Abstract
We conducted a retrospective matched cohort study to examine the impact of isolation of multi-drug-resistant (MDR) Acinetobacter baumannii on patient outcomes. Cases from whom MDR A. baumannii was isolated in a clinical culture (n = 118) were compared with controls from whom MDR A. baumannii was not isolated (n = 118). Cases and controls were matched according to ward, calendar month of hospitalization, and duration of hospitalization before culture. The following outcomes were compared in multivariable analysis: in-hospital mortality, length of stay, need for mechanical ventilation, and functional status at discharge. MDR A. baumannii was determined to be a pathogen in 72% of cases. In 36% of cases, the patient died, versus 21% of controls (odds ratio [OR] 2.21, 95% confidence interval [CI] 1.17–4.16, P = 0.014). Median length of stay for surviving cases was 17 days, versus 11 for surviving controls (multiplicative effect 1.55, 95% CI 0.99–2.44, P = 0.057). Fifty-two percent of cases required mechanical ventilation, versus 25% of controls (OR 3.72, 95% CI 1.91–7.25, P<0.001); 60% of surviving cases were discharged with reduced functional status, versus 38% of controls (OR 4.4, 95% CI 1.66–11.61, P = 0.003). In multivariable analysis, clinical isolation of MDR A. baumannii remained a significant predictor of mortality (OR 6.23, 95% CI 1.31–29.5, P = 0.021), need for mechanical ventilation (OR 7.34, 95% CI 2.24–24.0, P<0.001), and reduced functional status on discharge (OR 7.93, 95% CI 1.1–56.85, P = 0.039). Thus, MDR A. baumannii acquisition is associated with severe adverse outcomes, including increased mortality, need for mechanical ventilation, and reduced functional status.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Multi-drug-resistant (MDR) Acinetobacter baumannii is a nosocomial pathogen of increasing importance. Once considered an opportunistic pathogen of relatively low virulence affecting mostly patients in intensive care units (ICUs) [1], this organism has a selective advantage over many other nosocomial bacteria beause of its hardiness [2, 3], which allows cross-transmission, and its intrinsic and acquired resistance to many antimicrobial agents. Whereas multi-drug resistance is rarely found in community isolates of A. baumannii, the prevalence of the MDR phenotype among hospital isolates has increased during the last decade, and MDR A. baumannii has become a leading pathogen in many hospitals worldwide [4, 5]. Hospital outbreaks have been described from various geographical areas [6–9], and in some areas, this organism has become endemic [9]. It has been reported to be an important pathogen in wounded soldiers returning from Iraq and Afghanistan [10–12]. It has also been described infecting Iranian soldiers wounded during the Iran-Iraq conflict [13] and in trauma patients from mass casualty situations, such as suicide bombings [14], patients affected by the tsunami in Southeast Asia in December 2004 [15], and survivors of the earthquake in Turkey in 1999 [16]. Since these patients are often transferred to tertiary care centers, sometimes in distant geographic areas, they may become a source of transmission in previously non-endemic hospitals.
Since the late 1990s, MDR A. baumannii has been an important nosocomial pathogen in several Israeli hospitals, including ours [17, 18]. We have recently reported its complex epidemiology and individual risk factors for infections with MDR A. baumannii [18]. Infections caused by resistant organisms are thought to result in higher morbidity and mortality, prolonged hospitalization, and increased costs compared with infections caused by antibiotic-susceptible strains [19–21]. Data concerning the impact of MDR A. baumannii are insufficient and controversial. Although the reported crude mortality of patients with these organisms reaches 50%, the attributable mortality and other effects on patient outcomes are difficult to study, since the affected patients are often severely ill and have prolonged spells of hospitalization, and poor prognoses, irrespective of their infection. Indeed, reported attributable mortality ranges from 7% to 36% [1, 22–27].
The current study was designed to examine the impact of MDR A. baumannii acquisition on the outcomes of patients at our institution. The impact of resistant organisms may be related to two separate phenomena: replacement and addition of infections, i.e., in certain cases, resistant organisms merely replace the susceptible form as causative pathogens (replacement), whereas in other cases, resistant organisms cause additional infections (addition). In our institution, non-MDR A. baumannii rarely causes nosocomial infections. Thus, infections caused by MDR A. baumannii are additive and do not replace infections caused by a susceptible variant of the organism. We therefore elected to analyze the impact of MDR A. baumannii by using the additive model and to compare patients from whom MDR A. baumannii was isolated, with similar patients at risk from whom it was not [28]. On the basis of our earlier findings, we considered clinical isolation of A. baumannii as indicative of acquisition in the hospital [18].
