Abstract
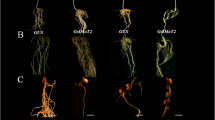
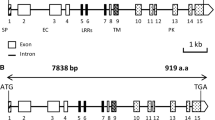
Development of symbiotic root nodules in legumes involves the induction and repression of numerous genes in conjunction with changes in the level of phytohormones. We have isolated several genes that exhibit differential expression patterns during the development of soybean nodules. One of such genes, which were repressed in mature nodules, was identified as a putative aldo/keto reductase and thus named Glycine max aldo/keto reductase 1 (GmAKR1). GmAKR1 appears to be a close relative of a yeast aldo/keto reductase YakC whose in vivo substrate has not been identified yet. The expression of GmAKR1 in soybean showed a root-specific expression pattern and inducibility by a synthetic auxin analogue 2,4-D, which appeared to be corroborated by presence of the root-specific element and the stress-response element in the promoter region. In addition, constitutive overexpression of GmAKR1 in transgenic soybean hairy roots inhibited nodule development, which suggests that it plays a negative role in the regulation of nodule development. One of the Arabidopsis orthologues of GmAKR1 is the ARF-GAP domain 2 protein, which is a potential negative regulator of vesicle trafficking; therefore GmAKR1 may have a similar function in the roots and nodules of legume plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Acevedo-Hernandez, G.J., Leon, P., and Herrera-Estrella, L.R. (2005). Sugar and ABA responsiveness of a minimal RBCS light-responsive unit is mediated by direct binding of ABI4. Plant J. 43, 506–519.
Boot, K., Van Der Zaal, B.J., Velterop, J., Quint, A., Mennes, A.M., Hooykaas, P., and Libbenga, K.R. (1993). Further characterization of expression of auxin-induced genes in Tobacco (Nicotiana tabacum) cell-suspension cultures. Plant Physiol. 102, 513–520.
Chakravarthy, S., Tuori, R.P., D’Ascenzo, M.D., Fobert, P.R., Despres, C., and Martin, G.B. (2003). The tomato transcription factor Pti4 regulates defense-related gene expression via GCC box and non-GCC box cis elements. Plant Cell 15, 3033–3050.
Charon, C., Sousa, C., Crespi, M., and Kondorosi, A. (1999). Alteration of enod40 expression modifies Medicago truncatula root nodule development induced by Sinorhizobium meliloti. Plant Cell 11, 1953–1966.
Chen, P.W., Chiang, C.M., Tseng, T.H., and Yu, S.M. (2006). Interaction between rice MYBGA and the gibberellin response element controls tissue-specific sugar sensitivity of alpha-amylase genes. Plant Cell 18, 2326–2340.
de Billy, F., Grosjean, C., May, S., Bennett, M., and Cullimore, J.V. (2001). Expression studies on AUX1-like genes in Medicago truncatula suggest that auxin is required at two steps in early nodule development. Mol. Plant Microbe Interact. 14, 267–277.
Fehlberg, V., Vieweg, M.F., Dohmann, E.M., Hohnjec, N., Puhler, A., Perlick, A.M., and Kuster, H. (2005). The promoter of the leghaemoglobin gene VfLb29: functional analysis and identification of modules necessary for its activation in the infected cells of root nodules and in the arbuscule-containing cells of my-corrhizal roots. J. Exp. Bot. 56, 799–806.
Foucher, F., and Kondorosi, E. (2000). Cell cycle regulation in the course of nodule organogenesis in Medicago. Plant Mol. Biol. 43, 773–786.
Guilfoyle, T., Hagen, G., Ulmasov, T., and Murfett, J. (1998). How does auxin turn on genes? Plant Physiol. 118, 341–347.
Higo, K., Ugawa, Y., Iwamoto, M., and Korenaga, T. (1999). Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res. 27, 297–300.
Hyndman, D., Bauman, D.R., Heredia, V.V., and Penning, T.M. (2003). The aldo-keto reductase superfamily homepage. Chem. Biol. Interact. 143–144, 621–631.
