Abstract
The purpose of this case report is to increase the knowledge about magnetic resonance spectrum of pyogenic abscesses of the brain. A 74-year-old woman presented with a left frontal lobe cystic mass, developed in the site of post-traumatic contusions after surgical evacuation of a subdural hematoma. MR imaging showed an ipsilateral mass lesion with a thin, regular rim of T1 high-intensity signal, T2 low-intensity signal, and gadolinium-enhancement. Diffusion-weighted imaging with measure of apparent diffusion coefficient value showed inhomogenous diffusion restriction in the lesion core. Perfusion-weighted imaging (PWI) demonstrated high relative cerebral blood volume (rCBV) in both the lesion wall and perilesional area, with a maximal rCBV ratio (rCBV of the lesion/rCBV of the normal contralateral white matter) of 5.65 and 0.58, respectively. As a result, surgery and pathology showed a pyogenic abscess. Cultures grew were Providencia stuartii species. In conclusion, a pyogenic brain abscess from P. stuartii may show high rCBV at PWI, thus mimicking a necrotic tumour.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The differential diagnosis between brain abscesses and necrotic tumours is not always possible by gadolinium-enhanced conventional magnetic resonance (MR) imaging [1–3]. Diffusion-weighted imaging with calculation of the apparent diffusion coefficient (DWI-ADC) had significantly contributed, since the core of pyogenic abscesses generally shows high-intensity signal on DWI and low mean ADC value due to the restricted diffusion of water molecules in the cavity containing bacteria, and inflamed cells. The core of necrotic tumours generally shows unrestricted diffusion [4–8]. However, also the specificity of DWI-ADC is not absolute, since some brain abscesses show a core of high mean ADC value, and some primary and metastatic brain tumours show a core of low mean ADC value [9–14]. Perfusion-weighted imaging (PWI) provides information on brain haemodynamics in normal and pathological conditions. Several studies have shown that the gadolinium-enhancing capsules of brain abscesses have a relative cerebral blood volume (rCBV) value lower than those of necrotic tumours, due to the lack of neoangiogenesis in the brain abscesses [9, 15–18]. However, these results should be considered preliminary, given the limited number of reported cases and aetiologies of brain abscesses [19].
The purpose of this case report is to increase awareness of the spectrum of brain abscesses by reporting DWI-ADC and PWI findings of an uncommon case of pyogenic brain abscess from Providencia stuartii.
Case report
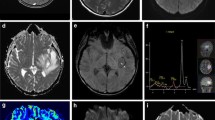
A 74-year-old semicomatouse woman was admitted to the Unit of Neurointensive Care of Azienda Ospedaliera di Rilievo Nazionale “Gaetano Rummo”, Benevento, Italy from a rehabilitation center, since a computed tomography (CT) of the brain (Fig. 1a) showed a hypodense cystic lesion in the left frontal lobe. This occurred 3 months after left fontal craniotomy for the evacuation of a traumatic subdural hematoma. At admission, the patient had a Glasgow Coma Scale score (GCSs) of 9, and a urinary catheter for neurological bladder, and was afebrile. Laboratory showed only an increased percentage of neutrophils (84.1%). History revealed episodes of pulmonary infections treated with antibiotics in the rehabilitation center. Brain MR imaging (Fig. 1b–h) confirmed a 54 × 54 × 45 mm cystic mass in the left frontal lobe, showing a well-definite thin rim of high-intensity signal on T1-weighted images, low-intensity signal on T2- and T2*-weighted images, and intense contrast-enhancement on gadolinium-enhanced T1-weighted images, and a core of heterogeneous signal intensity on both T1- and T2-weighted images without gadolinium-enhancement. The lesion core was heterogeneous also at isotropic DWI obtained with an echo-planar spin-echo technique with b values of 0 and 1,000 s/mm2, showing an inversely heterogeneous