Abstract
In addition to fatigue, many patients with chronic fatigue syndrome (CFS) experience chronic musculoskeletal pain. We aimed at examining the role of catastrophizing, coping, kinesiophobia, and depression in the chronic pain complaints and in the daily functioning of CFS patients. A consecutive sample of 103 CFS patients experiencing chronic widespread musculoskeletal pain completed a battery of questionnaires evaluating pain, daily functioning, and psychological characteristics (depression, kinesiophobia, pain coping, and catastrophizing). Thirty-nine patients participated in the 6–12-month follow-up, consisting of questionnaires evaluating pain and pressure pain algometry. Correlation and linear regression analyses were performed to identify predictors. The strongest correlations with pain intensity were found for catastrophizing (r = −.462, p < .001) and depression (r = −.439, p < .001). The stepwise multiple regression analysis revealed that catastrophizing was both the immediate main predictor for pain (20.2%) and the main predictor on the longer term (20.1%). The degree of depression was responsible for 10% in the observed variance of the VAS pain after 6–12 months. No significant correlation with pain thresholds could be revealed. The strongest correlations with daily functioning at baseline were found for catastrophizing (r = .435, p < .001) and depression (r = .481, p < .001). Depression was the main predictor for restrictions in daily functioning (23.1%) at baseline. Pain catastrophizing and depression were immediate and long-term main predictors for pain in patients with CFS having chronic widespread musculoskeletal pain. They were also correlated to daily functioning, with depression as the main predictor for restrictions in daily functioning at baseline.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic fatigue syndrome (CFS) is known to be a debilitating and complex disorder, characterized by extreme fatigue [1]. In addition to debilitating fatigue, the majority of patients with CFS (54.5% up to 84.4%) report chronic musculoskeletal pain [2, 3]. This chronic pain accounts for 26.0% up to 33.0% of their self-reported activity limitations and participation restrictions [4]. Despite the clinical relevance of this complaint, studies regarding chronic pain in CFS are scarce [5]. However, evidence for the role of kinesiophobia, depression, catastrophizing, and inadequate coping strategies in maintaining the chronic pain complaints has been provided in patients with fibromyalgia (FM), chronic low back pain, and other disorders associated with chronic musculoskeletal pain, e.g., [6, 7].
Pain catastrophizing
Catastrophizing, which is said to be the exaggerated and negative orientation towards pain, may have a role as a mediator to pain [8]. Evidence for the relation between catastrophizing and experienced pain and functional disabilities has been provided in different patient populations, as for example in FM [6]. Despite the similarities and the great overlap between CFS and FM [9], the role of catastrophizing in pain and in daily functioning has not extensively been studied in CFS.
Pain coping
“Coping” can be described as “the behavioral and cognitive attempts to tolerate or to deal with the pain” [10]. Active pain coping strategies have been defined as individuals' attempts to manage their pain through their own resources [11]. Active pain coping strategies such as persisting in tasks despite pain, exercising, and diverting attention from pain have been reported to be negatively associated with functional disabilities in patients with CFS [12]. In contrast, pain responses such as resting, negative thoughts, avoidance behavior, and guarding the painful body part have been labeled as passive coping [10, 11]. In CFS, passive coping strategies were found to be correlated with a reduced functionality, vitality, and mental health [13, 14]. Although these investigators already studied coping strategies, they did not study the influence of coping on chronic pain in CFS.
Kinesiophobia
Kinesiophobia is defined as “excessive, irrational, and debilitating fear of physical movement and activity resulting from a feeling of vulnerability to painful injury or re-injury” [15] and has been reported to be a common feature of patients with CFS, occurring in 45.5% to 65.0% of the patients [16, 17]. Nijs et al. [16] revealed a significant positive correlation between kinesiophobia and self-reported disabilities in activities and participation in patients with CFS. Up to now, no investigations focused on the association between kinesiophobia and pain intensity in these patients.
Depression
Depression is an often reported complaint in CFS patients [2, 18]. Hassett et al. [6] revealed depression and catastrophizing as significant predictors of pain in women with FM and women with rheumatoid arthritis, respectively responsible for 30.0% and 27.0% of the variance in pain. In CFS, this association between depression and pain has hardly been studied, except from the study of Morriss et al. [18] which could not reveal an association in CFS, but their patients did not fulfill the criteria [1] of the Center for Disease Control and Prevention.
