Abstract
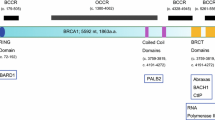
BRCA1 is a well-known tumor suppressor implicated in familial breast and ovarian cancer. Since its cloning in 1994, numerous studies have established BRCA1’s role in diverse cellular and biochemical processes, such as DNA damage repair, cell cycle control, and transcriptional regulation as well as ubiquitination. In addition, a number of recent studies have functionally linked this tumor suppressor to another important cellular regulator, microRNAs, which are short (19–22 nt) RNAs that were discovered in the nematode in 1993. Soon their presence and function were validated in mammals, and since then, the role of microRNAs has been actively investigated in almost all biological processes, including cancer. In this review, we will describe recent progress in the understanding of the BRCA1 function through microRNAs and the role of microRNAs in regulating BRCA1, with emphasis on the implication of these processes on the development and progression of cancer. We will also discuss the therapeutic potential of microRNA mimics or inhibitors of microRNAs to affect BRCA1 function.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aiyar, S.E., Cho, H., Lee, J., and Li, R. (2007). Concerted transcriptional regulation by BRCA1 and COBRA1 in breast cancer cells. Int. J. Biol. Sci. 3, 486–492.
Anderson, S.F., Schlegel, B.P., Nakajima, T., Wolpin, E.S., and Parvin, J.D. (1998). BRCA1 protein is linked to the RNA polymerase II holoenzyme complex via RNA helicase A. Nat. Genet. 19, 254–256.
Andreakos, E., Sacre, S., Foxwell, B.M., and Feldmann, M. (2005). The toll-like receptor-nuclear factor kappaB pathway in rheumatoid arthritis. Front. Biosci. 10, 2478–2488.
Bandi, N., Zbinden, S., Gugger, M., Arnold, M., Kocher, V., Hasan, L., Kappeler, A., Brunner, T., and Vassella, E. (2009). miR-15a and miR-16 are implicated in cell cycle regulation in a Rbdependent manner and are frequently deleted or down-regulated in non-small cell lung cancer. Cancer Res. 69, 5553–5559.
Bao, B., Ali, S., Banerjee, S., Wang, Z., Logna, F., Azmi, A.S., Kong, D., Ahmad, A., Li, Y., Padhye, S., et al. (2012). Curcumin analogue CDF inhibits pancreatic tumor growth by switching on suppressor microRNAs and attenuating EZH2 expression. Cancer Res. 72, 335–345.
Bhaumik, D., Scott, G.K., Schokrpur, S., Patil, C.K., Campisi, J., and Benz, C.C. (2008). Expression of microRNA-146 suppresses NF-kappaB activity with reduction of metastatic potential in breast cancer cells. Oncogene 27, 5643–5647.
Castellano, L., Giamas, G., Jacob, J., Coombes, R.C., Lucchesi, W., Thiruchelvam, P., Barton, G., Jiao, L.R., Wait, R., Waxman, J., et al. (2009). The estrogen receptor-alpha-induced microRNA signature regulates itself and its transcriptional response. Proc. Natl. Acad. Sci. USA 106, 15732–15737.
Chang, S., Wang, R.H., Akagi, K., Kim, K.A., Martin, B.K., Cavallone, L., Haines, D.C., Basik, M., Mai, P., Poggi, E., et al. (2011). Tumor suppressor BRCA1 epigenetically controls oncogenic microRNA-155. Nat. Med. 17, 1275–1282.
Cimmino, A., Calin, G.A., Fabbri, M., Iorio, M.V., Ferracin, M., Shimizu, M., Wojcik, S.E., Aqeilan, R.I., Zupo, S., Dono, M., et al. (2005). miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc. Natl. Acad. Sci. USA 102, 13944–13949.
Cittelly, D.M., Das, P.M., Salvo, V.A., Fonseca, J.P., Burow, M.E., and Jones, F.E. (2010). Oncogenic HER2{Delta}16 suppresses miR-15a/16 and deregulates BCL-2 to promote endocrine resistance of breast tumors. Carcinogenesis 31, 2049–2057.