Methods
Hospital setting, data collection, and microbiology
This study was performed at the Tel Aviv Sourasky Medical Center, Israel, an 1100-bed tertiary-care teaching hospital with 70,000 admissions annually. Approximately 82,500 clinical microbiological cultures are processed annually. This is a retrospective matched cohort study designed to identify the effect of clinical isolation of MDR A. baumannii on patient outcomes.
Cases were defined as patients from whom MDR A. baumannii was isolated from any clinical culture during the 6-month period from January 1st through June 30th, 2001. A control patient was matched to each case patient based on temporo-spatial factors according to the scheme described by Carmeli et al. [29]. Controls were randomly chosen from the list of patients who stayed on the same ward in the same calendar month as the matched case and who had been hospitalized for at least the same number of days as the matched case had been at the time of culture. Controls did not have MDR A. baumannii isolated in any clinical culture. As we wanted the cohort to include the entire population at risk for MDR A. baumannii isolation, we did not exclude patients from whom non-MDR A. baumannii had been previously isolated from either the case or the control group. Random control selection was performed as follows: a list of all possible controls was created, each candidate was assigned a random number, and the highest random number was chosen.
Cases and control patients were included only once. Data were collected from the patients’ records and from hospital computerized databases and entered into a prepared electronic abstraction sheet. The following information was recorded for each patient: age, sex, use of tobacco and alcohol prior to hospitalization, cause and ward of hospitalization, transfer from another institution or ward within our institution, ICU stay, underlying disorders, immunosuppressive therapy, severity of illness, functional capacity and neurological condition at the time of isolation of A. baumannii or matching and at discharge, placement of a Foley catheter, invasive devices, surgery, mechanical ventilation, dialysis, infection versus colonization (cases only), and antibiotic therapy. A. baumannii was isolated from clinical specimens submitted to the clinical microbiology laboratory and identified by using the Gram-Negative Identification Panel (Microscan, Dade Behring, Sacramento, Calif.). Susceptibilities were determined by automated microdilution broth testing (Neg/Urine Combo panel, Dade Behring). Resistance to imipenem and meropenem was confirmed by using the Kirby-Bauer disk diffusion method performed according to CLSI guidelines [30].
Definitions
We defined A. baumannii as MDR when the organism was resistant to all of the following agents: piperacillin/tazobactam, cefepime, ceftazidime, aztreonam, ciprofloxacin, gentamicin, and tobramycin). Isolates susceptible to amikacin, ampicillin-sulbactam, imipenem, meropenem, minocycline, and colistin could still be considered MDR. Infection was defined according to CDC definitions and modified to include community-acquired infections and to exclude asymptomatic bacteriuria [31]. Patients with MDR A. baumannii who did not fulfill criteria for infection were classified as being colonized.
Standard criteria were used to define underlying disorders. Disease was considered to be active if signs of disease were clinically apparent or if the patient received treatment for the disease. A patient was considered to be under immunosuppressive therapy if he had received chemotherapy within 3 weeks, if he was treated with at least 20 mg prednisone daily for at least 2 weeks before entering the study, or if he recently received anti-rejection or other immunosuppressive therapy.
Severity of illness attributable to co-morbidities was defined according to the McCabe score [32]. Functional capacity during the index hospitalization was divided into two categories: independent or requiring significant assistance in activities of daily living. Renal failure was defined as a serum creatinine level above 2 mg/dl. Neurological function was classified according to three conditions: fully conscious, confused/demented, or unconscious.
We recorded the number of antibiotics that each patient received from the time of admission until study inclusion; home antibiotic therapy was recorded separately. Recent hospitalization was defined as hospital stay up to 3 months before the index hospitalization. Surgery, mechanical ventilation, and any invasive procedure that took place up to 1 month prior to inclusion in the study were recorded.