So, in other chronic pain populations, evidence is provided for the role of these psychological factors in pain and in daily functioning, but not yet in CFS, since pain is hardly studied in this population. However, as described by Smeets et al. [19], psychological variables are widely known to be important outcome predictors (characteristics that predict outcome independent of therapy), but they may also act as therapy effect modifiers (characteristics that predict treatment effects) [20]. Effect modifiers can help clinicians select the best treatment for an individual patient, whereas predictors of outcome can be used by a clinician to provide patient-specific information on prognosis.
Therefore, the four key questions of the present study concern the role of (1) catastrophizing, (2) coping, (3) depression, and (4) kinesiophobia in daily functioning and in chronic widespread musculoskeletal pain in CFS patients with chronic widespread pain, both at baseline (first study phase) and 6–12 months later (second study phase).
Methods
Design overview
Patients fulfilling all study criteria were asked to participate in the study. A leaflet explaining the purpose of the research was handed out, before patients were asked to sign the informed consent. The study protocol and the informed consent were approved by the Ethical Committee of the University Hospital of Brussels.
At the initial contact, subjects completed several questionnaires: the CFS Symptom List, Medical Outcomes Short Form 36 Health Status Survey (SF-36), Chronic Fatigue Syndrome Activities and Participation Questionnaire (CFS-APQ), the Tampa Scale for Kinesiophobia–version CFS (TSK-CFS), Pain Coping Inventory (PCI), Beck Depression Inventory (BDI), and the Pain Catastrophizing Scale (PCS).
Six to 12 months after their participation, these patients were telephonically contacted a second time and asked for cooperation in the sequel of the study. In those that were willing to participate again, pain was evaluated by the visual analogue scale (VAS) score for “myalgias and arthralgias” of the CFS Symptom List, by the subitem “pain” of the SF-36 or Short Form Health Survey 36, and by pressure pain threshold assessment.
Settings and participants
Consecutive CFS patients visiting the Chronic Fatigue Clinic of the Vrije Universiteit Brussel were screened following several in- and exclusion criteria. A total of 103 CFS patients meeting all study requirements were recruited.
Inclusion criteria
All subjects fulfilled the Center for Disease Control and Prevention criteria for CFS [1]. Since this study focused on chronic, widespread musculoskeletal pain, all patients had to experience widespread pain. Pain is considered widespread when all of the following are present: pain in both the left and the right side of the body, pain both above and below the waist, and axial skeletal pain for at least 3 months [21]. Furthermore, all study participants had Dutch as their native language and were within the age range of 18 to 65 years.
Outcomes: self-reported measures of psychological factors (first phase)
The Dutch version of the BDI II was used for the assessment of depression. The Dutch BDI II is a self-reported questionnaire consisting of 21 items describing various symptoms of depression; items are scored on a 4-point scale. Total scores are counted by summing all individual item scores, and higher total scores reflect more severe depression. The BDI appears to be a reliable and valid tool for the assessment of depressive symptoms in chronic pain patients [22].
The Dutch PCS [23] is a self-reported questionnaire aiming at assessing pain catastrophizing both in clinical and non-clinical populations. It consists of 13 items describing different thoughts and feelings that individuals may experience when they are experiencing pain. Items are scored on a 5-point scale, and total scores are counted by adding up all individual item scores. Higher scores correspond to more severe catastrophic thoughts about pain. Given the evidence for the good psychometric quality of the PCS [23], the usage of the PCS was found to be appropriate for the present investigation.
The PCI contains 33 items, assessing six specific, cognitive, and behavioral pain coping strategies, that represent two higher-order pain coping dimensions: passive (ruminating, retreating, and resting) and active (transformation, distraction, and reducing demands) coping [10]. Patients were asked to rate the 33 items on a 4-point Likert scale ranging from 1 (hardly ever) to 4 (very often) in terms of the frequency with which strategies were suffering pain. Results of the different subscales were obtained by taking the mean score of the items belonging to that subscale. A higher score indicates a more frequent application of that specific coping strategy. The PCI has been found to be sufficiently sensitive and valid [11].
