Abstract
In rheumatoid arthritis (RA) patients, weekly intake of methotrexate (MTX) is the basic drug treatment. This observational study aims to investigate how many RA patients are adherent in terms of MTX intake and to identify determinants of non-adherence. Intake of MTX (orally or via injection) was recorded in 129 RA patients with an electronic monitoring system (MEMS®) during 16 weeks. In addition, two adherence questionnaires, the Medication Adherence Report Scale (MARS-5) and the Compliance-Questionnaire-Rheumatology (CQR) as well as a visual analogue scale (VAS) measuring MTX adherence, were administered to these patients. As possible determinants of adherence, data on demographics, disease and treatment characteristics, depression, illness cognitions, motivation, and social support were collected. Of all participants, 58 % were fully adherent and 75 % skipped at most one dose during 16 weeks. A better mental health status and suffering from comorbidities had a positive effect on adherence, while living alone had a negative effect. These three predictors explained 30 % of the variance in MTX adherence. Of the three self-report medication adherence measures, the VAS correlated the highest with the results of the electronic monitoring system (r = 0.552, p = 0.01). A relatively high adherence rate was observed in RA patients treated with MTX. The determinants identified by this study could be used to screen patients at risk for non-adherence. A simple VAS scale seems to be an acceptable way for a preliminary screening of MTX adherence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is the most prevalent inflammatory rheumatic disease causing joint pain, swelling, and morning stiffness, leading to the destruction of cartilage and bone loss, potentially resulting in important disability [1]. Basic drug treatment consists of disease modifying anti-rheumatic drugs (DMARDs) of which methotrexate (MTX) is the first choice.
To achieve the disease modifying effects, medication adherence is crucial. Assessing adherence and identifying reasons for (non-) adherence is therefore important from a clinical perspective. The way medication adherence is measured may however be responsible for diversity in results [2]. Self-reported adherence mostly results in higher adherence rates than electronic monitoring, which was only used in two studies in RA patients [3, 4].
This observational study aims to answer the following questions:
-
To what extent do RA patients take their MTX as prescribed?
-
Which factors indicate an increased risk of non-adherence and can thus be useful for screening purposes?
-
Which self-report measure, useable in daily practice, is the most valid, compared to an electronic monitoring system?
Materials and methods
Research field
One hundred forty-six patients who visited the ambulatory rheumatology unit (outpatient/day clinic) of Antwerp University Hospital (Belgium) between September and November 2013 and who met the following inclusion criteria were invited to participate:
-
Diagnosed RA according to the ACR/EULAR criteria of 2010 treatment with MTX
-
Sufficient knowledge of the Dutch language
-
Not suffering from severe mental disorders
Data collection
The study was approved by the ethics committee of Antwerp university hospital (B300201318114). Patients who met the inclusion criteria were asked by their rheumatologists or the rheumatology nurse to participate. An informed consent was obtained from all patients prior to the study.
Assessment of adherence
Adherence was measured using the Medication Event Monitoring System (MEMS®, MWV Healthcare). The intake behavior is quantified by the use of a medication container of which the cap includes a chip that registers the moment of opening [2]. A patient was considered fully adherent if over a period of 1 week the MEMS®container was opened once or more in accordance with the prescription. For each of the 16 consecutive weeks, every patient was given a score of 1 (opened) or 0 (not opened). The average of these 16 measurements, multiplied by 100, resulted in a score of medication adherence.
Adherence was also measured by means of three self-report measures. The CQR [5, 6] consists of 19 statements concerning medication intake in which the patient indicates to what extent (s) he agrees on a 4-point Likert scale. The multivariate weighed model of De Klerk and colleagues proposes two discriminant formulas to detect intake compliance (≤80 and ≤50 %) [6]. The MARS-5 is an adherence measure developed to use in patients suffering from a variety of chronic diseases [7]. It consists of five statements with regard to aspects of non-adherent behaviors. Patients indicate to what extent each statement applies to them on a 5-point Likert scale (1 = always, 2 = often, 3 = sometimes, 4 = rarely, and 5 = never). By summing up the answers, a score in between 5 and 25 is obtained. A visual analogue scale ranging from “in 0 % of the cases” to “in 100 % of the cases” was used to ask patients to what extent they took the MTX as prescribed. Patients gave a mark on a 10 cm line resulting in a score in between 0 and 100.
