Abstract
Climate change is causing fire regime shifts in ecosystems worldwide. Plant species with regeneration strategies strongly linked to a fire regime, such as obligate seeders, may be particularly threatened by these changes. It is unclear whether changes in fire regimes or the direct effects of climate change will be the dominant threats to obligate seeders in future. We investigated the relative importance of fire-related variables (fire return interval and fire severity) and environmental factors (climate and topography) on seedling establishment in the world’s tallest angiosperm, an obligate seeder, Eucalyptus regnans. Throughout its range, this species dominates the wet montane forests of south-eastern Australia and plays a keystone role in forest structure. Following major wildfires, we investigated seedling establishment in E. regnans within 1 year of fire as this is a critical stage in the regeneration niche of obligate seeders. Seedling presence and abundance were strongly related to the occurrence of fire but not to variation in fire severity (moderate vs. high severity). Seedling abundance increased with increasing fire return interval (range 26–300 years). First-year seedling establishment was also strongly associated with low temperatures and with high elevations, high precipitation and persistent soil water availability. Our results show that both climate and fire regimes are strong drivers of E. regnans seedling establishment. The predicted warming and drying of the climate might reduce the regeneration potential for some obligate seeders in future and these threats are likely to be compounded by changes in fire regimes, particularly increases in fire frequency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecosystems around the world are increasingly threatened by habitat loss, overexploitation, climate change, altered disturbance regimes, and synergies among these threats (Brook and others 2008; Driscoll and others 2012; Silvestrini and others 2011). Fires are becoming more frequent and severe in many ecosystems experiencing climate change (Liu and others 2010; Westerling and others 2011), increasing the risk of species extinction (Fisher and others 2009; Swab and others 2012). Assessments of the relative influence of climatic and fire regime variables are needed to understand what drives the distribution and abundance of species and how species might respond to future change (Bradstock and others 2012; Franklin 2010).
Among plant functional groups, obligate seeders are particularly vulnerable to decline under disrupted fire regimes because fire return intervals shorter than their maturation age, or greater than their lifespan, can cause local extinction (McCarthy and others 1999; Pausas and others 2004) and regional decline (Franklin and others 2005; Russell-Smith and others 2012). Altered fire regimes have been identified as the dominant threat to some obligate seeding species under future climate scenarios (Regan and others 2010; Swab and others 2012). In contrast, other studies have suggested that the direct effects of climate change (for example, increased temperature and decreased precipitation) may be more important than fire regime changes (Lawson and others 2010). Although it is clear that climatic and fire regime variables do not act in isolation (Lawson and others 2010; Swab and others 2012), understanding their relative and cumulative influence on plant species is necessary to predict the effects of global change (Fordham and others 2012; Keith and others 2008) and adapt management practices appropriately.
We conducted a landscape-scale study following major wildfires to answer the question: what is the relative importance of fire-related variables (fire return interval and fire severity) and environmental factors (climate and topography) on seedling establishment in the Australian forest obligate seeder Eucalyptus regnans F. Muell. (Myrtaceae)? We focused on seedling establishment within 1 year of fire because it is a critical stage in the life cycle of obligate seeders. Fire can kill adult plants and exhaust seed supply (Bradstock and others 2006) so quantifying the conditions promoting seedling establishment can help uncover mechanisms that mediate successful regeneration (for example, seed supply at the time of fire). Eucalyptus regnans is the tallest flowering plant in the world and the world’s tallest obligate seeder (Tng and others 2012; Wood and others 2010). Within its range in south-eastern Australia, the species dominates the wet montane forest ecosystem, but repeated disturbances and continual exploitation for pulp and timber have led to an extreme decline in its old-growth forest stage (Lindenmayer and others 2012a). It is currently unclear how E. regnans will respond to future global change because we have little empirical data on its current regeneration potential under different climatic and fire regime conditions. In the context of previous research on obligate seeders, our general aim was to expand our understanding of the relative influence of climatic and fire regime variables on regeneration in this plant functional group.
Methods
Study System
Our study took place in the Central Highlands region of Victoria, Australia (Figure 1), approximately 120 km north-east of Melbourne. Mean maximum temperatures throughout the region range from 12 to 27°C in summer and 2 to 15°C in winter, whereas mean minimum temperatures range from 8 to 13°C in summer and 1 to 6°C in winter (Mackey and others 2002). Mean annual precipitation ranges from 1,200 to 1,800 mm, depending strongly on elevation and distance from the coast, and there is a tendency for greater rainfall in winter than in summer (Mackey and others 2002).
In our study region, E. regnans (Mountain Ash) forest typically forms monotypic even-aged stands between 200 and 1,100 m above sea level (Boland and others 2006; Lindenmayer 2009). Characteristic understorey species in E. regnans forest include Pomaderris aspera, Acacia dealbata, Olearia argophylla, Cyathea australis, and Dicksonia antarctica, whereas common ground cover species include Polystichum proliferum, Pteridium esculentum, Tetrarrhena juncea, and Hydrocotyle hirta (Ashton 1976; Ashton and Attiwill 1994). Eucalyptus regnans individuals often achieve heights of 65 m, and trees over 100 m tall have been recorded (Ashton and Attiwill 1994; Tng and others 2012). Trees begin producing seed at 15–20 years of age (see Gill 1981) and may live for 350–500 years (Ashton 1981; Wood and others 2010). Eucalyptus regnans forest covers about 162,000 ha of the Central Highlands region (Stein, unpublished data), of which approximately 30% occurs in conservation reserves (DNRE 1998; Lindenmayer 2009). The remaining 70% is broadly designated for timber and pulp production, but special protection zones exclude harvesting from stream buffers, steep, or rocky terrain and important wildlife habitat such as old-growth forest (DNRE 1998; Lindenmayer 2009). However, salvage logging is often practiced in these areas following wildfire (Lindenmayer and Ough 2006).
