 0.75 μg/mL; 95% CI, 0.70–0.79 μg/mL), and the MIC values measured by the two methods correlated poorly (Rho, 0.319; p = 0.148). Nevertheless, the mean MIC values obtained by the Etest using lower inocula (mean of 0.77 and 0.74 μg/mL for 107 or 106 CFU/mL, respectively) and those measured by BMD were comparable and correlated significantly (p = 0.004 for 107 CFU/mL and p = 0.029 for 106 CFU/mL).
Inter-assay coefficient of variation of the Etest method
The precision of the Etest for SA vancomycin MIC measurement using three different bacterial inocula was evaluated next. The vancomycin MICs of 17 SA isolates (see above) were measured in two different runs using the same SA subcultures. We observed that the inter-assay CV of the Etest decreased steadily in parallel to that of bacterial inocula. Specifically, the mean of the differences between runs was 0.133 μg/mL (95% CI, −0.061 to 0.168 μg/mL) when the standard inoculums was used, 0.057 μg/mL (95% CI, −0.051 to 0.167 μg/mL) for a 1 × 107 inoculum, and 0.034 μg/mL (95% CI, −0.006 to 0.075 μg/mL) for a 1 × 106 inoculum.
Discussion
Here, we report on the development of a BMD method that incorporates incremental vancomycin concentrations within the susceptible range not only in between the conventional log2 dilutions, but also in between those on the Etest scale, whose precision and repeatability exceeds by far that generally accepted for dilution-based reference MIC methods (± 1 log2 dilution). In fact, the vancomycin MIC values measured within individual runs, across the two runs at one center, and across one run performed at two different centers differed by less than 0.1 μg/mL for most isolates and no more than 0.5 μg/mL for all isolates. BMD assays measuring vancomycin MICs at 0.25 μg/mL intervals, thus mimicking vancomycin concentrations on Etest strips, have been previously developed [8, 13, 14]. In one of these studies [8], the authors evaluated the standard deviation of the log2 MICs obtained by testing five isolates in quintuplicate in the same day (within-run precision) and found that this was below 1, this being slightly higher that than observed in the current study. In addition, the intra- and inter-assay CVs for our BMD method were around 20%, which is an acceptable figure when handling discrete variables [15].
It is of interest the fact that vancomycin MIC values measured when using frozen plates were comparable to those determined in non-cryopreserved plates. Thus, MIC plate batches may be prepared using the same lots of reagents, stored, and used at the investigators’ convenience, with no impact on vancomycin MIC values.
Previously published data indicated that cryopreservation of SA isolates beyond 6–12 months resulted in significant decreases of vancomycin MIC values with respect to those measured at baseline [8, 12]. This effect is noticeable even after 3 months of storage when the Etest is used for vancomycin MIC measurements [5]. With the idea of conducting a prospective multicenter study to evaluate the impact of vancomycin MICs, as measured by our BMD method, on SA bacteremia clinical outcome, and considering that MIC testing of cryopreserved isolates every 1–3 months would be a feasible schedule, we evaluated the effect of storage at −20 °C during 1 and 3 months on vancomycin MIC values. We found this storage period to have no impact on the vancomycin MIC values. This finding is of relevance to surveillance programs handling frozen isolates.
In agreement with previous studies [6,7,8, 13, 16], vancomycin MIC values measured by the Etest using a standard inoculum (0.5 McFarland) were higher than and correlated poorly with those measured by BMD. We tested the hypothesis that this is the result of the higher inoculum density used in the former method, as hinted by Charlton et al. [8]. To our knowledge, here, we show, for the first time, that this was, indeed, the case. In effect, vancomycin MIC values measured by the Etest when using lower inocula (either 107 or 106 CFU/mL) were comparable to and correlated significantly with those measured by BMD. Thus, vancomycin MIC values measured by the Etest and BMD can be harmonized simply by using smaller inocula in the former assay. This is, in our opinion, a relevant finding that must be taken into consideration in future prospective studies aimed at elucidating the impact of vancomycin MIC values on clinical outcomes, clarifying treatment decisions based upon vancomycin MICs within the susceptible range, and interpreting experimental evidences of vancomycin MIC creep. In addition, an improvement in the precision of the Etest method was achieved by decreasing the bacterial inocula.
Our study has several limitations. First, despite the inclusion of a wide range of SA clonal types in this study, selection biases cannot be completely ruled out (in particular for MRSA isolates). Second, the age of the colonies (and, thus, the phase of bacterial growth) used for BMD testing were not similar (the time elapsed between runs was 6 h). It is uncertain as to what extent this may have influenced vancomycin MIC measurements.
In summary, we developed a robust BMD assay for measuring SA vancomycin MICs within the susceptible range. Methods such as the one described herein, which certainly requires extensive inter-center validation, are needed to elucidate the impact of vancomycin MICs on the clinical outcomes of invasive SA infections.
