Abstract
Herpes simplex virus-2 encephalitis (HSV2E) in immunocompetent adults is exceptionally rare, and the subsequent onset of autoimmune encephalitis after HSV2E is even less common. This report presents the inaugural Chinese case of anti-N-methyl-d-aspartate receptor encephalitis (NMDARE) induced by HSV2E, confirmed via metagenomic next-generation sequencing (mNGS). The patient demonstrated a favorable response to intravenous immunoglobulin (IVIG) monotherapy. This case emphasizes the importance of considering autoimmune encephalitis in patients exhibiting new or recurrent neurological symptoms after HSV2E recovery. Comprehensive mNGS and neuronal antibody testing are essential for timely diagnosis. Moreover, IVIG monotherapy can serve as an effective treatment for NMDARE induced by HSV2, providing a viable alternative, particularly when steroid therapy is contraindicated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anti-N-methyl-d-aspartate receptor encephalitis (NMDARE), first identified in 2007, is an immune-mediated neuroinflammatory disease and represents the predominant form of autoimmune encephalitis (Dalmau et al. 2019). Characterized by psychiatric disorders, cognitive impairment, seizures, central hypoventilation, speech disturbances, and orofacial dyskinesia, NMDARE often manifests acutely or subacutely, predominantly affecting children and females of reproductive age, with an estimated incidence of 1.5 per 100,000 (Dalmau et al. 2018; Dalmau et al. 2019; Du et al. 2024). Diagnostic markers include antibodies against the GluN1 subunit of NMDAR, which induce crosslinking and internalization of the receptor, thus disrupting NMDAR-related synaptic transmission (Dalmau et al. 2019). Although tumors (especially ovarian teratoma) are established triggers, recent studies have identified infections, particularly viral, as significant postinfectious precipitants of NMDARE (Armangue et al. 2014). Herpes simplex virus type 2 (HSV2) from the Herpesviridae family, a primary agent of neonatal meningitis characterized by fever, headache, and seizures (Bergström et al. 2021), is rarely associated with encephalitis in immunocompetent adults. However, its link to NMDARE remains largely underexplored. This report documents the first Chinese case of NMDARE induced by HSV2 encephalitis (HSV2E), confirmed via metagenomic next-generation sequencing (mNGS). The case was distinguished by symptom recurrence and cerebrospinal fluid (CSF) anti-NMDAR antibodies detected six weeks post-onset, with a favorable outcome following successive intravenous immunoglobulin (IVIG) treatment.
Case presentation
A 25-year-old female with no prior medical history presented acutely with fever, headache, nausea, vomiting, memory deficits, and hallucinations. Initially, neurological examination indicated drowsiness, disorientation, and neck rigidity.
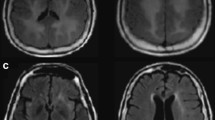
A comprehensive laboratory workup, including complete blood count, biochemical profiles, tumor markers, a systemic autoimmune panel, and infectious disease screening, revealed no significant abnormalities (Supplementary Table 1). Imaging studies, including computed tomography (CT) of the chest and abdomen, and cardiac ultrasound, were all unremarkable. Magnetic resonance imaging (MRI) of the brain displayed asymmetrical hyperintensities in the bilateral mesial temporal lobes on T2/fluid-attenuated inversion recovery (FLAIR) and diffusion-weighted imaging (DWI) (Fig. 1). CSF analysis showed elevated white blood cell (WBC) count (350 cells/µL; with 90% lymphocytes), high protein concentration (91.0 mg/dL), and normal glucose level. Bacterial cultures were negative. CSF mNGS was performed and only HSV2 was identified. Tests for neural antibodies were negative.
Patient’s brain MRI. Axial brain magnetic resonance imaging (MRI) upon admission reveals T1-weighted hypointensities (A), T2/fluid-attenuated inversion recovery (FLAIR) hyperintensities (B), and restricted diffusion on diffusion-weighted imaging (C) and apparent diffusion coefficient mapping (D) within the bilateral mesial temporal lobes, with greater prominence on the left side. A follow-up MRI conducted six weeks after the initial presentation shows an expansion of the previous lesions. Axial T2/FLAIR images highlight the enlarged hyperintensities across the bilateral mesial temporal lobes, along with a new lesion in the left parietal lobe (E-F)
Given the diagnostic evidence, HSV2E was strongly indicated. The patient underwent intravenous acyclovir treatment (10 mg/kg every 8 h for 21 days). Following three weeks of antiviral therapy, psychiatric symptoms resolved, leaving only mild memory impairment. A follow-up CSF analysis showed a reduction in WBC count (90 cells/µL; 98% lymphocytes) and protein level (48.0 mg/dL), with tests for neuronal antibodies remaining negative. The patient was subsequently discharged.