Statistical analysis
Statistical analysis was performed by using Stata version 7 (Stata, College Station, Tex.). All analyses were matched in order to correspond to the study design. Univariate analysis was performed by using the McNemar and paired Student’s t tests. Variables with a P-value of <0.2 in univariate analysis were included in the multivariate model. Multivariate analysis was performed by using conditional (fixed effect) logistic regression for dichotomized outcomes (in-hospital mortality, mechanical ventilation after inclusion in the study, surgery after inclusion in the study, functional status at discharge, and discharge disposition). For length of stay after inclusion in the study, matched linear regression (linear regression with absorbed variable) of the log-transformed length of stay was performed. The effect estimate in the mortality model was reported as the odds ratio (OR) and in the length-of-stay model as the multiplicative effect (ME; the anti-log of the β-coefficient [33]).
Variables with a P-value of <0.1 were retained in the multivariable model. Variables that were not retained in the model by this procedure were then tested for confounding by adding them one at a time to the model and examining their effects on the β-coefficients. Variables that caused substantial confounding (change in the β-coefficient of greater than 10%) were included in the final model.
In addition to examining statistical significance and confounding, effect modification between variables was evaluated by testing appropriate interaction terms for statistical significance. Colinearity was examined by interchanging variables and examining the effect on the model. All statistical tests were two-tailed. In the final multivariable models, a P-value of ≤0.05 was considered significant.
Results
Demographics and clinical characteristics
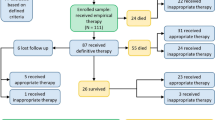
We identified 133 patients with a clinical culture of MDR A. baumannii. Four patients were not hospitalized adults (one was a child, and three were adult outpatients) and were therefore excluded. Charts were available for review for 120 cases. For two, no controls could be matched. Thus, a total of 236 patients were included in the study (118 cases and their matched controls). Of these, six (2.5%) had non-MDR A. baumanii isolated prior to inclusion in the study. MDR A. baumannii was initially isolated from the following sites: respiratory tract, 38 (32%); wounds, 23 (19.5%); urine, 22 (19%); blood 19, (16%); sterile fluids or catheter tips, 16 (13.5%).
Patients’ characteristics are displayed in Table 1. Cases were similar to their matched controls with respect to age (average 67.7 versus 64.4 years old) and sex distribution (60% versus 50% male). The groups were also similar with respect to tobacco and alcohol use and to the prevalence of lung disease, diabetes, kidney disease, liver disease, malignancy, and transplantation. The prevalence of ischemic heart disease was higher among cases than among controls (69% versus 52%, OR 2.33, P = 0.006), and cases tended to have been transferred from another institution more often than controls (20% versus 10%, OR 2.12, P = 0.065).
Clinical outcome of isolation of MDR A. baumannii
Mortality
A total of 42 case patients (36%) died in hospital. Among the 85 cases with clinical infection caused by A. baumannii, 41 (48%) died, and among the 33 patients in whom A. baumannii was classified as a colonizer, only one (3%) died. Among 118 control patients, 25 (21%) died. The crude fatality ratio was significantly higher among cases than among controls (OR 2.21, 95% CI 1.17–4.16, P = 0.014)
Other variables identified by univariate analysis as risk factors for in-hospital mortality included age >70 years (OR 3.2, P = 0.023), unconsciousness (OR 2.8, P = 0.048), a McCabe score of 3, indicating life expectancy of less than 6 months (OR 27, P<0.001), malignancy (OR 2.6, P = 0.04), presence of a Foley catheter (OR 5.5, P = 0.027), and A. baumannii infection rather than colonization (OR 6, P<0.001). Variables that approached statistical significance (0.05<P<0.1) as risk factors for mortality were surgery, mechanical ventilation, and poor functional status before isolation of A. baumannii (Table 2). In the multivariable analysis (Table 3), the following variables were included in the model for mortality: being a case patient (OR 6.23, P = 0.021), having malignant disease (OR 6.49, P = 0.064), and having a McCabe score of 3 (OR 64.4, P = 0.003).
Additional factors associated with mortality included need for mechanical ventilation after A. baumannii isolation, and need for antibiotic treatment after isolation. The fact that these covariates represent occurrences after the isolation of A. baumannii indicates that they are likely intermediate variables rather than confounders [34].
Length of hospital stay
The average hospital stay after study entry among surviving cases tended to be longer than that among surviving controls (28.34 ± 43 versus 17.5 ± 23.7 days, median 17 versus 11 days, P = 0.057). Among the cases defined as having clinical infection with A. baumannii but who survived to discharge, the average hospital stay was significantly longer (36.5 ± 53 days, P = 0.024; median 20 days).