The TSK-CFS aims at monitoring kinesiophobia in patients with CFS. It is a modification of the TSK-DV [24]. Each of the items on the questionnaire is provided with a 4-point Likert scale, with scoring alternatives ranging from “strongly agree” to “strongly disagree.” A total score is calculated (1–4 for each item) after inversion of the individual scores of items 4, 8, 12, and 16. Total scores on the Dutch TSK-CFS range between 17 and 68; higher scores indicate a higher degree of kinesiophobia. A total score greater than 37 indicates high fear of movement [17, 24].
Outcomes: self-reported measures of pain and functioning (first and second phases)
The CFS Symptom List is a self-reported, originally Dutch, measure for assessing symptom severity in CFS patients. It encompasses the 19 most frequently reported symptoms in a sample of 1,578 CFS patients [25]. In order to assess the severity of the symptoms included in the CFS Symptom List, visual analogue scales (100 mm) are used. In a previous study of 68 CFS patients, the internal consistency of the different items included in the Dutch CFS Symptom List was high [3]. The CFS Symptom List displayed excellent test–retest reliability, content, and concurrent validity [26].
The SF-36 assesses functional status and well-being or quality of life. The SF-36 contains eight subscales but we were only interested in the subscale bodily pain. Higher scores indicate less bodily pain; subscale scores range between 0 and 100. The psychometric properties of the SF-36 are well characterized; it has been documented to have reliability and validity in a wide variety of patient populations [27, 28].
The CFS-APQ is a self-administered questionnaire, which is aimed at monitoring activity limitations and participation restrictions in patients with CFS [29]. The scoring system of the CFS-APQ, as described elsewhere [30], generates two overall scores; the first one (CFS-APQ1) uses an importance verification to acknowledge that people value things differently, while the second total score (CFS-APQ2) does not take this importance verification into account. A CFS-APQ1 score of 1 indicates no activity limitations or participation restrictions while 16 represents the maximum score; for CFS-APQ2, the scores range between 1 and 4. Data supportive of the psychometric quality of the Dutch version of the CFS-APQ have been reported [30].
Outcomes: pressure pain thresholds (second phase)
Pressure pain thresholds (PPT) were measured with an analogue Fisher algometer (Force Dial model FDK 40, Wagner Instruments, Greenwich) in the skin web between thumb and index finger [31], 5 cm lateral to the spinous process of L3 [32], and at the proximal third of the calf, in order to test pain thresholds on non-specific locations both on the extremities and the trunk. All these sites were assessed in random order. The force is gradually increased at a rate of 1 kg/s until the subject indicates that the pain level has been reached. The threshold is determined as the mean of the two last values out of three consecutive (10 s in between) measurements, since this procedure has found to be reliable in healthy controls [32]. Pressure algometry has been found to be efficient and reliable in the exploration of physiopathological mechanisms involved in pain [33] and is useful for the evaluation of treatment outcome, as reviewed by Fischer [34].
Statistical analysis
All data were analyzed using SPSS 12.0© for Windows (Chicago, IL 60606 USA). Appropriate descriptive statistics were used: mean, median, frequency distribution, interquartile range, range and standard deviation for age, duration of the illness, symptom intensity (especially pain), degree of daily functioning, and the scores obtained with the other questionnaires.
In order to examine the associations between pain assessments and daily functioning on the one hand and cognitive and behavioral aspects on the other hand, Pearson and Spearman (for the variables on the ordinal level) correlation analyses were used. For interpreting correlation coefficients and defining predictors for pain and functioning, the coefficient of determination (of pain and daily functioning) for the different psychosocial factors was analyzed by multiple hierarchic regression analysis (stepwise). The significance level was set at .01 to help protect against potential type I errors.
Results
The descriptive statistics of the 103 included patients are presented in Table 1.
First phase (baseline)
In order to answer the four key questions of the present investigation, Table 2 presents the correlations between pain and daily functioning and the psychosocial determinants at baseline.