Assessment of possible determinants
We included sociodemographic characteristics, condition-related factors, and cognitions as potential determinants of medication adherence based on the evidence resulting from a systematic review [8]. In addition, we added therapy-related factors as there is also evidence for their importance [9]. Next, determinants such as quality of life, depression and anxiety, and patient-related characteristics such as social support and motivation were added based on more recent publications [10, 11].
Socio-demographic characteristics such as age, gender, living situation, and occupational status were collected by means of a structured questionnaire.
Condition-related factors retrieved from the medical file were disease activity of RA assessed by means of the Disease Activity Score 28 (DAS28) (remission status < 2,6) [1], daily functional limitations, and acquired damage to the joints, assessed by the HAQ [12] and comorbidities. To assess somatic symptoms, the validated Patient Health Questionnaire 15 (PHQ-15) was used [13]. The Short Form 36 (SF36), a validated generic instrument that measures quality of life, provided information on the patient’s physical and mental health status [14]. Depression was assessed using the nine items of the validated Patient Health Questionnaire 9 (PHQ-9) [15]. Anxiety was measured by means of the anxiety subscale of the validated Dutch version of the Brief Symptom Inventory, consisting of six items [16].
Therapy-related factors measured were disease duration, the dosage of MTX, the mode of patient follow up, the number of doctor visits during the last 6 months, the number of prescribed pills per day, and possible side effects.
Patient-related characteristics: The IPQ-K, the Dutch shortened version of the Illness Perception Questionnaire, was used to measure illness cognitions [17]. Eight statements were scored on a scale from 0 to 10. Social support was measured by means of a questionnaire concerning social support with the disease in general and more specific in relation to the medication intake [18]. The motivation to take MTX was assessed by means of the Treatment Self-Regulation Questionnaire (TSRQ), adapted to measure the medication adherence. Motivation can be autonomous (internally regulated) or coerced (externally regulated). Eleven items were rated on a 7-point Likert scale (1 = totally disagree and 7 = totally agree). A subscore for autonomous and for coerced motivation was compiled by calculating the average score of the corresponding items. By subtracting the score for coerced regulation from the score for autonomous regulation, a combined score was calculated [19].
Statistical analysis
Differences between categorical data were analyzed using chi-squared analysis, between continuous data with t tests. A logistic regression analysis with as dependent variable being fully adherent or not was carried out by a stepwise input of independent variables that showed a significant relationship of 0.01 or lower with MTX adherence in univariate analyses, until the best statistically significant model was found (p < 0.05). Correlations between the different measures of adherence were calculated with Pearson’s correlations. The statistical analyses were performed with SPSS 21.0.
Results
Sampling
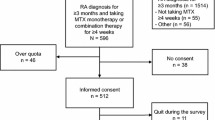
Figure 1 describes the process of inclusion.
The sample (n = 129) consisted for 60 % of women. The average age of the participants was 61 years. They had mean disease duration of 12 years and 65 % was in remission status. The MTX was taken orally by 64 % of the participants. Of the total sample, 43 % reported side effects of the MTX treatment.
Adherence with regard to methotrexate
Figure 2 gives a description of the number of doses of MTX missed over a period of 16 weeks. More than half of the participants (58 %) took faithfully each week the MTX. A quarter skipped the medication for at least 2 weeks, corresponding to less than 90 % adherence. Only 9 % failed more than three times to take their medication and was thus less than 80 % adherent.
The MARS-5 had an acceptable internal consistency (Cronbach’s alpha = 0.71). The mean score was 24.2 (SD = 1.7). The CQR showed a poor internal consistency in this population (Cronbach’s alpha = 0.51). Following the discriminant formula of De Klerk and colleagues [6] to detect 80 % taking compliance, 14.3 % of the participants were less than 80 % adherent. The visual analogue scale indicated a mean adherence of 94 % (SD = 10.0).
Factors associated with adherence to MTX
Table 1 reports on the predictors for adherence to MTX. A criterion of 100 % adherence was used to construct the dependent variable.
