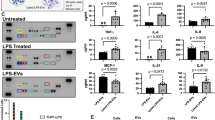
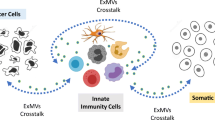
Abstract
The host immune response involves a variety of cell types, including specialized immune and non-immune cells. The delicate coordination among these cells via close communication is central for the proper operation of immune system. Cell-cell communication is mediated by a complex network that includes soluble factors such as cytokines, chemokines, and metabolites exported from cells, as well as membrane-bound receptors and their ligands. Cell-cell communication is also mediated by membrane vesicles (e.g., exosomes, ectosomes), which are either shed by distant cells or exchanged by cells that are making direct contact. Intercellular communication via extracellular membrane vesicles has drawn much attention recently, as they have been shown to carry various biomolecules that modulate the activities of recipient cells. In this review, I will discuss current views on cell-cell communication via extra-cellular membrane vesicles, especially shedded membrane vesicles, and their effects on the control of the immune system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allan, R.S., Waithman, J., Bedoui, S., Jones, C.M., Villadangos, J.A., Zhan, Y., Lew, A.M., Shortman, K., Heath, W.R., and Carbone, F.R. (2006). Migratory dendritic cells transfer antigen to a lymph node-resident dendritic cell population for efficient CTL priming. Immunity 25, 153–162.
Allison, A.C., and Eugui, E.M. (1983). The role of cell-mediated immune responses in resistance to malaria, with special reference to oxidant stress. Annu. Rev. Immunol. 1, 361–392.
Almqvist, N., Lonnqvist, A., Hultkrantz, S., Rask, C., and Telemo, E. (2008). Serum-derived exosomes from antigen-fed mice prevent allergic sensitization in a model of allergic asthma. Immunology 125, 21–27.
Bianco, N.R., Kim, S.H., Morelli, A.E., and Robbins, P.D. (2007). Modulation of the immune response using dendritic cell-derived exosomes. Methods Mol. Biol. 380, 443–455.
Blanchard, N., Lankar, D., Faure, F., Regnault, A., Dumont, C., Raposo, G., and Hivroz, C. (2002). TCR activation of human T cells induces the production of exosomes bearing the TCR/CD3/zeta complex. J. Immunol. 168, 3235–3241.
Boes, M., Cuvillier, A., and Ploegh, H. (2004). Membrane specializations and endosome maturation in dendritic cells and B cells. Trends Cell Biol. 14, 175–183.
Borges, F.T., Melo, S.A., Ozdemir, B.C., Kato, N., Revuelta, I., Miller, C.A., Gattone, V.H., 2nd, LeBleu, V.S., and Kalluri, R. (2013). TGF-beta1-containing exosomes from injured epithelial cells activate fibroblasts to initiate tissue regenerative responses and fibrosis. J. Am. Soc. Nephrol. 24, 385–392.
Bu, N., Wu, H., Sun, B., Zhang, G., Zhan, S., Zhang, R., and Zhou, L. (2011). Exosome-loaded dendritic cells elicit tumor-specific CD8+ cytotoxic T cells in patients with glioma. J. Neurooncol. 104, 659–667.
Bullerdiek, J., and Flor, I. (2012). Exosome-delivered microRNAs of “chromosome 19 microRNA cluster” as immunomodulators in pregnancy and tumorigenesis. Mol. Cytogenet. 5, 27.
Campbell, A.K., and Morgan, B.P. (1985). Monoclonal antibodies demonstrate protection of polymorphonuclear leukocytes against complement attack. Nature 317, 164–166.
Chalmin, F., Ladoire, S., Mignot, G., Vincent, J., Bruchard, M., Remy-Martin, J.P., Boireau, W., Rouleau, A., Simon, B., Lanneau, D., et al. (2010). Membrane-associated Hsp72 from tumor-derived exosomes mediates STAT3-dependent immunosuppressive function of mouse and human myeloid-derived suppressor cells. J. Clin. Invest. 120, 457–471.
Chaput, N., and Thery, C. (2011). Exosomes: immune properties and potential clinical implementations. Semin. Immunol. 33, 419–440.
Clayton, A., Turkes, A., Dewitt, S., Steadman, R., Mason, M.D., and Hallett, M.B. (2004). Adhesion and signaling by B cell-derived exosomes: the role of integrins. FASEB J. 18, 977–979.
Clayton, A., Mitchell, J.P., Court, J., Mason, M.D., and Tabi, Z. (2007). Human tumor-derived exosomes selectively impair lymphocyte responses to interleukin-2. Cancer Res. 67, 7458–7466.