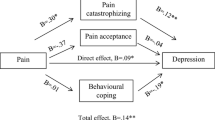
Catastrophizing, measured with the PCS, was significantly correlated with the two pain measurements and with the self-reported restrictions in activities and participation. These were, in fact, the highest correlations with the pain and functioning measurements (Table 2). Furthermore, the PCS was the main predictor for the pain measurement but could not predict daily functioning significantly (Table 3).
Coping. The PCI subscale “worrying” was positively related to the VAS pain score. Pain evaluated with the SF-36 correlated significantly with the PCI items “transforming pain,” “retreating,” “worrying,” and “resting.” The PCI subscales “transforming pain,” “worrying,” and “reducing demands” were positively associated with the activity limitations/participation restrictions, and “retreating” and “resting” were correlated with the amount of habitual daily activity (Table 2). On top of the main predictor PCS, the subscale “resting” is responsible for 4.7% of the variance of the SF-36 pain. “Resting” and “transforming the pain” were also responsible for about 14% of the variance on top of the main predictor BDI (Table 3).
Depression evaluated with the BDI showed high correlations with VAS pain, SF-36 pain, and activity limitations/participation restrictions and was the main predictor of activity limitations/participation restrictions (Table 2). BDI was not a significant predictor for the pain measurements (Table 3).
Kinesiophobia showed significant correlations with the two pain measurements and with activity limitations/participation restrictions (Table 2) but was not a significant predictor for pain or functioning (Table 3).
Following Table 4, pain is also related to age, use of analgesics, and the professional state. Restrictions in activities and participation correlated with the professional state and the use of antidepressants. Given the fact that the VAS pain was related to gender, the difference in pain intensity for men and women was analyzed by an independent t test. The mean VAS pain for women was 52.95 (±23.746). For men, the mean VAS pain was 35.60 (±20.406). Pain intensity was significantly different (p = .009). Therefore, a regression analysis was performed for the two sexes separately. The VAS pain was mainly predicted by “catastrophizing” in both men and women.
Second phase (6–12 months later)
Of the 103 patients of the first phase, 39 patients were willing to participate again. The reasons for the dropout of 61 subjects were the impossibility to be present in the 6 to 12 months after the first participation (25), refusal (11), practical obstacles as distance and time (11), impossibility to approach the subjects (10), and finally, absence on the appointment (4). The descriptive statistics of these patients are presented in Table 1.
Pearson correlation coefficients were calculated between the pain cognitions/behavior of the first phase and the pain measurements of the second phase. We found significant correlations with VAS pain for depression (r = .356, p = .036) and the “helplessness” subscale of the PCS (r = .340, p = .045). SF-36 pain was significantly correlated with “rumination” of the PCS (r = −.478, p = .008) and the total PCS score (r = −.415, p = .023). No significant correlations with pain thresholds were revealed.
For the significant correlations, a linear regression analysis was performed in order to define the predictive value of pain cognitions/behavior regarding pain assessments 6 to 12 months later. The predictive value of the pain cognitions/behavior is presented in Table 3.
Discussion
The goal of this study was to perform an investigation into the relation between psychosocial determinants and pain and daily functioning in CFS patients with chronic widespread pain. In other chronic pain populations, much more studies are conducted regarding the psychosocial background of pain, but in CFS, studies are currently lacking.
This study revealed an important relation between pain and psychological factors. Catastrophizing and depression were the most important predictors for pain intensity at the same time or 6–12 months later. Also, habitual activity and restrictions in activities and participations are related to these factors.
Pain
The coping strategy “worrying” was significantly correlated with pain severity, assessed by use of a VAS and by the SF-36, and in addition, the subscale “resting” correlated significantly with SF-36 bodily pain subscale score. These two subscales and the subitem “transforming pain” were furthermore related to the restrictions in activities and participation.
Catastrophizing and depression were significantly correlated with the pain measurements at the same time and 6 to 12 months later and also with activity limitations/participation restrictions. Kinesiophobia was associated with the SF-36 bodily pain subscale score and with the self-reported restriction. These findings indicate that the more pain and restrictions the patients experience, the more they deal with depressive and catastrophic thoughts and the more they use passive coping styles as resting and worrying. No significant correlations with pain thresholds were revealed.