Jez, J.M., Bennett, M.J., Schlegel, B.P., Lewis, M., and Penning, T.M. (1997). Comparative anatomy of the aldo-keto reductase superfamily. Biochem. J. 326, 625–636.
Jin, Y., and Penning, T.M. (2007). Aldo-keto reductases and bioactivation/detoxication. Annu. Rev. Pharmacol. Toxicol. 47, 263–292.
Johnson, C, Glover, G., and Arias, J. (2001). Regulation of DNA binding and trans-activation by a xenobiotic stress-activated plant transcription factor. J. Biol. Chem. 276, 172–178.
Katagiri, F., Lam, E., and Chua, N.H. (1989). Two tobacco DNA-binding proteins with homology to the nuclear factor CREB. Nature 340, 727–730.
Kim, Y.J., Kwak, C.I., Gu, Y.Y., Hwang, I.T., and Chun, J.Y. (2004). Annealing control primer system for identification of differentially expressed genes on agarose gels. Biotechniques 36, 424–426, 428, 430 passim.
Kim, Y.K., Son, O., Kim, M.R., Nam, K.H., Kim, G.T., Lee, M.S., Choi, S.Y., and Cheon, C.I. (2007a). ATHB23, an Arabidopsis class I homeodomain-leucine zipper gene, is expressed in the adaxial region of young leaves. Plant Cell Rep. 26, 1179–1185.
Kim, H.B., Lee, H., Oh, C.J., Lee, N.H., and An, C.S. (2007b). Expression of EuNOD-ARPI encoding auxin-repressed protein homolog is upregulated by auxin and localized to the fixation zone in root nodules of Elaeagnus umbellata. Mol. Cells 23, 115–121.
Klinedinst, S., Pascuzzi, P., Redman, J., Desai, M., and Arias, J. (2000). A xenobiotic-stress-activated transcription factor and its cognate target genes are preferentially expressed in root tip meristems. Plant Mol. Biol. 42, 679–688.
Lee, M.Y., Shin, K.H., Kim, Y.K., Suh, J.Y., Gu, YY., Kim, M.R., Hur, Y.S., Son, O., Kim, J.S., Song, E., et al. (2005). Induction of thioredoxin is required for nodule development to reduce reactive oxygen species levels in soybean roots. Plant Physiol. 139, 1881–1889.
Lescot, M., Dehais, P., Thijs, G., Marchal, K., Moreau, Y., Van de Peer, Y., Rouze, P., and Rombauts, S. (2002). PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res. 30, 325–327.
Li, Y., Liu, Z.B., Shi, X., Hagen, G., and Guilfoyle, T.J. (1994). An auxin-inducible element in soybean SAUR promoters. Plant Physiol. 106, 37–43.
Martin, H.J., Breyer-Pfaff, U., Wsol, V., Venz, S., Block, S., and Maser, E. (2006). Purification and characterization of akr1b10 from human liver: role in carbonyl reduction of xenobiotics. Drug Metab. Dispos. 34, 464–470.
Mathesius, U., Schlaman, H.R., Spaink, HP., Of Sautter, C., Rolfe, B.G., and Djordjevic, M.A. (1998). Auxin transport inhibition precedes root nodule formation in white clover roots and is regulated by flavonoids and derivatives of chitin oligosaccharides. Plant J. 14, 23–34.
Morita, T., Huruta, T., Ashiuchi, M., and Yagi, T. (2002). Characterization of recombinant YakC of Schizosaccharomyces pombe showing YakC defines a new family of aldo-keto reductases. J. Biochem. 132, 635–641.
Niggeweg, R., Thurow, C., Kegler, C., and Gatz, C. (2000). Tobacco transcription factor TGA2.2 is the main component of as-1-binding factor ASF-1 and is involved in salicylic acid- and auxin-inducible expression of as-1-containing target promoters. J. Biol. Chem. 275, 19897–19905.
