Abstract
This report describes clinicopathological findings, including genetic data of STAT6, in a solitary fibrous tumor (SFT)/hemangiopericytoma (HPC) of the central nervous system in an 83-year-old woman with a bulge in the left forehead. She noticed it about 5 months before, and it had grown rapidly for the past 1 month. Neuroradiological studies disclosed a well-demarcated tumor that accompanied the destruction of the skull. The excised tumor showed a prominent papillary structure, where atypical cells were compactly arranged along the fibrovascular core (‘pseudopapillary’). There was rich vasculature, some of which resembled ‘staghorn’ vessels. Mitotic figures were occasionally found. Whorls, psammoma bodies, or intra-nuclear pseudoinclusions were not identified. By immunohistochemistry, CD34 was strongly positive in the tumor cells, and STAT6 was localized in their nuclei. By reverse transcription-polymerase chain reaction (RT-PCR), an NAB2-STAT6 fusion gene, NAB2 exon6-STAT6 exon17, was detected, establishing a definite diagnosis of SFT/HPC. ‘Papillary’ SFT/HPC needs to be recognized as a possible morphological variant of SFT/HPC, and should be borne in mind in its diagnostic practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hemangiopericytoma (HPC) of the central nervous system (CNS) is a highly cellular and vascularized mesenchymal tumor with monotonous proliferation of tumor cells and well-developed ‘staghorn’ vessels [1]. Typically, the tumor cells are positive for CD34. On the other hand, solitary fibrous tumor (SFT) of the CNS is composed of spindle cells placed in fascicles among prominent collagen fibers with or without staghorn vessels [1, 2]. The tumor cells, albeit more intensely than HPC, are also positive for CD34. In contrast to soft tissue pathology where the use of ‘HPC’ has almost been abandoned because of the clinical and histologic similarities between the two entities, the neuropathology community still retains the use of ‘HPC’ because CNS HPCs are known to be clinically more aggressive than CNS SFTs [1–3].
Recently, the pathognomonic NAB2-STAT6 fusion gene located at chromosomal region 12q13 has been discovered for non-CNS SFT [4–7] as well as for CNS SFT and HPC [8], suggesting that SFT and HPC are likely a single entity (SFT/HPC), regardless of whether they are of non-CNS origin or of CNS origin. Furthermore, the aberrant, nuclear localization of STAT6, natively a cytoplasmic protein, which corresponds to the NAB2-STAT6 fusion gene, has been demonstrated for non-CNS SFT/HPC and CNS SFT/HPC by immunohistochemistry [8–10]. This immunoprofile is a useful surrogate marker of NAB2-STAT6 fusion, and thus considered highly specific for SFT/HPC [8–10].
In this report, we present a case of CNS SFT/HPC with a prominent papillary structure. Since this ‘papillary’ morphology is quite unusual for SFT/HPC, it was extremely difficult to make the histologic diagnosis; however, the nuclear expression of STAT6 and the NAB2-STAT6 fusion gene were demonstrated by immunohistochemistry and reverse transcription-polymerase chain reaction (RT-PCR), respectively, establishing a definite diagnosis of SFT/HPC.
Clinical summary
The patient was an 83-year-old woman with a bulge in the left forehead, which she had noticed about 5 months before. It had grown rapidly for the past 1 month, and she was admitted to our hospital. Head computed tomography (CT) (Fig. 1a–c) disclosed a well-demarcated tumor, which was about 5 cm in diameter and accompanied the destruction of the skull. The tumor was mildly enhanced by contrast medium (Fig. 1c). On head magnetic resonance imaging (MRI) (Fig. 1d–f), the tumor showed iso-intensity on T1 weighted image (T1WI, Fig. 1d) and hyper-intensity on fluid attenuated inversion recovery (FLAIR, Fig. 1e), and was homogenously enhanced by gadolinium (Fig. 1f). On positron emission tomography with 2-deoxy-2-[fluorine-18]fluoro-d-glucose integrated with computed tomography (FDG-PET/CT), the uptake of FDG by the tumor was mildly present, but other foci of abnormal uptake were not identified. Contrast-enhanced CT of the trunk and contrast-enhanced MRI of the whole spine excluded metastasis or dissemination. One month after the admission, the tumor was excised (Simpson grade II), which was then followed by irradiation for about 1.5 month. No recurrence was noted for the succeeding 1 year.
