Abstract
The Canarian pine (Pinus canariensis) exhibits a striking combination of high adult resistance to fire and intermediate serotiny. Hence, the study of its post-fire regeneration can support valuable new insights about functional adaptations to fire. Here, we analyse the first-year seedling establishment after fire in a P. canariensis forest on the northern slope of Tenerife, Canary Islands. The effects of fire severity and other explanatory variables on the seedling density recorded 9 months after fire were examined. We detected a clear unimodal relationship between seedling density and fire severity, with maximum regeneration associated with intermediate fire severity and no regeneration associated with very high crown damage. The results suggested that high severity fires may have caused the partial destruction of the aerial seed bank and/or the creation of unfavourable seedbed conditions for germination and seedling emergence. The density of large pine trees, reflecting seed availability, was the second most important factor explaining the distribution of seedlings. Cover of scorched needle litter on the ground correlated strongly and positively with pine seedling density and negatively with fire severity. The complete lack of regeneration at sites most strongly affected by fire does not represent a major threat for the stand recovery of the Canarian pine, due to the very high tree resistance to fire and the tremendous capacity of the Canarian pine to resprout after fire. The observed very high seedling densities at sites with intermediate fire impacts can probably be related to both the complete liberation of the seed bank (including seeds stored in serotinous cones) and favourable micro-environmental conditions for seed germination and seedling establishment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The importance of fire in influencing the structure and dynamics of Mediterranean ecosystems has long been highlighted (Trabaud et al. 1985; Naveh 1990; Moreno and Oechel 1994; Begon et al. 1996). Disturbances related to wildland fires also determine the regeneration of ecosystems made up of different pine species (Agee 1998; Fernandes et al. 2008), which employ various strategies in response to fire intensity and frequency (Keeley and Zedler 1998). Despite the abundant scientific investigations published for some Mediterranean pine species (Thanos et al. 1996; Eshel et al. 2000; De Las Heras et al. 2002; Ne’eman et al. 1992; Pausas et al. 2003; Daskalakou and Thanos 2004; Nathan and Ne’eman 2004; Vega et al. 2008), there exist very few studies on fire ecology for the Canarian pine (Höllermann 1993, 2000; Arévalo et al. 2001; Climent et al. 2004).
The Canarian pine, Pinus canariensis Chr. Sm. ex DC., is an endemic species of the Canary Islands and forms up to 60% of the forest area of the archipelago. Most of the traits associated with fire resistance, such as thick bark, long needles, large buds, a tall growth habit, self-pruning, deep rooting and longevity and/or capacity to resprout, are well developed in this species (Climent et al. 2004). It has also been reported that this pine produces a considerable proportion of serotinous cones, similar to Mediterranean serotinous pines such as Pinus halepensis and Pinus pinaster (Tapias et al. 2004). The degree of serotiny of the Canarian pine seems to be higher for stands in the north of Tenerife, reaching 60% (Tapias et al. 2004), than in the south of the island, which was related to higher levels of productivity and, in consequence, to higher frequency and intensity of fires in the north of the island (Climent et al. 2004). Although this unusual combination of resistance and recovery to fire is striking, quantitative information about the behaviour of the Canarian pine after fire is very scarce and was never studied in natural stands, only in pine plantations (Arévalo et al. 2001).
Fire severity reflects the impact of fire on vegetation and soil (Whelan 2002). However, there exist no generally accepted quantitative indicators or measures for fire severity. Commonly, the degree of fuel consumption in the understory or canopy layer (Pérez and Moreno 1998) or the level of crown damage (Pausas et al. 2003; Vega et al. 2008) is considered, but the degree of mortality of trees and shrubs has also been used to estimate fire severity (Chappell and Agee 1996). Fire intensity and severity usually vary considerably at the landscape scale, producing a patchiness of post-fire biotic and abiotic conditions (Turner et al. 1994), due to topographical and micro-climatic variations and fuel accumulation (Gómez-Tejedor et al. 1999; Miller and Urban 2000).
Several studies have examined regeneration patterns in relation to fire severity in Mediterranean pine forests (Pausas et al. 2003; Broncano and Retana 2004; Fernández et al. 2008; Vega et al. 2008). Initial seedling density can be negatively influenced by high levels of crown damage (Fernández et al. 2008; Vega et al. 2008). This has, on the one hand, been interpreted as a result of a partial destruction of the aerial seed bank due to high fire intensity. Other studies showed no clear relationship between seedling density and fire severity (Pausas et al. 2003; Broncano and Retana 2004). Seedling emergence after fire can be very high despite almost full crown damage for the serotinous P. halepensis (Tsitsoni 1997; Ne’eman et al. 1992), while Pinus ponderosa, a non-serotinous pine, showed very low regeneration levels in areas with completely damaged canopies (Bonnet et al. 2005).