Cubillos-Ruiz, J.R., Baird, J.R., Tesone, A.J., Rutkowski, M.R., Scarlett, U.K., Camposeco-Jacobs, A.L., Anadon-Arnillas, J., Harwood, N.M., Korc, M., Fiering, S.N., et al. (2012). Reprogramming tumor-associated dendritic cells in vivo using miRNA mimetics triggers protective immunity against ovarian cancer. Cancer Res. 72, 1683–1693.
Easton, D.F., Ford, D., and Bishop, D.T. (1995). Breast and ovarian cancer incidence in BRCA1-mutation carriers. Breast Cancer Linkage Consortium. Am. J. Hum. Genet. 56, 265–271.
Fackenthal, J.D., and Olopade, O.I. (2007). Breast cancer risk associated with BRCA1 and BRCA2 in diverse populations. Nat. Rev. Cancer 7, 937–948.
Fong, P.C., Boss, D.S., Yap, T.A., Tutt, A., Wu, P., Mergui-Roelvink, M., Mortimer, P., Swaisland, H., Lau, A., O’Connor, M.J., et al. (2009). Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N. Engl. J. Med. 361, 123–134.
Friedman, R.C., Farh, K.K., Burge, C.B., and Bartel, D.P. (2009). Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 19, 92–105.
Garcia, A.I., Buisson, M., Bertrand, P., Rimokh, R., Rouleau, E., Lopez, B.S., Lidereau, R., Mikaelian, I., and Mazoyer, S. (2011a). Down-regulation of BRCA1 expression by miR-146a and miR-146b-5p in triple negative sporadic breast cancers. EMBO Mol. Med. 3, 279–290.
Garcia, A.I., Cox, D.G., Barjhoux, L., Verny-Pierre, C., Barnes, D., Antoniou, A.C., Stoppa-Lyonnet, D., Sinilnikova, O.M., and Mazoyer, S. (2011b). The rs2910164:G>C SNP in the MIR146A gene is not associated with breast cancer risk in BRCA1 and BRCA2 mutation carriers. Hum. Mutat. 32, 1004–1007.
Gelmon, K.A., Tischkowitz, M., Mackay, H., Swenerton, K., Robidoux, A., Tonkin, K., Hirte, H., Huntsman, D., Clemons, M., Gilks, B., et al. (2011). Olaparib in patients with recurrent high-grade serous or poorly differentiated ovarian carcinoma or triplenegative breast cancer: a phase 2, multicentre, open-label, nonrandomised study. Lancet Oncol. 12, 852–861.
Gonzalez, M.E., Li, X., Toy, K., DuPrie, M., Ventura, A.C., Banerjee, M., Ljungman, M., Merajver, S.D., and Kleer, C.G. (2009). Downregulation of EZH2 decreases growth of estrogen receptornegative invasive breast carcinoma and requires BRCA1. Oncogene 28, 843–853.
Gorski, J.J., Savage, K.I., Mulligan, J.M., McDade, S.S., Blayney, J.K., Ge, Z., and Harkin, D.P. (2011). Profiling of the BRCA1 transcriptome through microarray and ChIP-chip analysis. Nucleic Acids Res. 39, 9536–9548.
Gregory, P.A., Bert, A.G., Paterson, E.L., Barry, S.C., Tsykin, A., Farshid, G., Vadas, M.A., Khew-Goodall, Y., and Goodall, G.J. (2008). The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat. Cell Biol. 10, 593–601.
Griffiths-Jones, S., Saini, H.K., van Dongen, S., and Enright, A.J. (2008). miRBase: tools for microRNA genomics. Nucleic Acids Res. 36, D154–158.
Harkin, D.P., Bean, J.M., Miklos, D., Song, Y.H., Truong, V.B., Englert, C., Christians, F.C., Ellisen, L.W., Maheswaran, S., Oliner, J.D., et al. (1999). Induction of GADD45 and JNK/SAPKdependent apoptosis following inducible expression of BRCA1. Cell 97, 575–586.