Nikiforova, V., Freitag, J., Kempa, S., Adamik, M., Hesse, H., and Hoefgen, R. (2003). Transcriptome analysis of sulfur depletion in Arabidopsis thaliana: interlacing of biosynthetic pathways provides response specificity. Plant J. 33, 633–650.
Oh, H.S., Son, O., Chun, J.Y., Stacey, G., Lee, M.S., Min, K.H., Song, E.S., and Cheon, C.I. (2001). The Bradyrhizobium japoni-cum hsfA gene exhibits a unique developmental expression pattern in cowpea nodules. Mol. Plant Microbe Interact. 14, 1286–1292.
Oldroyd, G.E., and Downie, J.A. (2008). Coordinating nodule morphogenesis with rhizobial infection in legumes. Annu. Rev. Plant Biol. 59, 519–546.
Peer, W.A., and Murphy, A.S. (2007). Flavonoids and auxin transport: modulators or regulators? Trends Plant Sci. 12, 556–563.
Rombauts, S., Dehais, P., Van Montagu, M., and Rouze, P. (1999). PlantCARE, a plant c/s-acting regulatory element database. Nucleic Acids Res. 27, 295–296.
Sakai, T., Takahashi, Y., and Nagata, T. (1996). Analysis of the promoter of the auxin-inducible gene, parC, of tobacco. Plant Cell Physiol. 37, 906–991.
Sieburth, LE., Muday, G.K., King, E.J., Benton, G., Kim, S., Metcalf, K.E., Meyers, L, Seamen, E., and Van Norman, J.M. (2006). SCARFACE encodes an ARF-GAP that is required for normal auxin efflux and vein patterning in Arabidopsis. Plant Cell 18, 1396–1411.
Stacey, G., Libault, M., Brechenmacher, L., Wan, J., and May, G.D. (2006). Genetics and functional genomics of legume nodulation. Curr. Opin. Plant Biol. 9, 110–121.
Subramanian, S., Stacey, G., and Yu, O. (2007). Distinct, crucial roles of flavonoids during legume nodulation. Trends Plant Sci. 12, 282–285.
Tiwari, S.B., Hagen, G., and Guilfoyle, T. (2003). The roles of auxin response factor domains in auxin-responsive transcription. Plant Cell 15, 533–543.
Ulmasov, T., Hagen, G., and Guilfoyle, T. (1994). The ocs element in the soybean GH2/4 promoter is activated by both active and inactive auxin and salicylic acid analogues. Plant Mol. Biol. 26, 1055–1064.
Ulmasov, T., Liu, Z.B., Hagen, G., and Guilfoyle, T.J. (1995). Composite structure of auxin response elements. Plant Cell 7, 1611–1623.
Ulmasov, T., Murfett, J., Hagen, G., and Guilfoyle, T.J. (1997). Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin response elements. Plant Cell 9, 1963–1971.
Wasson, A.P., Pellerone, F.I., and Mathesius, U. (2006). Silencing the flavonoid pathway in Medicago truncatula inhibits root nodule formation and prevents auxin transport regulation by rhizobia. Plant Cell 18, 1617–1629.
Xiang, C., Miao, Z.H., and Lam, E. (1996). Coordinated activation of as-1-type elements and a tobacco glutathione S-transferase gene by auxins, salicylic acid, methyl-jasmonate and hydrogen peroxide. Plant Mol. Biol. 32, 415–426.
Zhuang, X., Jiang, J., Li, J., Ma, Q., Xu, Y., Xue, Y., Xu, Z., and Chong, K. (2006). Over-expression of OsAGAP, an ARF-GAP, interferes with auxin influx, vesicle trafficking and root development. Plant J. 48, 581–591.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Hur, YS., Shin, KH., Kim, S. et al. Overexpression of GmAKR1, a stress-induced aldo/keto reductase from soybean, retards nodule development. Mol Cells 27, 217–223 (2009). https://doi.org/10.1007/s10059-009-0027-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0027-x