Clayton, A., Mitchell, J.P., Court, J., Linnane, S., Mason, M.D., and Tabi, Z. (2008). Human tumor-derived exosomes down-modulate NKG2D expression. J. Immunol. 180, 7249–7258.
Combes, V., Taylor, T.E., Juhan-Vague, I., Mege, J.L., Mwenechanya, J., Tembo, M., Grau, G.E., and Molyneux, M.E. (2004). Circulating endothelial microparticles in malawian children with severe falciparum malaria complicated with coma. JAMA 291, 2542–2544.
Combes, V., Coltel, N., Alibert, M., van Eck, M., Raymond, C., Juhan-Vague, I., Grau, G.E., and Chimini, G. (2005). ABCA1 gene deletion protects against cerebral malaria: potential pathogenic role of microparticles in neuropathology. Am. J. Pathol. 166, 295–302.
Cooper, A.M. (2009). Cell-mediated immune responses in tuberculosis. Annu. Rev. Immunol. 27, 393–422.
Denzer, K., Kleijmeer, M.J., Heijnen, H.F., Stoorvogel, W., and Geuze, H.J. (2000). Exosome: from internal vesicle of the multivesicular body to intercellular signaling device. J. Cell Sci. 113 Pt 19, 3365–3374.
Eken, C., Gasser, O., Zenhaeusern, G., Oehri, I., Hess, C., and Schifferli, J.A. (2008). Polymorphonuclear neutrophil-derived ectosomes interfere with the maturation of monocyte-derived dendritic cells. J. Immunol. 180, 817–824.
Fagiolo, E. (2004). Immunological tolerance loss vs. erythrocyte self antigens and cytokine network disregulation in autoimmune hemolytic anaemia. Autoimmun. Rev. 3, 53–59.
Fanaei, M., Monk, P.N., and Partridge, L.J. (2011). The role of tetraspanins in fusion. Biochem. Soc. Trans. 39, 524–528.
Filipazzi, P., Burdek, M., Villa, A., Rivoltini, L., and Huber, V. (2012). Recent advances on the role of tumor exosomes in immunosuppression and disease progression. Semin. Cancer Biol. 22, 342–349.
Fooksman, D.R., Vardhana, S., Vasiliver-Shamis, G., Liese, J., Blair, D.A., Waite, J., Sacristan, C., Victora, G.D., Zanin-Zhorov, A., and Dustin, M.L. (2010). Functional anatomy of T cell activation and synapse formation. Annu. Rev. Immunol. 28, 79–105.
Franciszkiewicz, K., Boissonnas, A., Boutet, M., Combadiere, C., and Mami-Chouaib, F. (2012). Role of chemokines and chemokine receptors in shaping the effector phase of the antitumor immune response. Cancer Res. 72, 6325–6332.
Fredrickson, E.K., and Gardner, R.G. (2012). Selective destruction of abnormal proteins by ubiquitin-mediated protein quality con trol degradation. Semin. Cell Dev. Biol. 23, 530–537.
Gasser, O., and Schifferli, J.A. (2004). Activated polymorphonuclear neutrophils disseminate anti-inflammatory microparticles by ecto-cytosis. Blood 104, 2543–2548.
Gasser, O., and Schifferli, J.A. (2005). Microparticles released by human neutrophils adhere to erythrocytes in the presence of complement. Exp. Cell Res. 307, 381–387.
Gasser, O., Hess, C., Miot, S., Deon, C., Sanchez, J.C., and Schifferli, J.A. (2003). Characterisation and properties of ectosomes released by human polymorphonuclear neutrophils. Exp. Cell Res. 285, 243–257.
Gastpar, R., Gehrmann, M., Bausero, M.A., Asea, A., Gross, C., Schroeder, J.A., and Multhoff, G. (2005). Heat shock protein 70 surface-positive tumor exosomes stimulate migratory and cytolytic activity of natural killer cells. Cancer Res. 65, 5238–5247.
Gibbings, D.J., Ciaudo, C., Erhardt, M., and Voinnet, O. (2009). Multivesicular bodies associate with components of miRNA effector complexes and modulate miRNA activity. Nat. Cell Biol. 11, 1143–1149.
Giri, P.K., and Schorey, J.S. (2008). Exosomes derived from M. Bovis BCG infected macrophages activate antigen-specific CD4+ and CD8+ T cells in vitro and in vivo. PLos One 3, e2461.