Heyn, H., Engelmann, M., Schreek, S., Ahrens, P., Lehmann, U., Kreipe, H., Schlegelberger, B., and Beger, C. (2011). MicroRNA miR-335 is crucial for the BRCA1 regulatory cascade in breast cancer development. Int. J. Cancer 129, 2797–2806.
Huen, M.S., Sy, S.M., and Chen, J. (2010). BRCA1 and its toolbox for the maintenance of genome integrity. Nat. Rev. Mol. Cell Biol. 11, 138–148.
Iorio, M.V., Casalini, P., Piovan, C., Di Leva, G., Merlo, A., Triulzi, T., Menard, S., Croce, C.M., and Tagliabue, E. (2009). microRNA-205 regulates HER3 in human breast cancer. Cancer Res. 69, 2195–2200.
Jiang, S., Zhang, H.W., Lu, M.H., He, X.H., Li, Y., Gu, H., Liu, M.F., and Wang, E.D. (2010). MicroRNA-155 functions as an OncomiR in breast cancer by targeting the suppressor of cytokine signaling 1 gene. Cancer Res. 70, 3119–3127.
John, B., Enright, A.J., Aravin, A., Tuschl, T., Sander, C., and Marks, D.S. (2004). Human microRNA targets. PLoS Biol. 2, e363.
Jones, K.L., and Buzdar, A.U. (2004). A review of adjuvant hormo nal therapy in breast cancer. Endocr. Relat. Cancer 11, 391–406.
Kong, W., He, L., Coppola, M., Guo, J., Esposito, N.N., Coppola, D., and Cheng, J.Q. (2010). MicroRNA-155 regulates cell survival, growth, and chemosensitivity by targeting FOXO3a in breast cancer. J. Biol. Chem. 285, 17869–17879.
Kurowska-Stolarska, M., Alivernini, S., Ballantine, L.E., Asquith, D.L., Millar, N.L., Gilchrist, D.S., Reilly, J., Ierna, M., Fraser, A.R., Stolarski, B., et al. (2011). MicroRNA-155 as a proinflammatory regulator in clinical and experimental arthritis. Proc. Natl. Acad. Sci. USA 108, 11193–11198.
Lehmann, U., Hasemeier, B., Romermann, D., Muller, M., Langer, F., and Kreipe, H. (2007). Epigenetic inactivation of microRNA genes in mammary carcinoma. Verh. Dtsch. Ges. Pathol. 91, 214–220.
Lewis, B.P., Burge, C.B., and Bartel, D.P. (2005). Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120, 15–20.
Li, D., Wang, Q., Liu, C., Duan, H., Zeng, X., Zhang, B., Li, X., Zhao, J., Tang, S., Li, Z., et al. (2012). Aberrant expression of miR-638 contributes to benzo(a) pyrene-induced human cell transformation. Toxicol. Sci. 125, 382–391.
Lian, H., Wang, L., and Zhang, J. (2012). Increased risk of breast cancer associated with CC genotype of Has-miR-146a Rs29 10164 polymorphism in Europeans. PLoS One 7, e31615.
Liu, Z., Lu, C.L., Cui, L.P., Hu, Y.L., Yu, Q., Jiang, Y., Ma, T., Jiao, D.K., Wang, D., and Jia, C.Y. (2012). MicroRNA-146a modulates TGF-beta1-induced phenotypic differentiation in human dermal fibroblasts by targeting SMAD4. Arch. Dermatol. Res. 304, 195–202.
Lossner, C., Meier, J., Warnken, U., Rogers, M.A., Lichter, P., Pscherer, A., and Schnolzer, M. (2011). Quantitative proteomics identify novel miR-155 target proteins. PLoS One 6, e22146.
Majid, S., Saini, S., Dar, A.A., Hirata, H., Shahryari, V., Tanaka, Y., Yamamura, S., Ueno, K., Zaman, M.S., Singh, K., et al. (2011). MicroRNA-205 inhibits Src-mediated oncogenic pathways in renal cancer. Cancer Res. 71, 2611–2621.