Based on the revealed correlations, the direction of the relation remains unclear. A multiple regression analysis was performed in order to define the contribution of the different predictors of pain and functioning, corrected for possible confounding factors. Following this analysis, the variance in pain at the same time can be predicted significantly by the four questionnaires for up to 34.5% and by the PCS for up to 22.6%, eventually accompanied by the subscale “resting” of the PCI. Activity limitations and participation restrictions can be predicted by the four questionnaires for 41.7%. The main predictors are depression (23.1%), resting (8.9%), and transforming pain (4.9%).
VAS pain 6 to 12 months later was mainly predicted by depression (12.7%), while pain assessed with the SF-36 was chiefly predicted by the catastrophizing subitem “rumination” (22.8%). These findings are slightly different from the result of the first phase, where the total score of the PCS was the main predictor for both pain assessments, and depression was not a significant predictor, although responsible for 23.1%. While several coping strategies were important predictors for pain at the same time, they become irrelevant in predicting pain intensity 6 to 12 months later.
Similar to the present findings, earlier investigations pushed catastrophizing and depression forward as the most important psychological predictors of pain in other chronic pain patients, accounting for 7% to 31% of the variance in pain ratings [6, 35]. In FM, the predictive value of pain catastrophizing and depression for pain was reported by Hassett et al. [6] (respectively 27% and 30%), and in female CFS patients, catastrophizing accounted for 41% of the observed variance in bodily pain, independent of depression [36]. Petrie et al. [37] mentioned that CFS patients with catastrophic thoughts experienced more fatigue and more disabilities, but they did not study the interaction with chronic pain in CFS.
This can be explained by the findings of Gracely et al. [38]. They suggest that pain catastrophizing is significantly associated with increased activity in brain areas related to anticipation of pain (medial frontal cortex, cerebellum), attention to pain (dorsal ACC, dorsolateral prefrontal cortex), emotional aspects of pain (claustrum, closely connected to the amygdala), and motor control. Catastrophizing would influence pain perception through altering attention and anticipation and heightening emotional responses to pain, leading to avoidance, hypervigilance, inactivity, and reduced pain tolerance.
To obtain a more accurate estimation of the predictive value of catastrophizing and depression for pain after 6–12 months, we reanalyzed the determination of VAS pain and SF-36 pain reported during the follow-up moment by correcting for the relationship with baseline pain. This was done by the use of blocks in a hierarchical multiple regression analysis. The baseline variables were entered as a covariate in a second block. In the case of the VAS pain, this leads to an R square change of .065 (p = .141), but in the case of the SF-36, pain was significant (p = .001) with an R square change of .289. So this means that the pain measured with the SF-36 was more predicted by the baseline SF-36 pain than by the psychological variables! The differences between the two pain measurements may be due to the fact that the VAS pain measures pain intensity during the last 24 h, while the SF-36 evaluated the last 2 weeks, which is more an average score. This inconsistent finding requires further research.
Daily functioning
In accordance to the role of depression in self-reported restrictions in our study, depression has been found to be an important predictor of disabilities in activities in patients with chronic low back pain [39]. Also in CFS patients, depression was related to restrictions in social functioning, mental health, and health perception [18].
As for kinesiophobia, earlier studies in CFS provided evidence for the relation with self-reported restrictions in activities [3] and with activity avoidance leading to the maintenance of symptoms and disabilities [17]. This activity avoidance and an “external locus of control” were significantly correlated to fatigue and impairments in the study of Ray et al. [12].
The relation between the coping styles “resting” and “worrying” and pain and daily functioning was also expected, given the fact that these passive or maladaptive coping styles [10] have been found to be associated with worse outcomes in CFS patients [12, 13]. Coping styles were even found to be major predictors of long-term outcome in CFS patients [40]. Furthermore, taking rest is obviously related to a restriction in physical activities. The present study, however, was the first to study kinesiophobia and coping strategies in relation to pain and daily functioning in CFS patients having chronic widespread pain, rather than patients with CFS in general.