Apart from fire severity, other factors such as topography, exposure, aspect and climatic conditions during the growing season after fire or micro-environmental conditions have been reported to influence post-fire regeneration in pine forests (Tsitsoni 1997; Ne’eman and Izhaki 1998; Eshel et al. 2000; Pausas et al. 2004; Daskalakou and Thanos 2004; Broncano and Retana 2004; Bonnet et al. 2005). Ash deposition under adult trees can have negative effects on pine post-fire regeneration (Eshel et al. 2000), but fire can also increase nutrient availability (Notario del Pino et al. 2008; Durán et al. 2008) and enhance seedling growth (Pausas et al. 2003). Bonnet et al. (2005) described the presence of scorched needle litter on blackened mineral soil as safe sites for ponderosa pine regeneration. Until now, neither the effects of fire severity nor environmental factors on post-fire regeneration for the Canarian pine have been studied.
The aim of this paper is to analyse early post-fire regeneration in natural stands of P. canariensis after an intense fire that affected 11,458 ha of pine forest on the northern slope of Tenerife in July 2007, relating the establishment of seedlings in the first year after fire to fire severity and other explanatory variables. Our working hypothesis was that seedling density depended on seed availability, which in turn was determined by the distribution of the aerial seed bank, fire severity and environmental factors such as altitude, substrate age and type. A second objective was to examine pine tree mortality.
Materials and methods
Study area
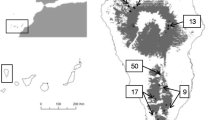
The study area of about 8,000 ha is located in the north of Tenerife and included the Corona Forestal Natural Park (28º18′40″N, 16º43′19″W), ranging from 300 to 2,700 m and occupying a total area of 46.613 ha (Martín Esquivel et al. 1995; Fig. 1). This area was burned in 1983 and again on the 27th of July 2007 (i.e. 24 years later), affecting 11.458 ha of mostly pine forest. The fire in 2007 spread very fast due to exceptionally high air temperatures (>40°C), very low air humidity (<20%) and strong eastern winds from Africa.
Two main types of pine forests, dominated exclusively by the endemic P. canariensis Chr. Sm. ex DC., can be distinguished on the northern slope of Tenerife. The first is the humid pine forest that occurs below 1,400 m and is strongly influenced by the north-east trade winds. The second is the dry pine forest that occurs above 1,400 m and is also found in the south of the island. The understory of the first type can be dense and is characterised by shrubs or small trees such as Erica arborea, Myrica faya, Ilex canariensis and Daphne gnidium, all species that are found in the evergreen laurel forest, situated below 1,000 m. Above 1,400 m, the understory is dominated by Chamaecytisus proliferus and Adenocarpus viscosus, two leguminous shrubs that are well adapted to more arid conditions. Abundant in both pine forests are Cistus symphytifolius, Lotus campylocladus and Asphodelus aestivus, as well as some annuals such as Wahlenbergia lobelioides, Tuberaria guttata, Trifolium ligusticum or Cardamine hirsuta (Rivas-Martínez et al. 1993). Annual precipitation reaches 800 mm at the most humid sites on the northern slopes at about 1,000 m, whereas fog drip can substantially increase overall precipitation (Marzol 2008). The drier sites typically receive 400–500 mm of precipitation per year. Mean annual temperature varies between 9 and 15°C within the altitudinal range of the Canarian pine.
According to Del Arco (2006), natural pine forests originally covered ca. 43,000 ha on Tenerife, of which the majority was destroyed during the first three centuries of European settlement. Since the beginning of the twentieth century, great efforts were made to restore the original pine forest distribution. Pine forests are now considered fundamental for ecosystem stability and conservation and are protected on all Canary Islands (Martín Esquivel et al. 1995). Therefore, they are no longer managed for timber production. However, some silvicultural activities still exist, such as thinning in an attempt to naturalise pine plantations. Pine stands in the study area are considered natural.
Sampling design
In the burned area, a total of 23 UTM quadrats (1 km × 1 km) were selected randomly after stratification by geological age of substrate (Carrecedo et al. 2004). Seven quadrats were situated on very young substrate (<3,000-year-old lava flows), and 16 were situated on older substrate (Fig. 1). The first substrate type is very abundant in the centre of the study area but not at the island level. Without stratification, this type would have been over-represented in this study.
Within each UTM quadrat, we randomly selected one permanent 10 m × 10 m (100 m2) plot; this plot size is frequently used to study post-fire regeneration in pine forests (Tsitsoni 1997; Arévalo et al. 2001; Pausas et al. 2003). Within these plots, we installed five subplots (1 m × 1 m in each corner and in the centre) to monitor pine seedling emergence and several micro-environmental variables following the sampling design of other studies (Ne’eman and Izhaki 1998; Elliott and Vosse 2005). Since this intense fire had burned all natural pine stands in the north and north-west of Tenerife, from the lower limit at the transition zone with the laurel forest at 600 m to the upper timber line at 2,000 m, and the closest unburned stands in the north of Tenerife consisted of pine plantations, no control plots could be found for comparison. Nevertheless, Srutek et al. (2002) described natural regeneration of pine stands at the tree line above 1,700 m near the study area that may be comparable to our results.