The following determinants were dichotomized: living situation (living alone, yes/no) and comorbidities (yes/no). Only independent variables that showed a significant univariate relationship with the dependent variable at a 0.01 level were entered in the logistic regression analysis: living alone, suffering from comorbidities, mental health status, and the illness cognitions identity and perceived emotional consequences. Suffering from comorbidities and a better mental health status had a positive influence on medication adherence. Living alone was negatively associated with adherence to MTX.
The association between electronic monitoring and self-report measures of adherence proved to be different.
The scores on the visual analogue scale for adherence correlated moderately with the results of the electronic adherence monitoring (r = 0.552, p < 0.001) although the VAS score seems to overestimate adherence (see Fig. 3).
The correlation between the MARS-5 questionnaire and the electronic monitoring system was weaker (r = 0.356, p < 0.001). As the internal consistency of the CQR was too low, 8 questions out of 19 were deleted (items 4, 5, 8, 9, 11, 12, 16, and 19) resulting in a good consistency (Cronbach’s alpha = 0.77). There was however no significant correlation between the results of the electronic monitoring system and the sum score of the remaining items. In addition, no association was found between the discriminant formula of De Klerk and colleagues [6] to detect 80 % taking compliance and the results of the 80 % adherence level measured with the electronic monitoring system (X 2 (2, N = 104) = 0.09, p = 0.77).
Discussion
A large study on MTX adherence showed that only 64 % of the participants took at least 80 % of the prescribed dose [20]. In the present study, 92 % of the patients took at least 80 % of the prescribed dose. In a Dutch study, full adherence for methotrexate was found in a small sample of patients [4]. The high adherence rate in our study may be due to a selection bias: possibly, patients who were less adherent did not want to participate or did not complete the medication registration. This concerns, however, only 17 patients out of 146. The fact that Belgian patients only contribute for a very small part to health care costs may also have played a role [8]. In addition, in the Belgian context, RA patients usually see their specialist every 6 months or even more frequently. This close follow may also have increased the adherence rate. Another issue is that this study collected data in only one health care center. A larger, multicenter study might have resulted in lower adherence rates. In addition, a large number of the participants had long disease duration and most patients were in remission. Finally, extending the MEMS measurement period might also increase the validity of the results.
With respect to predictors of adherence, the negative relationship between living alone and adherence indicates that social support has an impact on medication intake. This is consistent with previous findings [21]. The positive influence of comorbidity on adherence is also in accordance with other studies [22, 23]. Finally, a better mental health status was also associated to medication adherence in at least one other study [11].
Adherence scores based on a visual analogue scale showed a higher correlation with results from electronic monitoring than the data obtained with two other self-report measures. As a consequence, a VAS scale may be used in daily practice for a quick screening of medication adherence in patients. A weaker correlation was found between the MARS-5 and the electronic monitoring which is consistent with the results of an earlier study [2]. Finally, the sum score of the CQR did not show a significant association with the results of the electronic monitoring. The CQR may be more appropriate to identify determinants of treatment adherence.
References
Prevoo ML, van’t Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL (1995) Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 38(1):44–48
Sutton S, Kinmonth A-L, Hardeman W, Hughes D, Boase S, Prevost AT et al (2014) Does electronic monitoring influence adherence to medication? randomized controlled trial of measurement reactivity. Ann Behav Med Publ Soc Behav Med 48(3):293–299
Waimann CA, Marengo MF, de Achaval S, Cox VL, Garcia-Gonzalez A, Reveille JD et al (2013) Electronic monitoring of oral therapies in ethnically diverse and economically disadvantaged patients with rheumatoid arthritis: consequences of low adherence. Arthritis Rheum 65(6):1421–1429
