Abstract
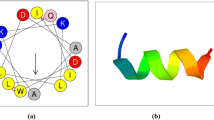
In response to infection, insects produce a variety of antimicrobial peptides (AMPs) to kill the invading pathogens. To study their physicochemical properties and bioactivities for clinical and commercial use in the porcine industry, we chemically synthesized the mature peptides Bombyx mori moricin and Hyalophora cecropia cecropin B. In this paper, we described the antimicrobial activity of the two AMPs. Moricin exhibited antimicrobial activity on eight strains tested with minimal inhibitory concentration values (MICs) ranging between 8 and 128 μg/ml, while cecropin B mainly showed antimicrobial activity against the Gramnegative strains with MICs ranging from 0.5 to 16 μg/ml. Compared to the potent antimicrobial activity these two AMPs displayed against most of the bacterial pathogens tested, they exhibited limited hemolytic activity against porcine red blood cells. The activities of moricin and cecropin B against Haemophilus parasuis SH 0165 were studied in further detail. Transmission electron microscopy (TEM) of moricin and cecropin B treated H. parasuis SH 0165 indicated extensive damage to the membranes of the bacteria. Insights into the probable mechanism utilized by moricin and cecropin B to eliminate pathogens are also presented. The observations from this study are important for the future application of AMPs in the porcine industry.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andersson, M., Boman, A., and Boman, H. (2003). Ascaris nematodes from pig and human make three antibacterial peptides: isolation of cecropin P1 and two ASABF peptides. Cell. Mol. Life Sci. 60, 599–606.
Boman, H.G., and Hultmark, D. (1981). Cell-free immunity in insects. Trends Biochem. Sci. 6, 306–309.
Brogden, K.A. (2005). Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 3, 238–250.
Cavaco, L.M., Hasman, H., and Aarestrup, F.M. (2011). Zinc resistance of Staphylococcus aureus of animal origin is strongly associated with methicillin resistance. Vet. Microbiol. 150, 344–348.
Chen, H.M., Chan, S.C., Lee, J.C., Chang, C.C., Murugan, M., and Jack, R.W. (2003). Transmission electron microscopic observations of membrane effects of antibiotic cecropin B on Escherichia coli. Microsc. Res. Tech. 62, 423–430.
Christensen, B., Fink, J., Merrifield, R., and Mauzerall, D. (1988). Channel-forming properties of cecropins and related model compounds incorporated into planar lipid membranes. Proc. Nat. Acad. Sci. USA 85, 5072–5076.
Durell, S.R., Raghunathan, G., and Guy, H.R. (1992). Modeling the ion channel structure of cecropin. Biophys. J. 63, 1623–1631.
Flyg, C., Dalhammar, G., Rasmuson, B., and Boman, H.G. (1987). Insect immunity: inducible antibacterial activity in Drosophila. Insect Biochem. 17, 153–160.
Frye, J.G., and Fedorka-Cray, P.J. (2007). Prevalence, distribution and characterisation of ceftiofur resistance in Salmonella enterica isolated from animals in the USA from 1999 to 2003. Int. J. Antimicrob. Agents 30, 134–142.
Gazit, E., Boman, A., Boman, H.G., and Shai, Y. (1995). Interaction of the mammalian antibacterial peptide cecropin P1 with phospholipids vesicles. Biochemistry 34, 11479–11488.
Hammer, M.U., Brauser, A., Olak, C., Brezesinski, G., Goldmann, T., Gutsmann, T., and Andrä, J. (2010). Lipopolysaccharide interaction is decisive for the activity of the antimicrobial peptide NK-2 against Escherichia coli and Proteus mirabilis. Biochem. J. 427, 477–488.
Hancock, R.E.W., and Rozek, A. (2002). Role of membranes in the activities of antimicrobial cationic peptides. FEMS Microbiol. Lett. 206, 143–149.
Hara, S., and Yamakawa, M. (1995a). A novel antibacterial peptide family isolated from the silkworm, Bombyx mori. Biochem. J. 310 651–656.
Hara, S., and Yamakawa, M. (1995b). Moricin, a novel type of antibacterial peptide isolated from the silkworm, Bombyx mori. J. Biol. Chem. 270, 29923–29927.
Hartmann, M., Berditsch, M., Hawecker, J., Ardakani, M.F., Gerthsen, D., and Ulrich, A.S. (2010). Damage of bacterial cell envelope by antimicrobial peptides Gramicidin S and PGLa as revealed by transmission and scanning electron microscopy. Antimicro. Agents Chemother. 54, 3132–3142.
Hemmi, H., Ishibashi, J., Hara, S., and Yamakawa, M. (2002). Solution structure of moricin, an antibacterial peptide, isolated from the silkworm Bombyx mori. FEBS Lett. 518, 33–38.
Hultmark, D., Steiner, H., Rasmuson, T., and Boman, H.G. (1980). Insect immunity: purification and properties of three inducible bactericidal proteins from hemolymph of immunized pupae of Hyalophora cecropia. Eur. J. Biochem. 106, 7–16.