At the plot level (100 m2), we recorded the following biotic and abiotic parameters: altitude, slope (degrees), age of substrate following the geological map of Carrecedo et al. (2004), tree DBH (diameter at breast height), total tree density (individuals with DBH > 5 cm), tree density by DBH ranges (DBH < 25 cm, 25 < DBH < 50 cm, DBH > 50 cm), total shrub cover (although the understory was heavily burned, pre-fire cover was roughly estimated by the structure of the burned individuals), basal area and post-fire mortality of pine trees (DBH > 5 cm).
Within the subplots, we visually estimated litter cover and cover of rocks (pieces > 30 cm in diameter), stones (pieces with diameter between 2 and 30 cm) and uncovered soil (pieces < 2 cm in diameter). Mean canopy cover was measured with a spherical convex densitometer (Lemmon 1956), always from the same corner of the subplot and in the same direction. Fallen cones and pine seeds at the soil surface were counted. All seedlings were tagged in May 2008, 9 months after fire and at the end of the rainy season and just before the summer drought started. P. canariensis does not maintain a soil seed bank but an aerial seed bank, and, consequently, all seedlings that emerged from seeds dispersed after fire come from normal or serotinous cones. Germination started with the first heavy winter rains that occurred in November and December 2007 (Fig. 2). There was a second peak of precipitation in February/March 2008. Regeneration was recorded in May 2008, and at 3 and 5 months, respectively, after the two peaks of germination. Therefore, we assume that most of the germinated seedlings were recorded, with the only missing seedlings being those desiccated early in the rainy season between January and March 2008 due to unfavourable micro-environmental conditions. However, we cannot exclude the effect of predation on seeds and seedlings in the first month after fire.
Fire severity was determined according to the degree of crown damage measured at the nearest six adult trees from the plot centre; damage was visually estimated by rating the percentage of green needles at the top of the crown with respect to the whole crown (see similar classifications, Pausas et al. 2003; Vega et al. 2008). Instead of dividing arbitrarily the severity gradient into two or three classes (Pausas et al. 2003; Vega et al. 2008), we decided to use a continuous variable.
Since P. canariensis has a good capacity to resprout after intense canopy fires, individuals with highly damaged crowns that retain neither green nor scorched needles are rarely dead but rather are able to produce new green needles from the stem or thick branches within a few months after fire. New green needles produced after the fire could be perfectly distinguished from partly undamaged green crowns.
Statistical analyses
In a first step, we constructed a correlation matrix using non-parametric Spearman rank correlation coefficients to explore relationships among explanatory variables. To avoid collinearity (multicollinearity) and eliminate redundant information, we then selected a set of explanatory variables to enter in the GAM and GLM analysis by eliminating those variables that were highly correlated with each other (r > 0.70) and exhibited low tolerance statistics (<0.3) in ordinary least squares regression analysis (OLS). This was done in a stepwise procedure, where the variable with the lowest tolerance value was always eliminated, until all the variables in the equation showed a tolerance of >0.4.
In a second step, we used non-parametric models, such as regression trees (Zuur et al. 2007) and generalised additive models (GAM; Hasti and Tibshirani 1990), to evaluate the effect of explanatory variables such as fire severity and environmental factors on seedling density. These regression techniques deal better with non-linearity than ordinary least squares regression (OLS) and can be used to find interactions missed by other methods in ecological studies (Zuur et al. 2007). Most of the diagnostic tests for fitting linear regression models have analogues in fitting GAMs, with the exception of concurvity, the non-parametric analogue of multicollinearity (Hasti and Tibshirani 1990). At present, most statistical programs provide no diagnostic tools for assessing the impact of concurvity on a fitted GAM. However, multicollinearity is strictly a problem in the design matrix and does not depend on the nature of the link to the response. Therefore, it can always be detected with an OLS procedure that provides collinearity diagnostics (Miles and Shevlin 2001; Wood 2006).
Regression trees, a relatively new technique in ecology, were used to explore the relationship between a single response variable (seedling density) and multiple explanatory variables (biotic and abiotic factors) (De’Ath and Fabricus 2000; Zuur et al. 2007). A regression tree is created by a process of repeatedly partitioning the observations into two homogenous groups based on the values (order) of an explanatory variable. This statistical technique also indicates the relative importance of different explanatory variables. Tree models are not affected by transformations of the explanatory variables. Cross-validation was used to select the optimal tree size and to avoid complex suboptimal trees.
Generalised additive models assume that the mean of the dependent variable depends on an additive predictor through a non-linear link function. Generalised additive models permit the response probability distribution to be any member of the exponential family of distributions. AIC (Akaike information criteria) with forward stepwise selection was used to obtain the optimal set of predictor variables, where the lowest AIC value for every possible combination of explanatory variables was chosen, starting with the explanatory variable with the best fit. The splines used were simplified to a maximum of four degrees of freedom. Partial predictions for significant predictors were plotted, and 95% confidence bands were constructed.
