Abstract
Background
Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis (AE) is a common cause of nonviral infectious encephalitis, which can be triggered by herpes simplex virus infection. Previous studies have shown that approximately 27% of herpes simplex encephalitis (HSE) patients produce anti-NMDAR antibodies within 3 months. Immunotherapy is recommended in this situation, but some symptoms usually remain in the 1-year follow-up.
Case presentation
A previously healthy 23-year-old Chinese young woman developed epileptic attack followed by psychiatric symptoms of confusion and irritation as well as cognitive deficits. Brain MRI showed hyperintense lesions of the right temporal lobe on DWI and T2 without contrast enhancement effects. Twenty-one days of acyclovir was administered based on the primary diagnosis of HSE. The anti-NMDAR antibody (IgG) was detected positively on day 11 after disease onset. She had improved cognitive function but suffered another grand mal epilepsy after the first course of intravenous immunoglobulin (IVIG) therapy combined with 1000 mg intravenous methylprednisolone. After discussion, another course of IVIG was started for 5 days. Her symptoms were well controlled with only mild cognitive deficits at the 1-year follow-up (mRS = 1).
Conclusions
Our case indicated that anti-NMDAR antibodies could develop earlier after HSE compared with previous data from adults. We suggested detecting AE antibodies simultaneously with each CSF analysis. Meanwhile, the second course of IVIG therapy was reasonable when symptoms were not controlled after the first course of IVIG combined with IV steroid treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis (AE) is now a widely accepted common type of autoimmune-mediated disorder in both children and adults and was first reported by Dalmau et al. in 2007 [1]. The typical clinical characteristics of AE are seizure, psychiatric disorders, cognitive deficits, autonomic dysfunction, central hypoventilation, language disorder, and dyskinesia [1,2,3,4,5]. The exact cause of this disease is uncertain, and it is frequently associated with a neoplastic condition (especially ovarian teratoma) [1]. On the other hand, AE can be triggered by infection of viruses, parasites, bacteria, and even Borrelia, and one of the most commonly reported AEs trigger is herpes simplex virus (HSV) infection [6,7,8]. According to previous studies, approximately 27% of herpes simplex encephalitis (HSE) patients produce anti-NMDAR antibodies within 3 months [8]. However, the condition of combined HSE and AE at the acute phase of disease and therapeutic management is still controversial. Here, we present the first Chinese case of an HSE patient whose anti-NMDAR antibody was positive on day 11 of the disease who received two courses of intravenous immunoglobulin (IVIG) therapy.
Case report
A 23-year-old Chinese young woman was found to have a seizure lasting approximately 10 min by her roommates. Two days later, she exhibited mood instability and increased irritability, presented as paroxysmal crying and laughing without any reason. On the fifth day, she was admitted to a psychiatric hospital. During the previous 5 days, she had experienced several minor focal seizures with mouth twitching. The brain MRI showed T2 and DWI hyperintense lesions of the right temporal lobe (Supplement 1). She had a fever of 37.8 degree centigrade on the sixth day. Ten days after the first symptom onset, she became calm under the control of sodium valproate and olanzapine. In the afternoon, she developed a generalized tonic-clonic seizure, and she was transferred to our hospital. Her past medical history was unremarkable. On examination, her vital signs were stable. She was responsive to verbal and painful stimulation but disoriented with regard to time and place. She was obtunded and confused by most of the questions we asked. The muscle tone was slightly decreased in her four limbs, but her strength was normal. Other physical examinations were unremarkable.
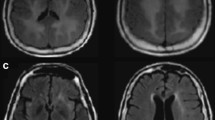
The repeated brain MRI on the day of admission showed a decline in the hyperintense signal on DWI and T2 (Fig. 1 and Supplement 2). Lumbar puncture on the second day of admission (11 days after symptom onset) showed normal intracranial pressure and clear cerebrospinal fluid (CSF). The CSF analysis indicated mild pleocytosis (48 × 106/L) with lymphocytic predominance. Glucose was slightly increased (4.8 mmol/L, reference range: 2.4–4.5 mmol/L). Protein and chloride levels were normal. HSV-1 PCR was positive in the CSF. IgM of the HSV was detected in the serum with 1.1 s/co (reference range < 1.0 s/co) as well as IgG (30 s/co, reference range < 1.0 s/co). Anti-NMDAR antibody (IgG) was positive both in the CSF and serum, with the same level of 1:32 by way of cell transfection analysis (CBA) detection methods. Additionally, the serum antibody of glutamic acid decarboxylase (GAD) was positive. A contrast-enhanced brain MRI was performed on the sixth day (15 days after symptom onset), which revealed T2, FLAIR, and DWI hyperintense lesions in the right lateral temporal lobe without a contrast enhancement effect (Fig. 1). Electroencephalography (EEG) showed diffuse slow waves, especially on the right temporal lobe. Both pelvic ultrasonography and serum antibody detection were negative for tumor screening.