The following variables were found to be risk factors for prolonged hospital stay in univariate analysis (Table 2): infection rather than colonization (ME 1.92, P = 0.02), male sex (ME 2.34, P = 0.009), age (considered as a continuous variable; ME 1.02, P = 0.03), unconsciousness (ME 1.54, P = 0.05), and mechanical ventilation before A. baumannii isolation (ME 2.89, P = 0.003).
In multivariable analysis (Table 3), only male sex was significantly associated with prolonged length of hospital stay (ME 1.95, P = 0.04). Other factors included in the model were age >70 years (ME 1.75, P = 0.08) and being a case (ME 1.4, P = 0.13).
Mechanical ventilation
Ninety-one patients (38%) had to be mechanically ventilated after entering the study (52% of cases versus 25% of controls). In univariate analysis, the following variables were found to be statistically significant predictors of mechanical ventilation: being a case (OR 3.72, P<0.001), having lung disease (OR 3, P = 0.02), being bed-ridden before A. baumannii isolation (OR 5.3, P = 0.008), unconsciousness (OR 11.05, P<0.001), having a Foley catheter (OR 6.33, P = 0.003), or a McCabe score of 3 (OR 3.23, P = 0.005). In multivariate analysis (Table 3), the following variables were found to be statistically significant as risk factors for mechanical ventilation: being a case (OR 7.34, P<0.001), having a Foley catheter (OR 5.84, P = 0.03), and having a McCabe score of 3 (OR 11.4, P = 0.005).
Functional status at discharge
Among surviving patients, cases were more likely than controls to be discharged with low functional status, i.e., requiring significant assistance in activities of daily living (60% versus 38%, OR 4.4, P = 0.003), and to be discharged to a nursing home (55% versus 42%, OR 3.33, P = 0.01).
In univariate analysis, the following variables were found to be predictors of poor functional status at discharge: having poor functional status before culture (OR 18, P = 0.005), age >70 years (OR 4, P = 0.03), and having a Foley catheter (OR 13, P = 0.013). In multivariable analysis, after adjustment for functional status before culture, being a case remained an independent predictor of poor functional status at discharge (OR 7.93, P = 0.039; Table 3).
Discussion
The literature regarding clinical outcomes of MDR A. baumannii acquisition among hospitalized patients is inconclusive regarding one principal question: are the adverse outcomes associated with the organism directly attributable to its acquisition, or is the presence of MDR A. baumannii merely a marker of poor outcome in patients with extensive comorbid conditions?
In an attempt to allay some of the confusion on the subject, Falagas et al. conducted a systematic review of published case-control studies and examined the effect of colonization or infection with A. baumannii on morbidity and mortality by comparing critically ill patients with A. baumannii with matched controls without A. baumannii [5]. Whereas their findings suggested increased attributable mortality and ICU stay in patients with A. baumannii infection and colonization, their ability to draw conclusions was limited by small sample sizes and by methodological heterogeneity in the included studies.
Our study, the largest to date on outcomes of MDR A. baumannii acquisition in hospitalized patients, adds to the currently limited supply of data on the topic. We have found that, after control for confounding variables, patients with MDR A. baumannii were significantly more likely to die, to require mechanical ventilation, and to be discharged with poor functional status than patients without MDR A. baumannii. Moreover, the findidng that the overwhelming majority of case patients who died and those mechanically ventilated had clinical infection with A. baumannii rather than just colonization (41 of 42 and 56 of 61, respectively) suggests that the organism is a cause, rather than a mere marker, of these deleterious outcomes. Furthermore, a certain proportion of colonized patients will ultimately develop infection and thereby potentially be at higher risk for adverse outcomes. Ideally, future analyses that can account for variation in status between colonization and infection over time will help to elucidate more clearly the precise effect of MDR A. baumannii isolation on outcomes.
That MDR A. baumannii should lead to poor outcomes is certainly plausible for a number of reasons. First, the patient populations affected by the organism are largely debilitated [35]. The insult of a bacterial infection added to multiple comorbidities may certainly result in clinical deterioration. Moreover, the types of infections commonly caused by A. baumannii, particularly pneumonia or bacteremia (occurring in over 50% of infected patients in our study), may be particularly severe. In addition, the high degree of antimicrobial resistance found among these pathogens may result in a deleterious delay in an institution of effective therapy, as empiric coverage may not include the agents ultimately to be proven necessary when culture and susceptibility data become available [36, 37]. Finally, the emergence of pan-resistant and nearly pan-resistant strains of A. baumannii may result in an inability to treat infections caused by this pathogen optimally with currently available antibiotics [38].