Hultmark, D., Engström, Å., Bennich, H., Kapur, R., and Boman, H.G. (1982). Insect immunity: isolation and structure of cecropin D and four minor antibacterial components from Cecropia pupae. Eur. J. Biochem. 127, 207–217.
Ishitsuka, Y., Pham, D.S., Waring, A.J., Lehrer, R.I., and Lee, K.Y.C. (2006). Insertion selectivity of antimicrobial peptide protegrin-1 into lipid monolayers: effect of head group electrostatics and tail group packing. Biochim. Biophys. Acta (BBA)-Biomembranes 1758, 1450–1460.
Jan, P.S., Huang, H.Y., and Chen, H.M. (2010). Expression of a synthesized gene encoding cationic peptide cecropin B in transgenic tomato plants protects against bacterial diseases. Appl. Environ. Microb. 76, 769–775.
Jenssen, H., Hamill, P., and Hancock, R.E.W. (2006). Peptide antimicrobial agents. Clin. Microbiol. Rev. 19, 491–511.
Kim, S.R., Hong, M.Y., Park, S.W., Choi, K.H., Yun, E.Y., Goo, T.W., Kang, S.W., Suh, H.J., Kim, I., and Hwang, J.S. (2010). Characterization and cDNA cloning of a cecropin-like antimicrobial peptide, papiliocin, from the swallowtail butterfly, Papilio xuthus. Mol. Cells 29, 419–423.
Kim, H.B., Baek, H., Lee, S.J., Jang, Y.H., Jung, S.C., Kim, A., and Choe, N.H. (2011). Prevalence and antimicrobial resistance of Salmonella spp. and Escherichia coli isolated from pigs at slaughterhouses in Korea. Afr. J. Microbiol. Res. 5, 823–830.
Klüver, E., Schulz-Maronde, S., Scheid, S., Meyer, B., Forssmann, W.G., and Adermann, K. (2005). Structure-activity relation of human β-defensin 3: influence of disulfide bonds and cysteine substitution on antimicrobial activity and cytotoxicity. Biochemistry 44, 9804–9816.
Kreil, G. (1994). Antimicrobial peptides from amphibian skin: an overview. Ciba Found. Symp. 186, 77–85.
Merrifield, R.B. (1963). Solid phase peptide synthesis. l. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 85, 2149–2154.
Oizumi, Y., Hemmi, H., Minami, M., Asaoka, A., and Yamakawa, M. (2005). Isolation, gene expression and solution structure of a novel moricin analogue, antibacterial peptide from a lepidop-teran insect, Spodoptera litura. Biochim. Biophys. Acta 1752, 83–92.
Sarmasik, A., and Chen, T.T. (2003). Bactericidal activity of cecro pin B and cecropin P 1 expressed in fish cells (CHSE-214): application in controlling fish bacterial pathogens. Aquaculture 220, 183–194.
Sato, H., and Feix, J.B. (2006). Peptide-membrane interactions and mechanisms of membrane destruction by amphipathic α-helical antimicrobial peptides. Biochim. Biophys. Acta 1758, 1245–1256.
Steinberg, D.A., Hurst, M.A., Fujii, C.A., Kung, A., Ho, J., Cheng, F., Loury, D.J., and Fiddes, J.C. (1997). Protegrin-1: a broad-spectrum, rapidly microbicidal peptide with in vivo activity. Antimicrob. Agents Chemother. 41, 1738–1742.
Steiner, H. (1982). Secondary structure of the cecropins: antibacterial peptides from the moth Hyalophora cecropia. FEBS Lett. 137, 283–287.
Steiner, H., Andreu, D., and Merrifield, R.B. (1988). Binding and action of cecropin and cecropin analogues: antibacterial peptides from insects. Biochim. Biophys. Acta 939, 260–266.
Van Hofsten, P., Faye, I., Kockum, K., Lee, J., Xanthopoulos, K., Boman, I., Boman, H., Engström, A., Andreu, D., and Merrifield, R. (1985). Molecular cloning, cDNA sequencing, and chemical synthesis of cecropin B from Hyalophora cecropia. Proc. Nat. Acad. Sci. USA 82, 2240.
Vanni, M., Merenda, M., Barigazzi, G., Garbarino, C., Luppi, A., Tognetti, R., and Intorre, L. (2011). Antimicrobial Resistance of Actinobacillus Pleuropneumoniae Isolated from Swine. Vet. Microbiol. 156, 172–177.
Veldhuizen, E.J.A., Rijnders, M., Claassen, E.A., Van Dijk, A., and Haagsman, H.P. (2008). Porcine β-defensin 2 displays broad antimicrobial activity against pathogenic intestinal bacteria. Mol. Immunol. 45, 386–394.
Wu, J.M., Jan, P.S., Yu, H.C., Haung, H.Y., Fang, H.J., Chang, Y.I., Cheng, J.W., and Chen, H.M. (2009). Structure and function of a custom anticancer peptide, CB1a. Peptides 30, 839–848.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hu, H., Wang, C., Guo, X. et al. Broad activity against porcine bacterial pathogens displayed by two insect antimicrobial peptides moricin and cecropin B. Mol Cells 35, 106–114 (2013). https://doi.org/10.1007/s10059-013-2132-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-2132-0