In a third step, parametric generalised linear models (GLM; McCullagh and Nelder 1989; Dobson 1990) were applied, including the same explanatory variables, using linear or quadratic terms in the case of unimodal relationships and best set selection with AIC for the optimal model. For generalised additive and linear models, we selected a Poisson error distribution using a log-link function, as recommended for count data in ecological analysis (Crawley 1993). The use and comparison of several statistical approaches on the same set of ecological data, as applied here, is recommended since it can improve the statistical and ecological conclusions (Zuur et al. 2007). The following statistical programs Brodgar 2.5.6. (www.brodgar.com) and STATISTICA 8 were used for the multivariate analyses.
Results
Variation of explanatory variables
The 23 sampling plots were located between 900 and 1,700 m, within the typical range for pine forests in the north of Tenerife, where the timber line is reached at 2,000 m (Table 1). Plots were not very inclined and were exposed mainly to the north and north-west. The amount of rocks, stones and uncovered soil varied considerably depending on the age of the lava flow; e.g. plots with the youngest lava flows had high rock cover (up to 95%). Basal area ranged between 4 and 75 m2/ha. Since we did not find any fallen trees in our plots, these values represent nearly pre-fire area basal. All pine trees (DBH > 5 cm) within the studied plots were still alive, resprouting from stem and larger branches, meaning that tree mortality = 0%. Post-fire mean canopy cover was 38% (16–59%). Mean density of pine trees (DBH > 5 cm) was 3.83 ind./0.01 ha, reaching 11 ind./0.01 ha in one plot.
The analysis of the correlation matrix revealed that basal area was positively correlated with the number of large trees (DBH > 25 cm) and negatively correlated with rock cover. Pine tree density showed a positive relationship with mean canopy cover, which was negatively correlated with altitude. The amount of scorched needles on the ground correlated positively with basal area, the number of larger trees and the number of cones. There were not any pine seeds found at the soil surface within the subplots (n = 115).
Fire severity, considered one of the most important explanatory variables in this study, varied considerably within the area affected by this canopy fire. Mean crown damage reached 77%, but within very short distances (200 m) we could detect areas with almost intact green crowns (50% affected) and areas with completely burned crowns. Fire severity exhibited a positive but not very strong correlation with pre-fire understory cover (Spearman rank coefficient r = 0.53, P = 0.01) and was also positively related, but not significantly, to higher density of small trees (r = 0.36, P = 0.088). A high correlation was found between fire severity and needle litter on the ground (r = 0.61, P = 0.002). To avoid collinearity for the GAM and GLM models, we skipped 10 of the 18 explanatory variables for the regression analysis (see Table 1).
Post-fire seedling density
A total of 564 established P. canariensis seedlings were recorded and tagged. The average seedling density was 4.9 ind./m2, with a maximum value of 16.2 ind./m2, which is equal to 162.000 ind./ha (Table 1; Fig. 3). No dead seedlings could be detected. The regression tree model with eight explanatory variables and seedling density as the response variable (Fig. 4) selected fire severity as the most important explanatory variable. Seedling density was strongly negatively influenced by a fire severity greater than or equal to 87.5%. That means that the highest seedling densities can be expected for plots with pine canopies affected less that 87.5%. The second most important variable in explaining seedling density was the number of large trees (DBH > 25 cm), which had a positive effect when number of trees was higher than 1.5 ind./0.01 ha. Both factors accounted for 42.6% of the original deviance. The fact that fire severity appeared again further down in the regression tree indicates that the relationship between the two variables is not linear. The highest seedling densities could be found in areas where tree canopies were damaged between 45 and 82.5% and where density of large trees was >1.5 ind./0.01 ha.
Regression tree model including eight explanatory variables and seedling density as response variable. The observations are repeatedly split into two groups. If a statement is true, follow the left side of a branch, and if false follow the right side. The numbers at the bottom of a terminal leaf represent the mean abundance value and the number of observations in that particular group (n)
Results of GAMs with seedling density as a response variable using the Poisson distribution and a log-link function are shown in Table 2. AIC methods and forward selection was applied to select the optimal model, which included the same two variables: fire severity and large trees (DBH > 25 cm). Fire severity was again the most important variable in explaining the variation in seedling density. The splines for each significant explanatory variable with the 95% confidence bands are represented in Fig. 5. As indicated by the rather large degrees of freedom for the smoother curve representing fire severity, the corresponding smoothing curve revealed a unimodal relationship between fire severity and seedling density, with a maximum seedling density for areas with 75% crown damage. From this point, seedling density decreased drastically with increasing fire severity. The shape of the smoother for the number of large trees indicates a nearly linear positive relationship. Analysis of the residuals of the optimal additive model showed no clear patterns and a good fit.