The pre-operative neuroradiological findings on head CT (a–c) and MRI (d–f). a The tumor is a bulging mass in the left forehead. It is well-demarcated and about 5 cm in diameter. b On CT in bone condition, the destruction of the skull is more evident. c The tumor is mildly enhanced by contrast medium (delayed phase). d On head MRI (T1WI), the tumor shows iso-intensity. e On head MRI (FLAIR), the tumor shows hyper-intensity. f The tumor is homogenously enhanced by gadolinium
Pathological findings
Formalin-fixed, paraffin-embedded sections prepared from the excised tumor served for histology and immunohistochemistry. All the sections for immunohistochemistry were stained with commercially available primary antibodies, using an immunoperoxidase method with diaminobenzidine as the chromogen. For detection of the NAB2-STAT6 fusion gene, total RNA was extracted from formalin-fixed, paraffin-embedded tissues, and RT-PCR was performed as was previously described [11, 12]. PCR primers, which had been designed as was reported [7], served for this analysis. Forward primers for exon 3, 6, 7 of NAB2 (5′-3′) are: F-NAB2-ex3, CCGAGAGAGCACCTACTTGTCC; F-NAB2-ex6B, TGCAGCAGACACTGATGGACGAG; F-NAB2-ex7, AGTTCGAGGAAGGGCTGCTGG.
Reverse primers for exon 3, 17, 18 of STAT6 (5′-3′) are: R-STAT6-ex3B, TCTCCAGCCAGTCACCCAGAAGA; R-STAT6-ex17B, CGGTCCCCCAGTGAGCGAATGG; R-STAT6-ex18B, GGTCATCTTGATGGTAGCTGGG.
A series of RT-PCR were performed in 3 × 3 ways of primer combination with amplification of GAPDH cDNA (110 base pairs) as a control of RNA quality. Direct sequencing of PCR products was performed with ABI BigDye terminator ver.3.1 (Applied Biosystems, Foster City, CA) and ABI Prism 3100-Avant Genetic analyzer (Applied Biosystems).
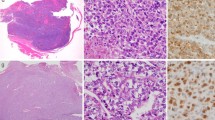
Grossly, the tumor was well-demarcated and whitish, involving the dura and the skull, but not the cerebrum, with extension to the skin (Fig. 2a). Histologically, the tumor showed a prominent papillary structure in which atypical cells were compactly arranged along the fibrovascular core (‘pseudopapillary’, Fig. 2b, c). In some areas, there was a solid pattern in which the tumor cells were compactly nested (Fig. 2d). The tumor had rich vasculature, some of which resembled ‘staghorn’ vessels (Fig. 2e). Mitotic figures were occasionally found (6–10/10 high power fields (HPFs): Fig. 2c, d). In a fraction of the tumor (<10 % in area), the tumor cells showed pleomorphism (Fig. 2f), but mitosis was not brisk. Necrosis was absent throughout the tumor. Whorls, psammoma bodies, or intra-nuclear pseudoinclusions were not identified. On reticulin silver impregnation, reticulin fibers were deposited in papillary areas (Fig. 3a, left column) as well as in solid areas (Fig. 3a, right column); but their deposition was not a pronounced feature. By immunohistochemistry, CD34 was strongly positive in the tumor cells (Fig. 3b), and STAT6 (Santa Cruz, sc-621, CA; ×100, PH6, autoclaving for antigen retrieval) was localized in their nuclei (Fig. 3c). Epithelial membrane antigen (EMA) was negative in the tumor cells. MIB-1 index (Ki-67) was around 10 %. On electron microscopy, the structures suggestive of desmosome or interdigitation were not identified. By RT-PCR, NAB2 exon 6-STAT6 exon17, which is one of the fusion genes so far reported for SFT/HPC [5], was detected (Fig. 4).