Martinez-Nunez, R.T., Louafi, F., Friedmann, P.S., and Sanchez-Elsner, T. (2009). MicroRNA-155 modulates the pathogen binding ability of dendritic cells (DCs) by down-regulation of DCspecific intercellular adhesion molecule-3 grabbing non-integrin (DC-SIGN). J. Biol. Chem. 284, 16334–16342.
Miki, Y., Swensen, J., Shattuck-Eidens, D., Futreal, P.A., Harshman, K., Tavtigian, S., Liu, Q., Cochran, C., Bennett, L.M., Ding, W., et al. (1994). A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 266, 66–71.
Moskwa, P., Buffa, F.M., Pan, Y., Panchakshari, R., Gottipati, P., Muschel, R.J., Beech, J., Kulshrestha, R., Abdelmohsen, K., Weinstock, D.M., et al. (2011). miR-182-mediated downregulation of BRCA1 impacts DNA repair and sensitivity to PARP inhibitors. Mol. Cell 41, 210–220.
Murchison, E.P., and Hannon, G.J. (2004). miRNAs on the move; miRNA biogenesis and the RNAi machinery. Curr. Opin. Cell Biol. 16, 223–229.
Nakasa, T., Shibuya, H., Nagata, Y., Niimoto, T., and Ochi, M. (2011). The inhibitory effect of microRNA-146a expression on bone destruction in collagen-induced arthritis. Arthritis Rheum. 63, 1582–1590.
Nelson, P., Kiriakidou, M., Sharma, A., Maniataki, E., and Mourelatos, Z. (2003). The microRNA world: small is mighty. Trends Biochem. Sci. 28, 534–540.
Nicoloso, M.S., Sun, H., Spizzo, R., Kim, H., Wickramasinghe, P., Shimizu, M., Wojcik, S.E., Ferdin, J., Kunej, T., Xiao, L., et al. (2010). Single-nucleotide polymorphisms inside microRNA target sites influence tumor susceptibility. Cancer Res. 70, 2789–2798.
Ohta, T., Sato, K., and Wu, W. (2011). The BRCA1 ubiquitin ligase and homologous recombination repair. FEBS Lett. 585, 2836–2844.
Pelletier, C., Speed, W.C., Paranjape, T., Keane, K., Blitzblau, R., Hollestelle, A., Safavi, K., van den Ouweland, A., Zelterman, D., Slack, F.J., et al. (2011). Rare BRCA1 haplotypes including 3’UTR SNPs associated with breast cancer risk. Cell Cycle 10, 90–99.
Pongsavee, M., Yamkamon, V., Dakeng, S., O-charoenrat, P., Smith, D.R., Saunders, G.F., and Patmasiriwat, P. (2009). The BRCA1 3′-UTR: 5711+421T/T_5711+1286T/T genotype is a possible breast and ovarian cancer risk factor. Genet. Test Mol. Biomarkers 113, 307–317.
Pullamsetti, S.S., Doebele, C., Fischer, A., Savai, R., Kojonazarov, B., Dahal, B.K., Ghofrani, H.A., Weissmann, N., Grimminger, F., Bonauer, A., et al. (2012). Inhibition of microRNA-17 improves lung and heart function in experimental pulmonary hypertension. Am. J. Respir. Crit. Care Med. 185, 409–419.
Puppe, J., Drost, R., Liu, X., Joosse, S.A., Evers, B., Cornelissen-Steijger, P., Nederlof, P., Yu, Q., Jonkers, J., van Lohuizen, M., et al. (2009). BRCA1-deficient mammary tumor cells are dependent on EZH2 expression and sensitive to polycomb repressive complex 2-inhibitor 3-deazaneplanocin A. Breast Cancer Res. 11, R63.
Ramus, S.J., and Gayther, S.A. (2009). The contribution of BRCA1 and BRCA2 to ovarian cancer. Mol. Oncol. 3, 138–150.
Rodriguez, A., Vigorito, E., Clare, S., Warren, M.V., Couttet, P., Soond, D.R., van Dongen, S., Grocock, R.J., Das, P.P., Miska, E.A., et al. (2007). Requirement of bic/microRNA-155 for normal immune function. Science 316, 608–611.