Limitations
Of course, the present results should be interpreted in the light of several study limitations. The biggest problem, however, was the great loss to follow-up. Only 39 of the original 103 CFS patients participated in the second phase. Therefore, there may be a matter of selection bias in the present study. Although the present sample was demographically comparable to the sample of the first phase and apparently representative for the CFS population (gender distribution, age, etc.), only the motivated and the better patients may have participated. If we compared all 39 participants of the second phase with 64 patients that only participated in the first phase for all questionnaires, we saw that for most variables, the dropout patients were comparable to the 39 participants that completed the study, despite for stomach pain and hypersensitivity to light of the CFS symptom list and role limitations due to physical health problems (SF-36). Future studies may try to deal with great losses to follow-up, by trying to obtain at least the self-reported measures on follow-up by mail, if participants cannot show up at further assessment appointments.
Another limitation in the present study is the use of the BDI II to assess depressive symptoms. A number of items of the BDI refer to somatic symptoms that are also part of CFS. This could artificially inflate the correlation between depression and pain/disability in CFS. For future research, it would be better to use the BDI primary care version (which does not use somatic symptoms) or the Hospital Anxiety and Depression Scale for example in further studies. The latter seems a valid screening tool for depression in patients with CFS [41].
Relevance for clinical practice
The approach of these patients is often a real challenge, because of the unknown source of the pain and the poor therapy adherence. One factor influencing individual success with such programs may be the adherence of the patient to the treatment, where adherence implies active voluntary involvement. Psychological variables, like catastrophizing and kinesiophobia, are expected to present barriers for active involvement of patients suffering chronic pain [42].
As catastrophizing was the most important predictor here, it could be recommended to inform your patient. Catastrophizing could indeed be the expression or the consequence of ignorance or incorrect illness perceptions [43]. Therefore, reassuring and giving appropriate information might be helpful, because information is determining for the eventual threatening appraisal of pain [44]. Pain education has already been shown to reduce catastrophic thinking in CFS patients [45]. Also, graded activity or graded exposure programs may be useful in reducing catastrophic thinking [46]. Given the association between these pain cognitions and pain, it can be hypothesized that an improvement in pain cognitions will lead to less pain on longer term, but this hypothesis needs further study.
On the other hand, it seems that cognitive behavior therapy (CBT) aimed at fatigue in CFS also leads to a reduction of pain. Surprisingly, changes in physical activity, in negative affectivity, or in body consciousness could not explain the decrease in pain severity after cognitive behavior therapy. Only a relationship between the decrease in fatigue and the decrease in pain was found. This implies that pain in CFS is part of the syndrome and is directly related to chronic fatigue [47]. Unfortunately, the role of catastrophizing was not studied in the latter study, since CBT also seems to reduce pain catastrophizing in patients with fibromyalgia [48]. Therefore, it would be interesting to study whether the reduced pain after CBT may be mediated by reduced catastrophizing.
Conclusion
In conclusion, pain catastrophizing, depression, kinesiophobia, and several passive coping styles were related to pain and functioning, but catastrophizing and depression were the main predictors for pain at the same time and 6 to 12 months later and for functioning in patients with CFS having chronic widespread musculoskeletal pain. Depression and catastrophizing are able to predict 23% of the pain variance 6 to 12 months later. Since these factors may be at the basis of chronic pain and act as important therapy barriers in CFS, it is important to consider these psychosocial factors in the approach of CFS patients experiencing chronic widespread pain.