The GLM approach using the Poisson distribution, AIC best set selection and the same explanatory variables, including a quadratic term of fire severity, yielded a model where fire severity (linear and quadratic terms), large trees, altitude and geological age of the substrate were selected (Table 3). Fire severity was the most important factor in explaining seedling distribution, followed by the variable number of large trees.
Seedling density was also strongly and positively related to the cover of scorched needles on the ground (Spearman rank correlation coefficient r = 0.74). However, this factor was not included in the previous models due to collinearity with fire severity and the number of larger trees.
Discussion
Fire severity and pine regeneration
All three statistical techniques used confirmed a quadratic response of post-fire pine seedling establishment to fire severity. The GAM smoothing curve showed that maximum seedling density of 14–16 ind./m2 was found in areas with crowns 70–80% affected by fire. Surprisingly, we could not find any pine seedlings in the seven plots highly affected by crown fire nor seeds on the ground, despite the intermediate level of serotinous cones (35%) reported for the burned pine stands in the north of Tenerife (Gil et al. 2002; Climent et al. 2004). This suggests that the canopy seed bank was either significantly damaged by fire or seedbed conditions were markedly unfavourable for seed germination and seedling emergence (or both factors). The observed negative correlation between fire severity and litter cover on the ground makes it difficult to disentangle the two effects on seedling density 9 months after fire. Nevertheless, because some favourable micro-sites might always be available for germination and establishment, the complete lack of pine regeneration at these plots suggests that destruction of seeds and cones in the canopy related to high fire intensity could be an important factor in explaining this pattern. However, only experimental studies in early post-fire conditions could clarify the importance of the scorched needle layer for pine regeneration.
There exist very few studies about the effect of fire on natural regeneration for P. canariensis. Very low sapling densities (0.014 ind./m2) in post-fire regeneration were reported for a pine plantation (Arévalo et al. 2001), probably due to low seed and cone production. Seedling/sapling densities (individual height ≤ 10 cm) in unburned natural stands at the tree line in the north-west of Tenerife were also reported to be rather low (0.34 ind./m2, Srutek et al. 2002). With respect to Mediterranean pine forests, several studies deal with fire severity and early post-fire regeneration. Pausas et al. (2003) and Broncano and Retana (2004) did not find a clear relation between fire severity (measured by crown tree damage level) and P. halepensis seedling density. Fernández et al. (2008) and Vega et al. (2008) found a weak and negative influence of crown damage level on initial P. pinaster seedling density after fire. In these latter cases, decreases were explained, on the one hand, by the partial destruction of the aerial seed bank.
Pausas et al. (2003) showed that fire severity strongly influenced post-fire soil conditions (e.g. needle litter cover, organic matter inputs) that may have implications for germination, emergence, growth and survival of seedlings. A positive effect of needle litter over burned mineral soil was confirmed for post-fire regeneration of Pinus ponderosa (Bonnet et al. 2005) and a negative effect of ash layer under adult trees for P. halepensis regeneration (Eshel et al. 2000). A positive effect of soil organic cover reduction on initial seedling density after fire was found for P. pinaster (Vega et al. 2008) and P. sylvestris (Hille and Den Ouden 2004). Regarding P. canariensis, we hypothesise that the consumption of litter by fire on the ground and the transformation into an ash layer, combined with the subsequent cover of fallen scorched needles, may favour pine seed germination and seedling establishment by increased nutrient availability (Notario del Pino et al. 2008) and favourable micro-climatic conditions, but the complete lack of a protecting needle layer could negatively influence pine regeneration. However, further studies should focus on the effects of micro-environmental conditions on the regeneration of the Canarian pine in the very early stages of post-fire succession.
Stand characteristics and environmental factors
The second most important variable explaining the distribution of pine seedlings was the number of large trees (DBH > 25 cm). This variable showed a linear relationship with seedling density and probably reflects the size of the aerial seed bank because the Canarian pine starts to produce flowers and cones quite late (age of first flowering 15–20 years, Tapias et al. 2004). Seed availability has been found to strongly determine post-fire regeneration of pine species (de Groot et al. 2004; Bonnet et al. 2005; Vega et al. 2008). Basal area, here not related to regeneration, possibly reflects better overall site productivity, while abundance of large trees indicates a certain degree of maturity of the stand. Mean canopy cover of natural stands can also be positively related to seed production or to favourable post-fire micro-environmental conditions for germination and survival of seedlings (Bonnet et al. 2005).
Serotiny and its influence on regeneration of P. canariensis has not been analysed in detail thus far (Tapias et al. 2004). Although serotiny was not measured in this study, the maximum seedling density of P. canariensis in areas with intermediate fire severity could indicate the liberation of the total aerial seed bank, including an important part stored in serotinous cones. For the clearly serotinous Mediterranean pine P. halepensis (Daskalakou and Thanos 2004; Tapias et al. 2004), massive regeneration has also been reported after fire (14.0 ind./m2, Tsitsoni 1997; 5–20 ind./m2, Ne’eman et al. 1992; 5.5 ind./m2, Thanos et al. 1996; 4 ind./m2, Daskalakou and Thanos 2004), values similar to the observed densities in the present study.