Wildfire (also called unplanned fire or bushfire) is the most common natural disturbance in the study region and the majority of fire events occur in summer to early autumn (Lindenmayer 2009; Mackey and others 2002). Fire intensity in E. regnans forest can be extreme (35,000–100,000 kWm−1) and, at such high intensities, fires usually kill trees resulting in stand-replacement from seedlings (Gill and Catling 2002). Variation in the intensity of any particular fire means that approximately half of the trees in a landscape will survive the fire (Mackey and others 2002). The mean fire return interval in E. regnans forest has been estimated as 75–150 years for tree-killing fires and 37–75 years for all fires (McCarthy and others 1999). Approximately 79% of the Central Highlands region was burned by wildfire in 1939 (Land Conservation Council 1994) and major wildfires have since occurred in 1983 and 2009 (Figure 1). The “Black Saturday” fires of February 2009 burned approximately 59,000 ha of E. regnans forest in the region (Stein, unpublished data).
Study Sites and Seedling Surveys
Following the fires of 2009, we surveyed E. regnans seedlings at 123 one-hectare sites throughout the study region spanning an elevational gradient of 327–1,054 m above sea level (Figure 1). Our sites were a subset of those established in 1997 for a long-term forest ecology and monitoring project (Lindenmayer and others 2003) that included only sites dominated by E. regnans (Lindenmayer and others 2000). Our sites included a range of fire return interval and burn categories (Table 1). As with many “natural experiments,” achieving a balanced design was not possible given the availability of sampling locations following the unexpected wildfires of 2009 (Driscoll and others 2010). However, a key strength of our study was the ability to examine the effects of this rare disturbance event using our controlled, landscape-scale sampling approach (Lindenmayer and others 2010; Romme and others 2011).
We grouped our sites into three fire return interval (FRI) categories based on stand age at the time of the 2009 fires (Lindenmayer and others 2000) (Table 1). We used stand age to estimate potential effects of fire return interval because average measures over successive fires were not available given the long temporal scale of fire frequency and available historical data. Sites in the “long” FRI category (N = 20) were established following fires that occurred at various times between the early 1700s and 1908 (old-growth forest). In the “intermediate” FRI category (N = 80), 94% of sites were established after the extensive fires of 1939, with the remainder established after fires in 1926 and 1932. In the “short” FRI category (N = 23), some sites were established following clearfell and slash burn logging or post-fire salvage logging between 1974 and 1983 (10 sites), 1955 (1 site), 1990 (1 site), and 1998 (1 site). The remaining 10 sites established naturally following the 1983 wildfires. Given the small number of sites affected by logging it was not possible to separate the effects of fire and mechanical disturbance but we consider this issue in the interpretation of our results. Some sites were multi-aged and we classified those according to the predominant age class in the stand.
Between April 15th, 2009 and November 25th, 2009, we surveyed all sites to assess fire severity and record the presence and abundance of E. regnans seedlings. We grouped sites into three burn categories (Table 1): unburned (N = 71); moderate severity (N = 39) where the majority of the understorey and midstorey was scorched but the canopy remained unburned; and high severity (N = 13) where the understorey, midstorey, and canopy had all been scorched or completely consumed. This burn category variable allowed us to examine effects of the occurrence of fire (unburned compared to the two burned categories) which is known to initiate regeneration in E. regnans, and to examine the effects of fire severity (moderate compared to high severity), which has largely unknown effects on E. regnans regeneration (Ashton and Martin 1996). At each site, three 1 × 1 m plots were established 40 m apart along a central transect and the number of E. regnans seedlings in each plot was recorded. Seedlings included all individuals visible on the surface and over 99% were less than 10 cm high.
We used spatial modeling software to derive values for climatic and topographic variables for each site. We included elevation in our suite of environmental variables because it governs the distribution of E. regnans forest in relation to other vegetation types (Lindenmayer 2009) and because it can be related to factors other than climate (for example, soil chemistry or herbivory, Moen and others 2008) that affect plant regeneration. Elevation was derived from a 20-m resolution digital elevation model (DEM) created in ANUDEM (Hutchinson 2011). Variation in slope can affect seedling establishment because of its influence on soil nutrients (Metz 2012). We derived values for slope (1–31° of inclination) from neighboring cell geometry in the DEM (Hutchinson, unpublished software). We included three variables to incorporate potential effects of variation in moisture limitation and heat stress which have widely documented influences on seedling establishment (for example, Bauweraerts and others 2013). A topographic wetness index (TWI) (Beven and Kirkby 1979) was calculated as a measure of available soil moisture (−4.34 to 6.95). Annual mean temperature (8–12°C) and precipitation (1,293–1,776 mm/y) were estimated from monthly mean climate surfaces from 1921 to 1995 for each cell in the DEM using the BIOCLIM module of the ANUCLIM software (Xu and Hutchinson 2011).