mean ADC at ADC maps. The focal gravity-dependent and medial portions of the core showed restricted diffusion (0.587 × 10−3 mm2/s), and the other portions showed unrestricted diffusion (1.17 × 10−3 mm2/s). Proton MR spectroscopy (1H-MRS) was not performed because of not completely patient’s collaboration. PWI was performed by a T2-weighted echo-planar spin-echo sequence during the passage of an intravenous bolus of 0.1 mmol/kg of Gadobutrol (Gadovist, Bayer-Schering, Germany), followed by 20 ml of saline solution, at a flow velocity of 5 ml/s. Hemodynamic evaluation was made by identifying multiple regions of interests (ROIs) from the rCBV colour map showing maximal perfusion (Fig. 1g). Multiple ROIs were measured in the wall of the lesion, in the perilesional area, and in the contralateral normal-appearing white matter, resulting in mean ROIs leading to rCBV ratio, i.e. rCBV of the lesion/rCBV of the normal contralateral white matter. Relative CBV was increased in the wall of the lesion and in the perilesional edema with a rCBV ratio of 5.65 and 0.58, respectively. Presumptive diagnoses included pyogenic abscess and infected necrotic tumour. Surgical aspiration of the lesion with resection of the wall was then performed by a left frontal craniotomy. Postoperatively, the patient showed a subtle clinical improvement, and 2 days later, she was discharged and transferred to the rehabilitation center. Pathology ruled out a neoplasm, and showed a pyogenic abscess. Cultures grew P. stuartii species, which showed a high susceptibility to meropenem at susceptibility test. The patient was then lost at follow-up.
Computed tomography (CT) and magnetic resonance (MR) of the brain. Unenhanced CT axial scan (a) show a large mass of low attenuation density in the left frontal lobe. Unenhanced T2-weighted (b), T2*-weighted (c), and T1-weighted (d), isotropic diffusion-weighted (DW) (e) and mean apparent diffusion (ADC) map (f) obtained with a b value = 1000 s/mm2, relative cerebral blood volume (rCBV) map (g), and gadolinium-enhanced T1-weighted (h) axial MR images show a thin, regular rim of high-intensity signal on T1-weighted images (white arrows), low-signal intensity on T2- and T2*-weighted images (black arrows), high rCBV (white arrowheads), and intense contrast-enhancement (black arrowheads), and an inhomogeneous core in all the images. The study plan of the perfusion-weighted images (g) were different from the conventional, DW and ADC maps ones (a–f, h), in order to avoid artifacts from frontal bones
Discussion
Brain abscesses are not frequent [20–22], accounting for 1–2% of intracranial space-occupying lesions in the developed countries, and 8% in developing countries. Brain abscesses may occur at any age but are most commonly encountered in the first four decades of life. About one-third of all brain abscesses are pyogenic. Pyogenic brain abscess is a localized focus of pus circumscribed by a fibrous-tissue capsule, usually secondary to an acute bacterial infection. The necrotic cavity contains a large amount of neutrophils and proteins. In up to 40% of cases, a primary infection is not recognized. Predisposing factors include diabetes mellitus and immune system disorders, however the patient presented herein was neither diabetic nor immunocompromised. The occurrence of brain abscesses is favoured by (1) haematogenous spread of bacteria from distal foci of infection such as endocarditis, and pneumonia, (2) contiguous foci of infection such as otitis, mastoiditis, and sinusitis, and (3) head injury with penetration of bone fragments or foreign bodies [23, 24]. The abscess formed from haematogenously disseminated infection is generally located at the gray-white matter junction in the distribution of the anterior or middle cerebral artery, most commonly in the frontal and parietal lobes [25]. Brain abscess developing at the site of previous intracerebral haematoma, such as in the patient presented herein, is a rare occurrence [26–28].