Very young substrate had a negative effect on pine seedling density. This environmental factor was also negatively correlated with basal area and the number of large trees, indicating a smaller aerial seed bank compared to more productive sites. Moreover, favourable micro-environmental conditions probably are scarce and hydric stress could increase with increasing rockiness. A negative effect of rockiness on post-fire regeneration was reported for P. pinaster (Vega et al. 2008).
Altitude influenced weakly and negatively seedling density independently from fire severity and the number of large trees. Since fire severity was not related to tree cover or altitude, we suggest that environmental factors such as precipitation and temperature could explain this weak trend. The area above 1,400 m is less influenced by the cloud layer caused by the trade winds and, therefore, the climate is drier and colder and favours the formation of dry pine forests. While analysing unburned natural Canarian pine stands along an elevational gradient between 1,700 and 2,000 m in the same region at La Guancha in the north-west of Tenerife, Srutek et al. (2002) observed a decrease in basal area, tree density, canopy cover and seedling/sapling density with altitude, which was interpreted as a response to environmental stress, i.e. decreases in productivity. The decrease in canopy cover could also represent lower levels of shading and, as a consequence, a reduced number of favourable micro-sites for germination and establishment of seedlings.
Results show that the influence of rockiness and altitude on seedling density was only significant when applying GLMs. Furthermore, we could confirm that GLMs also cope very well with non-linear relationships, if quadratic terms are included, but data should first be explored by means of regression trees or GAMs to detect non-linear relationships as suggested by Zuur et al. (2007), otherwise there is a risk of missing important explanatory variables.
Fire severity and tree mortality
According to the applied classification of fire severity by the degree of crown damage, the wildland fire of 2007 in the northern region of Tenerife could be characterised by a high severity ground fire (understory: 100% affected) and a medium-to-high severity canopy fire (crowns: 77% affected). Even in the seven plots where needles and small branches of the crowns were completely consumed (real crown fire), we could not detect any tree mortality, which reflects the extraordinary fire resistance of P. canariensis in burned natural stands. Although mentioned before (Ceballos and Ortuño 1976; Höllermann 2000) and related to several life history traits (Climent et al. 2004), here we confirm this functional adaptation with quantitative data. The ongoing monitoring of pine regeneration will reveal if all the trees that were highly affected by fire will survive the next years. Fire severity, therefore, negatively affects the vitality of the Canarian pine but rarely causes mortality of individuals.
Conclusions
With respect to post-fire seedling distribution in natural P. canariensis stands, we propose a hierarchical scheme: The aerial seed bank, i.e. seeds stored in normal and serotinous cones, determine the potential level of seed rain that can fall to the ground after release to be transformed into a seedling bank. Fire severity modifies this potential seed rain by liberating seeds enclosed in serotinous cones or by partially destroying seeds and cones in the canopy. In a second step, post-fire micro-environmental conditions such as the amount of pine needle litter on the ground or the degree of rockiness of the substrate determine the success of germination and establishment in the first year.
The complete lack of regeneration at sites most affected by fire does not represent a major threat for the stand recovery of the Canarian pine, due to its very high tree resistance to fire and its tremendous capacity to resprout after fire. The observed very high seedling densities at sites with intermediate fire impacts can probably be related to both the complete liberation of the seed bank (including seeds stored in serotinous cones) and favourable micro-environmental conditions for seed germination and seedling establishment. However, we hypothesise that regeneration at these sites will not be functional due to relatively fast canopy closure in the next years.
The occurrence of the striking combination of the two main strategies to cope with wildland fire, high adult resistance and variable light or intermediate serotiny, has been related to the unpredictable fire regimes deriving from volcanism in subtropical areas (Climent et al. 2004). This highlights the importance of serotinous trees for contributing to stand recruitment after fire. Our observation, that no recruitment seems to occur in highly burned areas where seeds released from serotinous cones would have high light availability for several years and, therefore, good chances to establish, calls into question this functional adaptation to fire in case of the Canarian pine. Long-term monitoring of post-fire regeneration will probably reveal which are the best conditions for pine seedling establishment after fire.