Analysis
To analyze the effect of fire return interval, burn category, and environmental variables on E. regnans seedling abundance, we used a generalized linear model (GLM) framework. Seedling abundance data from the three survey plots at each site were combined, giving a total count for each site as our response variable. We excluded one site (long FRI, high severity fire) from the analysis because its seedling abundance was 19 times higher than the mean of all sites with seedlings (mean 116.28). There was a high proportion of zeros in our seedling data (60%) so we used a hurdle model which is a GLM extension appropriate for count data with more zeros than expected by the Poisson distribution (Welsh and others 1996). Hurdle models include two parts: a “zero” part to model the data as presence (all positive values = 1) or absence (0) (that is, a binomial GLM with a logit link), and a truncated “count” part to model only the positive values in the data (Zeileis and others 2008). For the count part, we specified a negative binomial distribution with a log link to account for overdispersion (Welsh and others 1996). Models were fitted using the hurdle function (Zeileis and others 2008) in the pscl library 1.04.4 (Jackman 2012) for R 3.0 (R Development Core Team 2013).
For the zero part of the model, we fitted burn category (unburned, moderate severity, and high severity) as a main effect to examine the effect of the occurrence of fire, which initiates regeneration, and variation in fire severity which has a largely unknown effect on seedling presence (Ashton and Martin 1996). To parameterize the count part of the model, we fitted burn category and fire return interval as main effects. We also fitted an interaction between fire return interval and burn category but excluded the interaction term for short FRI because of limited data. Some of our climatic and topographic variables were correlated so we conducted a principal components analysis (PCA) using the princomp function in R to reduce the data into fewer, independent variables. The PCA included elevation, slope, TWI, temperature, and precipitation. We used the correlation matrix to standardize the variables as they were measured in different units. We fitted the first two principal components (see “Results” section) as main effects in the count part of the hurdle model to analyze the effect of environmental variables on seedling establishment. To account for variation in seedling abundance over the 7-month period of our surveys, we also fitted the number of days since fire at the time of the survey as a main effect. We conducted linear regression of each principal component separately on the other predictor variables (fire return interval × burn category + days since fire) to investigate multicollinearity. The adjusted R 2 values for these models were low (PC1 = 0.02, PC2 = −0.06) indicating that our environmental and fire-related variables were not confounded.
We obtained P values for the numeric parameters (principal components and days since fire) from the partial Wald test from the hurdle model summary. To obtain P values for burn category in the zero part and the interactive term in the count part, we compared the full model to a model without each term separately using likelihood ratio tests. We estimated seedling abundance from the model conditional on the presence of seedlings. To determine if seedling abundance differed among fire return interval and burn categories we calculated the relative number of seedlings between each category and 95% confidence intervals of the differences. Significant differences were inferred where, on the log scale, the confidence interval did not include zero. Differences reported on response scale (to compare effect sizes among the model terms) are indicated by confidence intervals that do not include one.
Results
We counted a total of 3,841 seedlings across all sites, excluding the site that we removed from the analysis that had 2,260 seedlings (total no. of seedlings per site in each burn category: unburned = 0.69; moderate severity = 84.36; high severity = 38.62). The log(theta) value in our hurdle model was small (0.286) indicating overdispersion and thus a more appropriate fit to the negative binomial than the Poisson error distribution. In the zero part of the hurdle model, burn category was a significant predictor of E. regnans seedling occurrence (P < 0.001; Table 2). Compared to unburned sites, there was a significantly greater probability of seedling presence on moderate severity (log(odds ratio) [95% confidence interval] = 3.77 [2.63, 4.91]) and high severity sites (2.40 [1.04, 3.76]). There was no difference in the probability of seedling presence between moderate and high severity sites (−1.37 [−2.81, 0.07]).
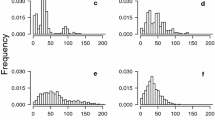
In the count part of the model, there was an interaction between burn category and fire return interval (P = 0.053; Table 2; Figure 2A). Given the presence of seedlings, there were significantly more seedlings (between 5 and 36 times more) on moderate and high severity sites compared with unburned sites in each FRI category (Figure 2B). There were no significant differences in seedling abundance between the moderate and high severity sites in any FRI category (Figure 2B). However, abundance estimates on burned sites were highly variable (Figure 2A), probably in part because of limited sample sizes in most categories (Table 1). There were significantly more seedlings in the long FRI category compared with the intermediate FRI category on unburned (6.19 times more) and moderate severity sites (2.39 times more) (Figure 2C). On high severity sites there were significantly more seedlings in the intermediate compared with the short FRI category (4.49 times more) (Figure 2C).
The interactive effect of burn category and fire return interval (FRI) on Eucalyptus regnans seedling abundance (conditional on presence) within 1 year of fire. A Model estimates and 95% confidence intervals, calculated at mean values of PC1, PC2, and days since fire, B difference in seedling abundance between burn categories for each FRI level and C difference in seedling abundance between FRI levels for each burn category. For B and C cases where the 95% confidence interval does not overlap zero indicate a significant difference. One long FRI, high severity site with 2,260 seedlings was removed from the analysis.
The first two principal components (PC) of the PCA explained a cumulative total of 83% of the variation in the environmental variables (PC1 = 56%, PC2 = 27%). PC1 had high loadings on temperature (loading = 0.583), precipitation (−0.558), and elevation (−0.586) and PC2 had high loadings on slope (−0.707) and TWI (0.706). Both principal components significantly affected E. regnans seedling abundance in the count part of the hurdle model (Table 2). The effect of PC1 showed that seedling abundance was greater at high elevation sites with low annual mean temperature and high precipitation, than at low elevation sites with high temperature and low precipitation (Figure 3A). The maximum effect size of PC1 (that is, the number of times more seedlings at the minimum PC value compared with the maximum, on the response scale [95% confidence interval]) was 9.96 [2.77, 35.78]. The effect of PC2 showed that seedling abundance was greater at sites with flatter terrain (low slope) and greater soil moisture availability (high TWI) than at sites with high slope and low TWI (Figure 3B). The maximum effect size of PC2 (maximum PC value compared with the minimum) was 10.10 [1.87, 54.42]. Our model also showed that seedling abundance significantly decreased over the 7-month sampling period (Table 2; Figure 4).