Providencia stuartii is a Gram-negative bacterium commonly found in soil, water, and sewage [29–31], and the most common of the five species found in the genus Providencia which include also P. rettgeri, P. alcalifaciens, P. rustigianii, and P. heimbachae. Notably, P. stuartii is the most common Providencia species capable of causing human infections. It is an opportunistic pathogen seen in patients with severe burns or long-term indwelling urinary catheters, putting elderly individuals at a greater risk for infections. The long-term-catheterized urinary tract may offer a particular niche to P. stuartii, which is otherwise an uncommon clinical isolate. Published accounts of bacteriuria in patients catheterized for long periods indicate that P. stuartii has often been found as frequently as familiar uropathogens such as Escherichia coli, Proteus mirabilis, Enterococcus, and Pseudomonas aeruginosa [29–31]. The disruption of the blood–brain barrier by haemorrhage may predispose the affected brain tissue to infection by haematogenous spread of bacteria [32]. The patient presented herein had a long-term catheterized urinary tract for neurological bladder, however there were no reports of bacteriuria neither of septicaemia. An alternative scenario is that the brain abscess was secondary to the spread of bacteria in the course of neurosurgical procedure for the evacuation of acute subdural hematoma.
At conventional unenhanced MR images, the collagenous capsule of the abscess can be seen as a regular rim of generally hypointensity or, less frequently, isointensity on T2-weighted images when compared to the white matter [1, 2], isointensity or slightly hyperintensity on T1-weighted images [1, 25]. High-intensity signal on T1-weighted images has been attributed to paramagnetic free radicals within phagocytosing macrophages [1]. Abscess core is typically hypointense on T1-weighted images and hyperintense on T2-weighted images, and may be inhomogeneous [35]. In our case, MR imaging revealed a thin rim of high-intensity signal on T1-weighted images, and low-intensity signal on T2-weighted images, and a core of heterogeneous signal on both T1- and T2-weighted images. Gradient-echo images ruled out the possibility of blood products, and this was confirmed at pathology. Heterogeneous signal on T1- and T2-weighted images in the abscess core might have been caused by different concentrations in protein content. Intravenous administration of gadolinium is followed by contrast-enhancement of the rim, which usually is regular and thin, such as in the patient reported herein. The contrast-enhancing rim may show a greater thickness on the side facing the gray matter where the inflammatory response is usually more intense [1]. However, all of these signs at conventional MR imaging are not specific and can also be encountered in necrotic tumours, such as high grade gliomas and brain metastases [1, 2].
At DWI-ADC, the necrotic core of pyogenic abscesses show high-intensity signal on isotropic DWI and low mean ADC values, those of high-grade gliomas and brain metastases generally produce a low-intensity signal on isotropic DWI and a high mean ADC value [4–8]. However, restricted diffusion of the core of ring-enhancing lesions is not pathognomonic of brain abscesses, since cases of glioblastoma multiforme [10, 11], and brain metastases [12–14] with high-intensity signal on isotropic DWI and low mean ADC have been reported, as well as cases of brain abscesses with a core of unrestricted diffusion [17, 36]. Notably, the viscosity of the abscess core, and consequently DWI-ADC findings, is determined by various factors including the concentrations of inflammatory cells and bacteria, different aetiological organisms, host immune response, difference of the necrotic or viable inflammatory cells, and age of the abscess, as well as the presence of gas [36]. The patient presented herein was not immunocompromised. P. stuarti had susceptibility to meropenem, however this antibiotic had not been administered before diagnosis. P. stuartii is an aerobic and facultative anaerobic bacterium, thus gases via anaerobic fermentation may be ruled out [37]. It is possible that the heterogeneous signal on isotropic DWI might reflect the density of the necrotic or viable inflammatory cells within the abscess, and the early age of the abscess. Furthermore, the large size of the cavity abscess might have precluded achievement of a threshold inflammatory cell density necessary for DWI-ADC changes.