Macroscopic (a) and histologic (b–f) findings. a The tumor is well-demarcated and whitish, with the destruction of the skull. b The tumor shows a prominent papillary structure. c Atypical cells are arranged along the fibrovascular core (‘pseudopapillary’). Mitotic figures are occasionally found (arrows). d There is also a solid pattern in which the tumor cells are compactly nested. Mitotic figures are occasionally found (arrows). e The tumor has rich vasculature. Some of the vessels resemble ‘staghorn’. f Pleomorphic cells are also a participant in the tumor. Original magnification: b 200×; c 600×; d 400×; e 100×; f 200×
Reticulin silver impregnation (a) and immunohistochemistry (b, c). a Reticulin fibers are deposited in papillary areas (left column) as well as in solid areas (right column); but this is not a pronounced feature. b The tumor cells are strongly positive for CD34. c The tumor cells show nuclear expression of STAT6. Original magnification: a 400×; b 200×; c 400×
RT-PCR analyses of NAB2-STAT6 fusion genes using paraffin embedded tissues. a PCR-amplified DNA fragments and bands (arrowheads) shown to be true fusion genes after sequencing (arrows in lanes 5, 7 and 10 show DNA fragments of about 486, 175 and 308 bp, respectively). M means the 100 bp ladder marker. Lane 1 shows the result of PCR using GAPDH primer pairs (110 bp), and lanes 2–10 show the result of PCR using primer pairs F-NAB2-ex3/R-STAT6-ex3B, F-NAB2-ex7/R-STAT6-ex3B, F-NAB2-ex6B/R-STAT6-ex3B, F-NAB2-ex3/R-STAT6-ex17B, F-NAB2-ex7/R-STAT6-ex17B, F-NAB2-ex6B/R-STAT6-ex17B, F-NAB2-ex3/R-STAT6-ex18B, F-NAB2-ex7/R-STAT6-ex18B, and F-NAB2-ex6B/R-STAT6-ex18B, respectively. bp base pairs. b Result of direct sequencing analysis of 308 bp fragment in lane 10 in a. Sequence analysis shows the fusion of NAB2 exon 6 and STAT6 exon 17. This sequence result is exactly similar to those of DNA fragments in lanes 5 and 7 in a
Discussion
In the present case, the location of the tumor and the prominent papillary structure could suggest a diagnosis of papillary meningioma (PM). PM is characterized by perivascular pseudopapillary pattern with the fibrovascular core, and the tumor cells can display abundant cytoplasm, frequent mitoses, necroses, and brain invasion [13, 14]. These morphologic features, albeit not all of them, were also noted in the present case. However, whorls, psammoma bodies, or intranuclear inclusions [14], which may have corroborated the diagnosis of PM, were absent. On the other hand, the staghorn vessels or CD34-immunoreactivity in the present case could indicate a diagnosis of SFT/HPC, but the prominent papillary structure was likely to defy the diagnosis. In such a situation, the nuclear expression of STAT6 by immunohistochemistry and the detection of NAB2 exon 6-STAT6 exon17 by RT-PCR allowed us to make a definite diagnosis of SFT/HPC. Furthermore, the high mitotic count (6–10/10 HPFs) and the moderate to high degree of nuclear atypia and cellularity suggested that the tumor, according to the 2007 WHO classification of tumors of the CNS [1], is a WHO grade III HPC (anaplastic HPC).
The papillary pattern of SFT/HPC is unusual, but there have been a few reports of such cases. Table 1 summarizes clinicopathological features of these cases including ours. Tsukamoto et al. [12] reported a STAT6-positive intra-orbital papillary tumor in a 22-year-old woman. The tumor showed a papillary pattern, and staghorn vessels typical of SFT/HPC were not present. However, the tumor cells were positive for CD34, and the nuclei were positive for STAT6. Although the NAB2-STAT6 fusion gene was not detected by RT-PCR, this tumor can be an SFT/HPC mainly on immunohistochemical grounds. Another papillary SFT/HPC was reported by Tomek et al. [15], which described a 66-year-old man with a spinal, extradural SFT/HPC. The tumor showed a unique retiform and papillary architecture, and staghorn vessels typical of SFT/HPC were absent. However, the tumor cells were positive for CD34 by immunohistochemistry. Although immunostaining for STAT6, let alone the genetic analyses for NAB2-STAT6, was not performed, this tumor is also a probable SFT/HPC with a papillary pattern. Taken together with the present case, the papillary pattern is a possible morphology in SFT/HPC. ‘Papillary’ SFT/HPC needs to be borne in mind in the diagnostic practice of SFT/HPC.