The estimated effect (and 95% point-wise confidence intervals) of principal components (PC) on Eucalyptus regnans seedling abundance (conditional on presence) for sites with intermediate fire return interval and moderate fire severity. A Precipitation and elevation were negatively correlated with temperature on PC1 and B slope and topographic wetness index (TWI) were negatively correlated on PC2.
Discussion
We aimed to determine the relative influence of fire-related variables and environmental factors on seedling establishment in E. regnans, an obligate seeder that dominates the wet montane forests of south-eastern Australia. A key result to emerge from our study was that climatic and topographic environmental variables and fire return interval were both strong drivers of first-year seedling establishment in E. regnans. Seedlings were more abundant in areas of flat terrain with high soil moisture, and at the high end of their elevational range where precipitation is high and temperature is low. We also found that post-fire seedling establishment in E. regnans seedlings was greater at sites with longer fire return intervals. Overall, our results suggest that the effects of changes in climate and fire regimes may both affect the regeneration potential and thus the distribution of E. regnans in the future. As the region becomes warmer and drier during this century (Christensen and others 2007) low elevations within the range of E. regnans could become unsuitable for seedling establishment, potentially driving an elevational range shift. This threat may be compounded by climate change-driven increases in fire frequency that are predicted for south-eastern Australia (Bradstock 2010).
Elevational range shifts have been documented in tree species from many ecosystems in response to climatic change over the past century (for example, Kharuk and others 2010; Lenoir and others 2008). Variation in seed dispersal is an important predictor of the likelihood that the range of a species can shift to track changes in climate (Berland and others 2011; Dullinger and others 2012). If there is a mismatch between the rate of climate change and the dispersal rate (or the ability to evolve higher dispersal Boeye and others 2013), local extinctions and range contractions may occur (Foden and others 2007; Nathan and others 2011). Early descriptive research reported that seed dispersal in E. regnans was limited to approximately one tree height (65 m) (Cunningham 1957) suggesting a limited capacity for a range shift. However, rates of long distance seed dispersal may increase in open habitats (for example, after fire) because obstacles are reduced (Nathan and others 2008). It will therefore be important to gain a more detailed understanding of seed dispersal in E. regnans, for example by quantifying dispersal kernels under different conditions. Interspecific competition (particularly with E. delegatensis, the dominant species at higher elevations) is also likely to influence the ability for E. regnans to undergo a range shift (Dullinger and others 2004). Our results showing strong sensitivity to climatic variation suggest that areas currently occupied by E. regnans forest at lower elevations could be transformed into a novel ecosystem state following future fires in warmer climates (Lindenmayer and others 2011; Westerling and others 2011). The loss of this keystone overstorey species would change the structure and function of the ecosystem and have negative impacts on over 40 vertebrate species that rely on the hollows that develop in E. regnans trees for nesting and shelter (Lindenmayer and others 2012b).
Although fire is essential for regeneration in E. regnans, among burned sites we found no effect of fire severity on the presence or abundance of seedlings. A range of factors have been suggested to control seedling establishment in E. regnans, and these have contrasting relationships with fire severity (conceptualized in Figure 5). These factors include increases in light (Ashton and Turner 1979), soil nutrients (Chambers and Attiwill 1994), seed harvest by ants (Ashton and Willis 1982), ectomycorrhizal associations (Launonen and others 1999), reduced inter- and intra-specific competition from adult plants (Ashton and Willis 1982), and decreased fungal attack on seedlings (Ashton and Willis 1982). Antagonisms among these factors may have masked any effects of fire severity in our study. For example, levels of available light and soil nutrients at our high severity sites may have been optimal for seedling growth (Ashton and Turner 1979; Weston and Attiwill 1990) but increased seed mortality during fire (Moreira and others 2010) may have concealed any positive effects of fire severity (Figure 5). Furthermore, seed reserves during initial establishment reduce the reliance on light and soil nutrients for growth (Pérez-Ramos and others 2012). The lower levels of light and greater structural complexity in ground cover that we observed on our moderate fire severity sites may therefore have had beneficial effects on seedlings by allowing them to retain water (Figure 5).
Factors affecting seedling establishment in Eucalyptus regnans and their relationship (positive = +, negative = −) with fire severity. *Although seed input increases after fire seeds may be killed under very high severity fire. Seed input may also increase with increasing fire return interval and the effects of fire return interval interact with fire severity.
The effects of fire severity on seedling establishment in other plant species are varied and often nonlinear (for example, Davies and others 2010). In two Mediterranean pine species, the highest levels of early seedling establishment occurred after low (Pinus pinaster, Maia and others 2012) or moderate severity fire (Pinus canariensis, Otto and others 2010). Both of these studies pointed to increased seed mortality as a key driver of low levels of seedling establishment after high severity fire. However, in another obligate seeding eucalypt species (E. delegatensis), first-year seedling density and growth were greatest after high severity fire, most likely because of reduced competition from surviving plants (Vivian and others 2008). The effects of fire severity on regeneration are therefore complex and can change over long time scales due to variation in growth and mortality rates (Pausas and others 2003).