The capsule of mature abscesses consists mainly of collagen fibers with low capillary density, and thus generally shows a reduced perfusion with low values of rCBV at PWI. High-grade gliomas and cerebral metastases are usually associated with increased neovascularization with high capillary density, and therefore high rCBV values in both the capsule and peritumoural areas [16, 17, 41–43]. This has been demonstrated to be useful in the differential diagnosis of brain abscesses presenting as ring-enhancing lesions and increased diffusion of the core, such as those from Toxoplasma gondii [9, 17]. However, also PWI is not specific and the possibility of a brain abscess presenting high rCBV has been already reported [19]. In the patient reported herein, the brain abscess had a thin capsule and a large central purulent collection. rCBV was high, including a maximal ratio value of 5.65 in the ring-enhancing rim, and 0.58 in the perilesional area, thus mimicking a necrotic tumour. The abscess had not a thick capsule with a small central purulent collection, i.e. an occurrence which potentially may have a substantially higher rCBV than a lesion with a large central cavity [15]. A possible explanation is that in the early capsular stage, brain abscess has the maximum of capillary density, and thus the high rCBV values detected result from the young age of the lesion. In a ring-enhancing lesions with low mean ADC values of the core and high rCBV, the possibility of an infected tumour should be also considered [44]. However, in the patient presented herein pathology ruled out neoplastic cells.
1H-MRS may be useful for the differential diagnosis between brain abscesses and necrotic tumours. Pyogenic abscesses and necrotic tumours both display lactate and lipid peaks produced by anaerobic glycolysis and cellular necrosis. However, in pyogenic brain abscesses, 1H-MRS shows acetate, and aminoacid peaks, i.e. the products of bacterial metabolism, as well as the absence of normal cerebral components such as N-acetyl-aspartate, choline, and creatine [45]. However, inherent problems of the technique and lengthy acquisition times limit the use of 1H-MRS, especially in noncompletely collaborative patients such as the one reported herein.
In conclusion, the possibility for a brain abscess to show heterogeneous diffusion of its core and increased rCBV of its wall, thus mimicking a necrotic tumour, should be taken in consideration, although rare. Despite the use of DWI-ADC and PWI, MR imaging does not present an absolute specificity in the differential diagnosis of ring-enhancing brain lesions.
References
Haimes AB, Zimmerman RD, Morgello S, Weingarten K, Becker RD, Jennis R, Deck MD (1989) MR imaging of brain abscesses. AJR Am J Roentgenol 152:1073–1085
Schwartz KM, Erickson BJ, Lucchinetti C (2006) Pattern of T2 hypointensity associated with ring-enhancing brain lesion can help to differentiate pathology. Neuroradiology 48:143–149
Masamed R, Meleis A, Lee EW, Hathout GM (2009) Cerebral toxoplasmosis: case review and description of a new imaging sign. Clin Radiol 64:560–563
Krabbe K, Gideon P, Wagn P, Hansen U, Thomsen C, Madsen F (1997) MR diffusion imaging of human intracranial tumours. Neuroradiology 39:483–489
Desprechins B, Stadnik T, Koerts G, Shabana W, Breucq C, Osteaux M (1999) Use of diffusion-weighted MR imaging in differential diagnosis between intracerebral necrotic tumours and cerebral abscesses. AJNR Am J Neuroradiol 20:1252–1257
Ebisu T, Tanaka C, Umeda M, Kitamura M, Naruse S, Higuchi T, Ueda S, Sato H (1996) Discrimination of brain abscess from necrotic or cystic tumours by diffusion-weighted echo planar imaging. Magn Reson Imaging 14:1113–1116