References
Giannini C, Rushing EJ, Hainfellner JA (2007) Haemangiopericytoma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (eds) WHO classification of tumours of the central nervous system, 4th edn. IARC, Lyon, pp 178–183
Burger PC, Scheithauer BW, Vogel FS (2002) Intracranial meninges. In: Burger PC, Scheithauer BW, Vogel FS (eds) Surgical pathology of the nervous system and its coverings. Churchill Livingstone, New York, pp 49–112
Scheithauer BW, Fuller GN, VandenBerg SR (2008) The 2007 WHO classification of tumors of the nervous system: controversies in surgical neuropathology. Brain Pathol 18:307–316
Chmielecki J, Crago AM, Rosenberg M, O’Connor R, Walker SR, Ambrogio L, Auclair D, McKenna A, Heinrich MC, Frank DA, Meyerson M (2013) Whole-exome sequencing identifies a recurrent NAB2-STAT6 fusion in solitary fibrous tumors. Nat Genet 45:131–132
Barthelmes S, Geddert H, Boltze C, Moskalev E, Bieg M, Sirbu H, Brors B, Wiemann S, Hartmann A, Agaimy A, Haller F (2014) Solitary fibrous tumors/hemangiopericytomas with different variants of the NAB2-STAT6 gene fusion are characterized by specific histomorphology and distinct clinicopathological features. Am J Pathol 184:1209–1218
Mohajeri A, Tayebwa J, Collin A, Nilsson J, Magnusson L, von Steyern FV, Brosjö O, Domanski HA, Larsson O, Sciot R, Debiec-Rychter M, Hornick JL, Mandahl N, Nord KH, Mertens F (2013) Comprehensive genetic analysis identifies a pathognomonic NAB2/STAT6 fusion gene, nonrandom secondary genomic imbalances, and a characteristic gene expression profile in solitary fibrous tumor. Genes Chromosom Cancer 52:873–886
Robinson D, Wu Y, Kalyana-Sundaram S, Cao X, Lonigro R, Sung Y, Chen C, Zhang L, Wang R, Su F, Iyer M, Roychowdhury S, Siddiqui J, Pienta K, Kunju L, Talpaz M, Mosquera J, Singer S, Schuetze S, Antonescu C, Chinnaiyan A (2013) Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat Genet 45:180–185
Schweizer L, Koelsche C, Sahm F, PR M, Capper D, Reuss DE, Pusch S, Habel A, Meyer J, Göck T, Jones DT, Mawrin C, Schittenhelm J, Becker A, Heim S, Simon M, Herold-Mende C, Mechtersheimer G, Paulus W, König R, Wiestler OD, Pfister SM, von Deimling A (2013) Meningeal hemangiopericytoma and solitary fibrous tumors carry the NAB2-STAT6 fusion and can be diagnosed by nuclear expression of STAT6 protein. Acta Neuropathol 125:651–658
Koelsche C, Schweizer L, Renner M, Warth A, Jones DT, Sahm F, Reuss DE, Capper D, Knösel T, Schulz B, Petersen I, Ulrich A, Renker EK, Lehner B, Pfister SM, Schirmacher P, von Deimling A, Mechtersheimer G (2014) Nuclear relocation of STAT6 reliably predicts NAB2-STAT6 fusion for the diagnosis of solitary fibrous tumour. Histopathology 65:613–622
Yoshida A, Tsuta K, Ohno M, Yoshida M, Narita Y, Kawai A, Asamura H, Kushima R (2014) STAT6 immunohistochemistry is helpful in the diagnosis of solitary fibrous tumors. Am J Surg Pathol 38:552–559
Tsukamoto Y, Nakata Y, Futani H, Fukunaga S, Kajimto K, Hirota S (2013) A rare case of clear cell sarcoma with 4 types of EWSR1-ATF1 fusions detected not in primary site but in metastatic site. Pathol Res Pract 209:803–807
Tsukamoto Y, Watanabe T, Nishimoto S, Kakibuchi M, Yamada Y, Kohashi K, Oda Y, Hirota S (2014) STAT6-positive intraorbital papillary tumor: a rare variant of solitary fibrous tumor? Pathol Res Pract 210:450–453
Wang XQ, Chen H, Zhao L, Li ST, Hu J, Mei GH, Jiang CC (2013) Intracranial papillary meningioma: a clinicopathologic study of 30 cases at a single institution. Neurosurgery 73:777–790
Perry A, Louis DN, Scheithauer BW, Budka H, von Deimling A (2007) Meningiomas. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (eds) WHO classification of tumours of the central nervous system, 4th edn. IARC, Lyon, pp 164–172
Tomek M, Bravi I, Mendoza N, Alsafi A, Mehta A, Molinaro L, Singh P, Radotra B, Dei Tos AP, Roncaroli F (2013) Spinal extradural solitary fibrous tumor with retiform and papillary features. Ann Diagn Pathol 17:281–287
Acknowledgments
We thank Dr. Akihiko Yoshida, the Department of Pathology and Clinical Laboratory, National Cancer Center Hospital, Tokyo, Japan, for his invaluable advice and suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This study has no conflict of interest, no funding, or no sponsorship.
Rights and permissions
About this article
Cite this article
Ishizawa, K., Tsukamoto, Y., Ikeda, S. et al. ‘Papillary’ solitary fibrous tumor/hemangiopericytoma with nuclear STAT6 expression and NAB2-STAT6 fusion. Brain Tumor Pathol 33, 151–156 (2016). https://doi.org/10.1007/s10014-015-0247-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10014-015-0247-z