Fire return interval is a critical parameter influencing the persistence of obligate seeders (Franklin and others 2005; Pausas and others 2004). In our study, fire return interval influenced the abundance of seedlings that established after fire, and these effects depended on fire severity. On unburned and moderately burned sites seedling abundance was greater after long fire return intervals compared with intermediate fire return intervals, whereas on severely burned sites seedling abundance was greater after intermediate compared with short fire return intervals. The latter result may have been driven by, or exacerbated by the effects of logging at some sites in the short FRI category. However, the difference between intermediate and long fire return intervals shows that increases in fire frequency without the effects of logging can reduce seedling establishment. Taken together, these results reflect an increase in seedling abundance with increasing forest age, indicating greater regeneration potential with longer fire return intervals.
Seed storage potential of E. regnans forest increases with forest age (Ashton 1975) providing an explanation for the general increase in seedling establishment with fire return interval. Yet it is unclear why certain aspects of the effect of fire return interval are expressed under different fire severities. The conditions immediately after fire have strong associations with fire severity (Figure 5) and a number of these conditions may interact with seed input and germination to explain our results. For example, the effect of greater seedling establishment after long compared with intermediate fire return intervals may have disappeared on severely burned sites if seed mortality during fire was high enough to equalize the seed input between the two FRI categories (Figure 5). Alternatively, high severity fire may have eliminated any structural differences in the forest floor (for example, ground cover creating safe sites for germination) between the intermediate and long FRI categories and this may have influenced seedling establishment. The range of possible explanations cannot be firmly separated in this study but provide important starting points for future work on the influence of micro-environmental characteristics and reproductive output (for example, seed fall) under different fire regimes.
The important message in the effect of fire return interval on seedling establishment in E. regnans is that increases in fire frequency could decrease regeneration potential. In many ecosystems, including Australian temperate forests, fire frequency is predicted to increase under climate change (Cary and others 2012; Liu and others 2010). The predicted shorter fire return intervals could compound the direct impacts of warming and drying on E. regnans seedling establishment. For other montane forest species, simulation models have shown that cumulative or synergistic effects of changes in climate and fire regimes can result in abrupt changes in ecosystem structure and function, particularly at low elevations (Fyllas and Troumbis 2009; Schumacher and Bugmann 2006). Approximately 36% of the total mainland E. regnans forest was burned in 2009. If another wildfire occurs in this area within the maturation time of E. regnans (15–20 years) the species would be unable to regenerate and the ecosystem would likely become dominated by Acacia and potentially other Eucalyptus species (Lindenmayer and others 2011). Beyond this critical maturation period, our results suggest that fire return intervals of 26–83 years do not result in the same seedling establishment rates that develop in old-growth forest after long fire return intervals. Our survey site with 2,260 seedlings provides evidence for the massive regeneration potential of old-growth forest. Old-growth forest can decrease fire spread across landscapes due to hump-shaped age-flammability relationships (see Kitzberger and others 2012). Protecting old-growth forest, and younger forest so it can become old-growth forest, will not only help maintain regeneration potential but may also moderate increases in fire frequency. Urgent changes to current forest management practices are essential to achieve this protection (Lindenmayer and others 2012a).
Our discussion has focussed on seedling abundance as a measure of regeneration potential in E. regnans but we recognize that seedling density can have negative effects on survival and growth through intraspecific competition (self-thinning) (Stankova and Diéguez-Aranda 2013; but see Woodruff and others 2002). If self-thinning was the sole process governing the survival and growth of seedlings after establishment there might be little consequence of our findings for future forest structure and adult tree density. However, our results provide evidence that density is strongly affected by environmental factors including climate, topography, and fire. Under extreme environmental conditions or fire regime scenarios (for example, higher temperatures or repeated frequent fire) seedling density could therefore decline below that required for successful regeneration.
Understanding the relative importance of fire regime and climatic factors on regeneration in obligate seeders is critical to predicting how future global change will affect their distribution and abundance. Previous studies have identified that potential changes in fire regimes are the dominant threat to obligate seeders (for example, Swab and others 2012), whereas other studies indicate that the direct effects of climate change are the main threatening factors (for example, Lawson and others 2010). Our study indicated that both climatic and topographic factors and fire return interval have a strong influence on first-year seedling establishment in E. regnans, broadening our understanding of the drivers of regeneration in obligate seeders. Longer term trends in seedling growth and survival may show different responses to variation in fire regimes, and we emphasize this as a priority for research.
References
Ashton DH. 1975. Studies of flowering behaviour in Eucalyptus regnans F. Muell. Aust J Bot 23:399–411.
Ashton DH. 1976. The development of even-aged stands of Eucalyptus regnans F. Muell. in central Victoria. Aust J Bot 24:397–414.
Ashton DH. 1981. Fire in tall open-forests (wet sclerophyll forests). In: Gill AM, Groves RH, Noble IR, Eds. Fire and the Australian Biota. Canberra: Australian Academy of Science. p 339–66.
Ashton DH, Attiwill PM. 1994. Tall open-forests. In: Groves RH, Ed. Australian vegetation. Melbourne: Cambridge University Press. p 157–96.
Ashton DH, Martin DG. 1996. Regeneration in a pole-stage forest of Eucalyptus regnans subjected to different fire intensities in 1982. Aust J Bot 44:393–410.
Ashton DH, Turner JS. 1979. Studies on the light compensation point of Eucalyptus regnans F. Muell. Aust J Bot 27:589–607.
Ashton DH, Willis EJ. 1982. Antagonisms in the regeneration of Eucalyptus regnans in the mature forest. In: Newman EI, Ed. The plant community as a working mechanism. London: British Ecological Society. p 113–28.
Bauweraerts I, Wertin TM, Ameye M, McGuire MA, Teskey RO, Steppe K. 2013. The effect of heat waves, elevated [CO2] and low soil water availability on northern red oak (Quercus rubra L.) seedlings. Glob Change Biol 19:517–28.