She received 10 mg/kg intravenous acyclovir every 8 h for 21 days on the first day of admission with a primary diagnosis of HSE. Five days of intravenous methylprednisolone (1000 mg/day) was commended on day 12 of the disease because of the positive anti-NMDAR antibodies. However, her symptoms were not controlled well. Intravenous immunoglobulin therapy (0.4 g/kg/day for 5 days for 1 course) was initiated after a week of admission. After 5 days of intravenous immunoglobulin therapy, she was oriented to time and place and able to communicate with us briefly. However, she suffered another generalized tonic-clonic seizure on day 24 since disease onset, even though she was taking oral sodium valproate regularly (500 mg bid). We repeated CSF HSV PCR, which was negative. The EEG was consistent with the prior result. The second course of intravenous immunoglobulin therapy was started after a discussion with her family. One week later (31 days of disease), she was discharged with improved response and cognitive function. For the follow-up, brain MRI was repeated 3 months after symptom onset and showed almost complete resolution of the hyperintense lesion. She was capable of performing activities of daily life but not the previous school study (mRS = 1) at the 1-year follow-up (the case is summarized in Fig. 2).
Discussion
AE is an autoimmune disease that has been gradually recognized as a common cause of noninfective encephalitis. The exact cause of this disease is still unclear, and teratomas are one of the most common relevant conditions [1]. Meanwhile, virus infection might be another common trigger of AE. The clinical symptoms varied based on sex and age [9]. Symptoms of behavior and psychical disorders are more common in adults, especially in females [10]. Therefore, adults with AE are at risk of being misdiagnosed as mental illness, similar to our patient. There are no fixed prodromes of AE, but virus infection is one of the most common prior presentations [2]. The first individual case of a young adult with HSE followed by AE was reported in 2013 by Frank Leypoldt et al. [11]. Then, series of similar cases but not common in teenagers and adults were subsequently published (summarized in Supplement 3) [11,12,13,14,15,16,17,18,19]. It has been considered less likely for HSE and AE to coincide because of the low incidence of HSE (2–4/1,000,000 population per year) [20]. Instead, HSV infection is considered a trigger of AE, which usually presents as a relapse of prior HSE symptoms [18]. The mechanism of this secondary immune reaction is not fully clear, and molecular mimicry, protein misfolding, and dysregulation of immune regulators are some of the potential mechanisms [21].
According to a retrospective cohort study, 30% of HSE patients suffered AE in the same course of disease [22]. The anti-NMDAR antibodies (IgG) as well as other unknown neurosurface antibodies were detected within 90 days after onset of the initial symptoms of HSE [23]. However, the relapse of symptoms usually occurred weeks earlier than the examination of autoimmune antibodies. Except for a case of a 2-month-old infant whose anti-NMDAR antibody was positive in CSF 7 days after the onset of HSE symptoms, the median time interval from HSE until probable AE was generally more than 30 days in adults [12]. The young lady in our case already developed the anti-NMDAR antibody (1:32 in CSF and serum) on day 11, which was earlier than previous data from adults. It is worth noting that Flora Schein et al. reported a case of a 52-year-old female whose anti-NMDAR antibody was positive on day 50+ of the disease with relapse of AE symptoms, but the antibody was also positive on the retrospective detection of the first CSF sample taken on day 4 since disease onset [18]. Therefore, we hypothesized that HSV could trigger the production of anti-NMDAR antibodies even earlier than in our case.