Gomez-Rodriguez, J., Readinger, J.A., Viorritto, I.C., Mueller, K.L., Houghtling, R.A., and Schwartzberg, P.L. (2007). Tec kinases, actin, and cell adhesion. Immunol. Rev. 218, 45–64.
Graham, G.J., and Locati, M. (2013). Regulation of the immune and inflammatory responses by the ‘atypical’ chemokine receptor D6. J. Pathol. 229, 168–175.
Helenius, A. (2011). Endosome maturation. EMBO J. 30, 3481–3500.
Hess, C., Sadallah, S., Hefti, A., Landmann, R., and Schifferli, J.A. (1999). Ectosomes released by human neutrophils are specialized functional units. J. Immunol. 163, 4564–4573
Hess, C., Sadallah, S., and Schifferli, J.A. (2000). Induction of neutrophil responsiveness to myeloperoxidase antibodies by their exposure to supernatant of degranulated autologous neutrophils. Blood 96, 2822–2827.
Horgan, C.P., and McCaffrey, M.W. (2011). Rab GTPases and microtubule motors. Biochem. Soc. Trans. 39, 1202–1206.
Huang, J.F., Yang, Y., Sepulveda, H., Shi, W., Hwang, I., Peterson, P.A., Jackson, M.R., Sprent, J., and Cai, Z. (1999). TCR-Mediated internalization of peptide-MHC complexes acquired by T cells. Science 286, 952–954.
Huber, V., Filipazzi, P., Iero, M., Fais, S., and Rivoltini, L. (2008). More insights into the immunosuppressive potential of tumor exosomes. J. Trans. Med. 6, 63.
Huotari, J., and Helenius, A. (2011). Endosome maturation. EMBO J. 30, 3481–3500.
Hwang, I., and Ki, D. (2011). Receptor-mediated T cell absorption of antigen presenting cell-derived molecules. Front Biosci. 16, 411–421.
Hwang, I., Huang, J.F., Kishimoto, H., Brunmark, A., Peterson, P.A., Jackson, M.R., Surh, C.D., Cai, Z., and Sprent, J. (2000). T cells can use either T cell receptor or CD28 receptors to absorb and internalize cell surface molecules derived from antigen-presenting cells. J. Exp. Med. 191, 1137–1148.
Hwang, I., Shen, X., and Sprent, J. (2003). Direct stimulation of naive T cells by membrane vesicles from antigen-presenting cells: distinct roles for CD54 and B7 molecules. Proc. Natl. Acad. Sci. USA 100, 6670–6675.
Jouvenet, N. (2012). Dynamics of ESCRT proteins. Cell. Mol. Life Sci. 69, 4121–4133.
Karlsson, M., Lundin, S., Dahlgren, U., Kahu, H., Pettersson, I., and Telemo, E. (2001). “Tolerosomes” are produced by intestinal epithelial cells. Eur. J. Immunol. 31, 2892–2900.
Kawamata, T., and Tomari, Y. (2010). Making RISC. Trends Biochem. Sci. 35, 368–376.
Kim, K., Wang, L., and Hwang, I. (2009). A novel flow cytometric high throughput assay for a systematic study on molecular mechanisms underlying T cell receptor-mediated integrin activation. PLoS One 4, e6044.
Laulagnier, K., Motta, C., Hamdi, S., Roy, S., Fauvelle, F., Pageaux, J.F., Kobayashi, T., Salles, J.P., Perret, B., Bonnerot, C., et al. (2004). Mast cell- and dendritic cell-derived exosomes display a specific lipid composition and an unusual membrane organization. Biochem. J. 380, 161–171.
Lee, Y., El Andaloussi, S., and Wood, M.J. (2012). Exosomes and microvesicles: extracellular vesicles for genetic information transfer and gene therapy. Hum. Mol. Genet. 21, R125–134.
Li, X.B., Zhang, Z.R., Schluesener, H.J., and Xu, S.Q. (2006). Role of exosomes in immune regulation. J. Cell. Mol. Med. 10, 364–375.
Liang, B., Peng, P., Chen, S., Li, L., Zhang, M., Cao, D., Yang, J., Li, H., Gui, T., Li, X., et al. (2013). Characterization and proteomic analysis of ovarian cancer-derived exosomes. J. Proteomics 80C, 171–182.
Liu, S., and Storrie, B. (2012). Are Rab proteins the link between Golgi organization and membrane trafficking? Cell. Mol. Life Sci. 69, 4093–4106.
Ludwig, A.K., and Giebel, B. (2012). Exosomes: small vesicles participating in intercellular communication. Int. J. Biochem. Cell Biol. 44, 11–15.