Berland A, Shuman B, Manson SM. 2011. Simulated importance of dispersal, disturbance, and landscape history in long-term ecosystem change in the Big Woods of Minnesota. Ecosystems 14:398–414.
Beven KJ, Kirkby MJ. 1979. A physically based, variable contributing area model of basin hydrology. Hydrol Sci Bull 24:43–69.
Boeye J, Travis JMJ, Stoks R, Bonte D. 2013. More rapid climate change promotes evolutionary rescue through selection for increased dispersal distance. Evol Appl 6:353–64.
Boland DJ, Brooker MIH, Chippendale GM, Hall N, Hyland BPM, Johnston RD, Kleinig DA, McDonald MW, Turner JD. 2006. Forest trees of Australia. 5th edn. Melbourne: CSIRO Publishing.
Bradstock RA. 2010. A biogeographic model of fire regimes in Australia: current and future implications. Glob Ecol Biogeogr 19:145–58.
Bradstock RA, Bedward M, Cohn JS. 2006. The modelled effects of differing fire management strategies on the conifer Callitris verrucosa within semi-arid mallee vegetation in Australia. J Appl Ecol 43:281–92.
Bradstock RA, Williams RJ, Gill AM. 2012. Future fire regimes of Australian ecosystems: new perspectives on enduring questions of management. In: Bradstock RA, Gill AM, Williams RJ, Eds. Flammable Australia: fire regimes, biodiversity and ecosystems in a changing world. Collingwood, VIC: CSIRO Publishing. p 307–24.
Brook BW, Sodhi NS, Bradshaw CJA. 2008. Synergies among extinction drivers under global change. Trends Ecol Evol 23:453–60.
Cary GJ, Bradstock RA, Gill AM, Williams RJ. 2012. Global change and fire regimes in Australia. In: Bradstock RA, Gill AM, Williams RJ, Eds. Flammable Australia: fire regimes, biodiversity and ecosystems in a changing world. Collingwood, VIC: CSIRO Publishing. p 149–69.
Chambers DP, Attiwill PM. 1994. The ash-bed effect in Eucalyptus regnans forest: chemical, physical and microbiological changes in soil after heating or partial sterilisation. Aust J Bot 42:739–49.
Christensen JH, Hewitson B, Busuioc A, Chen A, Gao X, Held I, Jones R, Kolli RK, Kwon W-T, Laprise R, Magaña Rueda V, Mearns L, Menéndez CG, Räisänen J, Rinke A, Sarr A, Whetton P. 2007. Regional climate projections. Solomon S et al., editors. Climate change 2007: the physical science basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge: Cambridge University Press. pp. 847–940
Cunningham TM. 1957. Seed production and seed fall of Eucalyptus regnans (F. Muell). Aust For 21:30–9.
Davies GM, Smith AA, MacDonald AJ, Bakker JD, Legg CL. 2010. Fire intensity, fire severity and ecosystem response in heathlands: factors affecting the regeneration of Calluna vulgaris. J Appl Ecol 47:356–65.
DNRE. 1998. Forest management plan for the Central Highlands. Melbourne: Department of Natural Resources and Environment, State Government of Victoria.
Driscoll DA, Felton A, Gibbons P, Felton AM, Munro NT, Lindenmayer DB. 2012. Priorities in policy and management when existing biodiversity stressors interact with climate-change. Clim Change 111:533–57.
Driscoll DA, Lindenmayer DB, Bennett AF, Bode M, Bradstock RA, Cary GJ, Clarke MF, Dexter N, Fensham R, Friend G, Gill AM, James S, Kay G, Keith DA, MacGregor C, Russell-Smith J, Salt D, Watson JEM, Williams RJ, York A. 2010. Fire management for biodiversity conservation: key research questions and our capacity to answer them. Biol Conserv 143:1928–39.
Dullinger S, Dirnböck T, Grabherr G. 2004. Modelling climate change-driven treeline shifts: relative effects of temperature increase, dispersal and invasibility. J Ecol 92:241–52.
Dullinger S, Gattringer A, Thuiller W, Moser D, Zimmermann NE, Guisan A, Willner W, Plutzar C, Leitner M, Mang T, Caccianiga M, Dirnböck T, Ertl S, Fischer A, Lenoir J, Svenning J-C, Psomas A, Schmatz DR, Silc U, Vittoz P, Hülber K. 2012. Extinction debt of high-mountain plants under twenty-first-century climate change. Nat Clim Change 2:619–22.
Fisher JL, Loneragan WA, Dixon K, Delaney J, Veneklaas EJ. 2009. Altered vegetation structure and composition linked to fire frequency and plant invasion in a biodiverse woodland. Biol Conserv 142:2270–81.
Foden W, Midgley GF, Hughes G, Bond WJ, Thuiller W, Hoffman MT, Kaleme P, Underhill LG, Rebelo A, Hannah L. 2007. A changing climate is eroding the geographical range of the Namib Desert tree Aloe through population declines and dispersal lags. Divers Distrib 13:645–53.
Fordham DA, Akçakaya HR, Araújo MB, Elith J, Keith DA, Pearson R, Auld TD, Mellin C, Morgan JW, Regan TJ, Tozer M, Watts MJ, White M, Wintle BA, Yates C, Brook BW. 2012. Plant extinction risk under climate change: are forecast range shifts alone a good indicator of species vulnerability to global warming? Glob Change Biol 18:1357–71.
Franklin J. 2010. Moving beyond static species distribution models in support of conservation biogeography. Divers Distrib 16:321–30.