References
Rodríguez-Créixems M, Alcalá L, Muñoz P, Cercenado E, Vicente T, Bouza E (2008) Bloodstream infections: evolution and trends in the microbiology workload, incidence, and etiology, 1985–2006. Medicine (Baltimore) 87:234–249
van Hal SJ, Lodise TP, Paterson DL (2012) The clinical significance of vancomycin minimum inhibitory concentration in Staphylococcus aureus infections: a systematic review and meta-analysis. Clin Infect Dis 54:755–771
Jacob JT, DiazGranados CA (2013) High vancomycin minimum inhibitory concentration and clinical outcomes in adults with methicillin-resistant Staphylococcus aureus infections: a meta-analysis. Int J Infect Dis 17:e93–e100
Kalil AC, Van Schooneveld TC, Fey PD, Rupp ME (2014) Association between vancomycin minimum inhibitory concentration and mortality among patients with Staphylococcus aureus bloodstream infections: a systematic review and meta-analysis. JAMA 312:1552–1564
Falcón R, Madrid S, Tormo N, Casañ C, Albert E, Gimeno C, Navarro D (2015) Intra- and interinstitutional evaluation of an Etest for vancomycin minimum inhibitory concentration measurement in Staphylococcus aureus blood isolates. Clin Infect Dis 61:1490–1492
Kruzel MC, Lewis CT, Welsh KJ, Lewis EM, Dundas NE, Mohr JF, Armitige LY, Wanger A (2011) Determination of vancomycin and daptomycin MICs by different testing methods for methicillin-resistant Staphylococcus aureus. J Clin Microbiol 49:2272–2273
Muñoz-Cobo B, Sancho-Tello S, Costa E, Bravo D, Torregrosa I, de Lomas JG, Navarro D (2011) Differences in vancomycin minimum inhibitory concentrations for Staphylococcus aureus obtained with the automated Phoenix™ system, the Clinical and Laboratory Standards Institute broth microdilution method and the standard Etest. Int J Antimicrob Agents 37:278–279
Charlton CL, Hindler JA, Turnidge J, Humphries RM (2014) Precision of vancomycin and daptomycin MICs for methicillin-resistant Staphylococcus aureus and effect of subculture and storage. J Clin Microbiol 52:3898–3905
Falcón R, Martínez A, Albert E, Madrid S, Oltra R, Giménez E, Soriano M, Vinuesa V, Gozalbo D, Gil ML, Navarro D (2016) High vancomycin MICs within the susceptible range in Staphylococcus aureus bacteraemia isolates are associated with increased cell wall thickness and reduced intracellular killing by human phagocytes. Int J Antimicrob Agents 47:343–350
Clinical and Laboratory Standards Institute (CLSI) (2012) Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; Approved standard—Ninth edition. CLSI document M07-A9. CLSI, Wayne
Clinical and Laboratory Standards Institute (CLSI) (2014) Performance standards for antimicrobial susceptibility testing; Twenty-fourth informational supplement. CLSI document M100-S24. CLSI, Wayne
Ludwig F, Edwards B, Lawes T, Gould IM (2012) Effects of storage on vancomycin and daptomycin MIC in susceptible blood isolates of methicillin-resistant Staphylococcus aureus. J Clin Microbiol 50:3383–3387
Shoji H, Maeda M, Shirakura T, Takuma T, Ugajin K, Fukuchi K, Ishino K, Niki Y (2015) More accurate measurement of vancomycin minimum inhibitory concentration indicates poor outcomes in meticillin-resistant Staphylococcus aureus bacteraemia. Int J Antimicrob Agents 46:532–537
Hos NJ, Jazmati N, Stefanik D, Hellmich M, AlSael H, Kern WV, Rieg S, Wisplinghoff H, Seifert H, Kaasch AJ (2017) Determining vancomycin Etest MICs in patients with MRSA bloodstream infection does not support switching antimicrobials. J Infect 74:248–259
Reed GF, Lynn F, Meade BD (2002) Use of coefficient of variation in assessing variability of quantitative assays. Clin Diagn Lab Immunol 9:1235–1239
Rybak MJ, Vidaillac C, Sader HS, Rhomberg PR, Salimnia H, Briski LE, Wanger A, Jones RN (2013) Evaluation of vancomycin susceptibility testing for methicillin-resistant Staphylococcus aureus: comparison of Etest and three automated testing methods. J Clin Microbiol 51:2077–2081
Acknowledgments
We are grateful to the residents of the Microbiology Service of Hospital Clínico Universitario. Estela Giménez holds a Río Hortega research contract from the Carlos III Health Institute (ISCIII) (ref. CM16/00200). Eva María Mateo is grateful to the Ministry of Economy and Competitiveness (MINECO, Spanish Government) for a postdoctoral contract “Juan de la Cierva” (ref. FJCI-2015-25992).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical statement
This work uses clinical bacterial isolates from human infections. No tissue material or other biological material was stored from the patients, only subcultured bacterial isolates. This local ethical committee does not require approval for the type of study in our setting. All information regarding these isolates was anonymized.
Funding
The current study received no direct funding.
Informed consent
Not applicable (as discussed with the institutional medical ethical committee).
Rights and permissions
About this article
Cite this article
Falcón, R., Mateo, E.M., Talaya, A. et al. Reproducible measurement of vancomycin MICs within the susceptible range in Staphylococcus aureus by a broth microdilution method with a “quasi-continuum” gradient of antibiotic concentrations. Eur J Clin Microbiol Infect Dis 36, 2355–2360 (2017). https://doi.org/10.1007/s10096-017-3067-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-017-3067-8