Abstract
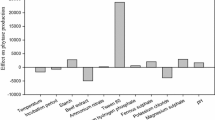
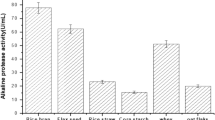
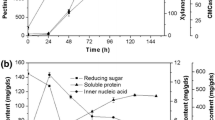
The effect of 8 factors [(with/without) daily mixing and moisture control, incubation time (t), temperature, ratio between dry substrate mass and bed’s cross section area (MA), inoculum size (spores/g), wheat germ content (WG), initial pH, and moisture content (M)] in the production of a prolyl endopeptidase (PEP) by Aspergillus niger ATCC 11414 in solid state fermentation (SSF) was tested. Contribution of all the factors was significant (p<0.05); main effects were those of MA, t, and M. The 4 interactions that presented high interaction severity indexes involved the WG. Under optimized conditions PEP and protease activity were 9.76±0.06 and 3.6×106±1.5×105 U/kg, respectively. The enzyme was partially purified (ammonium sulfate precipitation, dialysis, DEAE-Sepharose ionexchange); it has a molecular weight of 66 kDa (SDS-PAGE), and maximum activity was exhibited at pH 4 and 50°C. The enzyme is stable in a wide pH range (2.2–10) and at temperatures lower than 70°C.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Chevallier S, Goeltz P, Thibaultg P, Banvillel D, Gagnon J. Characterization of a prolyl endopeptidase from Flavobacterium meningosepticum. J. Biol. Chem. 267: 8192–8199 (1992)
Brömme D, Peters K, Fink S, Fittkau S. Enzyme-substrate interactions in the hydrolysis of peptide substrates by thermitase, subtilisin BPN, and proteinase K. Arch. Biochem. Biophys. 244: 439–446 (1986)
Fülöp V, Böcskei Z, Polgár L. Prolyl oligopeptidase: An unusual beta-propeller domain regulates proteolysis. Cell 94: 161–170 (1998)
Araki H, Ouchi H, Uesugi S, Hashimoto Y, Shimoda T. Prolyl endopeptidase and production thereof. EP19920111124 (1993)
Kubota K, Tanokura M, Takahashi K. Purification and characterization of a novel prolyl endopeptidase from Aspergillus niger. Proc. Jpn. Acad. Ser. B 81: 447–453 (2005)
Edens L, Dekker P, van der Hoeven R, Deen F, de Roos A, Floris R. Extracellular prolyl endoprotease from Aspergillus niger and its use in debittering of protein hidrolysates. J. Agr. Food Chem. 53: 7950–7957 (2005)
Stepniak D, Spaenij-Dekking L, Mitea C, Moester M, de Ru A, Baak-Pablo R, van Veelen P, Edens L, Frits K. Highly efficient gluten degradation with a newly identified prolyl endoprotease: Implications for celiac disease. Am. J. Physiol. -Gastr. L. 291: G621–G629 (2006)
Gass J, Bethune M, Siegel M, Spencer A, Khosla C. Combination enzyme therapy for gastric digestion of dietary gluten in patients with celiac sprue. Gastroenterology 133: 472–480 (2007)
Marti T, Molberg Ø, Li Q, Gray G, Khosla C, Sollid L. Prolyl endopeptidase-mediated destruction of T-cell epitopes in whole gluten: Chemical and immunological characterization. J. Pharmacol. Exp. Ther. 312: 19–26 (2005)
Lopez M, Edens L. Effective prevention of chill-haze in beer using an acid proline specific endoprotease from Aspergillus niger. J. Agr. Food Chem. 53: 7944–7949 (2005)
Aguilar CN, Favela-Torres E, Viniegra-González G, Augur C. Culture conditions dictate protease and tannase production in submerged and solid-state cultures of Aspergillus niger Aa-20. Appl. Biochem. Biotech. 102: 407–414 (2002)
Stamatis DH. TQM Engineering Handbook. Marcel Dekker, New York, NY, USA. pp. 230–235 (1997)
Rao RS, Kumar K, Prakasham S, Hobbs P. The Taguchi methodology as a statistical tool for biotechnological applications: A critical appraisal. Biotechnol. J. 3: 510–523 (2008)
Laemmli UK. Cleavage of structural proteins during assembly of the bacteriophage T4. Nature 227: 680–685 (1970)
Aoki H, Ahsan N, Matsuo K, Hagiwara T, Watabe S. Purification and characterization of collagenolytic proteases from hepatopancreas of northern shrimp (Pandalus eous). J. Agr. Food Chem. 51: 777–783 (2003)
Lowry O, Rosebrough N, Farr A, Rondall R. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–273 (1951)
Dubois M, Gilles DA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for the determination of sugars and related substances. Anal. Chem. 28: 350–356 (1956)
Brock F, Forsberg C, Buchanan J. Proteolytic activity of rumen microorganisms and effects of proteinase inhibitors. Appl. Environ. Microb. 44: 561–569 (1982)
Erlanger B, Kokowsky N, Cohen N. The preparation and properties of two new chromogenic substrate of trypsin. Arch. Biochem. Biophys. 95: 271–278 (1961)
Malathi S, Chakraborty R. Production of alkaline protease by a new Aspergillus flavus isolate under solid-substrate fermentation conditions for use as a depilation agent. Appl. Environ. Microb. 57: 712–716 (1991)
Chakraborty R, Srinivasan M, Sarkar SK, Raghvan KV. Production of acid protease by a new Aspergillus niger by solid substrate fermentation. J. Microbiol. Biotechn. 10: 17–30 (1995)
Agrawal D, Patidar P, Banerjee T, Patil S. Production of alkaline protease by Penicillium sp. under SSF conditions and its application to soy protein hydrolysis. Process Biochem. 39: 977–981 (2004)
Moon SH, Parulekar SJ. A parametric study of protease production in batch and fed-batch cultures of Bacillus firmus. Biotechnol. Bioeng. 37: 467–483 (1991)
Aikat K, Bhattacharyya BC. Protease extraction in solid state fermentation of wheat bran by a local strain of Rhizopus oryzae and growth studies by soft gel technique. Process Biochem. 35: 907–914 (2000)
Yang FC, Lin IH. Production of acid protease using thin stillage from a rice-spirit distillery by Aspergillus niger. Enzyme Microb. Tech. 23: 397–402 (1998)
Schaal R, Kupfahl C, Buchheidt D, Neumaier M, Findeisen P. Systematic identification of substrates for profiling of secreted proteases from Aspergillus species. J. Microbiol. Meth. 71: 93–100 (2007)
Kabashima T, Fujii M, Meng Y, Ito K, Yoshimoto T. Prolyl endopeptidase from Sphingomonas capsulata: Isolation and characterization of the enzyme and nucleotide sequence of the gene. Arch. Biochem. Biophys. 358: 141–148 (1998)
Szwajcer-dey E, Rasmussen J, Meldal M, Breddam K. Prolinespecific endopeptidases from microbial sources: Isolation of an enzyme from a Xanthomonas sp. J. Bacteriol. 174: 2454–2459 (1992)
Harwood V, Denson J, Robinson-Bidle K, Schreier H. Overexpression and characterization of a prolyl endopeptidase from the hyperthermophilic archaeon Pyrococcus furiosus. J. Bacteriol. 179: 3613–3618 (1997)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Esparza, Y., Huaiquil, A., Neira, L. et al. Optimization of process conditions for the production of a prolylendopeptidase by Aspergillus niger ATCC 11414 in solid state fermentation. Food Sci Biotechnol 20, 1323 (2011). https://doi.org/10.1007/s10068-011-0182-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10068-011-0182-7