Approximately six weeks post-HSV2E episode, the patient presented with prominent psychiatric symptoms and memory loss. A repeated MRI revealed increased T2/FLAIR hyperintensities in the bilateral mesial temporal lobes and a new hyperintense lesion in the left parietal lobe (Fig. 1, E-F). Upon readmission, neurological examination identified cognitive deficits and spatial disorientation. CSF analysis was negative for HSV2 via mNGS but positive for anti-NMDAR antibodies through a cell-based assay. Screening for occult tumors yielded no findings. The diagnosis was updated to postinfectious NMDARE. Due to concerns about the side effects of corticosteroids, the patient received IVIG at 0.4 g/kg/d for five days, leading to gradual symptom improvement. Subsequently, monthly IVIG treatments at 0.4 g/kg/d for five days were administered over six months. Eight months after initial onset, CSF analysis showed negative results for HSV2 via mNGS and absence of anti-NMDAR antibodies (Fig. 2). At the 3-year follow-up, the patient exhibited only mild memory impairment, not significantly impacting daily activities. MRI scans indicated slight lesion improvement with evidence of partial encephalomalacia.
Clinical course timeline for the patient. A timeline depicting the key events in the patient’s clinical journey, including significant symptoms, CSF analysis, diagnoses, and treatments. CSF, cerebrospinal fluid; D, day; HSV2E, herpes simplex virus 2 encephalitis; HSV-mNGS, herpes simplex virus-metagenomics next-generation sequencing; IgG, immunoglobulin G; IVIG, intravenous immunoglobulin; M, month; ND, not done; NMDAR, N-methyl-D-aspartate receptor; NMDARE, anti-N-methyl-D-aspartate receptor encephalitis; Red bar indicates fever; green bar indicates psychiatric disorder; black bar indicates cognitive deficits
Discussion
The patient initially manifested acute neuropsychiatric symptoms, corroborated by neuroimaging and CSF findings indicative of HSV2E. Following initial symptomatic improvement, the patient experienced severe psychiatric symptoms and cognitive decline. This deterioration was associated with radiological evidence of lesion enlargement on MRI, a positive anti-NMDAR antibody test, and a negative HSV2-mNGS result in the CSF, collectively suggesting a diagnosis of post-HSV2E NMDARE rather than an HSV2E recurrence.
Previous studies indicate a correlation between postinfectious NMDARE and viral infections, with the median time from HSV (mainly HSV1) encephalitis to NMDARE onset being less than three months (Armangue et al. 2018; Nosadini et al. 2017). Increasing reports show adult serum/CSF positivity for anti-NMDAR antibodies within 2 weeks post-HSV1 infection (Hu et al. 2021; Schein et al. 2017). The exact mechanisms underlying post-HSV encephalitis (HSVE) NMDARE remain undetermined. Proposed pathways involve molecular mimicry, antigen exposure, immune tolerance disruption, reduced toll-like receptor 3 function, bystander activation, and genetic predispositions (Galli et al. 2017; Manglani et al. 2021). Notably, post-HSV2E NMDARE is rarely documented. HSV2 is more commonly linked with genital herpes and is prevalent among neonates and immunocompromised individuals (Le Doare et al. 2015; Bradshaw et al. 2016). In adults, HSV2 CNS infections typically result in a milder herpes simplex meningitis, with most patients recovering within 2 weeks and exhibiting minimal or no neurological deficits (Miller et al. 2013). In contrast to HSV1 infections, which often result in severe encephalitis impacting the limbic system and temporal lobe, HSV2 infrequently causes encephalitis in adults, leading to minimal neuronal damage and neuroinflammation (Bergström et al. 2021). Reduced tissue injury and inflammation result in decreased autoantigen exposure and immune intolerance, making autoimmune responses secondary to HSV2 infections less probable. Consequently, the markedly lower incidence of postinfectious NMDARE following HSV2 infections compared to those caused by HSV1 is substantiated.