Luzio, J.P., Piper, S.C., Bowers, K., Parkinson, M.D., Lehner, P.J., and Bright, N.A. (2009). ESCRT proteins and the regulation of endocytic delivery to lysosomes. Biochem. Soc. Trans. 37, 178–180.
McLellan, A.D. (2009). Exosome release by primary B cells. Crit. Rev. Immunol. 29, 203–217.
Miyanishi, M., Tada, K., Koike, M., Uchiyama, Y., Kitamura, T., and Nagata, S. (2007). Identification of Tim4 as a phosphatidylserine receptor. Nature 450, 435–439.
Monteiro, A.C., Scovino, A., Raposo, S., Gaze, V.M., Cruz, C., Svensjo, E., Narciso, M.S., Colombo, A.P., Pesquero, J.B., Feres-Filho, E., et al. (2009). Kinin danger signals proteolytically released by gingipain induce Fimbriae-specific IFN-gamma- and IL-17-producing T cells in mice infected intramucosally with Porphyromonas gingivalis. J. Immunol. 183, 3700–3711.
Muller, W. (2006). Dissecting the cytokine network. Cell. Immunol. 244, 162–164.
Nieuwland, R., Berckmans, R.J., Rotteveel-Eijkman, R.C., Maquelin, K.N., Roozendaal, K.J., Jansen, P.G., ten Have, K., Eijsman, L., Hack, C.E., and Sturk, A. (1997). Cell-derived microparticles generated in patients during cardiopulmonary bypass are highly procoagulant. Circulation 96, 3534–3541.
Nolte-’t Hoen, E.N., Buschow, S.I., Anderton, S.M., Stoorvogel, W., and Wauben, M.H. (2009). Activated T cells recruit exosomes secreted by dendritic cells via LFA-1. Blood 113, 1977–1981.
Ostman, S., Taube, M., and Telemo, E. (2005). Tolerosome-induced oral tolerance is MHC dependent. Immunol. 116, 464–476.
Peterson, E.J. (2003). The TCR ADAPts to integrin-mediated cell adhesion. Immunol. Rev. 192, 113–121.
Peterson, R.A. (2012). Regulatory T-cells: diverse phenotypes integral to immune homeostasis and suppression. Toxicol. Pathol. 40, 186–204.
Pols, M.S., and Klumperman, J. (2009). Trafficking and function of the tetraspanin CD63. Exp. Cell Res. 315, 1584–1592.
Rabesandratana, H., Toutant, J.P., Reggio, H., and Vidal, M. (1998). Decay-accelerating factor (CD55) and membrane inhibitor of reactive lysis (CD59) are released within exosomes during In vitro maturation of reticulocytes. Blood 91, 2573–2580.
Rabinowits, G., Gercel-Taylor, C., Day, J.M., Taylor, D.D., and Kloecker, G.H. (2009). Exosomal microRNA: a diagnostic marker for lung cancer. Clin. Lung Cancer 10, 42–46.
Rana, S., Malinowska, K., and Zoller, M. (2013). Exosomal tumor microRNA modulates premetastatic organ cells. Neoplasia 15, 281–295.
Raposo, G., and Stoorvogel, W. (2013). Extracellular vesicles: exosomes, microvesicles, and friends. J. Cell Biol. 200, 373–383.
Raulet, D.H., Gasser, S., Gowen, B.G., Deng, W., and Jung, H. (2013). Regulation of ligands for the NKG2D activating receptor. Annu. Rev. Immunol. 31, 413–441.
Rieu, S., Geminard, C., Rabesandratana, H., Sainte-Marie, J., and Vidal, M. (2000). Exosomes released during reticulocyte maturation bind to fibronectin via integrin alpha4beta1. Eur. J. Biochem. 267, 583–590.
Roxrud, I., Stenmark, H., and Malerod, L. (2010). ESCRT & Co. Biology of the cell/under the auspices of the European Cell Biology Organization 102, 293–318.
Saas, P., and Perruche, S. (2012). Functions of TGF-beta-exposed plasmacytoid dendritic cells. Crit. Rev. Immunol. 32, 529–553.
Sadallah, S., Eken, C., and Schifferli, J.A. (2011). Ectosomes as immunomodulators. Semin. Immunopathol. 33, 487–495.
Sala-Valdes, M., Ailane, N., Greco, C., Rubinstein, E., and Boucheix, C. (2012). Targeting tetraspanins in cancer. Expert Opin. Ther. Targets 16, 985–997.