Why have different investigators arrived at differing conclusions regarding the role of A. baumannii in causing poor outcomes [1, 22–27]? The reasons for the differences in results can be divided into four basic categories: methodological, population, therapeutic, and bacterial. Methodological differences include those of study design, choice of comparison group (if any), control for confounding variables such as severity of underlying illness and comorbid conditions, and decisions regarding the matching of cases and controls according to specific attributes. Whereas these types of differences could certainly account for differing results, there is a lack of accord in results, even among studies employing similar methodology [5, 39].
Population differences, including medical versus surgical patients, adults versus children, outpatients versus inpatients, and differences in underlying comorbidity, may play a causative role in outcome differences among studies. Nevertheless, population differences are unlikely a major cause of the heterogeneity in results observed in outcome studies of MDR A. baumannii acquisition, as this is an organism isolated primarily among inpatients with extensive contact with the healthcare system and significant underlying comorbidity and antimicrobial exposure [18, 35]. Moreover, as the review by Falagas demonstrates, even studies of critically ill patients alone fail to yield definitive outcome results [5]. In our study, even after extensive efforts to control for differences in patient characteristics, we have found a significant and strong effect of MDR A. baumannii acquisition on outcomes.
Treatment differences among various cohorts of infected patients may likewise play a role in differing outcomes. Falagas et al. [37] have demonstrated worse outcomes in patients with A. baumannii bacteremia receiving inappropriate empirical therapy compared with those who received appropriate treatment. Even in established infection with an identified organism, and even when choices are within the susceptibility profile of the pathogen, differences in antibiotic selection may affect the outcome of treatment [40, 41]. Yet in MDR A. baumannii infection, the spectrum of susceptibility to presently available antibiotics is so narrow that little room is left for any variation in treatment options.
We postulate that the differences in observed outcomes of MDR A. baumannii infection and colonization reported in the literature are primarily attributable to differences in the bacteria themselves. In an earlier study at our institution, among 51 unique-patient MDR A. baumannii clinical strains available for genotyping, fully 10 distinct clones were identified [18]. A considerable degree of difference in genome has been observed between different genotypes of this organism. Moreover, the advent of whole-genome sequencing has allowed for quantitation and characterization of the wide array of resistance determinants that strains of A. baumannii are known to acquire [42]. While little is known about determinants of virulence in A. baumannii, there is evidence of virulence factors selectively present among resistant strains of other bacterial pathogens [43, 44]. Therefore, heterogeneity among genotypes, resistance determinants, and virulence factors might play the predominant role in the differences observed in outcome studies of MDR A. baumannii acquisition performed to date worldwide.
Our study adds to the growing body of data regarding the deleterious effects of MDR A. baumannii infection and supports the conclusion that this pathogen is independently associated with poor outcomes. Further studies are needed to elucidate the bacterial factors associated with differences in clinical outcomes.
References
Seifert H, Strate A, Pulverer G (1995) Nosocomial bacteremia due to Acinetobacter baumannii. Clinical features, epidemiology, and predictors of mortality. Medicine (Baltimore) 74:340–349
Getchell-White SI, Donowitz LG, Groschel DH (1989) The inanimate environment of an intensive care unit as a potential source of nosocomial bacteria: evidence for long survival of Acinetobacter calcoaceticus. Infect Control Hosp Epidemiol 10:402–407