Kim YJ, Chang KH, Song IC, Kim HD, Seong SO, Kim YH, Han MH (1998) Brain abscess and necrotic or cystic brain tumour: discrimination with signal intensity on diffusion-weighted MR imaging. AJR Am J Roentgenol 171:1487–1490
Noguchi K, Watanabe N, Nagayoshi T, Kanazawa T, Toyoshima S, Shimizu M, Seto H (1999) Role of diffusion-weighted echo-planar MRI in distinguishing between brain abscess and tumour: a preliminary report. Neuroradiology 41:171–174
Muccio CF, Bartolini A, Esposito G, Cerase A (2008) Cerebral abscesses and necrotic cerebral tumours: differential diagnosis by perfusion-weighted magnetic resonance imaging. Radiol med 113:747–757
Batra A, Tripathi RP (2004) Atypical diffusion-weighted magnetic resonance findings in glioblastoma multiforme. Australas Radiol 48:388–391
Hakyemez B, Erdogan C, Yildirim N, Parlak M (2005) Glioblastoma multiforme with atypical diffusion weighted MR finding. Br J Radiol 78:989–992
Holtås S, Geijer B, Strömblad LG, Maly-Sundgren P, Burtscher IM (2000) A ring-enhancing metastasis with central high signal on diffusion weighted imaging and low apparent diffusion coefficient. Neuroradiology 42:824–827
Hartmann M, Jansen O, Heiland S, Sommer C, Münkel K, Sartor K (2001) Restricted diffusion within ring enhancement is not pathognomonic for brain abscess. AJNR Am J Neuroradiol 22:1738–1742
Tung GA, Evangelista P, Rogg JM, Duncalùn JA 3rd (2001) Diffusion-weighted MR imaging of rim enhancing brain masses: is markedly decreased water diffusion specific for brain abscess? AJR Am J Roentgenol 177:709–712
Holmes TM, Petrella JR, Provenzale JM (2004) Distinction between cerebral abscesses and high-grade neoplasms by dynamic susceptibility contrast perfusion MRI. AJR Am J Roentgenol 183:1247–1252
Erdogan C, Hakyemez B, Yildirim N, Parlak M (2005) Brain abscess and cystic brain tumour. Discrimination with dynamic susceptibility contrast perfusion weighted MRI. J Comput Assist Tomogr 29:663–667
Haris M, Gupta RK, Singh A, Husain N, Husain M, Pandey CM, Srivastava C, Behari S, Rathore RK (2008) Differentiation of infective from neoplastic brain lesions by dynamic contrast-enhanced MRI. Neuroradiology 50:531–540
Chiang IC, Hsieh TJ, Chiu ML, Liu GC, Kuo YT, Lin WC (2009) Distinction between pyogenic brain abscess and necrotic brain tumour using 3-tesla MR spectroscopy, diffusion and perfusion imaging. Br J Radiol 82:813–820
Cianfoni A, Calandrelli R, De Bonis P, Pompucci A, Lauriola L, Colosimo C (2010) Nocardia brain abscess mimicking high grade necrotic tumour on perfusion-MRI: a case report. J Clin Neurosci 17:1080–1082
Yang S (1981) Brain abscess: a review of 400 cases. J Neurosurg 55:794–799
Strong AJ, Ingham HR (1983) Brain abscess. Br J Hosp Med 30:396–403
Osenbach RK, Loftus CM (1992) Diagnosis and management of brain abscess. Neurosurg Clin North Am 3:403–420
Patir R, Sood S, Bhatia R (1995) Post-traumatic brain abscess: experience of 36 patients. Br J Neurosurg 9:29–35
Stephanov S (1999) Brain abscesses from neglected open head injuries: experience with 17 cases over 20 years. Swiss Surg 5:288–292
Luthra G, Parihar A, Nath K, Jaiswal S, Prasad KN, Husain N, Husain M, Singh S, Behari S, Gupta RK (2007) Comparative evaluation of fungal, tubercular, and pyogenic brain abscesses with conventional and diffusion MR imaging and proton MR spectroscopy. AJNR Am J Neuroradiol 28:1332–1338
Nowak DA, Rodiek SO, Topka H (2003) Pyogenic abscess brain following haematogenous seeding of a thalamic haemorrhage. Neuroradiology 45:157–159