References
Fukuda K, Straus SE, Hickie I et al (1994) The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med 121:953–959
Jason LA, Torres-Harding SR, Carrico AW et al (2002) Symptom occurrence in persons with chronic fatigue syndrome. Biol Psychol 59:15–27
Nijs J, Aerts A, De Meirleir K (2006) Generalized joint hypermobility is more common in chronic fatigue syndrome than in healthy control subjects. J Manipulative Physiol Ther 29:32–39
Nijs J, Van de Velde B, De Meirleir K (2005) Pain in patients with chronic fatigue syndrome: does nitric oxide trigger central sensitisation? Med Hypotheses 64:558–562
Meeus M, Nijs J, Meirleir KD (2007) Chronic musculoskeletal pain in patients with the chronic fatigue syndrome: a systematic review. Eur J Pain 11:377–386
Hassett AL, Cone JD, Patella SJ et al (2000) The role of catastrophizing in the pain and depression of women with fibromyalgia syndrome. Arthritis Rheum 43:2493–2500
Vlaeyen JW, Linton SJ (2000) Fear-avoidance and its consequences in chronic musculoskeletal pain: a state of the art. Pain 85:317–332
Sullivan MJL, Bishop SR, Pivik J (1995) The pain catastrophizing scale: development and validation. Psychol Asses 7:524–532
Bradley LA, McKendree-Smith NL, Alarcon GS (2000) Pain complaints in patients with fibromyalgia versus chronic fatigue syndrome. Curr Rev Pain 4:148–157
Kraaimaat FW, Bakker A, Evers AWM (1997) Pijncoping-strategieën bij chronische pijnpatiënten: de ontwikkeling van de Pijn Coping Inventarisatielijst (PCI). Gedragstherapie 30:185–201
Kraaimaat FW, Evers AW (2003) Pain-coping strategies in chronic pain patients: psychometric characteristics of the pain-coping inventory (PCI). Int J Behav Med 10:343–363
Ray C, Jefferies S, Weir WR (1995) Coping with chronic fatigue syndrome: illness responses and their relationship with fatigue, functional impairment and emotional status. Psychol Med 25:937–945
Heijmans MJ (1998) Coping and adaptive outcome in chronic fatigue syndrome: importance of illness cognitions. J Psychosom Res 45:39–51
Ray C, Jefferies S, Weir WR (1997) Coping and other predictors of outcome in chronic fatigue syndrome: a 1-year follow-up. J Psychosom Res 43:405–415
Kori SH, Miller RP, Todd DD (1990) Kinesiophobia: a new view of chronic pain behavior. Pain Management 35–43
Nijs J, De Meirleir K, Duquet W (2004) Kinesiophobia in chronic fatigue syndrome: assessment and associations with disability. Arch Phys Med Rehabil 85:1586–1592
Silver A, Haeney M, Vijayadurai P et al (2002) The role of fear of physical movement and activity in chronic fatigue syndrome. J Psychosom Res 52:485–493
Morriss RK, Ahmed M, Wearden AJ et al (1999) The role of depression in pain, psychophysiological syndromes and medically unexplained symptoms associated with chronic fatigue syndrome. J Affect Disord 55:143–148
Smeets RJ, Maher CG, Nicholas MK et al (2009) Do psychological characteristics predict response to exercise and advice for subacute low back pain? Arthritis Rheum 61:1202–1209
Kraemer HC, Wilson GT, Fairburn CG et al (2002) Mediators and moderators of treatment effects in randomized clinical trials. Arch Gen Psychiatry 59:877–883
Wolfe F, Smythe HA, Yunus MB et al (1990) The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum 33:160–172
Bishop SR, Edgley K, Fisher R et al (1993) Screening for depression in chronic low back pain with the Beck Depression Inventory. Can J Rehab 7:143–148
Van Damme S, Crombez G, Bijttebier P et al (2002) A confirmatory factor analysis of the Pain Catastrophizing Scale: invariant factor structure across clinical and non-clinical populations. Pain 96:319–324
Vlaeyen JW, Kole-Snijders AM, Boeren RG et al (1995) Fear of movement/(re)injury in chronic low back pain and its relation to behavioral performance. Pain 62:363–372
De Becker P, McGregor N, De Meirleir K (2001) A definition-based analysis of symptoms in a large cohort of patients with chronic fatigue syndrome. J Intern Med 250:234–240
Nijs J, Thielemans A (2008) Kinesiophobia and symptomatology in chronic fatigue syndrome: a psychometric study of two questionnaires. Psychol Psychother 81:273–283
Ware JE Jr, Snow KK, Kosinki M et al (1993) SF-36 Health Survey manual and interpretation guide. The Health Institute, Boston
McHorney CA, Ware JE Jr, Raczek AE (1993) The MOS 36-Item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care 31:247–263