Wendt C, Dietze B, Dietz E, Ruden H (1997) Survival of Acinetobacter baumannii on dry surfaces. J Clin Microbiol 35:1394–1397
Gales AC, Jones RN, Forward KR, Linares J, Sader HS, Verhoef J (2001) Emerging importance of multidrug-resistant Acinetobacter species and Stenotrophomonas maltophilia as pathogens in seriously ill patients: geographic patterns, epidemiological features, and trends in the SENTRY Antimicrobial Surveillance Program (1997–1999). Clin Infect Dis 32 Suppl 2:S104–S113
Falagas ME, Bliziotis IA, Siempos II (2006) Attributable mortality of Acinetobacter baumannii infections in critically ill patients: a systematic review of matched cohort and case-control studies. Crit Care 10:R48
National Nosocomial Infections Surveillance (NNIS) (1996) National Nosocomial Infections Surveillance (NNIS) report, data summary from October 1986–April 1996, issued May 1996. A report from the National Nosocomial Infections Surveillance (NNIS) System. Am J Infect Control 24:380–388
Corbella X, Montero A, Pujol M, Domínguez MA, Ayats J, Argerich MJ, et al (2000) Emergence and rapid spread of carbapenem resistance during a large and sustained hospital outbreak of multiresistant Acinetobacter baumannii. J Clin Microbiol 38:4086–4095
Go ES, Urban C, Burns J, Kreiswirth B, Eisner W, Mariano N, et al (1994) Clinical and molecular epidemiology of Acinetobacter infections sensitive only to polymyxin B and sulbactam. Lancet 344:1329–1332
Landman D, Quale JM, Mayorga D, Adedeji A, Vangala K, Ravishankar J, et al (2002) Citywide clonal outbreak of multiresistant Acinetobacter baumannii and Pseudomonas aeruginosa in Brooklyn, NY: the preantibiotic era has returned. Arch Intern Med 162:1515–1520
Davis KA, Moran KA, McAllister CK, Gray PJ (2005) Multidrug-resistant Acinetobacter extremity infections in soldiers. Emerg Infect Dis 11:1218–1224
MMWR (2004) Acinetobacter baumannii infections among patients at military medical facilities treating injured U.S. service members, 2002–2004. MMWR Morb Mortal Wkly Rep 53:1063–1066
Aarabi B (1987) Comparative study of bacteriological contamination between primary and secondary exploration of missile head wounds. Neurosurgery 20:610–616
Heath CH OC, Lee RC, Pearman JW, McCullough C, Christiansen KJ (2003) A review of the Royal Perth Hospital Bali experience: an infection control perspective. Australian Infect Control 2:43–54
Maegele M, Gregor S, Steinhausen E, Bouillon B, Heiss MM, Perbix W, et al (2005) The long-distance tertiary air transfer and care of tsunami victims: injury pattern and microbiological and psychological aspects. Crit Care Med 33:1136–1140
Oncul O, Keskin O, Acar HV, Küçükardali Y, Evrenkaya R, Atasoyu EM, et al (2002) Hospital-acquired infections following the 1999 Marmara earthquake. J Hosp Infect 51:47–51
Paul M, Weinberger M, Siegman-Igra Y, Lazarovitch T, Ostfeld I, Boldur I, et al (2005) Acinetobacter baumannii: emergence and spread in Israeli hospitals 1997–2002. J Hosp Infect 60:256–60
Abbo A, Navon-Venezia S, Hammer-Muntz O, Krichali T, Siegman-Igra Y, Carmeli Y (2005) Multidrug-resistant Acinetobacter baumannii. Emerg Infect Dis 11:22–9
Acar JF (1997) Consequences of bacterial resistance to antibiotics in medical practice. Clin Infect Dis 24 Suppl 1:S17–S18
Cohen ML (1992) Epidemiology of drug resistance: implications for a post-antimicrobial era. Science 257:1050–1055
Schwaber MJ, Navon-Venezia S, Kaye KS, Ben-Ami R, Schwartz D, Carmeli Y (2006) Clinical and economic impact of bacteremia with extended-spectrum-beta-lactamase-producing Enterobacteriaceae. Antimicrob Agents Chemother 50:1257–1262
Beck-Sague CM, Jarvis WR, Brook JH, Culver DH, Potts A, Gay E, et al (1990) Epidemic bacteremia due to Acinetobacter baumannii in five intensive care units. Am J Epidemiol 132:723–733
Wisplinghoff H, Perbix W, Seifert H (1999) Risk factors for nosocomial bloodstream infections due to Acinetobacter baumannii: a case-control study of adult burn patients. Clin Infect Dis 28:59–66
Cisneros JM, Reyes MJ, Pachón J, Becerril B, Caballero FJ, García-Garmendía JL, et al (1996) Bacteremia due to Acinetobacter baumannii: epidemiology, clinical findings, and prognostic features. Clin Infect Dis 22:1026–1032