Dashti SR, Baharvahdat H, Sauvageau E, Chang SW, Stiefel MF, Park MS, Spetzler RF, Bambakidis NC (2008) Brain abscess formation at the site of intracerebral hemorrhage secondary to central nervous system vasculitis. Neurosurg Focus 24:E12
Thomas SG, Moorthy RK, Rajshekhar V (2009) Brain abscess in a non-penetrating traumatic intracerebral hematoma: case report and review of literature. Neurol India 57:73–75
Warren JW (1986) Providencia stuartii: a common cause of antibiotic-resistant bacteriuria in patients with long-term indwelling catheters. Rev Infect Dis 8:61–67
Woods TD, Watanakunakorn C (1996) Bacteremia due to Providencia stuartii: review of 49 episodes. South Med J 89:221–224
O’Hara CM, Brenner FW, Miller JM (2000) Classification, identification, and clinical significance of Proteus, Providencia, and Morganella. Clin Microbiol Rev 13:534–546
Siatouni A, Mpouras T, Boviatsis EJ, Gatzonis S, Stefanatou M, Sakas D (2007) Brain abscess following intracerebral haemorrhage. J Clin Neurosci 14:986–989
Parker JC Jr, Dyer MC (1985) Neurologic infections due to bacteria, fungi and parasites. In: Doris RL, Robertson DM (eds) Textbook of neuropathology. Williams & Wilkins, Baltimore, pp 632–703
Enzmann DR, Britt RH, Placone RC (1983) Staging of human brain abscess by computed tomograph. Radiology 146:703–708
Schwartz KM, Erickson BJ, Lucchinetti C (2006) Pattern of T2 hypointensity associated with ring-enhancing brain lesion can help to differentiate pathology. Neuroradiology 48:143–149
Lee EJ, Ahn KJ, Ha YS, Oh HE, Park CS, Song SY, Park NH, Kim MS (2006) Unusual findings in cerebral abscess: report of two cases. Br J Radiol 79:156–161
Barrow GI, Feltham RKA (2004) Cowan and Steel’s Manual for the identification of medical bacteria, 3rd edn. Cambridge University Press, Cambridge
Cartes-Zumelzu FW, Stavrou I, Castillo M, Eisenhuber E, Knosp E, Thurnher MM (2004) Diffusion weighted imaging in the assessment of brain abscesses therapy. AJNR Am J Neuroradiol 25:1310–1317
Fanning NF, Laffan EE, Shroff MM (2006) Serial diffusion-weighted MRI correlates with clinical course and treatment response in children with intracranial pus collections. Pediatr Radiol 36:26–37
Muccio C, Cerase A, Iorio G, Bartolini A, Savarese F, Esposito G (2008) La RM con tecnica di diffusione nel controllo della terapia degli ascessi cerebrali. Radiol Med 113(Suppl):404
Law M, Cha S, Knopp EA, Johnson G, Arnett J, Litt AW (2002) High-grade gliomas and solitary metastases: differentiation by using perfusion and proton spectroscopic MR imaging. Radiology 222:715–721
Bulakbasi N, Kocaoglu M, Farzaliyev A, Tayfun C, Ucoz T, Somuncu I (2005) Assessment of diagnostic accuracy of perfusion MR imaging in primary and metastatic solitary malignant brain tumours. AJNR Am J Neuroradiol 26:2187–2199
Rollin N, Guyotat J, Streichenberger N, Honnorat J, Tran Minh VA, Cotton F (2006) Clinical relevance of diffusion and perfusion magnetic resonance imaging in assessing intra-axial brain tumours. Neuroradiology 48:150–159
Chan JH, Tsui EY, Chau LF, Chan JH, Tsui EY, Chau LF, Chow KY, Chan MS, Yuen MK, Chan TL, Cheng WK, Wong KP (2002) Discrimination of an infected brain tumour from a cerebral abscess by combined MR perfusion and diffusion imaging. Comput Med Imaging Graph 26:19–23
Kimura T, Sako K, Gotoh TK, Tanaka T (2001) In vivo single-voxel proton MR spectroscopy in brain lesion with ring like enhancement. NMR Biomed 14:339–349
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muccio, C.F., Leonini, S., Esposito, G. et al. Pyogenic abscess from Providencia stuartii mimicking necrotic tumour at perfusion-weighted imaging. Neurol Sci 32, 919–923 (2011). https://doi.org/10.1007/s10072-011-0565-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-011-0565-9