References
Agee JK (1998) Fire and pine ecosystems. In: Richardson DM (ed) Ecology and biogeography of Pinus. Cambridge University Press, Cambridge, pp 193–218
Arévalo JR, Fernández-Palacios JM, Jiménez MJ, Gil P (2001) The effect of fire intensity on the understory composition of two Pinus canariensis reforested stands in Tenerife (Canary Islands). For Ecol Manage 148:21–29
Begon M, Harper JL, Townsend CR (1996) Ecology: individuals, populations, and communities, 3rd edn. Blackwell, Oxford
Bonnet VH, Schoettle AW, Shepperd WD (2005) Postfire environmental conditions influence the spatial pattern of regeneration for Pinus ponderosa. Can J For Res 35:37–47
Broncano MJ, Retana J (2004) Topography and forest composition affecting the variability in fire severity and post-fire regeneration occurring after a large fire in the Mediterranean basin. Int J Wildland Fire 13:209–216
Carrecedo JC, Guillou H, Paterne M, Scaillet S, Rodríguez E, Paris R, Pérez FJ, Hansen A, Bermejo JA, Pestana G, Cabrera JM, Rodríguez E, García A (2004) Avance de un Mapa de Peligros Volcánicos de Tenerife: Escenarios previsibles para una futura erupción en la isla. Caja Canarias, Obras Social y Cultural, Santa Cruz de Tenerife
Ceballos L, Ortuño F (1976) Vegetación y flora forestal de las Canarias Occidentales. Excmo. Cabildo Insular de Tenerife. Sta Cruz de Tenerife, pp 433
Chappell CB, Agee JK (1996) Fire severity and tree seedling establishment in Abies magnifica forests, Southern Cascades, Oregon. Ecol Appl 6:628–640
Climent J, Tapias R, Pardos JA, Gil L (2004) Fire adaptations in the Canary Islands pine (Pinus canariensis). Plant Ecol 171:185–196
Crawley MJ (1993) GLIM for ecologists. Blackwell Scientific Publications, Oxford
Daskalakou EN, Thanos CA (2004) Postfire regeneration of Aleppo pine—the temporal pattern of seedling recruitment. Plant Ecol 171:81–89
De Groot WJ, Bothwell PM, Taylor SW, Wotton BM, Stocks BJ, Alexander ME (2004) Jack pine regeneration and crown fires. Can J For Res 34:1634–1641
De las Heras J, Martínez-Sánchez JJ, González-Ochoa AI, Ferrandis P, Herranz JM (2002) Establishment of Pinus halepensis Mill. saplings following fire: effects of competition with shrub species. Acta Oecol 23:91–97
De′Ath G, Fabricus KA (2000) Classification and regression trees: a powerful yet simple technique for ecological data analysis. Ecology 81:3178–3192
Del Arco M (ed) (2006) Mapa de Vegetación de Canarias. GRAFCAN, Santa Cruz de Tenerife
Dobson AJ (1990) An introduction to generalized linear models. Chapman and Hall, London
Durán J, Rodríguez A, Fernández-Palacios JM, Gallardo A (2008) Changes in soil N and P availability in a Pinus canariensis fire chronosequence. For Ecol Manage 256:384–387
Elliott KJ, Vosse JM (2005) Effects of understory prescribed burning on shortleaf pine (Pinus echinata Mill.)/mixed-hardwood forests. J Torrey Bot Soc 132:236–251
Eshel A, Henig-Sever H, Ne’eman G (2000) Spatial variation of seedling distribution in an east Mediterranean pine woodland at the beginning of post-fire succession. Plant Ecol 148:175–182
Fernandes PM, Vega JA, Jiménez E, Rigolot E (2008) Fire resistance of European pines. For Ecol Manage 256:246–255
Fernández C, Vega JA, Fonturbel T, Jiménez E, Pérez-Gorostiaga P (2008) Effects of wildfire, salvage logging and slash manipulation on Pinus pinaster Ait. recruitment in Orense (NW Spain). For Ecol Manage 255:1294–1300
Gil L, Climent J, Nanos N, Mutke S, Ortiz I, Schiller G (2002) Cone morphology variation in Pinus canariensis Sm. Plant Syst Evol 235:35–51
Gómez-Tejedor JA, Estrela MJ, Millán MM (1999) A mesoscale model application to fire weather winds. Int J Wildland Fire 9:255–263
Hasti T, Tibshirani R (1990) Generalized additive models. Chapmann Hall, New York
Hille M, Den Ouden J (2004) Improved recruitment and early growth of Scots pine (Pinus sylvestris L.) seedlings after fire and soil scarification. Eur J Forest Res 123:213–218
Höllermann P (1993) Fire ecology in the Canary Island and Central California—a comparative outline. Erdkunde 47:177–184
Höllermann P (2000) The impact of fire in Canarian ecosystems 1983–1998. Erdkunde 54:70–75
Keeley JE, Zedler PH (1998) Evolution of life histories in Pinus. In: Richardson DM (ed) Ecology and biogeography of Pinus. Cambridge University Press, Cambridge, pp 219–250
Lemmon PE (1956) Densiometer for estimating forest overstory density. Forest Sci 2:314–320