HSVE typically follows a monophasic course with a relapse rate of 10–25%, but post-HSVE NMDARE can sometimes be mistaken for a recurrence of infectious encephalitis, causing diagnostic delays of 28–90 days and postponing treatment (Nosadini et al. 2017). Distinguishing between these conditions is vital due to their distinct therapeutic approaches. Key factors aid in this diagnostic differentiation. In the transition to NMDARE, patients exhibit movement disorders, such as choreoathetosis, dyskinesias, hemiballismus, and dystonia, more frequently than seizures compared to the HSVE phase (Nosadini et al. 2017). Furthermore, the clinical presentation of post-HSVE NMDARE varies with age; movement disorders are more common in children, while psychiatric symptoms predominate in adults (Armangue et al. 2018), consistent with our observations.
Therapeutic regimens for classical NMDARE and post-HSVE NMDARE are generally consistent. Initial treatments often include intravenous methylprednisolone alone or in combination with IVIG or plasma exchange (Armangue et al. 2014; Galli et al. 2017). Studies have observed that post-HSVE NMDARE patients tend to respond less favorably to immunotherapy and exhibit poorer prognoses, with a modified Rankin Score (mRS) ≥ 3 (Esposito et al. 2022), likely due to severe neuronal damage incurred during the initial HSVE phase (Armangue et al. 2018). In this case, due to potential steroid-associated adverse reactions, the patient declined steroid treatment following her NMDARE diagnosis. IVIG monotherapy was administered as an alternative primary treatment. Contrary to previous reports, the patient experienced gradual improvement with acute and maintenance IVIG treatment, administered monthly for seven months. Ultimately, she achieved a negative antibody status and nearly complete functional recovery, indicated by an mRS of 1. This favorable outcome can be attributed to early diagnosis, timely initiation of immunotherapy, mild neuronal damage, and a weaker secondary immune response induced by HSV2.
In summary, HSV2E is rarely observed in adults. NMDARE should be considered in patients experiencing health deterioration after initial recovery from HSV2E. Prompt reassessment using mNGS and evaluation of neural antibodies are essential for accurate differentiation and diagnosis. Timely immunotherapy contributes to a favorable prognosis. Additionally, IVIG monotherapy may be considered for NMDARE treatment post-HSV2E, particularly when steroids are contraindicated.
Data availability
No datasets were generated or analysed during the current study.
References
Armangue T, Leypoldt F, Málaga I, Raspall-Chaure M, Marti I, Nichter C, Pugh J, Vicente-Rasoamalala M, Lafuente-Hidalgo M, Macaya A, Ke M, Titulaer MJ, Höftberger R, Sheriff H, Glaser C, Dalmau J (2014) Herpes simplex virus encephalitis is a trigger of brain autoimmunity. Ann Neurol 75:317–323. https://doi.org/10.1002/ana.24083
Armangue T, Spatola M, Vlagea A, Mattozzi S, Cárceles-Cordon M, Martinez-Heras E, Llufriu S, Muchart J, Erro ME, Abraira L, Moris G, Monros-Giménez L, Corral-Corral Í, Montejo C, Toledo M, Bataller L, Secondi G, Ariño H, Martínez-Hernández E, Juan M, Marcos MA, Alsina L, Saiz A, Rosenfeld MR, Graus F, Dalmau J (2018) Frequency, symptoms, risk factors, and outcomes of autoimmune encephalitis after herpes simplex encephalitis: a prospective observational study and retrospective analysis. Lancet Neurol 17:760–772. https://doi.org/10.1016/s1474-4422(18)30244-8
Bergström P, Trybala E, Eriksson CE, Johansson M, Satir TM, Widéhn S, Fruhwürth S, Michno W, Nazir FH, Hanrieder J, Paludan SR, Agholme L, Zetterberg H, Bergström T (2021) Herpes Simplex Virus 1 and 2 Infections during Differentiation of Human Cortical Neurons. Viruses;13:2072. https://doi.org/10.3390/v13102072
Bradshaw MJ, Venkatesan A (2016) Herpes simplex Virus-1 encephalitis in adults: pathophysiology, diagnosis, and management. Neurotherapeutics 13:493–508. https://doi.org/10.1007/s13311-016-0433-7
Dalmau J, Graus F (2018) Antibody-mediated encephalitis. N Engl J Med 378:840–851. https://doi.org/10.1056/NEJMra1708712