Schorey, J.S., and Bhatnagar, S. (2008). Exosome function: from tumor immunology to pathogen biology. Traffic 9, 871–881.
Scolding, N.J., Morgan, B.P., Houston, W.A., Linington, C., Campbell, A.K., and Compston, D.A. (1989). Vesicular removal by oligodendrocytes of membrane attack complexes formed by activated complement. Nature 339, 620–622.
Scott, R.S., McMahon, E.J., Pop, S.M., Reap, E.A., Caricchio, R., Cohen, P.L., Earp, H.S., and Matsushima, G.K. (2001). Phagocytosis and clearance of apoptotic cells is mediated by MER. Nature 411, 207–211.
Segura, E., Guerin, C., Hogg, N., Amigorena, S., and Thery, C. (2007). CD8+ dendritic cells use LFA-1 to capture MHC-peptide complexes from exosomes in vivo. J. Immunol. 179, 1489–1496.
Taieb, J., Chaput, N., and Zitvogel, L. (2005). Dendritic cell-derived exosomes as cell-free peptide-based vaccines. Crit. Rev. Immunol. 25, 215–223.
Tamura, Y., Torigoe, T., Kutomi, G., Hirata, K., and Sato, N. (2012). New paradigm for intrinsic function of heat shock proteins as endogenous ligands in inflammation and innate immunity. Curr. Mol. Med. 12, 1198–1206.
Tan, A., Rajadas, J., and Seifalian, A.M. (2013). Exosomes as nanotheranostic delivery platforms for gene therapy. Adv. Drug Deliv. Rev. 65, 357–367.
Taylor, D.D., and Gercel-Taylor, C. (2011). Exosomes/microvesicles: mediators of cancer-associated immunosuppressive microenvironments. Semin. Immunopathol. 33, 441–454.
Thery, C., Regnault, A., Garin, J., Wolfers, J., Zitvogel, L., Ricciardi-Castagnoli, P., Raposo, G., and Amigorena, S. (1999). Molecular characterization of dendritic cell-derived exosomes. Selective accumulation of the heat shock protein hsc73. J. Cell Biol. 147, 599–610.
Thery, C., Ostrowski, M., and Segura, E. (2009). Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 9, 581–593.
Turola, E., Furlan, R., Bianco, F., Matteoli, M., and Verderio, C. (2012). Microglial microvesicle secretion and intercellular signaling. Front. Physiol. 3, 149.
Vestweber, D. (2007). Adhesion and signaling molecules controlling the transmigration of leukocytes through endothelium. Immunol. Rev. 218, 178–196.
Viaud, S., Thery, C., Ploix, S., Tursz, T., Lapierre, V., Lantz, O., Zitvogel, L., and Chaput, N. (2010). Dendritic cell-derived exosomes for cancer immunotherapy: what’s next? Cancer Res. 70, 1281–1285.
Walker, J.D., Maier, C.L., and Pober, J.S. (2009). Cytomegalovirusinfected human endothelial cells can stimulate allogeneic CD4+ memory T cells by releasing antigenic exosomes. J. Immunol. 182, 1548–1559.
Wubbolts, R., Leckie, R.S., Veenhuizen, P.T., Schwarzmann, G., Mobius, W., Hoernschemeyer, J., Slot, J.W., Geuze, H.J., and Stoorvogel, W. (2003). Proteomic and biochemical analyses of human B cell-derived exosomes. Potential implications for their function and multivesicular body formation. J. Biol. Chem. 278, 10963–10972.
Xia, P., and Wadham, C. (2011). Sphingosine 1-phosphate, a key mediator of the cytokine network: juxtacrine signaling. Cytokine Growth Factor Rev. 22, 45–53.
Yoshimura, A., and Muto, G. (2011). TGF-beta function in immune suppression. Curr. Top. Microbiol. Immunol. 350, 127–147.
Zech, D., Rana, S., Buchler, M.W., and Zoller, M. (2012). Tumorexosomes and leukocyte activation: an ambivalent crosstalk. Cell Commun. Signal. 10, 37.
Zomer, A., Vendrig, T., Hopmans, E.S., van Eijndhoven, M., Middeldorp, J.M., and Pegtel, D.M. (2010). Exosomes: fit to deliver small RNA. Commun. Integr. Biol. 3, 447–450.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hwang, I. Cell-cell communication via extracellular membrane vesicles and its role in the immune response. Mol Cells 36, 105–111 (2013). https://doi.org/10.1007/s10059-013-0154-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-0154-2