Martín Esquivel JL, García Court H, Redondo Rojas CE, García Fernández I, Carralero Jaime I (1995) La Red Canaria de Espacios Naturales Protegidos. Consejería de PolíticaTerritorial, Viceconsejería de Medio Ambiente, Gobierno de Canarias
Marzol MV (2008) Temporal characteristics and fog water collection during summer in Tenerife (Canary Islands, Spain). Atmos Res 87:352–361
McCullagh P, Nelder JA (1989) Generalized linear models. Chapman and Hall, London
Miles J, Shevlin M (2001) Applying regression and correlation. A guide for students and researchers. SAGE Publications LTD, London
Miller C, Urban DL (2000) Connectivity of forest fuels and surface fire regimes. Landscape Ecol 15:145–154
Moreno JM, Oechel WC (eds) (1994) The role of fire in Mediterranean-type ecosystems. Springer, Nueva York, Estados Unidos
Nathan R, Ne’eman G (2004) Spatiotemporal dynamics of recruitment in Aleppo pine (Pinus halepensis Miller). Plant Ecol 171:123–137
Naveh Z (1990) Fire in the Mediterranean—a landscape ecological perspective. In: Goldammer JF, Jenkins MJ (eds) Fire in ecosystems dynamics. Proceedings of the third international symposium in Freiburg, Frg, May 1989. Spb Academic Publishing By, The Hague, The Netherlands, pp 1–20
Ne’eman G, Izhaki I (1998) Stability of pre- and post-fire spatial structure of pine trees in Aleppo pine forest. Ecography 21:535–542
Ne’eman G, Lahav H, Izhaki I (1992) Spatial pattern of seedling one year after fire in a Mediterranean pine forest. Oecologia 91:365–370
Notario del Pino J, Dorta Almenar I, Rodríguez Rodríguez A, Arbelo Rodríguez C, Navarro Rivero FJ, Mora Hernández JL, Armas Herrera CM, Guerra García JA (2008) Analysis of the 1:5 soil:water extract in burnt soils to evaluate fire severity. Catena 74(3):246–255. doi:10.1016/j.catena.2008.03.001
Pausas J, Ouadah N, Ferran A, Gimeno T, Vallejo R (2003) Fire severity and seedling establishment in Pinus halepensis woodlands, eastern Ibernian Peninsula. Plant Ecol 169:205–213
Pausas J, Ribeiro E, Vallejo R (2004) Post-fire regeneration variability of Pinus halepensis in the eastern Iberian Peninsula. For Ecol Manage 203:251–259
Pérez B, Moreno JM (1998) Methods for quantifying fire severity in scrubland fires. Plant Ecol 139:91–101
Rivas-Martínez S, Wildpret W, Del Arco M, Rodríguez O, Pérez de Paz PL, García-Gallo A, Acebes JR, Díaz TE, Fenández F (1993) Las Comunidades vegetales de la Isla de Tenerife (Islas Canarias). Itinera 7:169–374
Srutek M, Dolezal J, Hara T (2002) Spatial structure and associations in a Pinus canariensis population at the Treeline, Pico del Teide, Tenerife, Canary Islands. Arct Antarct Alp Res 34:201–210
Tapias R, Climent J, Pardos JA, Gil L (2004) Life histories of Mediterranean pines. Plant Ecol 171:53–68
Thanos CA, Daskalakou EN, Nikolaidou S (1996) Early post-fire regeneration of a Pinus halepensis forest in Mount Párnis, Greece. J Veg Sci 7:273–280
Trabaud L, Michels C, Grosman J (1985) Recovery of burnt Pinus halepensis Mill. forests.ll. Pine reconstitution after wildfire. For Ecol Manage 13:167–179
Tsitsoni T (1997) Conditions determining natural regeneration after wildfires in the Pinus halepensis (Miller, 1768) forests of Kassandra Peninsula (North Greece). For Ecol Manage 92:199–208
Turner MG, Hargrove WW, Gardner RH, Romme WH (1994) Effects of fire on landscape heterogeneity in Yellowstone National Park, Wyoming. J Veg Sci 5:731–742
Vega JA, Fernández C, Pérez-Gorostiaga P, Fonturbel T (2008) The influence of fire severity, serotiny, and post-fire management on Pinus pinaster Ait. Recruitment in three burnt areas in Galicia (NW Spain). For Ecol Manage 256:1596–1603
Whelan RJ (2002) The ecology of fire. Cambridge University Press, Cambridge
Wood SN (2006) Generalized additive models. An introduction with R. Chapmann Hall, New York
Zuur AF, Ieno EN, Smith GM (2007) Analysing ecological data. Springer, USA
Acknowledgments
This study received full financial support from the Excmo. Cabildo Insular de Tenerife as a collaboration contract with the Ecology Department of the University of La Laguna and the Forest Anatomy, Physiology and Genetics unit of the E.T.S.I. Montes (Polytechnical University of Madrid).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Merino.
Rights and permissions
About this article
Cite this article
Otto, R., García-del-Rey, E., Muñoz, P.G. et al. The effect of fire severity on first-year seedling establishment in a Pinus canariensis forest on Tenerife, Canary Islands. Eur J Forest Res 129, 499–508 (2010). https://doi.org/10.1007/s10342-009-0347-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-009-0347-6