Dalmau J, Armangué T, Planagumà J, Radosevic M, Mannara F, Leypoldt F, Geis C, Lancaster E, Titulaer MJ, Rosenfeld MR, Graus F (2019) An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: mechanisms and models. Lancet Neurol 18:1045–1057. https://doi.org/10.1016/S1474-4422(19)30244-3
Du BQ, Lai QL, Li EC, Cai MT, Fang GL, Shen CH, Zhang YX, Ding MP (2024) Myelin oligodendrocyte glycoprotein antibody and N-methyl-d-aspartate receptor antibody overlapping syndrome: insights from the recent case reports. Clin Exp Immunol 215:27–36. https://doi.org/10.1093/cei/uxad109
Esposito S, Autore G, Argentiero A, Ramundo G, Principi N (2022) Autoimmune encephalitis after herpes simplex encephalitis: a still undefined condition. Autoimmun Rev 21:103187. https://doi.org/10.1016/j.autrev.2022.103187
Galli J, Clardy SL, Piquet AL (2017) NMDAR Encephalitis following herpes Simplex Virus Encephalitis. Curr Infect Dis Rep 19:1. https://doi.org/10.1007/s11908-017-0556-y
Hu S, Lan T, Bai R, Jiang S, Cai J, Ren L (2021) HSV encephalitis triggered anti-NMDAR encephalitis: a case report. Neurol Sci 42:857–861. https://doi.org/10.1007/s10072-020-04785-9
Le Doare K, Menson E, Patel D, Lim M, Lyall H, Herberg J (2015) Fifteen minute consultation: managing neonatal and childhood herpes encephalitis. Arch Dis Child Educ Pract Ed 100:58–63. https://doi.org/10.1136/archdischild-2014-306321
Manglani M, Poley M, Kumar A, McSherry G, Ericson JE (2021) Anti-NMDAR Encephalitis After Neonatal HSV-1 Infection in a Child With Low TLR-3 Function. Pediatrics;148:e2020035824. https://doi.org/10.1542/peds.2020-035824
Miller S, Mateen FJ, Aksamit AJ Jr (2013) Herpes simplex virus 2 meningitis: a retrospective cohort study. J Neurovirol 19:166–171. https://doi.org/10.1007/s13365-013-0158-x
Nosadini M, Mohammad SS, Corazza F, Ruga EM, Kothur K, Perilongo G, Frigo AC, Toldo I, Dale RC, Sartori S (2017) Herpes simplex virus-induced anti-N-methyl-d-aspartate receptor encephalitis: a systematic literature review with analysis of 43 cases. Dev Med Child Neurol 59:796–805. https://doi.org/10.1111/dmcn.13448
Schein F, Gagneux-Brunon A, Antoine JC, Lavernhe S, Pillet S, Paul S, Frésard A, Boutet C, Grange R, Cazorla C, Lucht F, Botelho-Nevers E (2017) Anti-N-methyl-D-aspartate receptor encephalitis after herpes simplex virus-associated encephalitis: an emerging disease with diagnosis and therapeutic challenges. Infection 45:545–549. https://doi.org/10.1007/s15010-016-0959-y
Acknowledgements
We thank the patient and her family for their understanding and support.
Funding
No funding was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Er-Chuang Li: Conceptualization, data curation, formal analysis, investigation, writing-original draft, writing-review & editing. Qi-Lun Lai: Conceptualization, data curation, formal analysis, investigation, writing-original draft, writing-review & editing. Tian-Yi Zhang: Conceptualization, data curation, formal analysis, investigation, writing-original draft, writing-review & editing. Bing-Qing Du: Data curation, formal analysis, funding acquisition, investigation, writing-review & editing. Jing Zhao: Data curation, formal analysis, funding acquisition, investigation, writing-review & editing. Meng-Ting Cai: Data curation, formal analysis, funding acquisition, investigation, writing-review & editing. Yin-Xi Zhang: Conceptualization, data curation, formal analysis, investigation, supervision, validation, writing-review & editing. Gao-Li Fang: Conceptualization, data curation, formal analysis, investigation, supervision, validation, writing-review & editing.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
No identifiable information was disclosed in writing this case report; further ethical board approval was waived in accordance with local regulations.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, EC., Lai, QL., Zhang, TY. et al. Type 2 herpes simplex virus-induced anti-N-methyl-d-aspartate receptor encephalitis responsive to immunoglobulin monotherapy. J. Neurovirol. (2024). https://doi.org/10.1007/s13365-024-01228-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13365-024-01228-5