Franklin J, Syphard AD, He HS, Mladenoff DJ. 2005. Altered fire regimes affect landscape patterns of plant succession in the foothills and mountains of southern California. Ecosystems 8:885–98.
Fyllas NM, Troumbis AY. 2009. Simulating vegetation shifts in north-eastern Mediterranean mountain forests under climatic change scenarios. Glob Ecol Biogeogr 18:64–77.
Gill AM. 1981. Adaptive responses of Australian vascular plant species to fires. In: Gill AM, Groves RH, Noble IR, Eds. Fire and the Australian Biota. Canberra: Australian Academy of Science. p 243–72.
Gill AM, Catling PC. 2002. Fire regimes and biodiversity of forested landscapes of southern Australia. In: Bradstock RA, Williams JE, Gill AM, Eds. Flammable Australia: the fire regimes and biodiversity of a continent. Cambridge: Cambridge University Press. p 351–69.
Hutchinson MF. 2011. ANUDEM version 5.3, user guide. Canberra: Fenner School of Environment and Society, Australian National University.
Jackman S. 2012. pscl: Political Science Computational Laboratory, Stanford University. Stanford, CA: Department of Political Science.
Keith DA, Akçakaya HR, Thuiller W, Midgley GF, Pearson RG, Phillips SJ, Regan HM, Araújo MB, Rebelo TG. 2008. Predicting extinction risks under climate change: coupling stochastic population models with dynamic bioclimatic habitat models. Biol Lett 4:560–3.
Kharuk VI, Ranson KJ, Im ST, Vdovin AS. 2010. Spatial distribution and temporal dynamics of high-elevation forest stands in southern Siberia. Glob Ecol Biogeogr 19:822–30.
Kitzberger T, Aráoz E, Gowda JH, Mermoz M, Morales JM. 2012. Decreases in fire spread probability with forest age promotes alternative community states, reduced resilience to climate variability and large fire regime shifts. Ecosystems 15:97–112.
Land Conservation Council. 1994. Melbourne area district 2 review: final recommendations. Melbourne: Land Conservation Council.
Launonen TM, Ashton DH, Keane PJ. 1999. The effect of regeneration burns on the growth, nutrient acquisition and mycorrhizae of Eucalyptus regnans F. Muell. (mountain ash) seedlings. Plant Soil 210:273–83.
Lawson DM, Regan HM, Zedler PH, Franklin J. 2010. Cumulative effects of land use, altered fire regime and climate change on persistence of Ceanothus verrucosus, a rare, fire-dependent plant species. Glob Change Biol 16:2518–29.
Lenoir J, Gégout JC, Marquet PA, de Ruffray P, Brisse H. 2008. A significant upward shift in plant species optimum elevation during the 20th Century. Science 320:1768–71.
Lindenmayer DB. 2009. Forest pattern and ecological process. A synthesis of 25 years of research. Melbourne: CSIRO Publishing.
Lindenmayer DB, Blanchard W, McBurney L, Blair D, Banks PB, Likens GE, Franklin JF, Laurance WF, Stein JAR, Gibbons P. 2012a. Interacting factors driving a major loss of large trees with cavities in a forest ecosystem. PLoS One 7(10):e41864.
Lindenmayer DB, Cunningham RB, Donnelly CF, Franklin JF. 2000. Structural features of old-growth Australian montane ash forests. For Ecol Manag 134:189–204.
Lindenmayer DB, Cunningham RB, MacGregor C, Incoll RD, Michael D. 2003. A survey design for monitoring the abundance of arboreal marsupials in the central highlands of Victoria. Biol Conserv 110:161–7.
Lindenmayer DB, Hobbs RJ, Likens GE, Krebs CJ, Banks SC. 2011. Newly discovered landscape traps produce regime shifts in wet forests. Proc Natl Acad Sci USA 108:15887–91.
Lindenmayer DB, Laurance WF, Franklin JF. 2012b. Global decline in large old trees. Science 338:1305–6.
Lindenmayer DB, Likens GE, Franklin JF. 2010. Rapid responses to facilitate ecological discoveries from major disturbances. Front Ecol Environ 8:527–32.
Lindenmayer DB, Ough K. 2006. Salvage logging in the montane ash eucalypt forests of the Central Highlands of Victoria and its potential impacts on biodiversity. Conserv Biol 20:1005–15.
Liu Y, Stanturf J, Goodrick S. 2010. Trends in global wildfire potential in a changing climate. For Ecol Manag 259:685–97.
Mackey BG, Lindenmayer DB, Gill AM, McCarthy MA, Lindesay J. 2002. Wildlife, fire and future climate: a forest ecosystem analysis. Collingwood: CSIRO Publishing.
Maia P, Pausas JG, Vasques A, Keizer JJ. 2012. Fire severity as a key factor in post-fire regeneration of Pinus pinaster (Ait.) in Central Portugal. Ann For Sci 69:489–98.
McCarthy MA, Gill AM, Lindenmayer DB. 1999. Fire regimes in mountain ash forest: evidence from forest age structure, extinction models and wildlife habitat. For Ecol Manag 124:193–203.
Metz MR. 2012. Does habitat specialization by seedlings contribute to the high diversity of a lowland fain forest? J Ecol 100:969–79.
Moen J, Cairns DM, Lafon CW. 2008. Factors structuring the treeline ecotone in Fennoscandia. Plant Ecol Divers 1:77–87.
Moreira B, Tormo J, Estrelles E, Pausas JG. 2010. Disentangling the role of heat and smoke as germination cues in Mediterranean Basin flora. Ann Bot 105:627–35.