Nijs J, Vaes P, Van Hoof E et al (2002) Activity limitations and participation restrictions in patients with chronic fatigue syndrome—construction of a disease specific questionnaire. J Chronic Fatigue Syndr 10:3–23
Nijs J, Vaes P, McGregor N et al (2003) Psychometric properties of the Dutch Chronic Fatigue Syndrome–Activities and Participation Questionnaire (CFS-APQ). Phys Ther 83:444–454
Whiteside A, Hansen S, Chaudhuri A (2004) Exercise lowers pain threshold in chronic fatigue syndrome. Pain 109:497–499
Farasyn A, Meeusen R (2003) Pressure pain thresholds in healthy subjects: influence of physical activity, history of lower back pain factors and the use of endermology as a placebo-like treatment. J Bodywork Mov Ther 7:53–61
Vanderweeen L, Oostendorp RA, Vaes P et al (1996) Pressure algometry in manual therapy. Man Ther 1:258–265
Fischer A (1998) Muscle pain syndromes and fibromyalgia. Pressure algometry for quantation of diagnosis and treatment outcome. J Musculoskelet Pain 6:1–32
Sullivan MJ, Thorn B, Haythornthwaite JA et al (2001) Theoretical perspectives on the relation between catastrophizing and pain. Clin J Pain 17:52–64
Nijs J, Van de Putte K, Louckx F et al (2008) Exercise performance and chronic pain in chronic fatigue syndrome: the role of pain catastrophizing. Pain Med. doi:10.1111/j.1526-4637.2007.00368.x
Petrie K, Moss-Morris R, Weinman J (1995) The impact of catastrophic beliefs on functioning in chronic fatigue syndrome. J Psychosom Res 39:31–37
Gracely RH, Geisser ME, Giesecke T et al (2004) Pain catastrophizing and neural responses to pain among persons with fibromyalgia. Brain 127:835–843
Verbunt JA, Seelen HA, Vlaeyen JW et al (2003) Fear of injury and physical deconditioning in patients with chronic low back pain. Arch Phys Med Rehabil 84:1227–1232
Wilson A, Hickie I, Lloyd A et al (1994) Longitudinal study of outcome of chronic fatigue syndrome. BMJ 308:756–759
Morriss RK, Wearden AJ (1998) Screening instruments for psychiatric morbidity in chronic fatigue syndrome. J R Soc Med 91:365–368
Mannion AF, Helbling D, Pulkovski N et al (2009) Spinal segmental stabilisation exercises for chronic low back pain: programme adherence and its influence on clinical outcome. Eur Spine 18:1881–91J
van Wilgen CP, van Ittersum MW, Kaptein AA et al (2008) Illness perceptions in patients with fibromyalgia and their relationship to quality of life and catastrophizing. Arthritis Rheum 58:3618–3626
Jackson T, Pope L, Nagasaka T et al (2005) The impact of threatening information about pain on coping and pain tolerance. Br J Health Psychol 10:441–451
Meeus M, Nijs J, Van Oosterwijck J et al (2010) Pain physiology education improves pain beliefs in patients with chronic fatigue syndrome compared with pacing and self-management education: a double-blind randomized controlled trial. Arch Phys Med Rehabil 91:1153–1159
Leeuw M, Goossens ME, van Breukelen GJ et al (2008) Exposure in vivo versus operant graded activity in chronic low back pain patients: results of a randomized controlled trial. Pain 138:192–207
Knoop H, Stulemeijer M, Prins JB et al (2007) Is cognitive behaviour therapy for chronic fatigue syndrome also effective for pain symptoms? Behav Res Ther 45:2034–2043
Alda M, Luciano-Devis JV, Andres E et al (2011) Effectiveness of cognitive behaviour therapy for the treatment of catastrophisation in patients with fibromyalgia: a randomised controlled trial. Arthritis Res Ther 13:R173
Acknowledgments
Mira Meeus was financially supported by a PhD grant supplied by the Higher Institute of Physiotherapy, Department of Health Sciences, Artesis University College Antwerp, Belgium (G 807) and co-financed by the Faculty of Physical Education and Physiotherapy–Vrije Universiteit Brussel (VUB), Belgium (OZR project OZ.R. 1234/MFYS Wer2) and is now supported as post-doctoral research fellow by the Research Foundation Flanders (FWO).
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meeus, M., Nijs, J., Van Mol, E. et al. Role of psychological aspects in both chronic pain and in daily functioning in chronic fatigue syndrome: a prospective longitudinal study. Clin Rheumatol 31, 921–929 (2012). https://doi.org/10.1007/s10067-012-1946-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-012-1946-z