Blot S, Vandewoude K, Colardyn F (2003) Nosocomial bacteremia involving Acinetobacter baumannii in critically ill patients: a matched cohort study. Intensive Care Med 29:471–475
Falagas ME, Kopterides P, Siempos II (2006) Attributable mortality of Acinetobacter baumannii infection among critically ill patients. Clin Infect Dis 43:389–390
Fournier PE, Richet H (2006) The epidemiology and control of Acinetobacter baumannii in health care facilities. Clin Infect Dis 42:692–699
Kaye KS, Engemann JJ, Mozaffari E, Carmeli Y (2004) Reference group choice and antibiotic resistance outcomes. Emerg Infect Dis 10:1125–1128
Carmeli Y, Eliopoulos GM, Samore MH (2002) Antecedent treatment with different antibiotic agents as a risk factor for vancomycin-resistant Enterococcus. Emerg Infect Dis 8:802–807
Clinical and Laboratory Standards Institute (2005) Performance standards for antimicrobial susceptibility testing; fifteenth informational supplement. M100S15. Clinical and Laboratory Standards Institute, Wayne, Pa., USA
Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM (1988) CDC definitions for nosocomial infections. Am J Infect Control 16:128–140
McCabe WR, Jackson GG (1962) Gram-negative bacteremia. Arch Intern Med 110:847–855
Carmeli Y, Troillet N, Karchmer AW, Samore MH (1999) Health and economic outcomes of antibiotic resistance in Pseudomonas aeruginosa. Arch Intern Med 159:1127–1132
Schwaber MJ, Carmeli Y (2006) Antimicrobial resistance and patient outcomes: the hazards of adjustment. Crit Care 10:164
Falagas ME, Kopterides P (2006) Risk factors for the isolation of multi-drug-resistant Acinetobacter baumannii and Pseudomonas aeruginosa: a systematic review of the literature. J Hosp Infect 64:7–15
Harbarth S, Garbino J, Pugin J, Romand JA, Lew D, Pittet D (2003) Inappropriate initial antimicrobial therapy and its effect on survival in a clinical trial of immunomodulating therapy for severe sepsis. Am J Med 115:529–535
Falagas ME, Kasiakou SK, Rafailidis PI, Zouglakis G, Morfou P (2006) Comparison of mortality of patients with Acinetobacter baumannii bacteraemia receiving appropriate and inappropriate empirical therapy. J Antimicrob Chemother 57:1251–1254
Navon-Venezia S, Ben-Ami R, Carmeli Y (2005) Update on Pseudomonas aeruginosa and Acinetobacter baumannii infections in the healthcare setting. Curr Opin Infect Dis 18:306–313
Sunenshine RH, Wright MO, Maragakis LL, Harris AD, Song X, Hebden J, et al (2007) Multidrug-resistant Acinetobacter infection mortality rate and length of hospitalization. Emerg Infect Dis 13:97–103
Paterson DL, Ko WC, Von Gottberg A, Mohapatra S, Casellas JM, Goossens H, et al (2004) Antibiotic therapy for Klebsiella pneumoniae bacteremia: implications of production of extended-spectrum beta-lactamases. Clin Infect Dis 39:31–37
Zimhony O, Chmelnitsky I, Bardenstein R, Goland S, Hammer Muntz O, Navon Venezia S, Carmeli Y (2006) Endocarditis caused by extended-spectrum-beta-lactamase-producing Klebsiella pneumoniae: emergence of resistance to ciprofloxacin and piperacillin-tazobactam during treatment despite initial susceptibility. Antimicrob Agents Chemother 50:3179–3182
Fournier PE, Vallenet D, Barbe V, Audic S, Ogata H, Poirel L, et al (2006) Comparative genomics of multidrug resistance in Acinetobacter baumannii. PLoS Genet 2:e7
Diep BA, Carleton HA, Chang RF, Sensabaugh GF, Perdreau-Remington F (2006) Roles of 34 virulence genes in the evolution of hospital- and community-associated strains of methicillin-resistant Staphylococcus aureus. J Infect Dis 193:1495–1503
Sahly H, Aucken H, Benedi VJ, Forestier C, Fussing V, Hansen DS, et al (2004) Increased serum resistance in Klebsiella pneumoniae strains producing extended-spectrum beta-lactamases. Antimicrob Agents Chemother 48:3477–3482
Acknowledgments
This study was funded by a grant from the United States-Israel Binational Science Foundation. The Foundation had no role in the design or conduct of any aspect of the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abbo, A., Carmeli, Y., Navon-Venezia, S. et al. Impact of multi-drug-resistant Acinetobacter baumannii on clinical outcomes. Eur J Clin Microbiol Infect Dis 26, 793–800 (2007). https://doi.org/10.1007/s10096-007-0371-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-007-0371-8