de Klerk E, van der Heijde D, Landewé R, van der Tempel H, Urquhart J, van der Linden S (2003) Patient compliance in rheumatoid arthritis, polymyalgia rheumatica, and gout. J Rheumatol 30(1):44–54
de Klerk E, van der Heijde D, van der Tempel H, van der Linden S (1999) Development of a questionnaire to investigate patient compliance with antirheumatic drug therapy. J Rheumatol 26(12):2635–2641
de Klerk E, van der Heijde D, Landewé R, van der Tempel H, van der Linden S (2003) The compliance-questionnaire-rheumatology compared with electronic medication event monitoring: a validation study. J Rheumatol 30(11):2469–2475
Salt E, Hall L, Peden AR, Home R (2012) Psychometric properties of three medication adherence scales in patients with rheumatoid arthritis. J Nurs Meas 20(1):59–72
Pasma A, van’t Spijker A, Hazes JMW, Busschbach JJV, Luime JJ (2013) Factors associated with adherence to pharmaceutical treatment for rheumatoid arthritis patients: a systematic review. Semin Arthritis Rheum 43(1):18–28
López-González R, León L, Loza E, Redondo M, de Yébenes MJ G, Carmona L (2015) Adherence to biologic therapies and associated factors in rheumatoid arthritis, spondyloarthritis and psoriatic arthritis: a systematic literature review. Clin Exp Rheumatol 33(4):559–569
Pascual-Ramos V, Contreras-Yáñez I (2013) Motivations for inadequate persistence with disease modifying anti-rheumatic drugs in early rheumatoid arthritis: the patient’s perspective. BMC Musculoskelet Disord 14:336
Cabrera-Marroquín R, Contreras-Yáñez I, Alcocer-Castillejos N, Pascual-Ramos V (2014) Major depressive episodes are associated with poor concordance with therapy in rheumatoid arthritis patients: the impact on disease outcomes. Clin Exp Rheumatol 32(6):904–913
Bruce B, Fries JF (2003) The Stanford health assessment questionnaire: dimensions and practical applications. Health Qual Life Outcomes 1:20
Kroenke K, Spitzer RL, Williams JBW (2002) The PHQ-15: validity of a new measure for evaluating the severity of somatic symptoms. Psychosom Med 64(2):258–266
Aaronson NK, Muller M, Cohen PD, Essink-Bot ML, Fekkes M, Sanderman R et al (1998) Translation, validation, and norming of the Dutch language version of the SF-36 health survey in community and chronic disease populations. J Clin Epidemiol 51(11):1055–1068
Martin A, Rief W, Klaiberg A, Braehler E (2006) Validity of the brief patient health questionnaire mood scale (PHQ-9) in the general population. Gen Hosp Psychiatry 28(1):71–77
Derogatis LR, Melisaratos N (1983) The brief symptom inventory: an introductory report. Psychol Med 13(03):595–605
de Raaij EJ, Schröder C, Maissan FJ, Pool JJ, Wittink H (2012) Cross-cultural adaptation and measurement properties of the brief illness perception questionnaire-Dutch language version. Man Ther 17(4):330–335
Van Camp YPM, Vrijens B, Abraham I, Van Rompaey B, Elseviers MM (2014) Adherence to phosphate binders in hemodialysis patients: prevalence and determinants. J Nephrol
Hurkmans EJ, Maes S, de Gucht V, Knittle K, Peeters AJ, Ronday HK et al (2010) Motivation as a determinant of physical activity in patients with rheumatoid arthritis. Arthritis Care Res 62(3):371–377
Harley CR, Frytak JR, Tandon N (2003) Treatment compliance and dosage administration among rheumatoid arthritis patients receiving infliximab, etanercept, or methotrexate. Am J Manag Care 9(6 Suppl):S136–S143
Scheurer D, Choudhry N, Swanton KA, Matlin O, Shrank W (2012) Association between different types of social support and medication adherence. Am J Manag Care 18(12):e461–e467
Borah BJ, Huang X, Zarotsky V, Globe D (2009) Trends in RA patients’ adherence to subcutaneous anti-TNF therapies and costs. Curr Med Res Opin 25(6):1365–1377
de Thurah A, Nørgaard M, Johansen MB, Stengaard-Pedersen K (2010) Methotrexate compliance among patients with rheumatoid arthritis: the influence of disease activity, disease duration, and co-morbidity in a 10-year longitudinal study. Scand J Rheumatol 39(3):197–205
Acknowledgments
This research received support from a scientific grant awarded by Merck Sharp & Dohme (MSD) Belgium.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None of the authors have a financial relationship with MSD nor other relationships or activities that could appear to have influenced the submitted work. The authors have full control of all primary data and agree to allow the journal to review their data if requested.
Rights and permissions
About this article
Cite this article
De Cuyper, E., De Gucht, V., Maes, S. et al. Determinants of methotrexate adherence in rheumatoid arthritis patients. Clin Rheumatol 35, 1335–1339 (2016). https://doi.org/10.1007/s10067-016-3182-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3182-4