Nathan R, Horvitz N, He Y, Kuparinen A, Schurr FM, Katul GG. 2011. Spread of North American wind-dispersed trees in future environments. Ecol Lett 14:211–19.
Nathan R, Schurr FM, Spiegel O, Steinitz O, Trakhtenbrot A, Tsoar A. 2008. Mechanisms of long-distance seed dispersal. Trends Ecol Evol 23:638–47.
Otto R, García-del-Rey E, Muñoz PG, Fernández-Palacios JM. 2010. The effect of fire severity on first-year seedling establishment in a Pinus canariensis forest on Tenerife, Canary Islands. Eur J For Res 129:499–508.
Pausas JG, Bradstock RA, Keith DA, Keeley JE, Network GF. 2004. Plant functional traits in relation to fire in crown-fire ecosystems. Ecology 85:1085–100.
Pausas JG, Ouadah N, Ferran A, Gimeno T, Vallejo R. 2003. Fire severity and seedling establishment in Pinus halepensis woodlands, eastern Iberian Peninsula. Plant Ecol 169:205–13.
Pérez-Ramos IM, Urbieta IR, Zavala MA, Marañón T. 2012. Ontogenetic conflicts and rank reversals in two Mediterranean oak species: implications for coexistence. J Ecol 100:467–77.
R Development Core Team. 2013. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org
Regan HM, Crookston JB, Swab R, Franklin J, Lawson DM. 2010. Habitat fragmentation and altered fire regime create trade-offs for an obligate seeding shrub. Ecology 91:1114–23.
Romme WH, Boyce MS, Gresswell R, Merrill EH, Minshall GW, Whitlock C, Turner MG. 2011. Twenty years after the 1998 Yellowstone fires: lessons about disturbance and ecosystems. Ecosystems 14:1196–215.
Russell-Smith J, Edwards AC, Price OF. 2012. Simplifying the savanna: the trajectory of fire-sensitive vegetation mosaics in northern Australia. J Biogeogr 39:1303–17.
Schumacher S, Bugmann H. 2006. The relative importance of climatic effects, wildfires and management for future forest landscape dynamics in the Swiss Alps. Glob Change Biol 12:1435–50.
Silvestrini RA, Soares-Filho BS, Nepstad D, Coe M, Rodrigues H, Assunção R. 2011. Simulating fire regimes in the Amazon in response to climate change and deforestation. Ecol Appl 21:1573–90.
Stankova TV, Diéguez-Aranda U. 2013. Simple and reliable models of density decrease with dominant height growth for even-aged natural stands and plantations. Ann For Sci 70:621–30.
Swab RM, Regan HM, Keith DA, Regan TJ, Ooi MKJ. 2012. Niche models tell half the story: spatial context and life-history traits influence species responses to global change. J Biogeogr 39:1266–77.
Tng DYP, Williamson GJ, Jordan GJ, Bowman DMJS. 2012. Giant eucalypts—globally unique fire-adapted rain-forest trees? New Phytol 196:1001–14.
Vivian LM, Cary GJ, Bradstock RA, Gill AM. 2008. Influence of fire severity on the regeneration, recruitment and distribution of eucalypts in the Cotter River Catchment, Australian Capital Territory. Austral Ecol 33:55–67.
Welsh AH, Cunningham RB, Donnelly CF, Lindenmayer DB. 1996. Modelling the abundance of rare species: statistical models for counts with extra zeros. Ecol Model 88:297–308.
Westerling AL, Turner MG, Smithwick EAH, Romme WH, Ryan MG. 2011. Continued warming could transform Greater Yellowstone fire regimes by mid-21st century. Proc Natl Acad Sci USA 108:13165–70.
Weston CJ, Attiwill PM. 1990. Effects of fire and harvesting on nitrogen transformations and ionic mobility in soils of Eucalyptus regnans forests of south-eastern Australia. Oecologia 83:20–6.
Wood SW, Hua Q, Allen KJ, Bowman DMJS. 2010. Age and growth of a fire prone Tasmanian temperate old-growth forest stand dominated by Eucalyptus regnans, the world’s tallest angiosperm. For Ecol Manag 260:438–47.
Woodruff DR, Bond BJ, Ritchie GA, Scott W. 2002. Effects of stand density of the growth of young Douglas-fir trees. Can J For Res 32:420–7.
Xu T, Hutchinson MF. 2011. ANUCLIM version 6.1, user guide. Canberra: Fenner School of Environment and Society, Australian National University.
Zeileis A, Kleiber C, Jackman S. 2008. Regression models for count data in R. J Stat Softw 27:1–21.
Acknowledgments
We thank John Stein for assistance with compiling spatial data and Sue McIntyre for constructive criticism on the manuscript. Valuable feedback from two anonymous reviewers and the editors helped to improve the manuscript. This project was supported by funds from the Australian Research Council, the Australian Government Department of Sustainability, Environment, Water, Population and Communities, the Victorian Government Department of Sustainability and Environment, Parks Victoria, Melbourne Water, the Thomas Foundation and the Earthwatch Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Contributions
This study was designed and conceived by ALS, DBL, DB, LM, SCB, PSB, WB, DAD, and AMG. ALS and WB developed and conducted the analysis. The field sites were established by DBL with input from DB and LM who also collected the field data. ALS wrote the manuscript and incorporated contributions from all authors.
Rights and permissions
About this article
Cite this article
Smith, A.L., Blair, D., McBurney, L. et al. Dominant Drivers of Seedling Establishment in a Fire-Dependent Obligate Seeder: Climate or Fire Regimes?. Ecosystems 17, 258–270 (2014). https://doi.org/10.1007/s10021-013-9721-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-013-9721-9