Additionally, the serum GAD antibody was detected in our patient. To our knowledge, except for one case of the Spanish retrospective study, which described the phenomenon of anti-NMDAR antibodies with coexisting GAD antibodies [12], our case is the second to reveal that both anti-NMDA receptor and GAD antibodies were present in HSE patients with relapsing symptoms. However, the GAD antibody was not detected in the CSF of our patient, and the serum titers were unknown by the limitation of test methods. The exact mechanism of this coexisting phenomenon is not clear. GAD is one of the intracellular antigens that can usually be detected in patients with limbic encephalitis (LE) [24]. Previous studies indicated that the GAD antibody was detectible in LE patients with CNS herpes virus type 6 (HHV-6) infection as well as patients with primary CNS cytomegalovirus (CMV) infection [25, 26]. Considering the clinical characteristics of our patient, we supposed GAD antibody positive, suggesting that the GABAergic system might be involved in the autoimmune reaction, which could be related to the intractable recurrent seizures of our patient.
Compared with infant or child patients, who mainly present choreoathetosis [27], teenage and adult patients more often complain of psychiatric and cognitive deficits with negative redetection of HSV DNA [16]. Brain MRI of the HSV-triggered AE was always negative in new necrotic lesions on the basis of previous lesions due to HSE. Mild enhancement effects of the previous lesions might exist, since viral infection of the central nervous system could disrupt the blood-brain barrier (BBB) [28].
Armangue et al. indicated that patients who developed AE post-HSE responded well to immunotherapy if they were older than 4 years [12]. Steroids are one of the first-line treatments for AE but are controversial in HSE management [29, 30]. Accumulating cases suggest that antiviral therapy combined with steroids may be reasonable since a virus can trigger the autoimmune reaction of patients. However, intravenous steroid therapy alone was not enough, regardless of whether dexamethasone (40 mg/day) was used in Schein’s case or methylprednisolone (1000 mg) was used in our case. IVIG is another first-line management for AE, with one course of 0.4 g/kg/day for 5 days generally being commended. If the symptoms could not be controlled well, another round of IVIG is acceptable in the management of typical AE [29]. Additionally, rituximab and/or cyclophosphamide could be another option for patients who do not respond well to first-line immunotherapy [31]. Based on the treatment strategy of our case, two courses of IVIG were also effective in AE following HSE. The prognosis of patients with AE triggered by HSE is not as good as that of typical AE cases, in which condition patients have a high chance of complete recovery. According to previous cases and studies, patients with HSE followed by AE were more likely to have residual mild to moderate symptoms, especially cognitive deficits with modified Rankin scale (mRS) scores of 2 to 3 [22].
In conclusion, our case indicated that anti-NMDAR antibodies could develop much earlier after HSE compared with previous data from adults. We suggested detecting AE antibodies simultaneously with each CSF analysis in patients who presented psychiatric disorders that were primarily diagnosed with HSV encephalitis. Meanwhile, the second course of IVIG therapy was reasonable when symptoms were not controlled after the first course of IVIG combined with IV steroid treatment. On the other hand, the GAD antibody was also positive in our case, except for the anti-NMDAR antibody. The mechanisms and clinical significance of combined intracellular and neurosurface antibody production after HSE will be further clarified in our future research.
References
Dalmau J, Tüzün E, Wu H-y, Masjuan J, Rossi JE, Voloschin A, Baehring JM, Shimazaki H, Koide R, King D, Mason W, Sansing LH, Dichter MA, Rosenfeld MR, Lynch DR (2007) Paraneoplastic anti-N-methyl-D-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol 61:25–36
Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R (2011) Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol 10:63–74
Lynch DR, Rattelle A, Dong YN, Roslin K, Gleichman AJ, Panzer JA (2018) Anti-NMDA receptor encephalitis: clinical features and basic mechanism. Adv Pharmacol 82:235–260
Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, Lai M, Dessain SK, Rosenfeld MR, Balice-Gordon R, Lynch DR (2008) Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol 7:1091–1098
Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, Cortese I, Dale RC, Gelfand JM, Geschwind M, Glaser CA, Honnorat J, Höftberger R, Iizuka T, Irani SR, Lancaster E, Leypoldt F, Prüss H, Rae-Grant A, Reindl M, Rosenfeld MR, Rostásy K, Saiz A, Venkatesan A, Vincent A, Wandinger KP, Waters P, Dalmau J (2016) A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol 15:391–404
Peng Y, Liu X, Pan S, Xie Z, Wang H (2017) Anti-N-methyl-D-aspartate receptor encephalitis associated with intracranial Angiostrongylus cantonensis infection: a case report. Neurol Sci 38:703–706
Martinez HR, Olguin-Ramirez LA, Camara-Lemarroy CR (2018) Lyme borreliosis as a trigger for NMDA receptor encephalitis? Neurol Sci 39:1815–1817
Iizuka T, Sakai F, Ide T, Monzen T, Yoshii S, Iigaya M, Suzuki K, Lynch DR, Suzuki N, Hata T, Dalmau J (2008) Anti-NMDA receptor encephalitis in Japan: long-term outcome without tumor removal. Neurology 70:504–511
Viaccoz A, Desestret V, Ducray F, Picard G, Cavillon G, Rogemond V, Antoine JC, Delattre JY, Honnorat J (2014) Clinical specificities of adult male patients with NMDA receptor antibodies encephalitis. Neurology 82:556–563
Nosadini M, Mohammad SS, Corazza F, Ruga EM, Kothur K, Perilongo G, Frigo AC, Toldo I, Dale RC, Sartori S (2017) Herpes simplex virus-induced anti-N-methyl-d-aspartate receptor encephalitis: a systematic literature review with analysis of 43 cases. Dev Med Child Neurol 59:796–805
Leypoldt F, Titulaer MJ, Aguilar E, Walther J, Bönstrup M, Havemeister S, Teegen B, Lütgehetmann M, Rosenkranz M, Magnus T, Dalmau J (2013) Herpes Simplex virus-1 Encephalitis Can Trigger anti-NMDA Receptor Encephalitis Case Report. Neurology 81:1637–1639
Armangue T, Spatola M, Vlagea A, Mattozzi S, Cárceles-Cordon M, Martinez-Heras E, Llufriu S, Muchart J, Erro ME, Abraira L, Moris G, Monros-Giménez L, Corral-Corral Í, Montejo C, Toledo M, Bataller L, Secondi G, Ariño H, Martínez-Hernández E, Juan M, Marcos MA, Alsina L, Saiz A, Rosenfeld MR, Graus F, Dalmau J, Aguilera-Albesa S, Amado-Puentes A, Arjona-Padillo A, Arrabal L, Arratibel I, Aznar-Laín G, Bellas-Lamas P, Bermejo T, Boyero-Durán S, Camacho A, Campo A, Campos D, Cantarín-Extremera V, Carnero C, Conejo-Moreno D, Dapena M, Dacruz-Álvarez D, Delgadillo-Chilavert V, Deyà A, Estela-Herrero J, Felipe A, Fernández-Cooke E, Fernández-Ramos J, Fortuny C, García-Monco JC, Gili T, González-Álvarez V, Guerri R, Guillén S, Hedrera-Fernández A, López M, López-Laso E, Lorenzo-Ruiz M, Madruga M, Málaga-Diéguez I, Martí-Carrera I, Martínez-Lacasa X, Martín-Viota L, Martín Gil L, Martínez-González MJ, Moreira A, Miranda-Herrero MC, Monge L, Muñoz-Cabello B, Navarro-Morón J, Neth O, Noguera-Julian A, Nuñez-Enamorado N, Pomar V, Portillo-Cuenca JC, Poyato M, Prieto L, Querol L, Rodríguez-Rodríguez E, Sarria-Estrada S, Sierra C, Soler-Palacín P, Soto-Insuga V, Toledo-Bravo L, Tomás M, Torres-Torres C, Turón E, Zabalza A (2018) Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: a prospective observational study and retrospective analysis. Lancet Neurol 17:760–772
Desena A, Graves D, Warnack W, Greenberg BM (2014) Herpes simplex encephalitis as a potential cause of anti-N-methyl-D-aspartate receptor antibody encephalitis: report of 2 cases. JAMA Neurol 71:344–346
Armangue T, Leypoldt F, Málaga I, Raspall-Chaure M, Marti I, Nichter C, Pugh J, Vicente-Rasoamalala M, Lafuente-Hidalgo M, Macaya A, Ke M, Titulaer MJ, Höftberger R, Sheriff H, Glaser C, Dalmau J (2014) Herpes simplex virus encephalitis is a trigger of brain autoimmunity. Ann Neurol 75:317–323