Sarasin-Filipowicz, M., Krol, J., Markiewicz, I., Heim, M.H., and Filipowicz, W. (2009). Decreased levels of microRNA miR-122 in individuals with hepatitis C responding poorly to interferon therapy. Nat. Med. 15, 31–33.
Scully, R., Anderson, S.F., Chao, D.M., Wei, W., Ye, L., Young, R.A., Livingston, D.M., and Parvin, J.D. (1997). BRCA1 is a component of the RNA polymerase II holoenzyme. Proc. Natl. Acad. Sci. USA 94, 5605–5610.
Segura, M.F., Hanniford, D., Menendez, S., Reavie, L., Zou, X., Alvarez-Diaz, S., Zakrzewski, J., Blochin, E., Rose, A., Bogunovic, D., et al. (2009). Aberrant miR-182 expression promotes melanoma metastasis by repressing FOXO3 and microphthalmia-associated transcription factor. Proc. Natl. Acad. Sci. USA 106, 1814–1819.
Shen, J., Ambrosone, C.B., DiCioccio, R.A., Odunsi, K., Lele, S.B., and Zhao, H. (2008). A functional polymorphism in the miR-146a gene and age of familial breast/ovarian cancer diagnosis. Carcinogenesis 29, 1963–1966.
Shen, J., Ambrosone, C.B., and Zhao, H. (2009). Novel genetic variants in microRNA genes and familial breast cancer. Int. J. Cancer 124, 1178–1182.
Shukla, V., Coumoul, X., Lahusen, T., Wang, R.H., Xu, X., Vassilopoulos, A., Xiao, C., Lee, M.H., Man, Y.G., Ouchi, M., et al. (2010). BRCA1 affects global DNA methylation through regulation of DNMT1. Cell Res. 20, 1201–1215.
Simon, J.A., and Lange, C.A. (2008). Roles of the EZH2 histone methyltransferase in cancer epigenetics. Mutat. Res. 647, 21–29.
Somasundaram, K., Zhang, H., Zeng, Y.X., Houvras, Y., Peng, Y., Wu, G.S., Licht, J.D., Weber, B.L., and El-Deiry, W.S. (1997). Arrest of the cell cycle by the tumour-suppressor BRCA1 requires the CDK-inhibitor p21WAF1/CiP1. Nature 389, 187–190.
Stittrich, A.B., Haftmann, C., Sgouroudis, E., Kuhl, A.A., Hegazy, A.N., Panse, I., Riedel, R., Flossdorf, M., Dong, J., Fuhrmann, F., et al. (2010). The microRNA miR-182 is induced by IL-2 and promotes clonal expansion of activated helper T lymphocytes. Nat. Immunol. 11, 1057–1062.
Suzuki, H.I., Yamagata, K., Sugimoto, K., Iwamoto, T., Kato, S., and Miyazono, K. (2009). Modulation of microRNA processing by p53. Nature 460, 529–533.
Tanic, M., Zajac, M., Gomez-Lopez, G., Benitez, J., and Martinez-Delgado, B. (2011). Integration of BRCA1-mediated miRNA and mRNA profiles reveals microRNA regulation of TRAF2 and NFkappaB pathway. Breast Cancer Res. Treat. s10549-011-1905-4.
Tavazoie, S.F., Alarcon, C., Oskarsson, T., Padua, D., Wang, Q., Bos, P.D., Gerald, W.L., and Massague, J. (2008). Endogenous human microRNAs that suppress breast cancer metastasis. Nature 451, 147–152.
Thai, T.H., Calado, D.P., Casola, S., Ansel, K.M., Xiao, C., Xue, Y., Murphy, A., Frendewey, D., Valenzuela, D., Kutok, J.L., et al. (2007). Regulation of the germinal center response by microRNA-155. Science 316, 604–608.
Tili, E., Croce, C.M., and Michaille, J.J. (2009). miR-155: on the crosstalk between inflammation and cancer. Int. Rev. Immunol. 28, 264–284.