Pistacchi M, Marsala SZ, Gioulis M, Sanson F, Giometto B (2015) Uncommon relapse after post-herpes simplex encephalitis: an atypical case report. Acta Neurol Belg 115:691–695
Moris G, Armangue T, Extremera VC, Conde CE, Erro ME, Rostasy K, Cuenca JCP, Turón-Viñas E, Málaga I, Muñoz-Cabello B, CarmenTorres-Torres SL, González-Gutiérrez-Solana L, González G, Casado-Naranjo I, Rosenfeld M, Graus F, Dalmau J, On behalf of the Spanish Prospective Multicentric Study of Autoimmunity in Herpes Simplex Encephalitis (2015) Autoimmune post-herpes simplex encephalitis of adults and teenagers. Neurology 85:1746–1743
Morris NA, Kaplan TB, Linnoila J, Cho T (2016) HSV encephalitis-induced anti-NMDAR encephalitis in a 67-year-old woman: report of a case and review of the literature. J Neuro-Oncol 22:33–37
Schein F, Gagneux-Brunon A, Antoine JC, Lavernhe S, Pillet S, Paul S, Frésard A, Boutet C, Grange R, Cazorla C, Lucht F, Botelho-Nevers E (2017) Anti-N-methyl-D-aspartate receptor encephalitis after herpes simplex virus-associated encephalitis: an emerging disease with diagnosis and therapeutic challenges. Infection 45:545–549
Dorcet G, Benaiteau M, Bost C, Mengelle C, Bonneville F, Martin-Blondel G, Pariente J (2020) Two cases of late-onset anti-NMDAr auto-immune encephalitis after herpes simplex virus 1 encephalitis. Front Neurol 11:38
Hjalmarsson A, Blomqvist P, Sköldenberg B (2007) Herpes simplex encephalitis in Sweden, 1990-2001: incidence, morbidity, and mortality. Clin Infect Dis 45:875–880
Fujinami RS, von Herrath MG, Christen U, Whitton JL (2006) Molecular mimicry, bystander activation, or viral persistence: infections and autoimmune disease. Clin Microbiol Rev 19(1):80–94
Pruss H, Finke C, Holtje M, Hofmann J, Klingbeil C, Probst C et al (2012) N-methyl-D-aspartate receptor antibodies in herpes simplex encephalitis. Ann Neurol 72:902–911
Westman G, Studahl M, Ahlm C, Eriksson BM, Persson B, Ronnelid J et al (2016) N-methyl-d-aspartate receptor autoimmunity affects cognitive performance in herpes simplex encephalitis. Clin Microbiol Infect 22:934–940
Malter MP, Helmstaedter C, Urbach H, Vincent A, Bien CG (2010) Antibodies to glutamic acid decarboxylase define a form of limbic encephalitis. Ann Neurol 67:470–478
Xu et al (2012) CMV-associated encephalitis and antineuronal autoantibodies - a case report. BMC Neurol 12:87
Niehusmann P, Widman G, Eis-Hubinger AM, Greschus S, Robens BK, Grote A et al (2016) Non-paraneoplastic limbic encephalitis and central nervous HHV-6B reactivation: causality or coincidence? Neuropathology 36:376–380
Hargrave DR, Webb DW (1998) Movement disorders in association with herpes simplex virus encephalitis in children: a review. Dev Med Child Neurol 40:640–642
Al-Obaidi MMJ, Bahadoran A, Wang SM, Manikam R, Raju CS, Sekaran SD (2018) Disruption of the blood brain barrier is vital property of neurotropic viral infection of the central nervous system. Acta Virol 62:16–27
Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, Honig LS, Benseler SM, Kawachi I, Martinez-Hernandez E, Aguilar E, Gresa-Arribas N, Ryan-Florance N, Torrents A, Saiz A, Rosenfeld MR, Balice-Gordon R, Graus F, Dalmau J (2013) Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol 12:157–165
Hau L, Csábi G, Tényi T (2015) Anti-N-methyl-D aspartate receptor encephalitis guideline to the challenges of diagnosis and therapy. Psychiatr Hung 30(4):402–408
Kong SS, Chen YJ, Su IC, Lin JJ, Chou IJ, Chou ML, Hung PC, Hsieh MY, Wang YS, Chou CC, Wang HS, Lin KL, CHEESE Study Group (2019) Immunotherapy for anti-NMDA receptor encephalitis: experience from a single center in Taiwan. Pediatr Neonatol 60:417–422
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, S., Lan, T., Bai, R. et al. HSV encephalitis triggered anti-NMDAR encephalitis: a case report. Neurol Sci 42, 857–861 (2021). https://doi.org/10.1007/s10072-020-04785-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-020-04785-9