Tsuruta, T., Kozaki, K., Uesugi, A., Furuta, M., Hirasawa, A., Imoto, I., Susumu, N., Aoki, D., and Inazawa, J. (2011). miR-152 is a tumor suppressor microRNA that is silenced by DNA hyper methylation in endometrial cancer. Cancer Res. 71, 6450–6462.
Turcatel, G., Rubin, N., El-Hashash, A., and Warburton, D. (2012). MIR-99a and MIR-99b modulate TGF-beta induced epithelial to mesenchymal plasticity in normal murine mammary gland cells. PLoS One 7, e31032.
Turner, M., and Vigorito, E. (2008). Regulation of B- and T-cell differentiation by a single microRNA. Biochem. Soc. Trans. 36, 531–533.
Wang, Q., Zhang, H., Kajino, K., and Greene, M.I. (1998). BRCA1 binds c-Myc and inhibits its transcriptional and transforming activity in cells. Oncogene 17, 1939–1948.
Wang, D., Huang, J., and Hu, Z. (2012). RNA helicase DDX5 regulates microRNA expression and contributes to cytoskeletal reorganization in basal breast cancer cells. Mol. Cell. Proteomics 11, M111. 011932.
Wu, H., Zhu, S., and Mo, Y.Y. (2009). Suppression of cell growth and invasion by miR-205 in breast cancer. Cell Res. 19, 439–448.
Xiao, B., Zhu, E.D., Li, N., Lu, D.S., Li, W., Li, B.S., Zhao, Y.L., Mao, X.H., Guo, G., Yu, P.W., et al. (2012). Increased miR-146a in gastric cancer directly targets SMAD4 and is involved in modulating cell proliferation and apoptosis. Oncol. Rep. 27, 559–566.
Xu, B., Wang, N., Wang, X., Tong, N., Shao, N., Tao, J., Li, P., Niu, X., Feng, N., Zhang, L., et al. (2011). MiR-146a suppresses tumor growth and progression by targeting EGFR pathway and in a p-ERK-dependent manner in castration-resistant prostate cancer. Prostate 72, 1171–1178.
Yang, J., Cao, Y., Sun, J., and Zhang, Y. (2010). Curcumin reduces the expression of Bcl-2 by upregulating miR-15a and miR-16 in MCF-7 cells. Med. Oncol. 27, 1114–1118.
Yu, X., Wu, L.C., Bowcock, A.M., Aronheim, A., and Baer, R. (1998). The C-terminal (BRCT). domains of BRCA1 interact in vivo with CtIP, a protein implicated in the CtBP pathway of transcriptional repression. J. Biol. Chem. 273, 25388–25392.
Yu, Z., Wang, C., Wang, M., Li, Z., Casimiro, M.C., Liu, M., Wu, K., Whittle, J., Ju, X., Hyslop, T., et al. (2008). A cyclin D1/microRNA 17/20 regulatory feedback loop in control of breast cancer cell proliferation. J. Cell Biol. 182, 509–517.
Zhao, J.L., Rao, D.S., Boldin, M.P., Taganov, K.D., O’Connell, R.M., and Baltimore, D. (2011). NF-kappaB dysregulation in microRNA-146a-deficient mice drives the development of myeloid malignancies. Proc. Natl. Acad. Sci. USA 108, 9184–9189.
Zhu, J.Y., Pfuhl, T., Motsch, N., Barth, S., Nicholls, J., Grasser, F., and Meister, G. (2009). Identification of novel Epstein-Barr virus microRNA genes from nasopharyngeal carcinomas. J. Virol. 83, 3333–3341.
Zilahi, E., Tarr, T., Papp, G., Griger, Z., Sipka, S., and Zeher, M. (2012). Increased microRNA-146a/b, TRAF6 gene and decreased IRAK1 gene expressions in the peripheral mononuclear cells of patients with Sjogren’s syndrome. Immunol. Lett. 141, 165–168.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Chang, S., Sharan, S.K. BRCA1 and MicroRNAs: Emerging networks and potential therapeutic targets. Mol Cells 34, 425–432 (2012). https://doi.org/10.1007/s10059-012-0118-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-012-0118-y